Abstract
Resilience is the capacity of ecosystems to recover from disturbance or to absorb disturbance without changing their structures and processes. While engineering resilience focuses solely on recovery from disturbance, ecological resilience also considers the possibility of a regime change after disturbance. A key element of resilience is the adaptive cycle in ecosystems, that is, the alternation of phases of growth, conservation, release, and renewal. Important mechanisms that make ecosystems resilient against disturbances are interactions over spatial and temporal scales, legacies of the pre-disturbance state, ecological stress memory, and the response diversity of plant communities.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Adaptive cycle
- Cross-scale interactions
- Disturbance legacies
- Early warning indicators
- Panarchy
- Recovery
- Regime shift
- Response diversity
- Stress memory
1 Introduction and Definitions
Disturbances are, by definition, discrete events in space and time which cause major changes in ecosystems, for example, in the form of plant mortality (see Chap. 2). Organisms, communities, and ecosystems are adapted to such changes and have a strong ability to recover from disturbances. This property is called resilience.
Resilience has received increasing attention in science in recent years, not least because of rapidly changing environmental conditions and new types of disturbances. The concept of resilience has become one of the most important research topics in the sustainability debate (Folke et al. 2004; Rockström et al. 2009). Fundamental research on mechanisms and limits of resilience is an active field that is developing rapidly. For example, achieving functional resilience is currently an important goal of risk research and experimental biodiversity research (Isbell et al. 2015; Kreyling et al. 2017). Furthermore, the concept of resilience is increasingly used as a guideline and target for the management of ecosystems (Biggs et al. 2012; Seidl 2014; see also Chap. 17). The diverse usages of the concept of resilience have led to a wide range of definitions (Brand and Jax 2007), which is why it is especially important to specify the meaning of resilience for the respective context or application (resilience of what? resilience to what? Carpenter et al. 2001). In general, the literature distinguishes three types of resilience: engineering resilience, ecological resilience, and social-ecological resilience (Nikinmaa et al. 2020).
1.1 Engineering Resilience
Engineering resilience focuses on the recovery after disturbance: the faster a system returns to its original state after a disturbance, the more resilient it is (Holling 1996). Engineering resilience assumes a predictable recovery path as well as constancy in the undisturbed state (ecological equilibrium; equilibrium assumption); systems, therefore, always recover along the same path and differ only in the speed of their recovery. As the name implies, this concept of resilience is often used in technical and engineering sciences, for example, to describe the development of material characteristics after stress. However, engineering resilience is also an important parameter in ecology: it can, for instance, be used to describe the recovery of tree growth after a drought period (Lloret et al. 2011; Zang et al. 2014). Tree growth is a narrowly defined indicator whose development has only one degree of freedom. The state of tree growth before a disturbance can be clearly defined, and therefore the requirements for the definition of engineering resilience are met in this example.
1.2 Ecological Resilience
The behaviour of communities and ecosystems is much more complex than materials, abiotic systems, or individual indicators such as tree rings. Many ecosystems are significantly affected by disturbance, but their characteristic functions remain intact despite disturbance or are restored relatively quickly after disturbance – thus, they are resilient. For example, after a fire, a forest stand will grow back into a forest stand, and after mowing a flowering mountain meadow will grow back into a flowering mountain meadow. Nevertheless, there are also disturbances which lead to regime shifts, especially if degradation has already occurred or if environmental conditions and resources change relatively quickly. If the ecological resilience of a system is exceeded, the system will change, for example, after a severe forest fire, trees may not regenerate sufficiently and there can be a transition towards open land. The concept of ecological resilience (Holling 1973, 1996; Gunderson 2000) considers this dynamic of alternative stable states, which are characterized by different structures and processes. Ecological resilience is thus the capacity of a system to absorb disturbances without changing the system’s typical structures and processes. It is important to note that the preservation of structures and processes does not necessarily mean a deterministic return of the system to the state before a disturbance. In European primaeval forests, for example, there are a multitude of developmental paths after a disturbance (Meigs et al. 2017). However, characteristic structures (e.g. a complex canopy structure) and processes (e.g. carbon uptake) are recovered (to varying degrees) in all these paths of natural ecosystem development after disturbance – the system is thus ecologically resilient (Seidl et al. 2014). Ecological resilience can be seen as a mechanism of dynamic stability (Turner et al. 1993): disturbances and transient changes are an inherent part of many ecosystems, without fundamentally changing them (see Chap. 3).
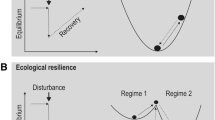
If a disturbance exceeds the ecological resilience of a system, its regime and thus its structures and processes change fundamentally. In most cases, this regime change is not gradual but abrupt and occurs when a threshold is exceeded. For example, precipitation-induced boundaries between forest and savannah or temperature-induced boundaries between forest and tundra are not gradual but are rather expressed as relatively discrete tipping points of the biosphere (Hirota et al. 2011; Scheffer et al. 2012). An often-used metaphor to describe the concept of ecological resilience is the ‘ball and cup’ model, in which the ball describes the current system state and a landscape of valleys (i.e. stable system states, attractors of the system) and crests (i.e. unstable system states) describes the different possible states of the system (Fig. 5.1). A disturbance causes an impulse on the system and pushes it from its resting point in the current attractor. The deeper and narrower the valley, the faster the system returns to the centre of the attractor after the disturbance (and the greater its resilience). If the impulse from the disturbance is so strong that the ball is moved from one attractor to the next, the disturbance exceeds the resilience of the system and results in a regime shift. We note that the ‘attractor landscape’ of a system (i.e. its valleys and crests in the ‘ball and cup’ model) is in most cases not static over time. Factors such as the extinction and immigration of species into a system or climatic changes may cause attractors (both in terms of strength and location) to change over time (Gunderson 2000; Seidl et al. 2016b). Different stable states of ecosystems are either alternating and thus reversible (e.g. the oligotrophic and eutrophic states of a lake) or they are largely irreversible (e.g. the nutrient enrichment from atmospheric deposition of ecosystems). Ecological resilience (i.e. the ability of a system to remain in a stable system state despite disturbance) is a neutral characteristic that is not per se good or bad. However, with regard to environmental changes such as climate change, the preservation of a certain system state is often the goal – here resilience is a desired property of the system, which can be further promoted by management measures. In contrast, restoration ecology often aims at restoring a system to its former state, possibly using disturbances to overcome the resilience of the current system. Examples are the liming of acidified lakes or the removal of topsoil from nutrient-polluted former nutrient-poor meadows (Fig. 5.2).
(a) Schematic representation of different attractors for resilient ecosystem states. Disturbances act as triggers and catalysts for regime change. (After Scheffer and Carpenter 2003.) (b) An example of the use of disturbances in nature conservation is the removal of nutrient-rich topsoil to restore resource-limited sand ecosystems on inland dunes in southern Germany in order to promote rare and endangered pioneer species that are weak competitors
1.3 Social-Ecological Resilience
Social-ecological resilience takes up the concept of ecological resilience and extends it from ecological systems to social-ecological systems (Folke 2006). Resilience here means the ability of these systems to maintain their structures and processes in the face of disturbance and, for example, provide ecosystem services to society in a sustainable manner despite disturbance (Folke et al. 2002; Brand and Jax 2007; Biggs et al. 2012). An important aspect is social adaptive capacity, that is, the ability to respond to external stressors and disturbances with social or political change (Adger 2000). This type of resilience will not be discussed further in this chapter. However, it is of importance in the context of disturbance management (Seidl et al. 2016b) and is addressed in Chap. 17.
1.4 Panarchy
Closely related to the idea of ecological resilience is the concept of panarchy, which was introduced by Holling and Gunderson (2002). It is a model that describes the dynamic organization of complex systems in space and time, and can be used for the characterization and quantification of resilience in ecosystems. The panarchy model distinguishes between different levels of a system, for example, in the context of a forest ecosystem – leaf, single tree, stand, and landscape. For each of these levels, the dynamics of the system can be described by an adaptive cycle through the phases of growth, conservation, release, and reorganization (Fig. 5.3). From the growth to the conservation phase, the potential of the ecosystem increases; it accumulates biomass, energy, and other system components, such as species, growth forms, or functional groups. At the same time, the interconnectedness between the individual system components also increases, that is, system behaviour is increasingly determined by interactions such as competition or mutualism. In an old-growth forest, for example, the competition for light between trees determines the regeneration dynamics and species composition more than external factors. However, these strong system-internal interactions combined with a simultaneous increase in system potential (e.g. accumulated biomass) also lead to the system becoming increasingly inflexible and thus susceptible to disturbance. If a disturbance occurs (e.g. reduction of live biomass because of fire) the potential of the ecosystem is reduced in the release phase. In the subsequent phase of reorganization, the system components recombine. This can either lead to a growth phase along the previous system trajectory or to a regime change and the beginning of a qualitatively different adaptive cycle (see Fig. 5.3; Scheffer and Carpenter 2003; Allen et al. 2014). The duration of individual phases of the adaptive cycle can vary considerably. While in a forest ecosystem the growth phase typically lasts several decades, depending on the location, the conservation phase can span many centuries. The release phase, on the other hand, often lasts only a few hours (windthrow), days (forest fire), or years (bark beetle outbreak), and the reorganization of the system usually takes a few years to a few decades. Panarchy is a hierarchically nested arrangement of adaptive cycles (Fig. 5.4). Key components of the panarchy model are the cross-scale connections between the individual adaptive cycles. Thus, structures and processes on subordinate scales (e.g. individual trees on the landscape that survive a forest fire) contribute to the resilience of the system in the reorganization phase by acting as a systemic memory and promoting the reorganization towards the previous structures and processes (e.g. by seed dispersal). At the same time, if critical thresholds are exceeded, disturbance processes can spread to higher levels in the release phase and thus lead to a regime shift. For example, large-scale bark beetle disturbances in forests result from individual infestation spots, that is, from small groups of infested trees (Peters et al. 2004; see Chap. 12). These feedbacks and interactions across scales are an important mechanism of system resilience and will therefore be discussed in more detail in Sect. 5.2.
The adaptive cycle forms the basis of panarchy in dynamic systems. (Holling and Gunderson 2002, redrawn)
Panarchy – A hierarchical arrangement of connected adaptive cycles at different spatial and temporal scales. (Modified from Allen et al. 2014)
2 Mechanisms of Resilience
2.1 Feedbacks and Interactions Across Scales
Ecological resilience results from the interactions of different organizational levels. These include the direct stress response of individual organisms within short periods of time as well as acclimatization processes and adaptations at the population level throughout evolutionary periods. Feedbacks and interactions across temporal and spatial scales can contribute to resilience as well as lead to regime change (Jentsch et al. 2002). Because of the important role of different levels in the context of resilience, it is not sufficient to consider a system at a single scale to describe its resilience. At least one level above and one level below the focal level of analysis should be considered (Walker et al. 2004). Processes at higher levels often have a preserving effect on ecosystems (Meyn et al. 2007), that is, they make an important contribution to their resilience. In forests, intensive disturbances at the stand level (1–10 ha) can lead to a massive loss of live plant biomass. However, at the landscape level (1000–100,000 ha), single individuals or individual stands usually survive even extreme events (Romme et al. 2011). As a consequence, these individuals or stands contribute to the recolonization and revegetation of the system after a disturbance via seed dispersal. Since the immediate vicinity of a disturbed area is often similar in its composition to the disturbed area before the disturbance event (e.g. see Palmer 2005), this feedback represents a systemic memory (Franklin et al. 2000) and gives the system ecological resilience. Many disturbances first develop locally before they spread to populations and entire landscapes. Examples are fires (which often develop from individual lightning strikes) and plant diseases. If individual thresholds are exceeded, the propagation rate of the disturbance changes non-linearly and amplification occurs at higher levels (Peters et al. 2004). Fires above a certain size are self-reinforcing by influencing their surrounding climate (e.g. increased wind development, pyrocumulus clouds) and drying out the combustible material on the ground via the heat that precedes the fire front. Tree mortality caused by bark beetle infestation increases disproportionately after a local threshold population is reached as the defence mechanisms of trees, as well as the populations of antagonists, are overrun by the exponentially increasing beetle population (Raffa et al. 2008; see Chap. 12). Spatial connectivity in the landscape (e.g. between habitat for bark beetles or combustible material for fire) plays an important role in reaching critical thresholds (Meyn et al. 2007) and can contribute to a positive amplification across scales (Seidl et al. 2016a).
2.2 Legacies of the Pre-disturbance State
In most cases, disturbances do not result in the complete destruction of all organisms inhabiting an area, but organic remains (biological legacies) of the ecosystem before the disturbance and undisturbed islands of intact vegetation persist in a matrix of disturbed areas (Franklin et al. 2000; White and Jentsch 2001). Organic remains after a disturbance include, for example, surviving organisms, seeds surviving in the soil (seed banks), microorganisms, fungi and insects, organic material such as humus and deadwood, and structural remains such as tree stumps or freshly exposed mineral soil. The amount and distribution of biological legacies significantly influence the type and speed of recovery from a disturbance, and thus the resilience of the ecosystem. Examples of such legacies are surviving old trees after a forest fire and unmowed parts of a meadow. Even a small percentage of surviving individuals or stands can make a significant contribution to the resilience of the composition, structure, and functioning of a landscape. For example, in forests of the temperate zone, the survival of trees on only 12% of the landscape area increases the carbon storage by 33.8% in the first 100 years after a disturbance (compared to forests with total loss of live trees). At the same time, these legacies of the system before disturbance triple the recolonization with late-successional species in the first century after disturbance (Seidl et al. 2014). Live tree legacies, therefore, contribute significantly to ecological resilience. Further forms of legacy are seed banks and the ability of certain species to resprout. Plants that can sprout from dormant buds after the death of aboveground plant parts often recover quickly after disturbance, as water and nutrients can be utilized by the established root network. Such plants oftentimes even benefit from disturbances as their competitors have been eliminated by the disturbance and more resources are available for their development (Buhk et al. 2007). In the Central Alps, aspen (Populus tremula L.) and downy oak (Quercus pubescens Willd.) are both able to resprout after fires, and thus benefit from fires in comparison with Scots pine (Pinus sylvestris L.), which is an obligate seeder (Wohlgemuth et al. 2018). Seeds can survive disturbances both in the soil (soil seed bank) and in tree crowns (canopy seed bank). The latter is called serotiny: seeds survive in closed, resinous cones in the canopy, which only open after the great heat produced by fire. This characteristic is an evolutionary adaptation to disturbances (see Chap. 6). For example, serotiny is shown by several pine species (Pinus halepensis Mill., P. pinaster Ait.) in the Mediterranean region and by black spruce (Picea mariana (Mill.) Britt.) in Alaska.
Frequently, not all developmental stages of plants are affected equally by disturbance, which means that less susceptible stages can serve as a system legacy and accelerate system recovery. Shade-tolerant tree species can, for example, regenerate under closed canopies even at low light levels and remain in a ‘waiting position’ for several decades until resources become available. This advanced regeneration is not affected by disturbances such as bark beetles or wind because the cambial layer of the young trees is still too thin to serve as breeding material for bark beetles and the susceptibility to wind is small owing to the high elasticity of young plants and low leverage because of low tree height. Consequently, advanced regeneration can play an important role in the recovery of forests following these disturbances. In the Bohemian Forest, for example, where mortality from bark beetle outbreaks in the last 25 years was up to 99% of canopy trees, this advanced regeneration led to a rapid recolonization (76% coverage 10 years after disturbance) by Norway spruce (Picea abies L.), which was already dominant before the disturbance (Zeppenfeld et al. 2015). The system thus proved to be very resilient to disturbance, which was largely due to the already existing regeneration under the canopy (Fig. 5.5).
2.3 Ecological Stress Memory and Acclimatization
Repeated disturbances can push ecosystems beyond a threshold so that a previous dynamic equilibrium can no longer be achieved. The consequences can be substantial changes in species composition and ecosystem services (see Chap. 18). However, some plant species have an individual ‘ecological stress memory’ and consequently can react relatively quickly and efficiently to disturbances (Walter et al. 2011). Such a memory could be the mechanism by which ecological communities can remain stable even under extreme climatic conditions. Ecological stress memory is defined as a plant reaction which, after being exposed to stress, buffers the plant against the influence of future stress. This mechanism of individual resilience is observed, for example, as a consequence of drought, frost, or heat stress (see Chap. 6).
Ecological stress memory occurs either in the form of acclimatization processes or as damage as a result of stress. Acclimatization occurs, for example, when trees that are repeatedly exposed to dry periods invest a larger proportion of their assimilates into root growth and are therefore better adapted to water limitation (Kozlowski and Pallardy 2002). However, if the frequency of disturbances is increased, plants may not be able to recover sufficiently between disturbance events. Because of the reduced assimilation capacity in periods of drought, trees are often forced to reduce their leaf area, which leads to lower radiation absorption and thus reduced photosynthesis in the following years (e.g. Allen et al. 2015; Johnstone et al. 2016). Whether trees acclimatize to stress or not also affects their reaction to new disturbance events: either disturbances can reduce resilience through accumulation of stress, resulting in an increased sensitivity to subsequent disturbance events (Scheffer et al. 2001), or acclimatization may persist after stress, resulting in reduced sensitivity or faster recovery to new stress events (Walter et al. 2011). Delayed or belated stress effects, which only become visible after a considerable time, can have negative effects on the resilience of plants to further disturbances. A delayed reaction to past extreme events such as summer drought can be observed, for example, in trees, with increased mortality occuring several years after drought (Bigler et al. 2007). Late ground frosts can trigger increased mortality in dwarf shrubs in the vegetation periods following the frost (Kreyling et al. 2010). Such delayed responses to disturbances indicate significant carry-over effects in plant fitness; the responses may be barely noticeable immediately after a disturbance but explain part of the reduced resilience after repeated disturbances (Buma 2015).
2.4 Response Diversity and Trade-Offs Within Plant Communities
Many plant communities consist of species with complementary traits in terms of competitive strength and tolerance to disturbance (White and Jentsch 2001). The phenomenon of community resilience often arises from this diversity in traits. Often, species of a community complement each other with respect to their tolerances to disturbance (e.g. drought, plant diseases, and herbivory), resulting in systems that are resilient to different disturbances. In addition, a trade-off between disturbance tolerance and competitive capacity has often developed over the course of evolution: competitive species often show little resistance to disturbance, while less competitive species can benefit from disturbance and the resulting removal of superior competitors (dominance reduction; Wohlgemuth et al. 2002) and release of resources (Davis et al. 2000; White and Jentsch 2001). As discussed above, the resilience of forests is determined, for example, by characteristics such as seed dispersal, serotiny, the ability to resprout, or shade tolerance, which vary between species. In many cases, these properties are negatively correlated: seed dispersal over long distances is a typical property of early-successional species in forests, which are, however, very light-demanding species. Shade-tolerant climax species, on the other hand, often have heavy seeds and thus disperse only over short distances. Therefore, a key to the resilience of ecosystems is their diversity, that is, the coexistence of species with different characteristics, niches, and life history strategies. While disturbances influence diversity (see Chap. 4), the opposite is also true: diversity determines the resilience of ecosystems to disturbance.
A key element for resilience is response diversity, that is, the variability in the responses of species to fluctuations and disturbances (Mori et al. 2013). A landscape consisting of species with similar niches and characteristics has a lower response diversity than a landscape in which species with very different niches and strategies occur (e.g. a mixture of pioneer species and late-successional species). Silva Pedro et al. (2015), for example, showed that carbon uptake and storage in species-rich forest landscapes (European beech forests in Hainich National Park, Germany) are significantly more resilient to disturbances than species-poor systems. Further investigations of the same forest ecosystem showed that mixtures of species with different characteristics and strategies (i.e. early-successional, intermediate, and late-successional species) also show higher productivity and thus recover faster from disturbances (Silva Pedro et al. 2016). Beta diversity (i.e. mixtures of species between stands) is at least as effective as alpha diversity (i.e. mixtures within a stand) for buffering disturbance impacts (Sebald et al. 2021).
In grasslands, the positive impact of high biodiversity on productivity and stability is attributed to different mechanisms (see Chap. 15), including: (1) the degree of asynchronous behaviour of species in a plant community in the face of disturbances, (2) insurance effects through complementary plant strategies from fast-growing to stress-tolerant species, (3) overcompensation of individual species during disturbances by reducing competitive pressure, and (4) the likelihood that a particularly productive species will occur when a high number of species are present (Yachi and Loreau 1999; Lehman and Tilman 2000; Loreau and de Mazancourt 2008; Hautier et al. 2014).
We note that the mechanisms of resilience in ecosystems are still far from being fully understood and are a current topic of research. Therefore, the processes listed here should be seen as examples rather than as a comprehensive list.
3 Measuring and Describing Resilience
Despite the growing theoretical understanding of resilience and the steadily increasing scientific literature on resilience in different systems, practical applications of the concept (e.g. in ecosystem management) are still rare. One of the reasons for the slow transdisciplinary spread of the concept is the difficulty in measuring and describing resilience and the multi-scaled nature of the concept, as described above (Seidl 2014). Therefore, many studies are currently dealing with the quantification of resilience (Isbell et al. 2015; Kreyling et al. 2017; Ingrisch and Bahn 2018). In this section, we address some important aspects in this regard.
3.1 Recovery After Disturbances
The recovery after a disturbance is an important indicator of the resilience of a system. In the ‘ball and cup’ model, the faster the ball is at the bottom of the cup, the faster the system approaches the centre of the attractor, and the higher is its resilience (Fig. 5.1). This property is probably the most directly measurable indicator of resilience. Frequently performed productivity measurements can be used in this context to gain insights into the resilience of a system (Isbell et al. 2015). For example, the resilience of managed spruce forests (based on stand volume) decreases at the margins of the natural distribution of spruce in Central Europe, with stand age and stand structure also influencing resilience (Seidl et al. 2017). It should be noted, however, that the speed of recovery is in most cases an indicator of engineering resilience and therefore does not allow any inference about possible regime shifts.
3.2 Position of the Attractor
Another resilience indicator besides the speed of recovery is the recovery pathway of the system towards the dominant attractor (Seidl et al. 2016b). This requires a precise knowledge of the location of possible attractors. However, the estimation of attractor positions (Fig. 5.1) is complicated by the above-mentioned multitude of possible development paths of a system after disturbance (Fig. 5.3). How do we know whether an observed development pathway leads back to the former attractor of the system – indicating resilience – or leads to an alternative regime? In order to answer this question, it is necessary to quantitatively describe the position of the attractors of a system. The Historical Range of Variability (HRV) is suitable for the quantitative description of system attractors (White and Jentsch 2001). The HRV quantifies possible system states and specifies probabilities (over time) or frequencies (proportion of landscape) of system states for relevant variables (Keane et al. 2009). HRV thus quantitatively describes the past attractors of the system. A comparison of the current state or current recovery trajectory with the HRV provides information about the ecological resilience of a system. Human disturbances have, for example, led to the situation where Mediterranean ecosystems in Europe are currently characterized by maquis shrubland systems over large areas (Fig. 5.6; Tinner et al. 2009). Without human disturbances, these systems would approach their natural attractor again under the prevailing Mediterranean climate, that is, evergreen oak forests (Quercus ilex L.) would reappear (Henne et al. 2015). The HRV of a system can only be described for long periods of time because of the necessity to include the full variation inherent to the system. Since these periods of time are often not sufficiently covered by empirical data, simulation models are needed in many cases to describe the HRV quantitatively (Cyr et al. 2009; Seidl et al. 2013). However, the orientation on the HRV is increasingly criticized in the light of global change, as historical reference conditions can no longer be used as models for future conditions under changing environmental conditions (Seidl et al. 2016b; Jentsch and White 2019). It should, therefore, be noted that the attractor landscape is not static and can change over time. When estimating the resilience under future climatic conditions, it is necessary to consider not only the historical but also the future variability of the system (Duncan et al. 2010; Seidl et al. 2016b; Jentsch and White 2019). Nevertheless, analyses of the past provide an important basis for the contextualization of current disturbance events (Janda et al. 2017). Increasing availability of palaeo-data is providing better information about past climate-related changes in the attractor landscape (Tinner et al. 2009; Henne et al. 2015).
Two alternative attractors of Mediterranean ecosystems in Southern Europe. (a) Open, shrubby maquis systems, which were created and are preserved by human disturbances. (Photo: M. Schweiss, Wikimedia Commons.) (b) Forests of Mediterranean oaks (e.g. Quercus ilex L.; Photo: F. Geller-Grimm, Wikimedia Commons) as they would appear without human disturbance (Henne et al. 2015)
3.3 Early-Warning Indicators
In applying the resilience concept, the question often arises of whether a regime shift is imminent or not. Many ecosystems recover relatively quickly after disturbance (high resilience), while others recover only over long periods of time (low resilience). In the case of ecosystems with low resilience, the question of a possible regime shift is increasingly being raised. If such a shift occurs, it can often have a large and long-lasting impact on ecosystems, which makes early-warning indicators of regime shifts particularly relevant.
The temporal autocorrelation of a system offers possible insights here. A system with high resilience (i.e. a deep valley in Fig. 5.1) quickly returns to the centre of the attractor after disturbance. The temporal autocorrelation of the system after disturbance (i.e. the temporal memory of a disturbance event) is low. At low resilience or near a tipping point, there are only weak gradients in the attractor landscape – the system returns only slowly to its starting point after disturbance. This slow return results in increasing temporal autocorrelation and is called ‘critical slowing down’; it is a possible early-warning indicator of a regime shift (Scheffer et al. 2009).
The spatial structure of ecosystems can also provide information about their resilience. For example, the further a disturbance radiates into a system, or the greater its spatial effect on surrounding areas, the lower the resilience of the system (Dai et al. 2013). This phenomenon is also expressed in increasing spatial autocorrelation in systems near tipping points (Dakos et al. 2010). Furthermore, an ecosystem-related regime change can often be first announced by changes in limited areas of the system, which recover only slowly, or not at all, from disturbances. Increasing spatial variability may therefore also indicate a regime change in some systems (Kéfi et al. 2014).
References
Adger WN (2000) Social and ecological resilience: are they related? Prog Hum Geog 24:347–364
Allen CR, Angeler DG, Garmestani AS, Gunderson LH, Holling CS (2014) Panarchy: theory and application. Ecosystems 17:578–589
Allen CD, Breshears DD, McDowell NG (2015) On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 6:article 129:1–55
Biggs R, Schlüter M, Biggs D, Bohensky EL, Burnsilver S, Cundill G, Dakos V, Daw TM, Evans LS, Kotschy K, Leitch AM, Meek C, Quinlan A, Raudsepp-Hearne C, Robards MD, Schoon ML, Schultz L, West PC (2012) Toward principles for enhancing the resilience of ecosystem services. Ann Rev Env Resour 37:421–448
Bigler C, Gavin DG, Gunning C, Veblen TT (2007) Drought induces lagged tree mortality in a subalpine forest in the Rocky Mountains. Oikos 116:1983–1994
Brand FS, Jax K (2007) Focusing the meaning(s) of resilience: resilience as a descriptive concept and a boundary object. Ecol Soc 12:23–23
Buhk C, Meyn A, Jentsch A (2007) The challenge of plant regeneration after fire in the Mediterranean Basin: scientific gaps in our knowledge on plant strategies and evolution of traits. Plant Ecol 192:1–19
Buma B (2015) Disturbance interactions: characterization, prediction, and the potential for cascading effects. Ecosphere 6:article 70
Carpenter S, Walker B, Anderies JM, Abel N (2001) From metaphor to measurement: resilience of what to what? Ecosystems 4:765–781
Cyr D, Gauthier S, Bergeron Y, Carcaillet C (2009) Forest management is driving the eastern north American boreal forest outside its natural range of variability. Front Ecol Environ 7:519–524
Dai L, Korolev KS, Gore J (2013) Slower recovery in space before collapse of connected populations. Nature 496:355–358
Dakos V, Nes EH, Donangelo R, Fort H, Scheffer M (2010) Spatial correlation as leading indicator of catastrophic shifts. Theor Ecol 3:163–174
Davis MA, Grime JP, Thompson K (2000) Fluctuating resources in plant communities: a general theory of invasibility. J Ecol 88:528–534
Duncan SL, McComb BC, Johnson KN (2010) Integrating ecological and social ranges of variability in conservation of biodiversity: past, present, and future. Ecol Soc 15:Article 5
Folke C (2006) Resilience: the emergence of a perspective for social-ecological systems analyses. Global Environ Chang 16:253–267
Folke C, Carpenter S, Elmqvist T, Gunderson L, Holling CS, Walker B (2002) Resilience and sustainable development: building adaptive capacity in a world of transformations. Ambio 31:437–440
Folke C, Carpenter S, Walker B, Scheffer M, Elmqvist T, Gunderson L, Holling CS (2004) Regime shifts, resilience, and biodiversity in ecosystem management. Annu Rev Ecol Evol 35:557–581
Franklin JF, Lindenmayer D, MacMahon JA, McKee A, Magnuson J, Perry DA, Waide R, Foster D (2000) Threads of continuity. Cons Pract 1:8–17
Gunderson LH (2000) Ecological resilience – in theory and application. Ann Rev Ecol Syst 69:473–439
Hautier Y, Seabloom EW, Borer ET, Adler PB, Harpole WS, Hillebrand H, Lind EM, MacDougall AS, Stevens CJ, Bakker JD, Buckley YM, Chu C-J, Collins SL, Daleo P, Damschen EI, Davies KF, Fay PA, Firn J, Gruner DS, Jin VL, Klein JA, Knops JMH, La Pierre KJ, Li W, McCulley RL, Melbourne BA, Moore JL, O’Halloran LR, Prober SM, Risch AC, Sankaran M, Schuetz M, Hector A (2014) Eutrophication weakens stabilizing effects of diversity in natural grasslands. Nature 508:521–525
Henne PD, Elkin C, Franke J, Colombaroli D, Calò C, La Mantia T, Pasta S, Conedera M, Dermody O, Tinner W (2015) Reviving extinct Mediterranean forests increases ecosystem potential in a warmer future. Front Ecol Environ 13:356–362
Hirota M, Holmgren M, Van Nes EH, Scheffer M (2011) Global resilience of tropical forest and savanna to critical transitions. Science 334:232–235
Holling CS (1973) Resilience and stability of ecological systems. Ann Rev Ecol Syst 4:1–23
Holling CS (1996) Engineering resilience versus ecological resilience. In: Schulze PC (ed) Engineering within ecological constraints. National Academy Press, Washington, DC, pp 31–44
Holling CS, Gunderson LH (2002) Resilience and adaptive cycles. Island Press, Washington, DC, pp 25–62
Ingrisch J, Bahn M (2018) Towards a comparable quantification of resilience. Trends Ecol Evol 33:251–259
Isbell F, Craven D, Connolly J, Loreau M, Schmid B, Beierkuhnlein C, Bezemer TM, Bonin C, Bruelheide H, De Luca E, Ebeling A, Griffin JN, Guo Q, Hautier Y, Hector A, Jentsch A, Kreyling J, Lanta V, Manning P, Meyer ST, Mori AS, Naeem S, Niklaus PA, Polley HW, Reich PB, Roscher C, Seabloom EW, Smith MD, Thakur MP, Tilman D, Tracy BF, van der Putten WH, van Ruijven J, Weigelt A, Weisser WW, Wilsey B, Eisenhauer N (2015) Biodiversity increases the resistance of ecosystem productivity to climate extremes. Nature 526:574–577
Janda P, Trotsiuk V, Mikoláš M, Bače R, Nagel TA, Seidl R, Seedre M, Morrissey RC, Kucbel S, Jaloviar P, Jasík M, Vysoký J, Šamonil P, Čada V, Mrhalová H, Lábusová J, Nováková MH, Rydval M, Matějů L, Svoboda M (2017) The historical disturbance regime of mountain Norway spruce forests in the Western Carpathians and its influence on current forest structure and composition. For Ecol Manag 388:67–78
Jentsch A, White PS (2019) A theory of pulse dynamics and disturbance in ecology. Ecology 100:e02734
Jentsch A, Beierkuhnlein C, White PS (2002) Scale, the dynamic stability of forest eco-systems, and the persistence of biodiversity. Silva Fenn 36:393–400
Johnstone JF, Allen CD, Franklin JF, Frelich LE, Harvey BJ, Higuera PE, Mack MC, Meentemeyer RK, Metz MR, Perry GLW, Schoennagel T, Turner MG (2016) Changing disturbance regimes, ecological memory, and forest resilience. Front Ecol Environ 14:369–378
Keane RE, Hessburg PF, Landres PB, Swanson FJ (2009) The use of historical range and variability (HRV) in landscape management. For Ecol Manag 258:1025–1037
Kéfi S, Guttal V, Brock WA, Carpenter SR, Ellison AM, Livina VN, Seekell DA, Scheffer M, van Nes EH, Dakos V (2014) Early warning signals of ecological transitions: methods for spatial patterns. PLoS One 9:10–13
Kozlowski TT, Pallardy SG (2002) Acclimation and adaptive responses of woody plants to environmental stresses. Bot Rev 68:270–334
Kreyling J, Beierkuhnlein C, Jentsch A (2010) Effects of soil freeze-thaw cycles differ between experimental plant communities. Basic Appl Ecol 11:65–75
Kreyling J, Dengler J, Walter J, Velev N, Ugurlu E, Sopotlieva D, Ransijn J, Picon-Cochard C, Nijs I, Hernandez P, Güler B, von Gillhaussen P, De Boeck HJ, Bloor JMG, Berwaers S, Beierkuhnlein C, Arfin Khan MAS, Apostolova I, Altan Y, Zeiter M, Wellstein C, Sternberg M, Stampfli A, Campetella G, Bartha S, Bahn M, Jentsch A (2017) Species richness effects on grassland recovery from drought depend on community productivity in a multisite experiment. Ecol Lett 20:1405–1413
Lehman CL, Tilman D (2000) Biodiversity, stability, and productivity in competitive communities. Am Nat 156:534–552
Lloret F, Keeling EG, Sala A (2011) Components of tree resilience: effects of successive low-growth episodes in old ponderosa pine forests. Oikos 120:1909–1920
Loreau M, De Mazancourt C (2008) Species synchrony and its drivers: neutral and nonneutral community dynamics in fluctuating environments. Am Nat 172:E48–E66
Meigs GW, Morrissey RC, Bače R, Chaskovskyy O, Čada V, Després T, Donato DC, Janda P, Lábusová J, Seedre M, Mikoláš M, Nagel TA, Schurman JS, Synek M, Teodosiu M, Trotsiuk V, Vítková L, Svoboda M (2017) More ways than one: mixed-severity disturbance regimes foster structural complexity via multiple developmental pathways. For Ecol Manag 406:410–426
Meyn A, White PS, Buhk C, Jentsch A (2007) Environmental drivers of large, infrequent wildfires: the emerging conceptual model. Prog Phys Geogr 31:287–312
Mori AS, Furukawa T, Sasaki T (2013) Response diversity determines the resilience of ecosystems to environmental change. Biol Rev 88:349–364
Nikinmaa L, Lindner M, Cantarello E, Jump AS, Seidl R, Winkel G, Muys B (2020) Reviewing the use of resilience concepts in forest sciences. Curr For Rep 6:61–80
Palmer MW (2005) Distance decay in an old-growth neotropical forest. J Veg Sci 16:161–166
Peters DPC, Pielke RA, Bestelmeyer BT, Allen CD, Munson-McGee S, Havstad KM (2004) Cross-scale interactions, nonlinearities, and forecasting catastrophic events. Proc Natl Acad Sci USA 101:15120–15125
Raffa KF, Aukema BH, Bentz BJ, Carroll AL, Hicke JA, Turner MG, Romme WH (2008) Cross-scale drivers of natural disturbances prone to anthropogenic amplification: the dynamics of bark beetle eruptions. Bioscience 58:501–517
Rockström J, Steffen W, Noone K, Persson A, Chapin FS, Lambin EF, Lenton TM, Scheffer M, Folke C, Schellnhuber HJ, Nykvist B, de Wit CA, Hughes T, van der Leeuw S, Rodhe H, Sörlin S, Snyder PK, Costanza R, Svedin U, Falkenmark M, Karlberg L, Corell RW, Fabry VJ, Hansen J, Walker B, Liverman D, Richardson K, Crutzen P, Foley JA (2009) A safe operating space for humanity. Nature 461:472–475
Romme WH, Boyce MS, Gresswell R, Merrill EH, Minshall GW, Whitlock C, Turner MG (2011) Twenty years after the 1988 Yellowstone fires: lessons about disturbance and ecosystems. Ecosystems 14:1196–1215
Scheffer M, Carpenter SR (2003) Catastrophic regime shifts in ecosystems: linking theory to observation. Trends Ecol Evol 18:648–656
Scheffer M, Carpenter S, Foley JA, Folke C, Walker B (2001) Catastrophic shifts in ecosystems. Nature 413:591–596
Scheffer M, Bascompte J, Brock WA, Brovkin V, Carpenter SR, Dakos V, Held H, van Nes EH, Rietkerk M, Sugihara G (2009) Early-warning signals for critical transitions. Nature 461:53–59
Scheffer M, Hirota M, Holmgren M, Van Nes EH, Chapin FS (2012) Thresholds for boreal biome transitions. Proc Natl Acad Sci USA 109:21384–21389
Sebald J, Thrippleton T, Rammer W, Bugmann H, Seidl R (2021) Mixing tree species at different spatial scales: the effect of alpha, beta and gamma diversity on disturbance impacts under climate change. J Appl Ecol 8:1749–1763
Seidl R (2014) The shape of ecosystem management to come: anticipating risks and fostering resilience. Bioscience 64:1159–1169
Seidl R, Eastaugh CS, Kramer K, Maroschek M, Reyer C, Socha J, Vacchiano G, Zlatanov T, Hasenauer H (2013) Scaling issues in forest ecosystem management and how to address them with models. Eur J For Res 132:653–666
Seidl R, Rammer W, Spies TA (2014) Disturbance legacies increase the resilience of forest ecosystem structure, composition, and functioning. Ecol Appl 24:2063–2077
Seidl R, Müller J, Hothorn T, Bässler C, Heurich M, Kautz M (2016a) Small beetle, large-scale drivers: how regional and landscape factors affect outbreaks of the European spruce bark beetle. J Appl Ecol 53:530–540
Seidl R, Spies TA, Peterson DL, Stephens SL, Hicke JA (2016b) Searching for resilience: addressing the impacts of changing disturbance regimes on forest ecosystem services. J Appl Ecol 53:120–129
Seidl R, Vigl F, Rössler G, Neumann M, Rammer W (2017) Assessing the resilience of Norway spruce forests through a model-based reanalysis of thinning trials. For Ecol Manag 388:3–12
Silva Pedro M, Rammer W, Seidl R (2015) Tree species diversity mitigates disturbance impacts on the forest carbon cycle. Oecologia 177:619–630
Silva Pedro M, Rammer W, Seidl R (2016) A disturbance-induced increase in tree species diversity facilitates forest productivity. Landsc Ecol 31:989–1004
Tinner W, van Leeuwen JFN, Colombaroli D, Vescovi E, van der Knaap WO, Henne PD, Pasta S, D’Angelo S, La Mantia T (2009) Holocene environmental and climatic changes at Gorgo Basso, a coastal lake in southern Sicily. Italy Quaternary Sci Rev 28:1498–1510
Turner MG, Romme WH, Gardner RH, Neill RVO, Kratz TK, O’Neill RV (1993) A revised concept of landscape equilibrium: disturbance and stability on scaled landscapes. Landsc Ecol 8:213–227
Walker B, Holling CS, Carpenter SR, Kinzig A (2004) Resilience, adaptability and transformability in social-ecological systems. Ecol Soc 9:article 5
Walter J, Nagy L, Hein R, Rascher U, Beierkuhnlein C, Willner E, Jentsch A (2011) Do plants remember drought? Hints towards a drought-memory in grasses. Environ Exp Bot 71:34–40
White PS, Jentsch A (2001) The search for generality in studies of disturbance and ecosystem dynamics. Prog Bot 62:399–449
Wohlgemuth T, Bürgi M, Scheidegger C, Schütz M (2002) Dominance reduction of species through disturbance – a proposed management principle for central European forests. For Ecol Manag 166:1–15
Wohlgemuth T, Doublet V, Nussbaumer C, Feichtinger L, Rigling A (2018) Baumartenwechsel in den Walliser Waldföhrenwälder verstärkt nach grossen Störungen. Schweiz Z Forstwes 169:279–289
Yachi S, Loreau M (1999) Biodiversity and ecosystem productivity in a fluctuating environment: the insurance hypothesis. Proc Natl Acad Sci USA 96:1463–1468
Zang C, Hartl-Meier C, Dittmar C, Rothe A, Menzel A (2014) Patterns of drought tolerance in major European temperate forest trees: climatic drivers and levels of variability. Glob Change Biol 20:3767–3779
Zeppenfeld T, Svoboda M, Derose RJ, Heurich M, Müller J, Čížková P, Starý M, Bače R, Donato DC (2015) Response of mountain Picea abies forests to stand-replacing bark beetle outbreaks: neighbourhood effects lead to self-replacement. J Appl Ecol 52:1402–1411
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Seidl, R., Jentsch, A., Wohlgemuth, T. (2022). Disturbance Resilience. In: Wohlgemuth, T., Jentsch, A., Seidl, R. (eds) Disturbance Ecology. Landscape Series, vol 32. Springer, Cham. https://doi.org/10.1007/978-3-030-98756-5_5
Download citation
DOI: https://doi.org/10.1007/978-3-030-98756-5_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-98755-8
Online ISBN: 978-3-030-98756-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)