Abstract
Norepinephrine (NE) is a monoamine neurotransmitter and is mainly released from the locus coeruleus in the brain. Locus coeruleus has about 20,000 neurons that produce NE and project to many different regions of the brain. The α1 and α2 receptors have been found to influence attention, fear, working memory, and spatial learning. β1 and β2 have been found to work on fear memory, auditory fear, memory retrieval, and spatial reference. Tyrosine hydroxylase is the rate-limiting enzyme in both NE and dopamine synthesis. Based on the specific receptor affinity, noradrenergic drugs are classified into selective or direct-acting drugs and nonselective or indirectly acting drugs.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
History of Norepinephrine
Norepinephrine (NE) is a neurotransmitter of the brain that has a vital role in the modulation of cognition, arousal, attention, and stress responses. In the periphery, NE functions as a hormone as a part of the “fight or flight” response in the sympathetic nervous system [1]. NE was discovered in the peripheral nervous system (PNS), along with epinephrine. German physiologist, Otto Loewi, demonstrated that a substance released into the blood subsequent to sympathetic ganglion stimulation increased the heart rate, which was later identified as epinephrine. NE was considered an inactive precursor of epinephrine; however, Ulf von Euler, a Swedish physiologist, demonstrated that NE was also involved in the sympathetic response in 1940 [2, 3].
Marthe Vogt, in 1954, expanded the role of NE as a neurotransmitter in the central nervous system (CNS). In 1962, Falck and Hillarp applied histochemistry to brain structures, and NE was identified in both PNS and CNS. Dahlstrom and Fuxe, in 1964, demonstrated that locus coeruleus (LC) is the primary source of NE in the CNS through their experiments [2, 4]. Alpha-2 adrenoceptors were first identified on peripheral nerve endings in 1974 and on cell bodies and dendrites of central noradrenergic neurons in 1975 [5, 6].
Neurochemical Profile
The noradrenergic system in the brain has two primary ascending projections that arise from the brainstem: the dorsal noradrenergic bundle (DNB) and ventral noradrenergic bundle (VNB). DNB originates from LC and is mainly composed of noradrenergic neurons that project to the cerebral cortex, hippocampus, and cerebellum and intersects with the projections from the VNB to innervate the hypothalamus amygdala, and spinal cord. The VNB also innervates the midbrain and medulla along with the amygdala and hypothalamus. VNB plays a crucial role in controlling vegetative functions and endocrine regulation [7,8,9].
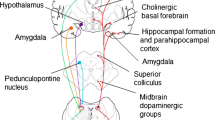
NE is released mainly from the LC in the brain. LC, a small pontine nucleus that is located in the lateral wall of the brainstem by the fourth ventricle. LC has about 20,000 neurons that produce central NE, while the medullary nuclei primarily function peripherally. Most of the noradrenergic neurons of the brain are found in LC. LC projects to most parts of the brain, forming both synaptic and nonsynaptic contacts, with projections to the forebrain, cerebellum, brainstem, and spinal cord. NE pathways have a wide innervation in the brain from olfactory tubercle to spinal cord and are shown below in Fig. 9.1 [10,11,12].
LC neurons produce NE from two types of NE cells, the large multipolar cells (35 μM) and smaller fusiform cells (20 μM). Their distribution in the LC is distinct—fusiform cells are located in the dorsal LC and multipolar cells in the ventral LC [13]. In addition to NE, galanin is also expressed in up to 80% of LC neurons, which is involved in regulating wake/sleep states, feeding, nociception, and parental behavior [14, 15].
The unique characteristic of LC is the diversity of projections and intense collateralization of the axons. LC projects to most parts of the brain and modulates information from various systems like sympathetic, parasympathetic, cortical, and limbic centers [12]. The sympathetic and parasympathetic nervous systems and their neurotransmitters are shown below in Fig. 9.2. The sympathetic nervous system and neuroendocrine chromaffin cells of the adrenal medulla synthesize and release NE and other catecholamines [2, 16].
Factors contributing to noradrenergic transmission are the diversity of noradrenergic receptors, tonic and phasic neurotransmission, and the nonlinear relationship between innervation and performance [17]. The noradrenergic system promotes wakefulness, arousal and facilitates sensory signal detection. Most studies have shown that NE influences cognition and behavior like working memory, attention, behavioral flexibility, and long-term mnemonic processing [18].
During anxiety or stress, the released NE and epinephrine are bound to adrenergic receptors throughout the body that exert effects like dilating pupils and bronchioles, increasing heart rate and constricting blood vessels, increasing renin secretion from the kidneys, and inhibiting peristalsis. NE plays a crucial role in metabolic effects like stimulating glycogenolysis and gluconeogenesis and inducing ketogenesis and lipolysis [19, 20].
Norepinephrine Receptors
NE acts through alpha and beta receptors, which are α1, α2, β1, β2, β3 adrenoreceptors. The various types of α1 receptors include α1a, α1b, and α1d receptors, which are found in LC, amygdala, thalamus, cerebral cortex, olfactory bulb, and dentate gyrus. α1 receptors act through the Gq protein signaling pathway, and the receptor pathway is shown below in Fig. 9.3. NE receptor and their mechanism of action are shown below in Table 9.1 [21, 22].
Noradrenaline has an excitatory action through its post-synaptic α1 and β-adrenoreceptors and inhibitory action through presynaptic α2-adrenoreceptors [17]. α2-adrenoreceptor is commonly found in prefrontal cortical areas, and NE has the highest affinity and lower affinity for α1 and β-adrenoreceptors, respectively. Thus the distribution and relationship of adrenoreceptors are variable, with moderate levels of NE activating α2 receptors, while higher levels activate lower-affinity α1 and β-adrenoreceptors [23,24,25].
α1 and α2 receptors have been found to influence attention, fear, working memory, and spatial learning. β1 and β2 have been found to work on fear memory, auditory fear, memory retrieval, and spatial reference [18].
The stimulation or inhibition of these functions is dependent on the agonism or antagonism of the adrenergic receptors. The α1 and β-adrenoreceptors promote neurotransmission and plasticity, thus enhancing the stimulatory effects in the CNS. At the same time, α2 receptors have inhibitory effects in the CNS, like decreasing NE release and reducing neuronal excitability [18].
In the periphery, acetylcholine (Ach) stimulates the release of adrenaline and noradrenaline. Ach binds to nicotinic receptors on adrenal chromaffin cells that generate action potentials via voltage-gated sodium and potassium channels. Calcium influx into the cytosol leads to binding of NE vesicles to the cell membrane causing NE release into the blood [26].
NE is removed from the synaptic cleft through presynaptic reuptake. NE transporters (NET) are found on presynaptic terminals that mediate the NE reuptake that can either be stored in the vesicles or undergo degradation [18, 27].
Norepinephrine Metabolism
NE is a monoamine neurotransmitter, and the chemical structure of Norepinephrine is shown below in Fig. 9.4. Tyrosine is an aromatic amino acid that acts as a precursor for the synthesis of dihydroxyphenylalanine (DOPA) by the action of tyrosine hydroxylase. Tyrosine hydroxylase is the rate-limiting enzyme in NE and dopamine synthesis. Dopamine is synthesized by the action of DOPA decarboxylase; dopamine is transported into vesicles through vesicular monoamine transporter (VMAT). Dopamine β hydroxylase (DBH) converts dopamine to NE. NE is converted to epinephrine via phenylethanolamine-N-methyltransferase, both in the CNS and adrenal medulla [2, 28]. The synthesis of NE is shown below in Fig. 9.5.
NE is degraded intracellularly or in the synaptic cleft by the enzymes monoamine oxidase (MAO) or catechol-O-methyltransferase (COMT). MAO oxidizes norepinephrine, and COMT metabolizes deaminated norepinephrine by O-methylation. MAO and COMT are found in adrenal chromaffin cells, while sympathetic nerves have only MAO. MAO has two isoforms—MAO-A and MAO-B. MAO-A is mainly found in noradrenergic neurons, while MAO-B is found in serotonergic cells. COMT is found in all organs, and degradation of NE to vanillylmandelic acid (VMA), is completed in the liver [2, 7, 16, 29].
Medications Acting on the Norepinephrine System
Adrenergic drugs can bind to one or more of the receptors to produce physiological actions. Based on the specific receptor affinity, they are classified into selective or direct-acting drugs and nonselective or indirectly acting drugs [21]. Medications acting on the NE system are shown below in Table 9.2 [30]. Direct-acting drugs are bronchodilators, and vasopressors, while indirect-acting drugs are amphetamines, desipramine, atomoxetine, and phenelzine [31, 32].
Selective Drugs
Alpha-1 Receptor Drugs
Phenylephrine, an alpha-1 receptor agonist, is commonly used as a decongestant and vasopressor in cases of hypotension due to septic shock. Another drug, oxymetazoline, is used as a decongestant and to treat rosacea. Common side effects include reflex bradycardia [33, 34].
Alpha-1 receptor antagonists like prazosin and terazosin are used in the treatment of hypertension and benign hypertrophy of the prostate because of their vasodilatory effect [30].
Alpha-2 Receptor Drugs
Clonidine, an alpha-2 receptor agonist, is used to treat hypertension and attention deficit hyperactivity disorder (ADHD). Nonapproved indications include post-traumatic stress disorder (PTSD), sleep disorders, anxiety, hot flashes associated with menopause, restless leg syndrome, and other illnesses. Methyldopa is a centrally acting sympatholytic and is used in the treatment of hypertension and gestational hypertension. It decreases the adrenergic outflow by alpha-2 agonistic action from CNS, thus reducing the total peripheral resistance and systemic blood pressure. As alpha-2 agonistic activity does not affect the cardiac output or renal blood flow, it is preferred in hypertensive patients with renal insufficiency. Dexmedetomidine is used in the intensive care unit for sedation, which does not induce respiratory depression. Common adverse effects of alpha-2 agonists include dry mouth, sedation, hypotension, respiratory depression, and somnolence [35, 36].
Alpha-2 receptor antagonists like mirtazapine are used as an antidepressant. It is a noradrenergic and specific serotonergic antidepressant, which acts by antagonizing both alpha-2 autoreceptors and heteroreceptors and also blocks the 5-HT2 and 5-HT3 receptors [30].
Beta-1 Receptor Drugs
Beta-1 receptors are commonly found in cardiac myocytes, cardiac nodal tissues, cardiac conduction pathways, and kidneys. Dobutamine is a beta-1 receptor agonist that is indicated in the treatment of cardiogenic shock and heart failure. Common side effects include hypertension, tachycardia, palpitations, anxiety, and tachyarrythmias [37, 38].
Beta-1 blockers: Cardio-selective blockers include atenolol, betaxolol, bisoprolol, esmolol, metoprolol, and nebivolol. They are clinically used for hypertension, heart failure, chronic stable angina, post-myocardial infarction, and decreased left ventricular function after myocardial infarction. They are also used in the treatment of arrhythmias, glaucoma, migraine prophylaxis, anxiety, and essential tremor [21, 39]. Common adverse effects include hypotension, bradycardia, heart failure, decreased exercise capacity, and atrioventricular nodal block. Noncardiac side effects include headache, fatigue, dizziness, nausea, vomiting, abdominal discomfort, dry mouth and eyes, sexual dysfunction, confusion, and memory loss.
Nonselective beta-1 blockers include propranolol, timolol, sotalol, and nadolol. Beta-1 blockade decreases heart rate, myocardial contractility by slowing AV conduction and suppresses automaticity. Beta-2 blockade reduces peripheral vascular resistance and can cause bronchospasm and hypoglycemia. Propranolol is used in hypertension, myocardial infarction, angina, idiopathic hypertrophic subaortic sclerosis, migraine, and vascular headaches. It also used for anxiety and in improving tremors by blocking the peripheral beta-2 adrenergic receptors. Common side effects include hypotension, bradycardia, dizziness, depression, memory loss, impotence, and rebound hypertension with sudden withdrawal. It is contraindicated in asthma, bradycardia, and heart failure [21, 40].
Beta-2 Receptor Drugs
Beta 2 receptors are commonly found in airway smooth muscles and cardiac muscles, uterine muscles, alveolar type II cells, mast cells, mucous glands, epithelial cells, vascular endothelium, eosinophils, lymphocytes, and skeletal muscles [41]. Short-acting Beta-2 agonists (SABAs) are albuterol, levalbuterol, metaproterenol, and terbutaline. Beta-2 agonists that act as bronchodilators are indicated for the treatment of obstructive lung diseases, such as COPD, asthma, or emphysema [41, 42].
Other uses of SABAs like albuterol are in the treatment of hyperkalemia, and terbutaline is used to postpone preterm labor and in the management of vascular extravasation. Long-acting B2 agonists (LABAs) are used in the maintenance treatment of patients with COPD, chronic bronchitis, and emphysema. Common LABAs include salmeterol, formoterol, and arformoterol. Olodaterol is an ultralong-acting beta-2 agonist used in the management of COPD and asthma [43, 44]. Adverse effects include anxiety, tachycardia, tremors, palpitations, nausea, vomiting, constipation, dizziness, and fatigue [37].
Beta-3 Receptor Drugs
Beta-3 receptors are located in the gallbladder, urinary bladder, and brown adipose tissue. They enhance lipolysis in adipose tissue and promote thermogenesis in skeletal muscle. They cause relaxation of the urinary bladder, thus preventing urination. Mirabegron is used in the treatment of overactive bladder (urinary incontinence, urinary frequency) [21].
Indirect Acting Agonists
Atomoxetine is a selective noradrenaline reuptake inhibitor, which is used as a first-line alternative when unresponsive to stimulants or in tic disorders that are aggravated by stimulants. As atomoxetine does not affect the dopamine level associated with motor activity, hence they do not worsen Tourette’s or tics. As it has a sedative effect, the dose is split for the evening to improve sleep symptoms, and it has a 24-h duration of action that can take up to 4–6 weeks to see the clinical benefit [45, 46].
Nonselective Drugs
Norepinephrine is used for the treatment of shock and hypotension. Epinephrine (adrenaline) is used in the treatment of cardiac arrest, anaphylaxis, and croup, while dopamine is used in the treatment of hypotension, bradycardia, and cardiac arrest. Isoprenaline is used in treating bradycardia and heart block [40].
Carvedilol and labetalol are nonselective and have both beta-receptor and alpha-blocking activity. Beta-1 blockade decreases heart rate, myocardial contractility by slowing AV conduction and suppresses automaticity. Beta-2 blockade reduces peripheral vascular resistance and can cause bronchospasm and hypoglycemia. Alpha-1 receptor blockade causes arterial smooth muscle relaxation and vasodilatation. Carvedilol is used in hypertension and heart failure and reduces cardiovascular mortality after myocardial infarction. It is also used in the treatment of migraine and vascular headaches [21].
Other Clinical Aspects
LC neuronal loss is the most commonly seen early feature in many neurodegenerative diseases like Alzheimer’s disease, Parkinson’s disease, and progressive supranuclear palsy.
Alzheimer’s Disease
Alzheimer’s disease is the most common neurodegenerative dementia that is characterized by progressive loss of memory and cognition. Although the disease pathology is considered multifactorial, the main factor might be the interaction between noradrenaline and acetylcholine. Proposed mechanisms of disease development and progression are tau protein hyperphosphorylation assembles into neurofibrillary tangles, which accumulate in LC, causing neurodegeneration and cell death. The rostral cortically projecting neurons are widely affected, leading to impaired cognition and dementia. Alzheimer’s disease is seen as a cholinergic dysfunction due to nucleus basalis of Meynert (nbM) degeneration. However, there is a significant loss of LC neurons and is directly correlated with the severity of the disease. Other factors include decreased NE levels that affect the anti-inflammatory and neuroprotective effects within the CNS. Impaired transmission of NE within the hippocampus leads to hyperphosphorylated tau protein and noradrenergic axonal degeneration [47, 48].
In Alzheimer’s disease pathogenesis, noradrenaline plays a crucial role as a neuroinflammatory moderator through microglial activation. Furthermore, it negatively affects the transcription of the inflammatory gene in microglia and astrocytes, which express adrenergic receptors [49].
Patients with Down’s syndrome and Alzheimer’s disease and patients with Down’s syndrome who later on develop Alzheimer’s disease have decreased levels of noradrenaline metabolites, which correlate with behavioral and psychological signs and symptoms of dementia [50, 51].
Parkinson’s Disease
Parkinson’s disease is the second most common neurodegenerative disease and a movement disorder that is characterized by bradykinesia, rigidity, tremor, postural instability, and cognitive decline in late stages. Loss of the dopaminergic nigrostriatal neurons is the main neuropathological abnormality that underlies the motor symptoms of the disease. In addition to the dopaminergic system, the noradrenergic system is also involved in the impairment of cognitive and emotions seen in Parkinson’s disease. The pathogenesis involves noradrenergic neuronal degeneration in LC and alpha-2 autoreceptor losing its protective effects on both noradrenergic and dopaminergic systems. Within LC, alpha-synuclein accumulates, leading to low NE levels and nigrostriatal pathway degeneration due to loss of NE neuroprotective effects [47].
Lewy body pathology is the unique feature of Parkinson’s disease, which occurs commonly in substantia nigra pars compacta and LC, that is associated with cell loss. Furthermore, Lewy body pathology in substantia nigra is preceded by the pathology in LC, indicating the dysfunction of noradrenaline even before affecting the dopaminergic systems in Parkinson’s disease [48, 52, 53].
Attention Deficit Hyperactivity Disorder (ADHD)
ADHD is a neurobehavioral disorder characterized by inattention, hyperactivity, and impulsivity, affecting 8% to 12% of children worldwide [54, 55]. The proposed mechanism involved in ADHD is an imbalance in dopaminergic and noradrenergic systems metabolism and neurotransmission in the prefrontal cortex and other subcortical regions. The deficiency in phosphoinositide 3-kinase (PI3K-gamma) enzyme causes an imbalance in norepinephrine/dopamine ratio and synaptic plasticity dysregulation. Impaired NET function and prefrontal dysfunctional noradrenergic transmission is involved in inattention and hyperactivity [56].
For parents of preschool children, a parent training program (PTP) is recommended. For moderately severe symptoms of ADHD in school-age children, consider PTP for parents and cognitive behavioral therapy (CBT) or social skills training, while CBT should be considered for adolescents [46]. Stimulants are the first-line agents in their treatment because of their rapid onset of action, safety, and efficacy in both children and adolescents [56]. Methylphenidate is the first-line therapy with a response rate of 60–80%. Atomoxetine is used if the child is intolerant to methylphenidate or has tics and anxiety.
Schizophrenia
Schizophrenia is a chronic mental illness characterized by both positive and negative symptoms, which include inattention, delusions, hallucinations, blunted affect, and disorganized thoughts. Alterations in the signaling of the dopaminergic and glutaminergic system appear to be the main cause, and there is evidence of noradrenergic system involvement as increased NE levels in CSF and blood in patients with paranoia and elevated NE markers in the post-mortem of schizophrenic patients [47].
Noradrenergic drugs can either treat or worsen the symptoms of schizophrenia. Blockage of alpha-1 adrenoceptors decreases the positive symptoms, while alpha-2 adrenoceptors blockage has been found to decrease the negative and cognitive symptoms [57]. Clonidine, methyldopa, and propranolol reduce the positive symptoms in schizophrenia by reducing the NE transmission in the brain. Desipramine and methylphenidate are found to worsen positive symptoms by increasing NE levels [47].
Depression
Depression is a common disorder that affects thoughts, mood, cognition, and behavior. The most common clinical symptoms include depleted energy, sadness, guilt, hopelessness, impaired concentration, and suicidal ideality [58, 59]. NE levels are elevated among other biomolecules in the periphery. Tyrosine hydroxylase expression was found to be increased in the LC, and levels of urinary NE were also increased [60].
References
Schwarz LA, Luo L. Organization of the locus coeruleus-norepinephrine system. Curr Biol. 2015;25(21):R1051–6. https://doi.org/10.1016/j.cub.2015.09.039. PMID: 26528750.
Drouin C. Reference module in neuroscience and biobehavioral psychology || Norepinephrine; 2017. https://doi.org/10.1016/B978-0-12-809324-5.03130-8.
Von Euler US. Sympathin in adrenergic nerve fibres. J Physiol. 1946;105:26.
Dahlstrom A, Fuxe K. Evidence for the existence of monoamine-containing neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of brain stem neurons. Acta Physiol Scand Suppl. 1964;232:231–55.
Dubocovich ML, Langer SZ. Negative feedback regulation of noradrenaline release by nerve stimulation in the perfused cat’s spleen: differences in potency of phenoxybenzamine in blocking the pre- and post-synaptic adrenergic receptors. J Physiol. 1974;237:505–19. https://doi.org/10.1113/jphysiol.1974.sp010495.
Svensson TH, Bunney BS, Aghajanian GK. Inhibition of both noradrenergic and serotonergic neurons in brain by the alpha-adrenergic agonist clonidine. Brain Res. 1975;92(2):291–306. https://doi.org/10.1016/0006-8993(75)90276-0.
Montoya A, Bruins R, Katzman MA, Blier P. The noradrenergic paradox: implications in the management of depression and anxiety. Neuropsychiatr Dis Treat. 2016;12:541–57. https://doi.org/10.2147/NDT.S91311. PMID: 27042068; PMCID: PMC4780187.
Rho HJ, Kim JH, Lee SH. Function of selective neuromodulatory projections in the mammalian cerebral cortex: comparison between cholinergic and noradrenergic systems. Front Neural Circuits. 2018;12:47. https://doi.org/10.3389/fncir.2018.00047. PMID: 29988373; PMCID: PMC6023998.
Weinshenker D, Schroeder JP. There and back again: a tale of norepinephrine and drug addiction. Neuropsychopharmacology. 2007;32(7):1433–51. https://doi.org/10.1038/sj.npp.1301263. Epub 2006 Dec 13. PMID: 17164822.
Aston-Jones G, Waterhouse B. Locus coeruleus: from global projection system to adaptive regulation of behavior. Brain Res. 2016;1645:75–8.
Pickel VM, Segal M, Bloom FE. A radioautographic study of the efferent pathways of the nucleus locus coeruleus. J Comp Neurol. 1974;155:15–42.
Szabadi E. Functional neuroanatomy of the central noradrenergic system. J Psychopharm. 2013;27:659–93.
Swanson LW. The locus coeruleus: a cytoarchitectonic, Golgi and immunohistochemical study in the albino rat. Brain Res. 1976;110:39–56.
Holets VR, Hokfelt T, Rokaeus A, Terenius L, Goldstein M. Locus coeruleus neurons in the rat containing neuropeptide Y, tyrosine hydroxylase or galanin and their efferent projections to the spinal cord, cerebral cortex and hypothalamus. Neuroscience. 1988;24:893–906.
Lang R, Gundlach AL, Holmes FE, Hobson SA, Wynick D, Hokfelt T, Kofler B. Physiology, signaling, and pharmacology of galanin peptides and receptors: three decades of emerging diversity. Pharmacol Rev. 2015;67:118–75.
Fung MM, Viveros OH, O’Connor DT. Diseases of the adrenal medulla. Acta Physiol. 2008;192(2):325–35. https://doi.org/10.1111/j.1748-1716.2007.01809.x. Epub 2007 Nov 16. PMID: 18021328; PMCID: PMC2576282.
Rogawski MA, Aghajanian GK. Activation of lateral geniculate neurons by locus coeruleus or dorsal noradrenergic bundle stimulation: selective blockade by the alpha 1-adrenoceptor antagonist prazosin. Brain Res. 1982;250(1):31–9.
Gannon M, Che P, Chen Y, Jiao K, Roberson ED, Wang Q. Noradrenergic dysfunction in Alzheimer’s disease. Front Neurosci. 2015;9:220. https://doi.org/10.3389/fnins.2015.00220. PMID: 26136654; PMCID: PMC4469831.
O’Donnell J, Zeppenfeld D, McConnell E, Pena S, Nedergaard M. Norepinephrine: a neuromodulator that boosts the function of multiple cell types to optimize CNS performance. Neurochem Res. 2012;37(11):2496–512. https://doi.org/10.1007/s11064-012-0818-x.
Terbeck S, Savulescu J, Chesterman LP, Cowen PJ. Noradrenaline effects on social behaviour, intergroup relations, and moral decisions. Neurosci Biobehav Rev. 2016;66:54–60. https://doi.org/10.1016/j.neubiorev.2016.03.031. Epub 2016 Apr 25. PMID: 27126289; PMCID: PMC4899514.
Farzam K, Kidron A, Lakhkar AD. Adrenergic drugs. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2021. [Updated 2021 Jul 18].
Strosberg AD. Structure, function, and regulation of adrenergic receptors. Protein Sci. 1993;2(8):1198–209. https://doi.org/10.1002/pro.5560020802. PMID: 8401205; PMCID: PMC2142449.
Aoki C, Go CG, Venkatesan C, Kurose H. Perikaryal and synaptic localization of alpha 2A-adrenergic receptor-like immunoreactivity. Brain Res. 1994;650(2):181–204.
Avery RA, Franowicz JS, Studholme C, van Dyck CH, Arnsten AF. The alpha-2A-adrenoceptor agonist, guanfacine, increases regional cerebral blood flow in dorsolateral prefrontal cortex of monkeys performing a spatial working memory task. Neuropsychopharmacology. 2000;23(3):240–9.
Birnbaum SG, Yuan PX, Wang M, et al. Protein kinase C overactivity impairs prefrontal cortical regulation of working memory. Science. 2004;306(5697):882–4.
Borges R, Gandía L, Carbone E. Old and emerging concepts on adrenal chromaffin cell stimulus-secretion coupling. Pflugers Arch. 2018;470(1):1–6. https://doi.org/10.1007/s00424-017-2082-z. Epub 2017 Nov 6.
Mandela P, Ordway GA. The norepinephrine transporter and its regulation. J Neurochem. 2006;97(2):310–33. https://doi.org/10.1111/j.1471-4159.2006.03717.x.
Hussain LS, Reddy V, Maani CV. Physiology, noradrenergic synapse. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2021. [Updated 2021 May 9]. https://www.ncbi.nlm.nih.gov/books/NBK540977/.
Dorszewska J, Prendecki M, Oczkowska A, Rozycka A, Lianeri M, Kozubski W. Polymorphism of the COMT, MAO, DAT, NET and 5-HTT genes, and biogenic amines in Parkinson’s disease. Curr Genomics. 2013;14(8):518–33. https://doi.org/10.2174/1389202914666131210210241. PMID: 24532984; PMCID: PMC3924247.
Bylund DB. Encyclopedia of the neurological sciences || Norepinephrine; 2014. p. 614–6.
Ciccarone D. Stimulant abuse: pharmacology, cocaine, methamphetamine, treatment, attempts at pharmacotherapy. Prim Care. 2011;38(1):41–58.
Giovannitti JA, Thoms SM, Crawford JJ. Alpha-2 adrenergic receptor agonists: a review of current clinical applications. Anesth Prog. 2015;62(1):31–9.
Atkinson HC, Potts AL, Anderson BJ. Potential cardiovascular adverse events when phenylephrine is combined with paracetamol: simulation and narrative review. Eur J Clin Pharmacol. 2015;71(8):931–8.
Hatton RC, Winterstein AG, McKelvey RP, Shuster J, Hendeles L. Efficacy and safety of oral phenylephrine: systematic review and meta-analysis. Ann Pharmacother. 2007;41(3):381–90.
Ming X, Mulvey M, Mohanty S, Patel V. Safety and efficacy of clonidine and clonidine extended-release in the treatment of children and adolescents with attention deficit and hyperactivity disorders. Adolesc Health Med Ther. 2011;2:105–12.
Yasaei R, Saadabadi A. StatPearls. Treasure Island, FL: StatPearls Publishing; 2021. Clonidine. [PubMed: 29083638].
Almadhoun K, Sharma S. StatPearls. Treasure Island, FL: StatPearls Publishing; 2021. Bronchodilators. [PubMed: 30085570].
Dubin A, Lattanzio B, Gatti L. The spectrum of cardiovascular effects of dobutamine—from healthy subjects to septic shock patients. Rev Bras Ter Intensiva. 2017;29(4):490–8. [PMC free article: PMC5764562] [PubMed: 29340539].
Ladage D, Schwinger RHG, Brixius K. Cardio‐selective beta‐blocker: pharmacological evidence and their influence on exercise capacity. Cardiovasc Ther. 2013;31(2):76–83.
Smith MD, Maani CV. StatPearls. Treasure Island, FL: StatPearls Publishing; 2020. Norepinephrine. [PubMed: 30725944].
Abosamak NER, Shahin MH. Beta 2 receptor agonists/antagonists. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2022. [Updated 2022 May 8]. https://www.ncbi.nlm.nih.gov/books/NBK559069/.
Barisione G, Baroffio M, Crimi E, Brusasco V. Beta-adrenergic agonists. Pharmaceuticals (Basel). 2010;3(4):1016–44. [PMC free article: PMC4034018] [PubMed: 27713285].
Allon M, Dunlay R, Copkney C. Nebulized albuterol for acute hyperkalemia in patients on hemodialysis. Ann Intern Med. 1989;110(6):426–9. [PubMed: 2919849].
Motazedian S, Ghaffarpasand F, Mojtahedi K, Asadi N. Terbutaline versus salbutamol for suppression of pretermlabor: a randomized clinical trial. Ann Saudi Med. 2010;30(5):370–5. [PMC free article: PMC2941249] [PubMed: 20697169].
NCCMH NCCfMH. Attention deficit hyperactivity disorder: diagnosis and management of ADHD in children, young people and adults (CG 72). London: National Institute for Health and Clinical Excellence; 2008.
Ougrin D, Chatterton S, Banarsee R. Attention deficit hyperactivity disorder (ADHD): review for primary care clinicians. London J Prim Care (Abingdon). 2010;3(1):45–51. https://doi.org/10.1080/17571472.2010.11493296.
Borodovitsyna O, Flamini M, Chandler D. Noradrenergic modulation of cognition in health and disease. Neural Plast. 2017;2017:6031478. https://doi.org/10.1155/2017/6031478.
Holland N, Robbins TW, Rowe JB. The role of noradrenaline in cognition and cognitive disorders. Brain. 2021;144(8):2243–56. https://doi.org/10.1093/brain/awab111.
Feinstein DL, Heneka MT, Gavrilyuk V, Dello RC, Weinberg G, Galea E. Noradrenergic regulation of inflammatory gene expression in brain. Neurochem Int. 2002;41(5):357–65.
Dekker AD, Coppus AM, Vermeiren Y, et al. Serum MHPG strongly predicts conversion to Alzheimer’s disease in behaviorally characterized subjects with Down syndrome. J Alzheimers Dis. 2015a;43(3):871–91.
Dekker AD, Strydom A, Coppus AM, et al. Behavioural and psychological symptoms of dementia in Down syndrome: early indicators of clinical Alzheimer’s disease? Cortex. 2015b;73:36–61.
Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord. 2015;30(12):1591–601.
Zarow C, Lyness SA, Mortimer JA, Chui HC. Neuronal loss is greater in the locus coeruleus than nucleus basalis and substantia nigra in Alzheimer and Parkinson diseases. Arch Neurol. 2003;60(3):337–41.
Barkley RA, Fischer M, Smallish L, Fletcher K. The persistence of attention-deficit/hyperactivity disorder into young adulthood as a function of reporting source and definition of disorder. J Abnorm Psychol. 2002;111(2):279–89. https://doi.org/10.1037/0021-843X.111.2.279.
Fayyad J, De Graaf R, Kessler R, Alonso J, Angermeyer M, Demyttenaere K, et al. Cross-national prevalence and correlates of adult attention-deficit hyperactivity disorder. Br J Psychiatry. 2007;190(5):402–9. https://doi.org/10.1192/bjp.bp.106.034389.
Mannem N, Mehta T, Nagarajan E, et al. ADHD and its therapeutics. Curr Dev Disord Rep. 2021;8:175–83. https://doi.org/10.1007/s40474-020-00215-9.
Svensson TH. Alpha-adrenoceptor modulation hypothesis of antipsychotic atypicality. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(7):1145–58.
Ahmadimanesh M, Balarastaghi S, Rashedinia M, et al. A systematic review on the genotoxic effect of serotonin and norepinephrine reuptake inhibitors (SNRIs) antidepressants. Psychopharmacology. 2020;237:1909–15. https://doi.org/10.1007/s00213-020-05550-8.
Zhong Z, Wang L, Wen X, Liu Y, Fan Y, Liu Z. A meta-analysis of effects of selective serotonin reuptake inhibitors on blood pressure in depression treatment: outcomes from placebo and serotonin and noradrenaline reuptake inhibitor controlled trials. Neuropsychiatr Dis Treat. 2017;13:2781–96.
Peacock BN, Scheiderer DJ, Kellermann GH. Biomolecular aspects of depression: a retrospective analysis. Compr Psychiatry. 2017;73:168–80. https://doi.org/10.1016/j.comppsych.2016.11.002. Epub 2016 Nov 10.
Acknowledgments
All the pictures in this chapter were prepared using BioRender premium software. The tables were redrawn using information from the reference article mentioned alongside them.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Murala, S., Bollu, P.C. (2022). Norepinephrine. In: Bollu, P.C. (eds) Neurochemistry in Clinical Practice. Springer, Cham. https://doi.org/10.1007/978-3-031-07897-2_9
Download citation
DOI: https://doi.org/10.1007/978-3-031-07897-2_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-07896-5
Online ISBN: 978-3-031-07897-2
eBook Packages: MedicineMedicine (R0)