Abstract
Liquid biopsy has emerged as one of the main pillars for personalized oncology. The term englobes body-fluid samples which contain tumor-derived material such as circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and circulating extracellular vesicles (cEVs). Potential clinical application of liquid biopsy analyses includes cancer screening, detection of minimal residual disease and recurrence, therapy selection, and evaluation of acquired resistance. Despite the great developments of technology focused on circulating biomarkers characterization only cfDNA testing is nowadays implemented for the therapy selection in some advanced tumors. This can be partially explained by the fact that there is still a lack of global standardization of procedures both in the pre-analytical and analytical steps. In the present chapter, we summarize the different strategies for addressing the study of liquid biopsy taking into account their pros and cons to be applied in a clinical context and we also discuss the main technical and clinical challenges in the field of circulating biomarkers and personalized oncology.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Circulating tumor biomarkers
- Liquid biopsy
- Personalized oncology
- Circulating tumor cells
- Extracellular vesicles
1 Introduction: Clinical Relevance of Liquid Biopsy for Personalized Oncology
Advances in molecular biology have clearly changed the way to manage cancer, allowing us to coin the term “personalized oncology.” This term refers to the individualized diagnosis, treatment and disease monitoring based on the specific molecular characteristics of each tumor [1]. In this context, the analysis of tissue remains the gold standard for making cancer diagnosis and characterization, but this procedure has limitations such as access difficulty, mainly to biopsy metastasis, the lack of representativity of the tumor heterogeneity and the possibility of follow-up the clonal evolution under the therapy pressure [2]. Therefore, the analysis of circulating biomarkers has emerged as a key tool reaching more personalized management of cancer patients [3].
The term “liquid biopsy” was first used by Pantel and Panabiéres in 2010 to refer to the analysis of circulating tumor cells (CTCs) in blood from cancer patients [4]. Currently, the concept is generally employed to talk about the sampling and analysis of tumor-derived material present in different body-fluids, mainly blood, but also other body fluids such as saliva, urine, cerebrospinal fluid, ascites, or pleural effusions [3]. Circulating biomarkers present in fluid biopsies comprise CTCs, circulating tumor DNA (ctDNA), circulating cell-free RNA (cfRNA), circulating extracellular vesicles (cEVs), and other circulating elements such as immune cells or tumor-educated platelets, among others. These circulating biomarkers have shown great potential for cancer screening, molecular diagnosis, predicting the patients’ prognosis, assessing minimal residual disease after surgery, the therapy selection/monitoring and for characterizing the mechanisms of resistance in different tumor types [3].
In the clinical setting, ctDNA has been recently implemented to analyze driver mutations that condition the response to targeted therapies and some molecular tests have been approved as a companion diagnostic in the context of advanced breast, non-small-cell lung (NSCLC), prostate and ovarian tumors. NSCLC was the first tumor in which the analysis of ctDNA for tumor phenotyping was included in the guidelines. Thus, genomic alterations in EGFR, ALK, ROS1, BRAF, MET, and RET must be analyzed in tissue or ctDNA to determine the appropriate treatment [5] (National Comprehensive Cancer Network (NCCN). Non-Small Cell Lung Cancer. Version 4.2020. NCCN Clinical Practice Guidelines in Oncology. Accessed January 6, 2020. nccn.org/professionals/physician_gls/pdf/nscl.pdf). In advanced breast cancer, NCCN guidelines recommend the analysis of PIK3CA status using tissue samples or ctDNA to guide the administration of Alpelisib [6] (National Comprehensive Cancer Network (NCCN). Invasive Breast Cancer. Version 4.2020. NCCN Clinical Practice Guidelines in Oncology. Accessed January 6, 2020. nccn.org/professionals/physician_gls/pdf/breast.pdf). Also, in advanced colorectal cancer, several studies have highlighted the feasibility of interrogating RAS and BRAF status to guide anti-EGFR therapy [7, 8]. The value of ctDNA analyses for the response assessment has been reported in many tumors such as melanomas or breast cancer, and for different targeted and non-targeted therapies [9,10,11,12]. Plasma ctDNA has been also explored as a prognostic biomarker to stratify the risk of recurrence in localized tumors after curative surgery, indicating those patients with a need of more intensive adjuvant therapy [13, 14]. In the same line, cfDNA studies have shown value as diagnostic tools. For this purpose, the identification of methylated patterns has been successfully applied to detect the presence of different tumor types [15]. Actually, the detection of methylation in the promoter region of the SEPT9 gene in plasma cfDNA (Epi proColon test) represents the first blood-based test approved by the FDA for the screening of CRC [16, 17].
CTC research is considered the start-point of the liquid biopsy field. Early in the formation and growth of a primary tumor, cells are released into the bloodstream. Several groups are studying the clinical benefit of CTC monitoring [18]. CTCs have been validated as a prognostic marker in metastatic breast cancer and other solid tumors such as prostate, colorectal, and lung cancer, showing even more accuracy than conventional imaging methods for response evaluation [19]. However, there are still technical challenges to using CTC monitoring to detect minimal residual disease in patients at early stages. On the other hand, the molecular characterization of CTCs is of great interest to guide the selection of targeted therapies since it allows clinicians to have a dynamic view of different molecular targets such as ERBB2, EGFR, AR or PD-L1, among others [19, 20].
On the other hand, the field of cEVs and miRNAs is continuously increasing due to their relevant function during the process of carcinogenesis and tumor spread. They can be detected in different body fluids and have shown great potential as cancer biomarkers for diagnostic and prognostic purposes. In particular, EVs contain both proteins and nucleic acids that can serve to increase tumor detection sensitivity [21, 22]. However, one of the main limitations for cEVs based approaches is the absence of tumor-specific markers to identify the tumor-derived EVs. Only hot shot protein 60 (HSP60) and Glypican-1 (GPC1) have been identified as potential identifiers for detecting EVs from colorectal, pancreatic, and breast cancer detection [23]. Thus, studies based on cEVs and in fluid samples are still in an infancy stage and further validation in clinical studies is required to clarify their impact on precision oncology.
Finally, other blood elements such as tumor-educated platelets (TEPs) or the different circulating immune cells have been evaluated as liquid biopsy biomarkers for prediction or monitoring of therapy responses, but their application is still far from the clinical routine [24, 25].
In the present chapter, we summarize the analytical strategies developed to interrogate the presence of CTCs, ctDNA, and cEVs as the main type of circulating markers with clinical interest for personalized oncology.
2 Strategies for CTCs Isolation and Characterization
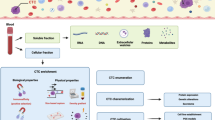
Circulating tumor cells are present in the bloodstream at a low proportion, about 1 CTC per 106–107 leukocytes [26] and with a very short half-life (1–2.4 h) [27, 28]. Due to the low concentration in blood, CTCs identification and characterization require methods with high analytical sensitivity and specificity [28], being their isolation technically challenging. In the last years, a high number of promising CTC-detection technologies have been developed, focused on the differential features between CTCs and the surrounding normal blood cells, including physical properties (size, density, electric charges, deformability) and biological properties (cell surface protein expression, viability) [29]. Here, we divide the different methods to enrich CTCs into two main principles: antigen-dependent methods and antigen-independent methods (Fig. 21.1).
2.1 CTCs Isolation Strategies
2.1.1 Antigen-Dependent
Antigen-dependent isolation approaches are the most common methods employed and they are based on the presence of specific surface markers by CTCs (called positive enrichment) or by blood cells (negative enrichment).
Positive enrichment, the most employed strategy is usually carried out using antibodies that recognize epithelial cell adhesion molecule (EpCAM) [29] conjugated with magnetic nanoparticles. Among the current EpCAM-based technologies, CellSearch® system (Menarini, Silicon Biosystem, Bologna, Italy) [30] has become the “gold standard” for the CTC-detection methods. CellSearch® system employs anti-EpCAM-coated ferrofluid nanoparticles for the selection of EpCAM positive cells. Next, an immunostaining step discriminates CTCs from leukocytes based on the positive expression of cytokeratins and the absence of CD45 staining together with morphologic criteria. Although a high number of alternatives that employ magnetic nanoparticles conjugated with anti-EpCAM antibodies are also available [31], until now CellSearch® system is the unique method approved by the Food and Drug Administration (FDA) for clinical use in metastatic breast, prostate and colorectal cancer [32,33,34].
Recently, new positive enrichment methods are being developed, in which the specific surface markers are immobilized on the surface of microfluidic chips [31] to increase the contact between the cells and, therefore, to enhance capture efficiency. However, the isolation in all these approaches is based on the EpCAM expression, therefore they are not able to detect CTCs that show no EpCAM expression, for example, CTCs of non-epithelial tumors such as sarcomas or CTC that have undergone epithelial-to-mesenchymal (EMT) transition [35, 36].
Negative enrichment methods employ magnetic nanoparticles conjugated with antibodies against the common leukocyte antigen CD45 [37] or other antigens expressed in blood cells and represent a good alternative to avoid the limitations of the EpCAM-dependent isolation. They allow isolating CTCs independently of any CTC surface marker expression however due to the low proportion of CTCs and the recent observation that CTCs travel into the bloodstream coated with blood cells [38], the resulted recovery rate is often relatively low [31].
2.1.2 Antigen-Independent
Antigen-independent methods are based on physical properties of CTCs such as density, electric charges (DEP, dielectrophoresis), size, and deformability, among others. The principal advantage and difference with the antigen-dependent methods are that they do not require specific surface markers on CTCs, so they also allow the isolation of CTCs with a low epithelial phenotype. Density-based methods were the first techniques developed. These methods allow to processing of high volumes of blood (about 25 mL) with a quick processing time; however, they generally show a low efficiency and purity of the sample obtained [31]. The size-based methods are the most common. They are based on the fact that tumor cells are larger than blood cells [39, 40] and, therefore, they can be isolated using filter-based strategies (such as ISET assay (Rarecells Diagnostics, Paris, France) [41]), microfluidic chips (such as Parsortix system (Angle, UK) [42]) and methods based on centrifugal forces [43]. The different charges between blood cells and CTCs can also be employed in their isolation. DEP field forces are employed to move CTCs independently to other blood cells, being a highly specific method [44].
Antigen-independent methods are generally easy to implement, however they depend on the availability of advanced materials or assistive engineering technologies for better clinical application [20]. Interestingly, new methods combining antigen-based capture with the advantages of microfluidics methods, such as CTC-iChip are being developed for increasing the isolation efficacy [45]; however, nowadays a robust and standardized platform to capture CTCs for clinical application remains a challenge.
Finally, it is important to remark that small volumes processed with the methods here described may be a serious limitation for the detection of these rare events, especially in cancer patients without metastases, in which the number of CTCs is expected to be very low. To solve this problem, some “in vivo” approaches such as GILUPI Nanodetector® [46] or Diagnostic leukapheresis (DLA) can be employed [47].
2.1.3 Single CTCs Isolation
After enrichment, the CTC fraction usually still contains a substantial number of leukocytes [29]. This background of leukocytes is seen in all CTC enrichment platforms being the posterior molecular analyses of CTCs a challenge. Therefore, after the detection of CTCs, there are some platforms that allow the isolation of pure CTCs at a single level by the use of micromanipulation or via dielectron force manipulation (such as the DEPArray system (Menarini, Silicon Biosystem, Bologna, Italy), among other strategies [48].
2.2 CTCs Characterization
After enrichment, a variety of approaches can be employed to distinguish and characterize the CTCs. Analysis of CTCs at the proteome, genome, and transcriptome level provides valuable information about the molecular heterogeneity of these cells and more precise characterization of the disease [49]. Furthermore, CTCs can also be used for functional studies “in vitro” and “in vivo” models, allowing to study the biological process and characteristics of CTCs as well as test the response to different therapies.
2.2.1 Protein Expression
After enrichment, immunohistochemical or immunofluorescent (IHC or IF) assays can be used to distinguish CTCs from nonspecifically captured cells. The most commonly used antibodies are cytokeratins combined with markers such as CD45 that identify the background blood cells [50]. In addition, other surface proteins can be analyzed by IF that could be key candidates for targeted therapies. Thus, certain protein expression in CTCs has been studied, such as PD-L1 in lung cancer patients [51] and ER and HER2 in breast cancer, among others [52]. In addition, a microfluidic western blot technology for proteomic phenotyping of CTCs has also been developed, however, the number of proteins included is scarce [53].
2.2.2 Genomic Analyses
Genomic analyses at the DNA level allow for the detection of driver mutations in enriched CTCs samples [54]. Real-time polymerase chain reaction (RT-PCR), digital droplet PCR (ddPCR), and next-generation sequencing (NGS) are the most employed methods; however, results obtained present a low sensitivity because of “masking” the tumor profile by wild-type DNA from leukocytes [55].
More comprehensive analyses can be carried out using CTCs isolated at a single level followed by amplification of the whole genome, providing a valuable tool in order to know more about the heterogeneity of the tumor [55], as well as to predict the response of therapy. For example, the genomic profile of single CTCs can be employed to generate a copy number abnormalities (CNA)-based classification that can differentiate chemosensitive from chemorefractory patients in small cell lung cancer [56]. In contrast, technical limitations of CTCs isolation efficiency and the difficulties of performing whole-genome analyses on rare cells have limited the number of CTCs genomic profile studies [50] in comparison with cfDNA studies.
In another hand, gene-expression analyses in CTCs could be useful to know the nature and extent of tumor heterogeneity, linking phenotypic differences with genetic and epigenetic aberrations [57]. However, RNA is less stable and more difficult to preserve in comparison to DNA. Hence, RNA degradation constitutes a major challenge for CTCs analyses in multicenter clinical studies. Until now few single-CTCs transcriptome studies have been performed.
2.2.3 Functional Analyses
CTCs can also be characterized in functional studies. Some strategies for CTCs isolation offer the possibility to isolate viable CTCs and apply innovative culturing technologies to study fundamental characteristics of CTCs such as invasiveness, kinetic activity, and responses to different therapies [28]. In vitro models have been successfully reported, however, to obtain a cell culture high number of CTCs are required, and few patients have the CTCs number required. So far, it has been possible to obtain a short-term and long-term expansion of CTCs from breast, colorectal, lung, and prostate cancer, among others [58]. These cell lines can be used for drug screening, but the process of establishing these cell lines is not yet rapid enough to enable studies to inform treatment decisions for the donor patient [59].
In another hand, CTC-derived explant (CDX) models have emerged recently. For their generation, CTCs are enriched from the blood of patients and injected into immunocompromised mice to generate tumors and expand the initial material. Thus, CDXs constitute a valuable tool for clinical drug development [60]. These CDXs have been successfully generated in small cell lung [61], colorectal [62], breast [63], and prostate cancer [64], among others. Their main limitation is the time required to develop the CDXs models, usually several months.
3 Strategies for cfDNA/ctDNA Characterization
Although the mechanisms by which this tumor DNA reaches the circulation are not fully described, there are currently two accepted processes to explain its release. The passive mechanism implies that cells release DNA into the circulation as a consequence of cell death phenomena (necrosis or apoptosis). In this sense, the usual size of ctDNA is 167 bp, in line with the size of nucleosomal DNA that normally appears in apoptotic phenomena, but fragments that represent nucleosomal dimers or trimers can also appear. The second mechanism that allows the appearance of DNA in circulation is associated with an active release by tumor cells and may constitute a communication mechanism, although this process is not known in detail [65, 66]. Once in circulation, cfDNA is eliminated in the liver, kidney, and spleen, with an approximate half-life in the circulation of 16 min [66]. In cancer patients, ctDNA is found in a variable but normally very low (1–0.01%) percentage in relation to all cfDNA, which is usually less than 1 ng/μL. As already mentioned, this fraction varies depending on the stage, location, or degree of vascularization of the tumor, but also other physiological conditions such as tissue damage or marked exercise. Thus, tumors with multiple metastatic locations and highly vascularized will have higher levels of ctDNA [67].
3.1 cfDNA Isolation and Quantification
Before describing detection techniques, it is important to focus on cfDNA isolation methods. For plasma isolation, the most recommended protocol includes double centrifugation: the first centrifugation at 1200/1600×g for 10 min and then the second centrifugation at 5000/6000×g for another 10 min to ensure the elimination of any cellular debris. Once the plasma is isolated, it must be stored at −80 °C until its use and avoid several processes of freezing and thawing of the sample [51, 68]. For cfDNA isolation, we can use traditional extraction methods such as phenol-chloroform or alcoholic precipitation, which normally have very high yields. However, these approaches require more processing time than commercial extraction kits, which are mainly based on affinity columns, magnetic particle capture, capture by filtration, and methods based on the phenol-chloroform strategy. There are several studies that have compared the efficiency of different commercial isolation kits [68, 69]. The main differences observed between them are the recovery efficiency and the size of the isolated fragments. In some studies, the recovery results have been favorable to kits that use magnetic particles, such as MagNA Pure (Roche Diagnostics, Basel, Switzerland), compared to those that use affinity columns. One of the most widely used column-based isolation kits is the QIAamp DNA Blood Mini Kit (Qiagen, Hilden, Germany), which has shown cfDNA recoveries of 80–90%. Another column kit that has shown good results is the NucleoSpin Plasma XS (Marcherey Nagel, Düren, Germany), which is capable of recovering DNA fragments >50 bp in very small volumes [70].
Four main strategies are commonly used to characterize the concentration and size of isolated cfDNA: spectrometry, fluorometry, electrophoresis, and PCR-based techniques. The most specific and sensitive of the four options is the assessment of cfDNA quantity by PCR-based strategies to detect conserved sequences in the genome [71].
3.2 ctDNA Characterization
CfDNA analyses allow to identify mutations of interest (including resistance mutations) to guide the therapeutic decisions in several cancer types [72], the detection of cancer at early stages and the presence of minimal residual disease [59, 73, 74] as well as the assessment of the tumor mutational burden [75].
Thus, after isolation, ctDNA can be assessed to investigate for molecular alterations by two different approaches: single-gene analysis (PCR-based methods) or genome-wide analysis (through NGS strategies) (Table 21.1). During the last years, the development and improvement of these technologies has allowed the implementation of ctDNA analyses into the clinical routine. Thus, four tests have been approved by the FDA for clinical use. Two PCR-based assays, the therascreen® PIK3CA PCR Kit (Qiagen, Hilden, Germany) for breast cancer patients [76] and the Cobas® EGFR Mutation Test v2 (Roche Molecular Systems, Inc., Basel, Switzerland) for NSCLC patients [77] and two NGS-based assays, the FoundationOne Liquid CDx test (Foundation Medicine, Inc., MA, EEUU) for patients with solid malignant neoplasm [78] and the Guardant360 CDx (Guardant Health, Inc., CA, EEUU) for NSCLC patients [79]. All of the kits allow identifying patients who may benefit from treatments based on specific targeted therapies.
However, due to the low concentration of ctDNA in total cfDNA, ctDNA analyses involve a challenge for detecting genetic alterations (point mutations, CNAs or small indels) at the early stages of tumor development [80]. Epigenetic analyses on cfDNA have increased relevance to improving ctDNA detection in the early phases of the disease.
3.2.1 PCR-Based Techniques
PCR-based techniques were the first assays that allow to detect single or a low number of point mutations using highly sensitive and specific techniques with a rather fast and cost-effective rate. Real-time PCR (RT-PCR) was the first assay employed, reporting specific known mutations, but with a limited sensitivity (0.1–1%) [81]. In the last years, new technologies such as digital PCR (dPCR) methods, which include droplet digital PCR (ddPCR) and BEAMing (beads, emulsions, amplification and magnetics), showed high concordance with results obtained in tumor tissue [8, 82], and improved the sensitivity (0.01–0.1%) and specificity (100%).
Nevertheless, the main limitation of ctDNA analyses using PCR-based techniques is the requirement of previous information about the tumor type and the mutations characterizing this tumor. Therefore, PCR-based techniques are commonly employed to select targeted therapies, monitor the patients’ evolution or detect resistant mutations during the treatment.
3.2.2 NGS
The second approach is focused on a genome-wide analysis of CNAs or point mutations through next-generation sequencing (NGS) strategies. Based on the assay panel size, there are single-locus/multiplexed assays, targeted sequencing, and genome-wide sequencing [83]. Genome-wide characterization allows a more complete and patient-specific genotyping to assess tumor heterogeneity and to follow the clonal evolution across the treatment [83]. The principal limitations of NGS-based strategies are the high cfDNA input requirement and general present lower specificity (80–99%) [67, 81].
Among these approaches, ctDNA can be analyzed by specific panels covering a high number of targeted genes (by NGS panels) or analyzing the total genome by whole-genome sequencing (WGS) or whole-exome sequencing (WES). WES and WGS based methods allow the detection of all possible aberrations in DNA, although it has limited analytical sensitivity in cfDNA applications. This phenomenon could be due to the efficiency by which the genetic regions of interest can be captured/enriched from cfDNA and the higher error rate of sequencing reactions [84].
3.2.3 Epigenetic Alterations
In addition to genetic alterations, different types of epigenetic marks have been explored in cancer as specific to the malignant process. These marks have been mainly explored in tissue samples but their interest in cancer diagnosis and monitoring using liquid biopsy has increased exponentially during the last 5 years [85].
DNA methylation is the most studied epigenetic modification. This covalent modification consists of the incorporation of a methyl group to the 5′ carbon of cytosines in cytosine-phosphate-guanine (CpG) dinucleotides to generate 5-methylcytosine (5mC) [86]. The detection strategies of DNA methylation patterns can be divided into sodium bisulfite conversion dependent or independent [87]. The most used are the bisulfite conversion dependent and are based on the fact that after sodium bisulfite treatment, 5mC cannot be converted into uracils [88].
For interrogating DNA of both CTCs or cfDNA different techniques have been successfully applied such as methylation-specific PCR (MSP), methylation-sensitive high-resolution melting (MS-HRM), quantitative methylation-specific PCR (qMSP) and digital PCR (dPCR) such as methyl-BEAMing and droplet digital PCR (ddPCR) [89,90,91,92]. These PCR based approaches are directed to analyze a low number of CpG, while other strategies like methylation microarrays [93, 94] or genome-wide bisulfite-based approaches based on NGS provide a more comprehensive view of the methylome using both cfDNA from cancer patients or DNA isolated from CTCs, even at the single-cell level [95, 96].
4 Advances in Circulating Extracellular Vesicles Analyses
The extracellular environment contains a large number of mobile membrane-limited vesicles secreted from different cells called “extracellular vesicles” (EVs) [97,98,99]. Although current research focuses primarily on two major types of EVs (exosomes and microvesicles (MVs)), EVs also include other vesicular structures such as large apoptotic bodies (Abs) as well as retrovirus-like particles (RLPs), exosome-like vesicles and membrane particles [97, 99, 100].
EVs represent a tool for intercellular communication in the body [97, 101,102,103,104] being present in a variety of body fluids including blood, urine, saliva, cerebrospinal fluid, lymphatics, tears, saliva and nasal secretions, ascites, and semen [101, 105, 106], which make EVs an interesting cancer biomarker. They carry different types of cellular content such as lipids, proteins, metabolites, receptors, effector molecules, and nucleic acids like DNA and RNA (mRNA and microRNA) [107, 108]. This content can be translated to another cell [107,108,109,110] promoting different mechanisms including tumor progression by favoring angiogenesis and tumor cell migration in metastases [111,112,113]. Actually, EVs have shown to be valuable tools as biomarkers for longitudinal monitoring, defining tumor type, stage, progression, and treatment response [114, 115].
Exosomes are small EVs that generally possess a diameter of ~40–100 nm and a buoyant density of 1.13–1.19 g/mL [101, 116,117,118]. They are generated through a double invagination of the plasma membrane and the following formation of intracellular multivesicular bodies (MVBs) containing intraluminal vesicles (ILVs) [119]. These vesicular bodies are sorted by the endosomal network to their appropriate destinations, including lysosomal degradation, recycling, or exocytosis, releasing his ILVs content as exosomes [98, 106, 120, 121]. On the other hand, microvesicles arise through direct outward budding and fission of the plasma membrane, in a process called ectocytosis which produces microvesicles, microparticles, and large vesicles in the size range of ~50 nm to 1 μm in diameter [106, 113].
4.1 Isolation Methods
Currently, available purification methods are not capable of fully discriminating between exosomes and MVs [97, 122]. This lack of sufficient specificity and sensitivity makes more challenging their implementation in routine clinical practice [123, 124]. In fact, nowadays, there is no consensus on a “gold standard” method for EV isolation and purification [125]. Therefore, it is of utmost importance to improve and establish guidelines for EV isolation and analysis [125, 126], since depending on the method employed the amount, type and purity of the EVs recovered is different. Here we summarize some of the options for their isolation and characterization (Fig. 21.2).
4.1.1 Ultracentrifugation Techniques
4.1.1.1 Differential Ultracentrifugation
This is the most commonly employed EVs isolation method [127,128,129]. It is based on a succession of differential centrifugal forces to separate the particles: firstly, a low centrifugal force (300–400×g) to sediment a main portion of the cells, a 2000×g to remove cell debris, a 10,000×g to remove the aggregates with a buoyant density higher than the EVs and a final high force (100,000×g) that concentrate EVs in the resulting supernatant [126, 130]. However, this protocol is not unified and can vary depending on the volume and viscosity of the sample, which can affect the speed of centrifugation and the time needed for the obtention of the EVs [130, 131]. This strategy needs costly instrumentation, is time-consuming, requires a large amount of sample, and the recovery is normally low and contaminated with non-vesicular materials [132], which affect the purity of the samples in terms of the omics, RNA, and functional EVs analysis [133].
4.1.1.2 Density Gradient Centrifugation
This is a stricter strategy based on size and density [126] where the separation occurs in the presence of a preconstructed density gradient, typically made of sucrose or iodoxinol [134], resulting in differences in the osmotic pressure which can potentially affect the EVs [134, 135]. Density Gradient centrifugation is very effective in separating EVs from protein aggregates and non-membranous particles. Although it reports higher purity, it counts with limitations associated with ultracentrifugation [136] such as low recovery [137].
4.1.2 Size-Based Techniques
4.1.2.1 Ultrafiltration
This is the most commonly used size-based technique and consists of the separation of particles using semipermeable membranes with defined pore size or molecular weight cut [134]. While the larger particles are retained, the smaller ones passed through the filter into the filtrate [138]. Ultrafiltration is less time-consuming than ultracentrifugation and does not require special equipment [139]. However, the use of shear force may result in the deformation, clogging, or trapping in the unit or breaking up of large vesicles which may potentially skew the results of downstream analysis [139,140,141].
4.1.2.2 Size-Exclusion Chromatography (SEC)
This technique lies in sorting vesicles and other molecules based on their size by filtration through a gel. The gel is composed of spherical beads which contain pores of a specific size distribution through which small particles can penetrate. When the sample enters the gel, small molecules slow down the movement into the pores, causing them to elute later, while large molecules are excluded from entering the pores [142, 143]. Despite SEC methods enabling more accurate EVs purification [144] and preserving vesicle integrity and biological activity, they require run times of several hours, are not easily scalable, and cannot be used for high throughput applications [145].
4.1.2.3 Field-Flow Fractionation (FFF)
In this separation technique, a force field is applied perpendicular to a sample flow, to enable separation based on different sizes and molecular weights. When the perpendicular force field is applied, analytes in the sample are driven toward the boundary. Brownian motion creates a counteracting motion such that smaller particles tend to reach an equilibrium position further away from the boundary. This type of separation spans a broad size range and could be applied to a wide variety of eluents [136].
4.1.2.4 Hydrostatic Filtration Dialysis (HFD)
In HFD, based on the traditional dialysis separation method, the sample is forced throw a dialysis tube with a mesh of membrane with a molecular weight cutoff of 1000 kDa by hydrostatic pressure. As a result, larger particles like exosomes and other EVs remain in the tube where they can be collected. Apart from showing an efficient enrichment of the vesicles in comparison with the differential centrifugation protocol, it counts with a superior cost-efficiency with a faster workflow too [146].
4.1.3 Precipitation Agents
4.1.3.1 Polyethylene Glycol (PEG) Precipitation
By introducing a water-excluding polymer, such as polyethylene glycol (PEG) into the sample, exosomes can be settled out of biological fluids [139]. The water molecules “tides-up” causing exosomes, and the rest of the less soluble molecules, to precipitate out the solution [139]. This isolation method is quick, easy to use, requiring little technical expertise or any specialized equipment [134, 141]. Furthermore, it is compatible with a large number of samples. However, although it could be an easy option to integrate into clinical usage, its lack of selectivity, causes PEG polymers to be not exclusive to EVs and have other contamination substances [128, 133, 141].
4.1.3.2 Lectin Induced Agglutination
As an alternative to PEG, lectins are a family of proteins that bind carbohydrate moieties of other particles at a very high specificity [122]. Like PEG precipitation methods, the lectin precipitation methods are not time-consuming and do not need much expertise but have the problem of other soluble components. Hence, Lectin-induced exosome agglutination was explored for urinary exosome isolation [147].
It is also important to remark that several commercially available kits based on precipitation agents have been produced like ExoQuick (System Biosciences, CA, EEUU) [148] and ExoSpin (Cell Guidance System, Cambridge, UK) [149], which are based on PEG precipitation or ExoGAG (NasasBiotech, A Coruña, Spain) [123] that is a reactive that bonds with the glycosaminoglycans (GAGs) presented in the surface of EVs.
4.1.4 Immunoaffinity Captured-Based Techniques
They rely on the use of antibodies to capture the EVs based on the presence of lipids, proteins, and polysaccharides exposed on their surface [138, 150]. The fact that these techniques are primarily marker-dependent could be a constraint because the specificity of the assay relies on the specificity of the antibody used and thus tend to underestimate counts [134]. On the other hand, it presents a higher EVs purity than other methods based on other techniques [141]. Some examples of immunoaffinity capture-based techniques not exclusive to EVs are the Enzyme-Linked Immunosorbent Assay (ELISA) used to isolate exosomes from urine, plasma, and serum and Magneto-Immunoprecipitation that in comparison with ELISA has a higher isolation efficiency [122].
4.1.5 Microfluidic Based Isolation Techniques
Microfluidic-based isolation techniques are presented as a way to establish the use of EVs in clinical practice; however, its implementation is obstructed by issues such as scalability, validation, and standardization. They consist of the isolation of EVs based on their physical and biochemical properties simultaneously [134]. With their use, significant reductions in sample volume, reagent consumption, and isolation time are obtained because they can reproduce numerous laboratory processes on a microscale with high accuracy and specificity [149].
4.2 cEVs Cargo Profiling
EVs have a tremendous potential to be used in the field of liquid biopsy due to the molecules enclosed in them, which turn them into a useful circulating biomarker [117]. These molecules are basically DNAs, RNAs, multiple proteins, and metabolites [151] (Fig. 21.3). The identification of EVs-RNAs has been improved in the last years. The RNA cargo includes protein-coding transcripts (mRNAs) and many types of non-coding RNAs, including miRNA, long non-coding RNAs (LncRNAs), circular RNAs (circRNAs), small nucleolar RNA (snoRNAs), small nuclear RNAs (snRNAs), transfer RNA (tRNAs), ribosomal RNAs (rRNAs), and piwi-interacting RNAs (piRNAs) [152, 153]. Besides, EVs harbor different types of DNA, including single-stranded (ssDNA), double-stranded (dsDNA), mitochondrial DNA (mtDNA), and even viral DNA [154]. Importantly, the analysis of dsDNA in exosomes reflects the mutational status of parental tumor cells, thus is potentially useful for early detection of cancer and metastasis and also for tumor phenotyping [155, 156].
The protein content of cEVs has been also explored to find diagnostic and prognostic biomarkers. Current tools used to study EV-proteins include Western blot, enzyme-linked immunosorbent assays (ELISA), flow cytometry, and mass spectrometry, among others. Thus, for example, higher levels of ANXA2 were described in cEVs isolated from plasma samples of patients with EC than healthy controls. The presence of therapeutic targets such as PD-L1 is also feasible in the fraction of cEVs although its clinical meaning is not totally understood [157, 158]. Also, Melo et al. demonstrated the interest of Glypican-1 (GPC1) positive exosomes for identifying early and late-stage pancreatic cancer from healthy individuals or patients with benign disease [23].
5 Alternative Circulating Biomarkers
In recent years the potential of tumor-educated blood platelets as a non-invasive tumor biomarker has been demonstrated [159, 160]. Platelets are involved in the progression and spread of various solid cancers, and their RNA molecular signatures can provide specific information about the presence, location, and molecular characteristics of the tumors [161]. Preliminary studies indicate that platelet RNA may complement the information obtained with other non-invasive biomarkers for cancer diagnosis, potentially improving early-tumors detection and facilitating dynamic monitoring of the disease [161]. In fact, recent advances in the characterization of platelet-mRNA using high-throughput techniques revealed that, in the presence of malignant disease, there was an increase from 10 to more than 1000 altered mRNAs in platelets. In fact, clinically relevant fusions such as EML4-ALK rearrangements have been described in platelets from patients with non–small cell lung carcinoma (NSCLC) [162]. Besides platelets can intake plasma proteins that promote tumor growth and vascularization, such as basic fibroblast growth factor (FGF) or vascular endothelial growth factor (VEGF) [163, 164].
For platelets isolation, there are some important points that should be taken into account. Many drugs can interfere with platelet studies (for example, antihistamines, aspirin, non-steroidal anti-inflammatory drugs). Furthermore, systemic factors such as chronic or transient inflammatory diseases, or cardiovascular events and other noncancerous diseases, can also influence the platelet mRNA profile. Therefore, for blocking platelet activation during the isolation procedure, strong mechanical forces should be avoided and platelet inhibitors such as Citrate or HEPES can be also used. The recommended isolation method is double centrifugation. The first centrifugation at 150–300×g to obtain platelet-rich plasma and the second to collect the platelet fraction is generally performed at 300–800×g.
On the other hand, in the era of immunotherapy, several works have described the interest of analyzing the immune cells present in the bloodstream. The isolation and characterization of these cells are preferentially performed by flow cytometry and the selected cell fraction can be analyzed by different strategies to characterize the proteins and DNA/RNA content. Of note, a correlation between the neutrophil to lymphocyte ratio has been described as a mark of the immunotherapy activity in terms of survival rates [165, 166]. Besides, the T-cell receptor (TCR) repertoire, which consists of the number of T cells with specific TCRs, has also been described as a predictor biomarker in patients under immunotherapy treatment) [167]. The analysis of PD-1 expression on circulating lymphocytes has been linked to better immune responses in melanoma and renal cell carcinoma [168]. Among the different subpopulations of immune cells CD8+/CD73+ subset of lymphocytes has been associated with worse survival and poor clinical benefits in patients with melanoma under immunotherapy [169]. Also, in melanoma low levels of myeloid-derived suppressor cells were associated with better response to immunotherapy [170].
6 Challenges for the Clinical Application
The possibility of finding non-invasive circulating biomarkers that provide comprehensive information about the molecular characteristics of each tumor is of incredible interest for oncologists [171]. However, 20 years after the field of liquid biopsy started to grow only ctDNA analyses are being used in a clinical context. Numerous studies have shown the potential of new technologies for detecting genetic alterations associated with ctDNA, with promising preliminary clinical results. However, the implementation of liquid biopsy analyses is being slow due to the need for very high-sensitive technologies and more economic sources to cover the PCR or NGS-based studies. Besides, liquid biopsy tests lack standardized workflows, and this impacts reproducibility and, therefore, on the robustness of the tests [171]. Preanalytical steps, including sample collection, processing, and storage, are important factors conditioning this reproducibility [172, 173]. The specificity is also a critical point, since, for example, the detection of mutations in cancer-associated genes is not a guarantee of their tumoral origin. Thus, the existence of clonal hematopoiesis should be taken into consideration when interpreting NGS results on cfDNA analysis in order to avoid false positives [174]. In addition to genetic alterations, epigenetic marks will play an important role to translate the cfDNA analyses to diagnosis or screening scenarios [85]. Besides, fragmentomics also appears as a promising strategy to identify tumors specific patterns in cfDNA from cancer patients [175].
Although ctDNA has emerged as the leading circulating biomarker, the analysis of other circulating biomarkers such as CTCs and cEVs can provide more biological information about tumor dissemination and the development of resistance mechanisms. In addition, the field of CTCs should go behind the enumeration and validate the CTC phenotyping as a surrogate of the solid tumor. For that, techniques should improve their versatility and sensitivity to be able to have more CTCs numbers for molecular characterization [18]. In this context, single-CTCs characterization is opening new perspectives for the definition and interpretation of tumor heterogeneity and its biological impact on tumor aggressiveness. For advancing in cEVs validation as a clinical tool the implementation of easy and reproducible techniques is a clear challenge in the close future [176]. Besides, the development of novel strategies for cEVs isolation which cover EV subgroups in a pure fraction will be also a key point for the field development [21, 176].
Overall, the incorporation of liquid biopsy analyses into the clinical context requires the generation of guidelines and harmonized procedures. This will allow the development of interventional clinical trials to demonstrate the clinical benefit of including liquid biopsy for the management of cancer patients.
7 Conclusions
The application of liquid biopsy-based biomarkers is being broadly explored in many clinical contexts to manage cancer patients due to its minimal invasiveness and its value to obtain comprehensive and dynamic information about tumors. Several technologies have been developed during the last 20 years to address the study of different circulating elements, mainly CTCs, cfDNA, and cEVs. Sensitivity and reproducibility are two of the most valuable characteristics which are mandatory to characterize the tumoral material present in body fluids. The analysis of CTCs needs still improvement in these two aspects, and for this reason, CTCs studies are mainly focused on translational research to understand the dissemination process although different clinically relevant markers can be characterized in this tumor circulation population. Fortunately, cfDNA analyses, through PCR or NGS-based approaches, are nowadays being incorporated into the clinical practice to select targeted therapies in advanced tumors opening new avenues for personalized treatments. Other circulating elements such as cEVs or educated platelets represent promising biomarkers to complement the current alternatives to address the study of liquid biopsies in oncology.
References
Letai A (2017 Sep 8) Functional precision cancer medicine-moving beyond pure genomics. Nat Med 23(9):1028–1035
Parikh AR, Leshchiner I, Elagina L, Goyal L, Levovitz C, Siravegna G et al (2019) Liquid versus tissue biopsy for detecting acquired resistance and tumor heterogeneity in gastrointestinal cancers. Nat Med 25(9):1415–1421
Siravegna G, Marsoni S, Siena S, Bardelli A (2017 Sep) Integrating liquid biopsies into the management of cancer. Nat Rev Clin Oncol 14(9):531–548
Pantel K, Alix-Panabières C (2010 Sep) Circulating tumour cells in cancer patients: challenges and perspectives. Trends Mol Med 16(9):398–406
Leighl NB, Page RD, Raymond VM, Daniel DB, Divers SG, Reckamp KL et al (2019 Aug 1) Clinical utility of comprehensive cell-free DNA analysis to identify genomic biomarkers in patients with newly diagnosed metastatic non-small cell lung cancer. Clin Cancer Res 25(15):4691–4700
André F, Ciruelos EM, Juric D, Loibl S, Campone M, Mayer IA et al (2021) Alpelisib plus fulvestrant for PIK3CA-mutated, hormone receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: final overall survival results from SOLAR-1. Ann Oncol 32(2):208–217
Vidal J, Muinelo L, Dalmases A, Jones F, Edelstein D, Iglesias M et al (2017 June 1) Plasma ctDNA RAS mutation analysis for the diagnosis and treatment monitoring of metastatic colorectal cancer patients. Ann Oncol 28(6):1325–1332
García-Foncillas J, Tabernero J, Élez E, Aranda E, Benavides M, Camps C et al (2018) Prospective multicenter real-world RAS mutation comparison between OncoBEAM-based liquid biopsy and tissue analysis in metastatic colorectal cancer. Br J Cancer 119(12):1464–1470
Hrebien S, Citi V, Garcia-Murillas I, Cutts R, Fenwick K, Kozarewa I et al (2019) Early ctDNA dynamics as a surrogate for progression-free survival in advanced breast cancer in the BEECH trial. Ann Oncol 30(6):945–952
Lee JH, Menzies AM, Carlino MS, McEvoy AC, Sandhu S, Weppler AM et al (2020) Longitudinal monitoring of ctDNA in patients with melanoma and brain metastases treated with immune checkpoint inhibitors. Clin Cancer Res 26(15):4064–4071
Moss EL, Gorsia DN, Collins A, Sandhu P, Foreman N, Gore A et al (2020 Aug 10) Utility of circulating tumor DNA for detection and monitoring of endometrial cancer recurrence and progression. Cancers 12(8):2231
Muinelo-Romay L, Casas-Arozamena C, Abal M (2018 Aug 7) Liquid biopsy in endometrial cancer: new opportunities for personalized oncology. Int J Mol Sci 19:2311
Tie J, Cohen JD, Lo SN, Wang Y, Li L, Christie M et al (2021) Prognostic significance of postsurgery circulating tumor DNA in nonmetastatic colorectal cancer: individual patient pooled analysis of three cohort studies. Int J Cancer 148(4):1014–1026
Radovich M, Jiang G, Hancock BA, Chitambar C, Nanda R, Falkson C et al (2020) Association of circulating tumor DNA and circulating tumor cells after neoadjuvant chemotherapy with disease recurrence in patients with triple-negative breast cancer: preplanned secondary analysis of the BRE12-158 randomized clinical trial. JAMA Oncol 6(9):1410–1415
Liu X, Ren J, Luo N, Guo H, Zheng Y, Li J et al (2019) Comprehensive DNA methylation analysis of tissue of origin of plasma cell-free DNA by methylated CpG tandem amplification and sequencing (MCTA-Seq). Clin Epigenetics 11(1):93
Pickhardt PJ (2016) Emerging stool-based and blood-based non-invasive DNA tests for colorectal cancer screening: the importance of cancer prevention in addition to cancer detection. Abdom Radiol (New York) 41(8):1441–1444
Issa IA, Noureddine M (2017 July 28) Colorectal cancer screening: an updated review of the available options. World J Gastroenterol 23(28):5086–5096
Habli Z, AlChamaa W, Saab R, Kadara H, Khraiche ML (2020 Jul) Circulating tumor cell detection technologies and clinical utility: challenges and opportunities. Cancers 17:12(7)
Vasseur A, Kiavue N, Bidard F-C, Pierga J-Y, Cabel L (2021) Clinical utility of circulating tumor cells: an update. Mol Oncol 15(6):1647–1666
Zhong X, Zhang H, Zhu Y, Liang Y, Yuan Z, Li J et al (2020) Circulating tumor cells in cancer patients: developments and clinical applications for immunotherapy. Mol Cancer 19(1):15
Herrero C, Abal M, Muinelo-Romay L (2020) Circulating extracellular vesicles in gynecological tumors: realities and challenges. Front Oncol 10:565666
Liang Y, Lehrich BM, Zheng S, Lu M (2021) Emerging methods in biomarker identification for extracellular vesicle-based liquid biopsy. J Extracell Vesicles 10(7):e12090
Melo SA, Luecke LB, Kahlert C, Fernandez AF, Gammon ST, Kaye J et al (2015 Jul 9) Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature 523(7559):177–182
Int Veld SGJG, Wurdinger T (2019) Tumor-educated platelets. Blood 133(22):2359–2364
Griffiths JI, Wallet P, Pflieger LT, Stenehjem D, Liu X, Cosgrove PA et al (2020) Circulating immune cell phenotype dynamics reflect the strength of tumor-immune cell interactions in patients during immunotherapy. Proc Natl Acad Sci U S A 117(27):16072–16082
Allan AL, Vantyghem SA, Tuck AB, Chambers AF, Chin-Yee IH, Keeney M (2005 May) Detection and quantification of circulating tumor cells in mouse models of human breast cancer using immunomagnetic enrichment and multiparameter flow cytometry. Cytometry A 65(1):4–14
Alix-Panabières C, Pantel K (2016) Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov 6(5):479–491
Pantel K, Speicher MR (2016 Mar 10) The biology of circulating tumor cells. Oncogene 35(10):1216–1224
Alix-Panabières C, Pantel K (2013 Jan) Circulating tumor cells: liquid biopsy of cancer. Clin Chem 59(1):110–118
Riethdorf S, O’Flaherty L, Hille C, Pantel K (2018) Clinical applications of the CellSearch platform in cancer patients. Adv Drug Deliv Rev 125:102–121
Rushton AJ, Nteliopoulos G, Shaw JA, Coombes RC (2021 Feb 26) A review of circulating tumour cell enrichment technologies. Cancers 13:13(5)
Hayes DF, Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Miller MC et al (2006 Jul 15) Circulating tumor cells at each follow-up time point during therapy of metastatic breast cancer patients predict progression-free and overall survival. Clin Cancer Res 12(14 Pt 1):4218–4224
de Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H et al (2008 Oct 1) Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res 14(19):6302–6309
Cohen SJ, Punt CJA, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY et al (2008 Jul 1) Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol 26(19):3213–3221
Grover PK, Cummins AG, Price TJ, Roberts-Thomson IC, Hardingham JE (2014 Aug) Circulating tumour cells: the evolving concept and the inadequacy of their enrichment by EpCAM-based methodology for basic and clinical cancer research. Ann Oncol 25(8):1506–1516
Kowalik A, Kowalewska M, Góźdź S (2017) Current approaches for avoiding the limitations of circulating tumor cells detection methods-implications for diagnosis and treatment of patients with solid tumors. Transl Res 185:58–84.e15
Liu Z, Fusi A, Klopocki E, Schmittel A, Tinhofer I, Nonnenmacher A et al (2011 May 19) Negative enrichment by immunomagnetic nanobeads for unbiased characterization of circulating tumor cells from peripheral blood of cancer patients. J Transl Med 9:70
Szczerba BM, Castro-Giner F, Vetter M, Krol I, Gkountela S, Landin J et al (2019) Neutrophils escort circulating tumour cells to enable cell cycle progression. Nature 566(7745):553–557
Vona G, Sabile A, Louha M, Sitruk V, Romana S, Schütze K et al (2000 Jan) Isolation by size of epithelial tumor cells: a new method for the immunomorphological and molecular characterization of circulatingtumor cells. Am J Pathol 156(1):57–63
Hao S-J, Wan Y, Xia Y-Q, Zou X, Zheng S-Y (2018) Size-based separation methods of circulating tumor cells. Adv Drug Deliv Rev 125:3–20
Farace F, Massard C, Vimond N, Drusch F, Jacques N, Billiot F et al (2011 Sep 6) A direct comparison of CellSearch and ISET for circulating tumour-cell detection in patients with metastatic carcinomas. Br J Cancer 105(6):847–853
Miller MC, Robinson PS, Wagner C, O’Shannessy DJ (2018) The Parsortix™ cell separation system: a versatile liquid biopsy platform. Cytometry A 93(12):1234–1239
Hou HW, Warkiani ME, Khoo BL, Li ZR, Soo RA, Tan DS-W et al (2013) Isolation and retrieval of circulating tumor cells using centrifugal forces. Sci Rep 3:1259
Gascoyne PRC, Shim S (2014 Mar 12) Isolation of circulating tumor cells by dielectrophoresis. Cancers 6(1):545–579
Nagrath S, Sequist LV, Maheswaran S, Bell DW, Irimia D, Ulkus L et al (2007 Dec 20) Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 450(7173):1235–1239
Dizdar L, Fluegen G, van Dalum G, Honisch E, Neves RP, Niederacher D et al (2019) Detection of circulating tumor cells in colorectal cancer patients using the GILUPI CellCollector: results from a prospective, single-center study. Mol Oncol 13(7):1548–1558
Fischer JC, Niederacher D, Topp SA, Honisch E, Schumacher S, Schmitz N et al (2013 Oct 8) Diagnostic leukapheresis enables reliable detection of circulating tumor cells of nonmetastatic cancer patients. Proc Natl Acad Sci U S A 110(41):16580–16585
Valihrach L, Androvic P, Kubista M (2018 Mar) Platforms for single-cell collection and analysis. Int J Mol Sci 11:19(3)
Lawson DA, Kessenbrock K, Davis RT, Pervolarakis N, Werb Z (2018) Tumour heterogeneity and metastasis at single-cell resolution. Nat Cell Biol 20(12):1349–1360
Agarwal A, Balic M, El-Ashry D, Cote RJ (2018 Mar) Circulating tumor cells. Cancer J 24(2):70–77
Mondelo-Macía P, García-González J, León-Mateos L, Anido U, Aguín S, Abdulkader I et al (2021) Clinical potential of circulating free DNA and circulating tumour cells in patients with metastatic non-small-cell lung cancer treated with pembrolizumab. Mol Oncol 15(11):2923–2940
Cortés-Hernández LE, Eslami-S Z, Pantel K, Alix-Panabières C (2020) Molecular and functional characterization of circulating tumor cells: from discovery to clinical application. Clin Chem 66(1):97–104
Sinkala E, Sollier-Christen E, Renier C, Rosàs-Canyelles E, Che J, Heirich K et al (2017) Profiling protein expression in circulating tumour cells using microfluidic western blotting. Nat Commun 8:14622
Maheswaran S, Sequist LV, Nagrath S, Ulkus L, Brannigan B, Collura CV et al (2008 Jul 24) Detection of mutations in EGFR in circulating lung-cancer cells. N Engl J Med 359(4):366–377
Punnoose EA, Atwal S, Liu W, Raja R, Fine BM, Hughes BGM et al (2012 Apr 15) Evaluation of circulating tumor cells and circulating tumor DNA in non-small cell lung cancer: association with clinical endpoints in a phase II clinical trial of pertuzumab and erlotinib. Clin Cancer Res 18(8):2391–2401
Carter L, Rothwell DG, Mesquita B, Smowton C, Leong HS, Fernandez-Gutierrez F et al (2017) Molecular analysis of circulating tumor cells identifies distinct copy-number profiles in patients with chemosensitive and chemorefractory small-cell lung cancer. Nat Med 23(1):114–119
Krebs MG, Metcalf RL, Carter L, Brady G, Blackhall FH, Dive C (2014 Mar 21) Molecular analysis of circulating tumour cells—biology and biomarkers. Nat Rev Clin Oncol 11(3):129–144
Guo T (2016 Aug 31) Culture of circulating tumor cells - holy grail and big challenge. Int J Cancer Clin Res 3:63
Pantel K, Alix-Panabières C (2019) Liquid biopsy and minimal residual disease - latest advances and implications for cure. Nat Rev Clin Oncol 16(7):409–424
Lallo A, Schenk MW, Frese KK, Blackhall F, Dive C (2017 Aug) Circulating tumor cells and CDX models as a tool for preclinical drug development. Transl Lung Cancer Res 6(4):397–408
Hodgkinson CL, Morrow CJ, Li Y, Metcalf RL, Rothwell DG, Trapani F et al (2014 Aug) Tumorigenicity and genetic profiling of circulating tumor cells in small-cell lung cancer. Nat Med 20(8):897–903
Schölch S, García SA, Iwata N, Niemietz T, Betzler AM, Nanduri LK et al (2016 May 10) Circulating tumor cells exhibit stem cell characteristics in an orthotopic mouse model of colorectal cancer. Oncotarget 7(19):27232–27242
Pereira-Veiga T, Abreu M, Robledo D, Matias-Guiu X, Santacana M, Sánchez L et al (2019) CTCs-derived xenograft development in a triple negative breast cancer case. Int J Cancer 144(9):2254–2265
Faugeroux V, Pailler E, Oulhen M, Deas O, Brulle-Soumare L, Hervieu C et al (2020) Genetic characterization of a unique neuroendocrine transdifferentiation prostate circulating tumor cell-derived eXplant model. Nat Commun 11(1):1884
Jahr S, Hentze H, Englisch S, Hardt D, Fackelmayer FO, Hesch RD et al (2001 Feb 15) DNA fragments in the blood plasma of cancer patients: quantitations and evidence for their origin from apoptotic and necrotic cells. Cancer Res 61(4):1659–1665
Kustanovich A, Schwartz R, Peretz T, Grinshpun A (2019) Life and death of circulating cell-free DNA. Cancer Biol Ther 20(8):1057–1067
Keller L, Belloum Y, Wikman H, Pantel K (2021) Clinical relevance of blood-based ctDNA analysis: mutation detection and beyond. Br J Cancer 124(2):345–358
Meddeb R, Pisareva E, Thierry AR (2019) Guidelines for the preanalytical conditions for analyzing circulating cell-free DNA. Clin Chem 65(5):623–633
van der Leest P, Boonstra PA, Ter Elst A, van Kempen LC, Tibbesma M, Koopmans J et al (2020 May 13) Comparison of circulating cell-free DNA extraction methods for downstream analysis in cancer patients. Cancers 12:1222
Franczak C, Filhine-Tresarrieu P, Gilson P, Merlin J-L, Au L, Harlé A (2019 Feb 1) Technical considerations for circulating tumor DNA detection in oncology. Expert Rev Mol Diagn 19(2):121–135
Mauger F, Dulary C, Daviaud C, Deleuze J-F, Tost J (2015 Sep) Comprehensive evaluation of methods to isolate, quantify, and characterize circulating cell-free DNA from small volumes of plasma. Anal Bioanal Chem 407(22):6873–6878
Kilgour E, Rothwell DG, Brady G, Dive C (2020) Liquid biopsy-based biomarkers of treatment response and resistance. Cancer Cell 37(4):485–495
Cohen JD, Li L, Wang Y, Thoburn C, Afsari B, Danilova L et al (2018 Feb 23) Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science (New York, NY). 359(6378):926–930
Abbosh C, Birkbak NJ, Swanton C (2018) Early stage NSCLC - challenges to implementing ctDNA-based screening and MRD detection. Nat Rev Clin Oncol 15(9):577–586
Wang Z, Duan J, Cai S, Han M, Dong H, Zhao J et al (2019 May 1) Assessment of blood tumor mutational burden as a potential biomarker for immunotherapy in patients with non-small cell lung cancer with use of a next-generation sequencing cancer gene panel. JAMA Oncol 5(5):696–702
Martínez-Sáez O, Chic N, Pascual T, Adamo B, Vidal M, González-Farré B et al (2020) Frequency and spectrum of PIK3CA somatic mutations in breast cancer. Breast Cancer Res 22(1):45
Malapelle U, Sirera R, Jantus-Lewintre E, Reclusa P, Calabuig-Fariñas S, Blasco A et al (2017) Profile of the Roche cobas® EGFR mutation test v2 for non-small cell lung cancer. Expert Rev Mol Diagn 17(3):209–215
Woodhouse R, Li M, Hughes J, Delfosse D, Skoletsky J, Ma P et al (2020) Clinical and analytical validation of FoundationOne liquid CDx, a novel 324-gene cfDNA-based comprehensive genomic profiling assay for cancers of solid tumor origin. PLoS One 15(9):e0237802
Laufer-Geva S, Rozenblum AB, Twito T, Grinberg R, Dvir A, Soussan-Gutman L et al (2018) The clinical impact of comprehensive genomic testing of circulating cell-free DNA in advanced lung cancer. J Thoracic Oncol 13(11):1705–1716
Ren AH, Fiala CA, Diamandis EP, Kulasingam V (2020) Pitfalls in cancer biomarker discovery and validation with emphasis on circulating tumor DNA. Cancer Epidemiol Biomarker Prev 29(12):2568–2574
Elazezy M, Joosse SA (2018) Techniques of using circulating tumor DNA as a liquid biopsy component in cancer management. Comput Struct Biotechnol J 16:370–378
Bando H, Kagawa Y, Kato T, Akagi K, Denda T, Nishina T et al (2019) A multicentre, prospective study of plasma circulating tumour DNA test for detecting RAS mutation in patients with metastatic colorectal cancer. Br J Cancer 120(10):982–986
Chen M, Zhao H (2019) Next-generation sequencing in liquid biopsy: cancer screening and early detection. Hum Genomics 13(1):34
Szilágyi M, Pös O, Márton É, Buglyó G, Soltész B, Keserű J et al (2020 Sep 17) Circulating cell-free nucleic acids: main characteristics and clinical application. Int J Mol Sci 21:6827
Rodriguez-Casanova A, Costa-Fraga N, Bao-Caamano A, López-López R, Muinelo-Romay L, Diaz-Lagares A (2021) Epigenetic landscape of liquid biopsy in colorectal cancer. Front Cell Dev Biol 9:622459
Portela A, Esteller M (2010 Oct) Epigenetic modifications and human disease. Nat Biotechnol 28(10):1057–1068
Huang J, Wang L (2019 Nov) Cell-free DNA methylation profiling analysis-technologies and bioinformatics. Cancers 6:11(11)
Frommer M, McDonald LE, Millar DS, Collis CM, Watt F, Grigg GW et al (1992 Mar 1) A genomic sequencing protocol that yields a positive display of 5-methylcytosine residues in individual DNA strands. Proc Natl Acad Sci U S A 89(5):1827–1831
Li M, Chen W-D, Papadopoulos N, Goodman SN, Bjerregaard NC, Laurberg S et al (2009 Sep) Sensitive digital quantification of DNA methylation in clinical samples. Nat Biotechnol 27(9):858–863
Chimonidou M, Strati A, Tzitzira A, Sotiropoulou G, Malamos N, Georgoulias V et al (2011 Aug) DNA methylation of tumor suppressor and metastasis suppressor genes in circulating tumor cells. Clin Chem 57(8):1169–1177
Boeckx N, Op de Beeck K, Beyens M, Deschoolmeester V, Hermans C, De Clercq P et al (2018) Mutation and methylation analysis of circulating tumor DNA can be used for follow-up of metastatic colorectal cancer patients. Clin Colorectal Cancer 17(2):e369–e379
Barault L, Amatu A, Bleeker FE, Moutinho C, Falcomatà C, Fiano V et al (2015 Sep) Digital PCR quantification of MGMT methylation refines prediction of clinical benefit from alkylating agents in glioblastoma and metastatic colorectal cancer. Ann Oncol 26(9):1994–1999
Gallardo-Gómez M, Moran S, Páez de la Cadena M, Martínez-Zorzano VS, Rodríguez-Berrocal FJ, Rodríguez-Girondo M et al (2018) A new approach to epigenome-wide discovery of non-invasive methylation biomarkers for colorectal cancer screening in circulating cell-free DNA using pooled samples. Clin Epigenetics 10:53
Friedlander TW, Ngo VT, Dong H, Premasekharan G, Weinberg V, Doty S et al (2014 May 15) Detection and characterization of invasive circulating tumor cells derived from men with metastatic castration-resistant prostate cancer. Int J Cancer 134(10):2284–2293
Li W, Li Q, Kang S, Same M, Zhou Y, Sun C et al (2018) CancerDetector: ultrasensitive and non-invasive cancer detection at the resolution of individual reads using cell-free DNA methylation sequencing data. Nucleic Acids Res 46(15):e89
Gkountela S, Castro-Giner F, Szczerba BM, Vetter M, Landin J, Scherrer R et al (2019) Circulating tumor cell clustering shapes dna methylation to enable metastasis seeding. Cell. 176(1–2):98–112.e14
Raposo G, Stoorvogel W (2013 Feb 18) Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 200(4):373–383
György B, Szabó TG, Pásztói M, Pál Z, Misják P, Aradi B et al (2011 Aug) Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cell Mol Life Sci 68(16):2667–2688
Colombo M, Raposo G, Théry C (2014) Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol 30:255–289
Théry C, Ostrowski M, Segura E (2009 Aug) Membrane vesicles as conveyors of immune responses. Nat Rev Immunol 9(8):581–593
van der Meel R, Krawczyk-Durka M, van Solinge WW, Schiffelers RM (2014 Jun) Toward routine detection of extracellular vesicles in clinical samples. Int J Lab Hematol 36(3):244–253
Raposo G, Nijman HW, Stoorvogel W, Liejendekker R, Harding CV, Melief CJ et al (1996 Mar 1) B lymphocytes secrete antigen-presenting vesicles. J Exp Med 183(3):1161–1172
Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D et al (1998 May) Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med 4(5):594–600
Yáñez-Mó M, Siljander PR-M, Andreu Z, Zavec AB, Borràs FE, Buzas EI et al (2015) Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles 4:27066
Kalluri R (2016 Apr 1) The biology and function of exosomes in cancer. J Clin Invest 126(4):1208–1215
Akers JC, Gonda D, Kim R, Carter BS, Chen CC (2013 May) Biogenesis of extracellular vesicles (EV): exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies. J Neuro-Oncol 113(1):1–11
Ratajczak J, Miekus K, Kucia M, Zhang J, Reca R, Dvorak P et al (2006 May) Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia 20(5):847–856
Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO (2007 Jun) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9(6):654–659
Lai RC, Yeo RWY, Tan KH, Lim SK (2013) Exosomes for drug delivery: a novel application for the mesenchymal stem cell. Biotechnol Adv. 31(5):543–551
Zaborowski MP, Balaj L, Breakefield XO, Lai CP (2015 Aug 1) Extracellular vesicles: composition, biological relevance, and methods of study. Bioscience 65(8):783–797
Hood JL, San RS, Wickline SA (2011 Jun 1) Exosomes released by melanoma cells prepare sentinel lymph nodes for tumor metastasis. Cancer Res 71(11):3792–3801
Rak J (2010 Nov) Microparticles in cancer. Semin Thromb Hemost 36(8):888–906
Cocucci E, Racchetti G, Meldolesi J (2009 Feb) Shedding microvesicles: artefacts no more. Trends Cell Biol 19(2):43–51
Zhang H-G, Grizzle WE (2011 Mar 1) Exosomes and cancer: a newly described pathway of immune suppression. Clin Cancer Res 17(5):959–964
Logozzi M, de Milito A, Lugini L, Borghi M, Calabrò L, Spada M et al (2009 Apr 17) High levels of exosomes expressing CD63 and Caveolin-1 in plasma of melanoma patients. PLoS One 4(4):e5219
Ciardiello C, Cavallini L, Spinelli C, Yang J, Reis-Sobreiro M, de Candia P et al (2016 Feb 6) Focus on extracellular vesicles: new Frontiers of cell-to-cell communication in cancer. Int J Mol Sci 17(2):175
Carvalho J, Oliveira C (2014) Extracellular vesicles - powerful markers of cancer EVolution. Front Immunol 5:685
Nosova VP, Alekseeva LM, Bobkov II, Mumladze RB, Popova OA (1989) Changes in the rheologic properties of the blood in peptic ulcer. Sov Med. 4:87–90
Théry C, Zitvogel L, Amigorena S (2002 Aug) Exosomes: composition, biogenesis and function. Nat Rev Immunol 2(8):569–579
Kahlert C, Kalluri R (2013 Apr) Exosomes in tumor microenvironment influence cancer progression and metastasis. J Mol Med (Berlin, Germany) 91(4):431–437
van Niel G, D’Angelo G, Raposo G (2018) Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol 19(4):213–228
Doyle LM, Wang MZ (2019) Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells 8(7):727
Herrero C, de la Fuente A, Casas-Arozamena C, Sebastian V, Prieto M, Arruebo M et al (2019 Dec 12) Extracellular vesicles-based biomarkers represent a promising liquid biopsy in endometrial cancer. Cancers 11:2000
Mathai RA, Vidya RVS, Reddy BS, Thomas L, Udupa K, Kolesar J et al (2019 Mar) Potential utility of liquid biopsy as a diagnostic and prognostic tool for the assessment of solid tumors: implications in the precision oncology. J Clin Med 18:8(3)
Lötvall J, Hill AF, Hochberg F, Buzás EI, di Vizio D, Gardiner C et al (2014) Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles 3:26913
Théry C, Amigorena S, Raposo G, Clayton A (2006 Apr) Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol Chapter 3:Unit 3.22
Hiemstra TF, Charles PD, Gracia T, Hester SS, Gatto L, Al-Lamki R et al (2014 Sep) Human urinary exosomes as innate immune effectors. J Am Soc Nephrol 25(9):2017–2027
Zarovni N, Corrado A, Guazzi P, Zocco D, Lari E, Radano G et al (2015 Oct 1) Integrated isolation and quantitative analysis of exosome shuttled proteins and nucleic acids using immunocapture approaches. Methods (San Diego, Calif). 87:46–58
Gardiner C, di Vizio D, Sahoo S, Théry C, Witwer KW, Wauben M et al (2016) Techniques used for the isolation and characterization of extracellular vesicles: results of a worldwide survey. J Extracell Vesicles 5:32945
Momen-Heravi F, Balaj L, Alian S, Mantel P-Y, Halleck AE, Trachtenberg AJ et al (2013 Oct) Current methods for the isolation of extracellular vesicles. Biol Chem 394(10):1253–1262
Livshits MA, Livshts MA, Khomyakova E, Evtushenko EG, Lazarev VN, Kulemin NA et al (2015 Nov 30) Isolation of exosomes by differential centrifugation: theoretical analysis of a commonly used protocol. Sci Rep 5:17319
Webber J, Clayton A (2013) How pure are your vesicles? J Extracell Vesicles 2
van Deun J, Mestdagh P, Sormunen R, Cocquyt V, Vermaelen K, Vandesompele J et al (2014) The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling. J Extracell Vesicles 3
Li P, Kaslan M, Lee SH, Yao J, Gao Z (2017) Progress in exosome isolation techniques. Theranostics 7(3):789–804
Kowal J, Arras G, Colombo M, Jouve M, Morath JP, Primdal-Bengtson B et al (2016 Feb 23) Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc Natl Acad Sci U S A 113(8):E968–E977
Shao H, Im H, Castro CM, Breakefield X, Weissleder R, Lee H (2018) New technologies for analysis of extracellular vesicles. Chem Rev 118(4):1917–1950
Tauro BJ, Greening DW, Mathias RA, Ji H, Mathivanan S, Scott AM et al (2012 Feb) Comparison of ultracentrifugation, density gradient separation, and immunoaffinity capture methods for isolating human colon cancer cell line LIM1863-derived exosomes. Methods (San Diego, Calif). 56(2):293–304
Zhang M, Jin K, Gao L, Zhang Z, Li F, Zhou F et al (2018 Sep) Methods and technologies for exosome isolation and characterization. Small Methods 2(9):1800021
Zeringer E, Barta T, Li M, Vlassov A (2015 Apr 1) v. Strategies for isolation of exosomes. Cold Spring Harb Protoc 2015(4):319–323
Liga A, Vliegenthart ADB, Oosthuyzen W, Dear JW, Kersaudy-Kerhoas M (2015 Jun 7) Exosome isolation: a microfluidic road-map. Lab Chip 15(11):2388–2394
Batrakova EV, Kim MS (2015 Dec 10) Using exosomes, naturally-equipped nanocarriers, for drug delivery. J Control Release. 219:396–405
Grubisic Z, Rempp P, Benoit H (1967 Sep) A universal calibration for gel permeation chromatography. J Polym Sci B Polym Lett 5(9):753–759
Feng Y, Huang W, Wani M, Yu X, Ashraf M (2014) Ischemic preconditioning potentiates the protective effect of stem cells through secretion of exosomes by targeting Mecp2 via miR-22. PLoS One 9(2):e88685
Roura S, Gámez-Valero A, Lupón J, Gálvez-Montón C, Borràs FE, Bayes-Genis A (2018) Proteomic signature of circulating extracellular vesicles in dilated cardiomyopathy. Lab Investig. 98(10):1291–1299
Gámez-Valero A, Monguió-Tortajada M, Carreras-Planella L, Franquesa M, Beyer K, Borràs FE (2016) Size-exclusion chromatography-based isolation minimally alters extracellular vesicles’ characteristics compared to precipitating agents. Sci Rep 6:33641
Musante L, Tataruch D, Gu D, Benito-Martin A, Calzaferri G, Aherne S et al (2014 Dec 23) A simplified method to recover urinary vesicles for clinical applications, and sample banking. Sci Rep 4:7532
Samsonov R, Shtam T, Burdakov V, Glotov A, Tsyrlina E, Berstein L et al (2016 Jan) Lectin-induced agglutination method of urinary exosomes isolation followed by mi-RNA analysis: application for prostate cancer diagnostic. Prostate 76(1):68–79
Hu JL, Wang W, Lan XL, Zeng ZC, Liang YS, Yan YR et al (2019) CAFs secreted exosomes promote metastasis and chemotherapy resistance by enhancing cell stemness and epithelial-mesenchymal transition in colorectal cancer. Mol Cancer 18(1):91
Małys MSS, Aigner C, Schulz SMM, Schachner H, Rees AJJ, Kain R (2021 Apr 28) Isolation of small extracellular vesicles from human sera. Int J Mol Sci 22(9):4653
Konoshenko MY, Lekchnov EA, Vlassov AV, Laktionov PP (2018) Isolation of extracellular vesicles: general methodologies and latest trends. BioMed Res Int. 2018:8545347
Huang T, Song C, Zheng L, Xia L, Li Y, Zhou Y (2019) The roles of extracellular vesicles in gastric cancer development, microenvironment, anti-cancer drug resistance, and therapy. Mol Cancer 18(1):62
Nolte-‘t Hoen ENM, Buermans HPJ, Waasdorp M, Stoorvogel W, Wauben MHM, ‘t Hoen PAC (2012 Oct) Deep sequencing of RNA from immune cell-derived vesicles uncovers the selective incorporation of small non-coding RNA biotypes with potential regulatory functions. Nucleic Acids Res 40(18):9272–9285
Kim KM, Abdelmohsen K, Mustapic M, Kapogiannis D, Gorospe M (2017) RNA in extracellular vesicles. Wiley Interdiscip Rev RNA 8(4):10.1002/wrna.1413
Elzanowska J, Semira C, Costa-Silva B (2021) DNA in extracellular vesicles: biological and clinical aspects. Mol Oncol 15(6):1701–1714
Kahlert C, Melo SA, Protopopov A, Tang J, Seth S, Koch M et al (2014 Feb 14) Identification of double-stranded genomic DNA spanning all chromosomes with mutated KRAS and p53 DNA in the serum exosomes of patients with pancreatic cancer. J Biol Chem 289(7):3869–3875
Thakur BK, Zhang H, Becker A, Matei I, Huang Y, Costa-Silva B et al (2014 Jun) Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res 24(6):766–769
Ricklefs FL, Alayo Q, Krenzlin H, Mahmoud AB, Speranza MC, Nakashima H et al (2018) Immune evasion mediated by PD-L1 on glioblastoma-derived extracellular vesicles. Sci Adv. 4(3):eaar2766
Wu F, Gu Y, Kang B, Heskia F, Pachot A, Bonneville M et al (2021 Jun) PD-L1 detection on circulating tumor-derived extracellular vesicles (T-EVs) from patients with lung cancer. Transl Lung Cancer Res 10(6):2441–2451
Nilsson RJA, Balaj L, Hulleman E, van Rijn S, Pegtel DM, Walraven M et al (2011 Sep 29) Blood platelets contain tumor-derived RNA biomarkers. Blood 118(13):3680–3683
Luo C-L, Xu Z-G, Chen H, Ji J, Wang Y-H, Hu W et al (2018) LncRNAs and EGFRvIII sequestered in TEPs enable blood-based NSCLC diagnosis. Cancer Manag Res 10:1449–1459
Best MG, Vancura A, Wurdinger T (2017) Platelet RNA as a circulating biomarker trove for cancer diagnostics. J Thromb Haemost 15(7):1295–1306
Park C-K, Kim J-E, Kim M-S, Kho B-G, Park H-Y, Kim T-O et al (2019 Aug) Feasibility of liquid biopsy using plasma and platelets for detection of anaplastic lymphoma kinase rearrangements in non-small cell lung cancer. J Cancer Res Clin Oncol 145(8):2071–2082
Roweth HG, Battinelli EM (2021) Lessons to learn from tumor-educated platelets. Blood 137(23):3174–3180
Klement GL, Yip T-T, Cassiola F, Kikuchi L, Cervi D, Podust V et al (2009 Mar 19) Platelets actively sequester angiogenesis regulators. Blood 113(12):2835–2842
Jiang T, Bai Y, Zhou F, Li W, Gao G, Su C et al (2019) Clinical value of neutrophil-to-lymphocyte ratio in patients with non-small-cell lung cancer treated with PD-1/PD-L1 inhibitors. Lung Cancer (Amsterdam, Netherlands) 130:76–83
Bryant AK, Sankar K, Strohbehn GW, Zhao L, Elliott D, Qin A et al (2022) Prognostic and predictive value of neutrophil-to-lymphocyte ratio with adjuvant immunotherapy in stage III non-small-cell lung cancer. Lung Cancer (Amsterdam, Netherlands) 163:35–41
Gibney GT, Weiner LM, Atkins MB (2016 Dec) Predictive biomarkers for checkpoint inhibitor-based immunotherapy. Lancet Oncol 17(12):e542–e551
Kim CG, Hong MH, Kim KH, Seo I-H, Ahn B-C, Pyo K-H et al (2021 Jan) Dynamic changes in circulating PD-1+ CD8+ T lymphocytes for predicting treatment response to PD-1 blockade in patients with non-small-cell lung cancer. Eur J Cancer 143:113–126
Capone M, Fratangelo F, Giannarelli D, Sorrentino C, Turiello R, Zanotta S et al (2020) Frequency of circulating CD8+CD73+T cells is associated with survival in nivolumab-treated melanoma patients. J Transl Med 18(1):121
Meyer C, Cagnon L, Costa-Nunes CM, Baumgaertner P, Montandon N, Leyvraz L et al (2014 Mar) Frequencies of circulating MDSC correlate with clinical outcome of melanoma patients treated with ipilimumab. Cancer Immunol Immunother 63(3):247–257
De Mattos-Arruda L, Siravegna G (2021) How to use liquid biopsies to treat patients with cancer. ESMO Open 6(2):100060
Gerber T, Taschner-Mandl S, Saloberger-Sindhöringer L, Popitsch N, Heitzer E, Witt V et al (2020) Assessment of pre-analytical sample handling conditions for comprehensive liquid biopsy analysis. J Mol Diagn 22(8):1070–1086
Fleischhacker M, Schmidt B (2020 Jun 25) Pre-analytical issues in liquid biopsy – where do we stand? J Lab Med 44(3):117–142
Hu Y, Ulrich BC, Supplee J, Kuang Y, Lizotte PH, Feeney NB et al (2018) False-positive plasma genotyping due to clonal hematopoiesis. Clin Cancer Res 24(18):4437–4443
YMD L, DSC H, Jiang P, RWK C (2021 Apr 9) Epigenetics, fragmentomics, and topology of cell-free DNA in liquid biopsies. Science 372(6538):eaaw3616
Ayers L, Pink R, Carter DRF, Nieuwland R (2019) Clinical requirements for extracellular vesicle assays. J Extracell Vesicles 8(1):1593755
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Mondelo-Macía, P., Rodríguez-Ces, A.M., Suárez-Cunqueiro, M.M., Romay, L.M. (2022). Methods for the Detection of Circulating Biomarkers in Cancer Patients. In: Caballero, D., Kundu, S.C., Reis, R.L. (eds) Microfluidics and Biosensors in Cancer Research. Advances in Experimental Medicine and Biology, vol 1379. Springer, Cham. https://doi.org/10.1007/978-3-031-04039-9_21
Download citation
DOI: https://doi.org/10.1007/978-3-031-04039-9_21
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-04038-2
Online ISBN: 978-3-031-04039-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)