Abstract
Ultrasonography (US) allows a quantitative assessment of synovitis in patients with chronic inflammatory arthritis. US assessment of synovitis is essential for at least three reasons: (i) for diagnosing synovitis, (ii) for therapy monitoring, and (iii) for predicting relapse in patients in remission. A number of scoring systems, improving both reliability and responsiveness of US, have been proposed. In most of the published studies, grayscale and Doppler findings have been graded independently and each elementary component had its dedicated scoring system. However, there is still a lack of an expert-derived consensus, especially on the core set of joints to scan. Recently, the EULAR-OMERACT (European League Against Rheumatism-Outcome Measures in Rheumatology) group for musculoskeletal US has agreed on the use of a four-grade semiquantitative scoring system for both B-mode-detected and Doppler-mode-detected synovitis, which have demonstrated good multi-examiner intra-observer and inter-observer reliability in rheumatoid arthritis patients. The use of contrast-enhanced ultrasonography (CEUS) improves the sensitivity of Doppler US in the identification of abnormal vascularization in joint inflammation, allowing a better characterization of the pannus, in terms of differentiation between hypervascularity, hypovascularity, and avascularity.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- B-mode ultrasonography
- Doppler mode
- Scoring systems
- Therapy monitoring
- Contrast-enhanced ultrasonography
1 Introduction
In chronic inflammatory joint diseases, synovial tissue is the major site of inflammation. Latest reports indicate that ultrasonography may be an important imaging technique to determine the degree of synovitis in inflamed joints of patients with chronic inflammatory arthritis. Furthermore, the use of power Doppler technique with high-frequency probes allows for a sensitive detection of synovial proliferation with abnormal blood flow, especially in the small joints of the hands and feet.
Power Doppler technique analyzes Doppler changes induced by the moving red blood cells and is particularly helpful for the quantification of low blood flows at synovial tissue level. Contrast-enhanced ultrasound (CEUS) may be a promising tool to evaluate inflammatory arthritis, because of its ability to provide dynamic imaging, and high sensitivity for angiogenesis. Angiogenesis is emerging as a key player in the pathogenesis of many chronic inflammatory arthritis. A number of scoring systems, improving reliability and consequently the responsiveness of US in clinical trials, have been proposed. However, there is still a lack of an expert-derived consensus, especially on the core set of joints to scan. We have attempted to summarize the emerging B-mode ultrasound, color/power Doppler ultrasound, and CEUS imaging techniques and their applications in quantifying synovial inflammation.
2 Musculoskeletal Ultrasound Scoring Methods
In patients with rheumatoid arthritis, sonographic findings of synovial inflammation were found predictive for irreversible joint damage (i.e., bone erosions), and they can be significantly changed by disease-modifying antirheumatic drugs. Color and power Doppler techniques have shown to be of diagnostic value in the detection of vascularity in intra-articular synovial tissue and provide a measure of neovascularization within the synovial lining of tendons and within tendons themselves. The ultrasound quantification of synovial inflammation is essential for at least three reasons: (i) for diagnosing active synovitis, (ii) for therapy monitoring, and (iii) as a predictive factor for relapse in patients in remission.
2.1 Ultrasound Scoring Systems
Several semiquantitative scoring systems using grayscale findings and power Doppler signals have been proposed (Table 26.1). In most of the published studies, grayscale and Doppler findings have been graded independently and each elementary component had its dedicated scoring system.
Recently, the EULAR-OMERACT (European League Against Rheumatism-Outcome Measures in Rheumatology) group for musculoskeletal ultrasound has agreed on the use of a four-grade semiquantitative scoring system for both B-mode-detected and Doppler-detected synovitis, which have demonstrated good multi-examiner intra-observer and inter-observer reliability in RA patients.
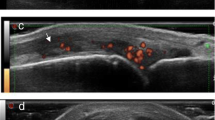
In 2003, Szkudlarek et al. developed a four-step semiquantitative US grading system for joint effusion, synovial thickening, and power Doppler signal at synovium level in five preselected small joints of patients with RA: second and third metacarpophalangeal joints, second proximal interphalangeal joint, and first and second metatarsophalangeal joints. Joint effusion was defined as a compressible anechoic intracapsular area and the amount of fluid semiquantitatively scored as follows: grade 0: no effusion; grade 1: minimal amount; grade 2: moderate amount (without distension of the joint capsule); and grade 3: extensive amount (with distension of the joint capsule). Synovial thickening was defined as a non-compressible hypoechoic intracapsular area scored as follows: grade 0: none; grade 1: minimal synovial thickening filling the angle between the periarticular bones, without bulging over the line linking tops of the bones; grade 2: synovial thickening bulging over the line linking tops of the periarticular bones without extension along the bone diaphysis; and grade 3: synovial thickening bulging over the line linking tops of the periarticular bones with extension to at least one of the bone diaphysis. Semiquantitative grading of the PD signal in the synovium was described as follows: grade 0: no flow; grade 1: single-vessel signals; grade 2: confluent vessel signals in less than half of the area of the synovium; and grade 3: vessel signals in more than half of the area of the synovium (Figs. 26.1 and 26.2).
Rheumatoid arthritis. Active synovitis detected using a longitudinal dorsal scan at metacarpophalangeal joint level. Both images (a) and (b) show representative examples of synovitis grade 3 using grayscale scoring system. Using power Doppler assessment grade 2 and grade 3 can be assigned, respectively, to (a) and (b). p proximal phalanx, m metacarpal bone
In 2000, Wakefield et al. described the first semiquantitative scoring system for the assessment of bone erosions. A bone erosion was defined as an interruption of the bony cortex with an irregular floor documented in longitudinal and transverse planes. The size of the definite bone erosion was measured using its maximal diameter and the following scoring system was proposed: small erosion: <2 mm; moderate erosion: 2–4 mm; and large erosion: >4 mm (Fig. 26.3).
In a recent study by Hurnakova J et al., cartilage damage of the metacarpal head was assessed in patients with rheumatoid arthritis and in patients with osteoarthritis using a very-high-frequency probe (up to 22 MHz) and the following five-grade semiquantitative scoring system: 0 = normal hyaline cartilage; 1 = loss of the sharpness of the cartilage superficial margin; 2 = partial-thickness defect of the cartilage layer; 3 = full-thickness defect of the cartilage layer with normal subchondral bone profile; and 4 = complete loss of the cartilage layer and subchondral bone damage (Fig. 26.4).
Rheumatoid arthritis. Metacarpal head on longitudinal dorsal scan. Representative examples of different grades of cartilage damage. (a) Normal hyaline cartilage. (b) Loss of the sharpness of the cartilage superficial margin. (c) Partial-thickness defect of the cartilage layer. (d) Full-thickness defect of the cartilage layer with normal subchondral bone profile. (e) Complete loss of the cartilage layer and subchondral bone damage. p proximal phalanx; m metacarpal bone
For tenosynovitis and tendon damage a taskforce of the OMERACT US group agreed on a four-grade semiquantitative scoring system (i.e., grade 0, normal; grade 1, minimal; grade 2, moderate; grade 3, severe). Both longitudinal and transverse planes should be used to assess both inflammatory findings and tendon ruptures.
The data acquired by all scoring systems depend on both acquisition and interpretation processes and the following practical tips are fundamental to consider, especially when a comparison between a previous or future ultrasound examination is, respectively, requested or planned.
As regards the acquisition process, it may be based on anatomic or pathologic references: in other words, the sonographer may save the images using standard scanning planes described using fixed anatomic landmarks, or according to the maximal expression of synovitis.
The images acquired using standard scans are easily compared, but they may underestimate or completely miss synovitis; conversely the method based on pathologic findings may lead to the acquisition of images with different anatomic backgrounds, but they display the outcome measure which is essential for therapy monitoring. Thus, ultrasound examination should not be limited to placing the probe in a selected number of scanning planes, but it should entail the movement of the probe from one side of the anatomic site under examination to the other side, to look for the maximal expression of synovial inflammation.
Interpretation of ultrasound findings indicative of synovitis for therapy monitoring must consider the following main issues: intra- and inter-observer reliability and sensitivity to change. In fact, interpretation based only on presence/absence is likely to provide a higher degree of intra- and inter-observer agreement, but it may miss early improvements due to treatment; conversely adopting a semiquantitative scoring system allows for a more sensitive-to-change method which distinguishes different grades of synovitis. In other words, presence/absence approach misses the benefit obtained by a treatment inducing a change from synovitis grade 3 at baseline to grade 1 at follow-up examination, because it requires the complete disappearance of the ultrasound findings indicative of synovial inflammation to record an improvement.
2.2 Examined Joints
Several core sets of joints have been proposed to assess rheumatoid arthritis activity with ultrasound; however, to date, there is no clear consensus on the optimal joint count to use in daily clinical practice. To include ultrasound in the clinical routine, it is of major importance to scan the lowest number of joints and tendons that is able to give relevant information on the inflammatory process at patient level.
Hammer et al. published a study, in which a comprehensive US score including 78 joints was compared with reduced joint counts (7-, 12-, 28-, and 44-joint scores) at different time points, during biologic agent treatment. They found high correlation between the reduced joint scores and the 78-joint score at all examination time points for power Doppler ultrasound.
In the development of scoring system, Scheel et al. examined semiquantitatively (0–3) and quantitatively synovitis in the clinically most affected metacarpophalangeal and proximal interphalangeal joints of the hands of rheumatoid patients. They found no significant differences between semiquantitative scores and quantitative measurements and concluded that the examination of a reduced number of joints is preferable, in view of the shorter examination time required.
Naredo et al. investigated the validity of reduced joint counts including large and small joints on both sides. A 12-joint score including bilateral wrist, metacarpophalangeal and proximal interphalangeal joints of the second and third fingers, and knees was used to determine effusion, synovitis, and PD activity. Such a scoring system correlated highly with a corresponding 60-joint score. In fact, 12-joint score reflected the overall joint inflammation in patients with RA and is therefore useful for monitoring treatment.
Luz et al. proposed a novel ultrasound scoring system for hand and wrist joints for evaluation of patients with early RA. Such a scoring system involved the assessment of the wrist and second and third metacarpophalangeal and proximal interphalangeal joints. The score consisted of inflammation parameters (synovial proliferation, power Doppler signal, and tenosynovitis) and joint damage parameters (bone erosion and cartilage damage). The method proved to be a useful tool for monitoring inflammation and joint damage in patients with early RA, demonstrating significant correlations with longitudinal changes in disease activity and functional status.
More recently, 705 patients with definite RA were investigated and a selection of eight joints (bilateral wrist and metacarpophalangeal joints of second, third, and fifth fingers) was found simple and efficient to detect synovitis in daily practice.
In 2009, Backhaus et al. proposed a seven-joint US composite scoring system, including wrist, metacarpophalangeal and proximal interphalangeal joint of the second and third fingers, and metatarsophalangeal joint of the second and fifth toes. The joints were examined by B-mode and power Doppler ultrasound for synovitis, tenosynovitis/paratendonitis, and erosions (Table 26.2).
B-mode ultrasound synovitis was scored semiquantitatively according to Scheel et al., while the power Doppler signal was assessed using the scoring system of Szkudlarek et al. Tenosynovitis/paratendonitis and bone erosions in B-mode ultrasound were recorded on a binary basis (presence/absence). The authors concluded that the use of this score would provide a fast overview of disease activity in daily clinical practice and would be helpful in monitoring treatment.
To date the seven-joint US composite scoring system proposed by Backhaus et al. represents the most comprehensive (not only joints are included but also tendons; not only inflammatory findings are evaluated, but also bone erosions) and validated (not only in cross-sectional studies in comparison with clinical and other imaging data, but also in longitudinal studies testing its responsiveness) approach for assessing patients with rheumatoid arthritis. This accounts for its being the most used score in rheumatological clinical practice. From a practical point of view, its main limitation lies on the fact that it is based on a fixed set of anatomic structures to scan (Table 26.2). Since the anatomic structures are frequently affected joints in rheumatoid arthritis, this score works very well when assessing a cohort of patients with rheumatoid arthritis. However, in a specific single patient it may miss the most clinically involved joints. Thus, a possible solution in daily clinical practice could be to scan the seven joints indicated by Backhaus et al. together with the most clinically inflamed joint at the time of the visit.
2.3 Color and/or Power Doppler Ultrasound Methods
Color and/or power Doppler ultrasound techniques have gained importance because of their ability to assess abnormal blood flow at synovial tissue level, a key feature of the inflammatory process in patients with chronic arthritis. Apart from the outcome measure for monitoring disease activity during treatment (Fig. 26.5), Doppler findings have been proposed as predictors for relapse in patients in clinical remission and have been found able to predict erosive progression both in patients with early RA and in patients with low disease activity or remission.
Rheumatoid arthritis. Wrist synovitis. Ultrasound changes induced by intra-articular steroid injection assessed using a dorsal longitudinal view. (a) Baseline assessment showing active wrist synovitis. (b) Two weeks after the injection the same view allows the detection of the disappearance of ultrasound signs of synovitis. (c) One month after the injection, the image shows the persistent absence of ultrasound signs of synovitis. ca capitate bone, lu lunate bone, ra radius, t common extensor tendons of the fingers
Color and/or power Doppler ultrasound techniques are operator- and machine-dependent techniques. The following practical aspects should be considered during a Doppler examination of joint and periarticular structures. First, the patient must find a comfortable position during the scanning to avoid an increase of pressure at the anatomic site under examination and consequent false-negative findings. Second, the sonographer should reduce as much as possible the compression on the tissues with the probe. Third, the Doppler parameters should be set to obtain the maximal sensitivity for the detection of synovial blood flow. Although they may vary using different ultrasound systems, the mean values of the main Doppler parameters can be the following:
-
Doppler frequency: 6 MHz for large joints, i.e., knee; 9 MHz for smaller joints, i.e., wrist; and 11 MHz for very small joints and superficial structures, i.e., distal interphalangeal joints and tendons of the fingers and toes.
-
Pulse repetition frequency (PRF): ranging from 750 Hz to 1.3 KHz.
-
Doppler gain: the highest value not generating random noise artifacts.
2.4 Doppler Quantitative Assessment of Synovial Blood Flow
Using different machines, Doppler modalities, and settings may have a considerable influence on the quantification of inflammation by ultrasound in patients with chronic arthritis, and this must be taken into account when a follow-up examination is performed. Semi- or fully automated software tools for quantitative assessment of active synovitis may help reducing the inter-observer variability. Quantitative Doppler scoring systems include the count of color pixels or the color pixel intensity, in a region of interest (ROI), outlined by the sonographer, in synovial tissue, inside the joint capsule, using dedicated post-processing software and spectral Doppler analysis with measurement of the resistive index (RI) (Fig. 26.6).
Spectral Doppler analysis can be used to obtain a quantitative estimation of the grade of the synovial perfusion. The normal flow at the level of soft tissues is characterized by high resistance (i.e., high values of RI), because the diastolic velocity has been considered to be zero. Conversely, the inflammatory process is characterized by an increased perfusion and permeability of vessels together with neovascularization, and consequently an increase of the diastolic velocity and low values of RI indicate an inflammation process.
Thus, RI value allows quantitative measurements of the synovial blood flow providing an estimation of the synovial activity in patients with chronic arthritis: the lower the value of RI, the higher the grade of synovitis.
3 Role of CEUS in the Assessment of Inflammatory Arthritis
Contrast-enhanced US (CEUS) consists of a suspension of stabilized gas-filled spheres, which once introduced into the bloodstream, intravenously, generate high-intensity signals which can be detected by the transducer. These tiny microbubbles are smaller than red blood cells, which thereby can be introduced into the vascular system, and keep stable during the whole process of examination. CEUS microbubbles are “blood pool” contrast agents because once injected inside the vessels, they remain inside the lumen and do not diffuse in the extra-arterial tissue or in the cells, showing the exact vascular patterns and neoangiogenesis in the inflamed synovium. This is important because angiogenesis is reported to be the earliest sign of rheumatoid arthritis and other inflammatory rheumatic diseases.
The more recent type of US contrast agents consists of stabilized microbubbles of a sulfur hexafluoride gas (SonoVue®, Bracco, Milan, Italy). The use of CEUS improves the sensitivity of CDUS and PDUS in the identification of abnormal vascularization in joint inflammation, allowing a more exact measurement of the synovitis and a better characterization of the pannus, in terms of differentiation between hypervascularity, hypovascularity, and avascularity. This method has been shown to correlate with the histopathological quantitative and morphologic estimation of microvascular proliferation in synovial tissue.
3.1 Quantitative Analysis of CEUS
After an intravenous injection of the contrast microbubbles, a time-intensity curve (TIC) in the region of interest (ROI) is displayed by the software, showing an S-shaped wash-in and a nearly exponential washout (Fig. 26.7). The enhancement curves can be compared to those built with gadolinium DTPA in magnetic resonance studies. After the bolus injection, the slope of the ascending and descending curve, the time to peak, the maximum intensity, and the area under the TIC curve are the most common parameters calculated by the software, representative of the perfusion kinetics, and therefore they allow a detailed evaluation and quantification of synovial inflammation. Another approach of quantitative CEUS analysis of the vascular perfusion of synovium is pixel-based level and in a study a linear relationship was discovered between the parameters of quantitative CEUS and the frequencies of some interleukins in patients with psoriatic arthritis.
3.2 Applications of Quantitative Analysis of CEUS in Inflammatory Arthritis
Many studies have demonstrated that CEUS may be an excellent tool in the early diagnosis in inflammatory diseases, such as rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis, as well as in degenerative disorders. Most of these studies have been performed in patients with rheumatoid arthritis. The capacity of CEUS compared to that of B-mode and power Doppler ultrasound for detection of vascularity in joints of rheumatoid patients was evaluated by the International Arthritis Contrast Ultrasound (IACUS) study group in a multicenter trial of five European centers. CEUS has been found more sensitive than color and power Doppler ultrasound in the differentiation between active and inactive synovitis. CEUS significantly improves the detection of synovial vascularization at the knee in rheumatoid patients and the area under the curve correlates with the clinical and laboratory findings of disease activity and with the degree of knee inflammation, being significantly higher in patients with clinically active synovitis compared with those with inactive synovitis. Apart from synovitis, CEUS was found to be useful in the detection of pathologic intra- and peritendinous vascularity associated with tenosynovitis; in the evaluation of vascularized erosive lesions, which are a sign of progressive active disease; and in the demonstration of vascularized synovial lining of the inflamed bursa. Similar results have been found in patients with spondylarthritis. It has been demonstrated that the perfusion kinetics of CEUS, such as refilling time, peak intensity, regional blood flow, and slope, are associated with vascular synovial pattern in patients with psoriatic arthritis. There is also evidence confirming that the hypervascularity can be shown in the inflamed sacroiliac joints with spondylarthritis.
3.3 Clinical Application of CEUS in Therapeutic Monitoring in Inflammatory Arthritis
The quantification of synovitis is a key aspect to support therapy decisions in daily practice in patients with inflammatory arthritis and imaging findings contribute to estimating synovial inflammation activity. In the EFSUMB (European Federation of Societies for Ultrasound in Medicine and Biology) guidelines, CEUS is described as a method whose findings may change as a result of adequate treatment. A number of studies confirmed that CEUS can play a role in the therapeutic monitoring in inflammatory arthritis and in the evaluation of true remission (Table 26.3). CEUS has been shown to be able to detect changes in synovial perfusion after intra-articular steroid injection in patients with RA and in patients treated with tumor necrosis factor alpha (TNFα) inhibitors. In these patients, CEUS was found to be useful in the short-term follow-up, as it seems to provide an indication of the presence or absence of residual disease activity.
3.4 Limitations
Apart from the advantages, some drawbacks must be considered when using CEUS to assess synovitis for therapy monitoring in clinical practice. In fact, CEUS imaging allows the assessment of only one joint per each dose of contrast medium administered intravenously which limits the examination to one or very few target districts. Moreover, the contrast agent administration involves an increase in the running costs, and carries a risk, albeit minimal, of side effects.
Further Readings
Albrecht K, Grob K, Lange U, Müller-Ladner U, Strunk J. Reliability of different Doppler ultrasound quantification methods and devices in the assessment of therapeutic response in arthritis. Rheumatology (Oxford). 2008;47(10):1521–6.
Ammitzbøll-Danielsen M, Janta I, Torp-Pedersen S, Naredo E, Østergaard M, Terslev L. Three-dimensional Doppler ultrasound findings in healthy wrist and finger tendon sheaths - can feeding vessels lead to misinterpretation in Doppler-detected tenosynovitis? Arthritis Res Ther. 2016;18:70.
Backhaus M, Ohrndorf S, Kellner H, Strunk J, Backhaus TM, Hartung W, Sattler H, Albrecht K, Kaufmann J, Becker K, Sörensen H, Meier L, Burmester GR, Schmidt WA. Evaluation of a novel 7-joint ultrasound score in daily rheumatologic practice: a pilot project. Arthritis Rheum. 2009;61(9):1194–201. https://doi.org/10.1002/art.24646.
Brown AK, Conaghan PG, Karim Z, Quinn MA, Ikeda K, Peterfy CG, et al. An explanation for the apparent dissociation between clinical remission and continued structural deterioration in rheumatoid arthritis. Arthritis Rheum. 2008;58:2958–67.
Brown AK, Quinn MA, Karim Z, Conaghan PG, Peterfy CG, Hensor E, Wakefield RJ, O'Connor PJ, Emery P. Presence of significant synovitis in rheumatoid arthritis patients with disease-modifying antirheumatic drug-induced clinical remission: evidence from an imaging study may explain structural progression. Arthritis Rheum. 2006;54:3761–73.
Bruyn GA, Iagnocco A, Naredo E, Balint PV, Gutierrez M, Hammer HB, Collado P, Filippou G, Schmidt WA, Jousse-Joulin S, Mandl P, Conaghan PG, Wakefield RJ, Keen HI, Terslev L, D'Agostino MA, OMERACT Ultrasound Working Group. OMERACT definitions for Ultrasonographic pathologies and elementary lesions of rheumatic disorders 15 years on. J Rheumatol. 2019;46:1388–93.
Bruyn GAW, Hanova P, Iagnocco A, et al. Ultrasound definition of tendon damage in patients with rheumatoid arthritis. Results of a OMERACT consensus-based ultrasound score focusing on the diagnostic reliability. Ann Rheum Dis. 2014;73:1929–34.
Carotti M, Galeazzi V, Catucci F, Zappia M, Arrigoni F, Barile A, Giovagnoni A. Clinical utility of eco-color-power Doppler ultrasonography and contrast enhanced magnetic resonance imaging for interpretation and quantification of joint synovitis: a review. Acta Biomed. 2018;89(1-S):48–77.
Carotti M, Salaffi F, Manganelli P, Salera D, Simonetti B, Grassi W. Power Doppler sonography in the assessment of synovial tissue of the knee joint in rheumatoid arthritis: a preliminary experience. Ann Rheum Dis. 2002;61:877–82.
Carotti M, Salaffi F, Morbiducci J, Ciapetti A, Bartolucci L, Gasparini S, et al. Colour Doppler ultrasonography evaluation of vascularization in the wrist and finger joints in rheumatoid arthritis patients and healthy subjects. Eur J Radiol. 2012;81:1834–8.
D'Agostino MA, Terslev L, Aegerter P, Backhaus M, Balint P, Bruyn GA, Filippucci E, Grassi W, Iagnocco A, Jousse-Joulin S, Kane D, Naredo E, Schmidt W, Szkudlarek M, Conaghan PG, Wakefield RJ. Scoring ultrasound synovitis in rheumatoid arthritis: a EULAR-OMERACT ultrasound taskforce-part 1: definition and development of a standardised, consensus-based scoring system. RMD Open. 2017;3(1):e000428. https://doi.org/10.1136/rmdopen-2016-000428. eCollection
Filippucci E, Cipolletta E, Mashadi Mirza R, Carotti M, Giovagnoni A, Salaffi F, Tardella M, Di Matteo A, Di Carlo M. Ultrasound imaging in rheumatoid arthritis. Radiol Med. 2019;124:1087–100.
Filippucci E, Di Geso L, Grassi W. Progress in imaging in rheumatology. Nat Rev. Rheumatol. 2014;10:628–34.
Filippucci E, Salaffi F, Carotti M, Grassi W. Doppler ultrasound imaging techniques for assessment of synovial inflammation. Rep Med Imaging. 2013;6:83–91. https://doi.org/10.2147/RMI.S32950.
Fiocco U, Cozzi L, Chieco-Bianchi F, Rigon C, Vezzù M, Favero E, et al. Vascular changes in psoriatic knee joint synovitis. J Rheumatol. 2001;28:2480–6.
Fiocco U, Stramare R, Coran A, Grisan E, Scagliori E, Caso F, Costa L, Lunardi F, Oliviero F, Bianchi FC, Scanu A, Martini V, Boso D, Beltrame V, Vezzù M, Cozzi L, Scarpa R, Sacerdoti D, Punzi L, Doria A, Calabrese F, Rubaltelli L. Vascular perfusion kinetics by contrast-enhanced ultrasound are related to synovial microvascularity in the joints of psoriatic arthritis. Clin Rheumatol. 2015;34:1903–12.
Fiocco U, Stramare R, Martini V, Coran A, Caso F, Costa L, et al. Quantitative imaging by pixel-based contrast-enhanced ultrasound reveals a linear relationship between synovial vascular perfusion and the recruitment of pathogenic IL-17A-F IL-23+ CD161 + CD4+ T helper cells in psoriatic arthritis joints. Clin Rheumatol. 2017;36:391–9.
Foltz V, Gandjbakhch F, Etchepare F, Rosenberg C, Tanguy ML, Rozenberg S, et al. Power Doppler ultrasound, but not low-field magnetic resonance imaging, predicts relapse and radiographic disease progression in rheumatoid arthritis patients with low levels of disease activity. Arthritis Rheum. 2012;64:67–76.
Freeston JE, Wakefield RJ, Conaghan PG, Hensor EM, Stewart SP, Emery P. A diagnostic algorithm for persistence of very early inflammatory arthritis: the utility of power Doppler ultrasound when added to conventional assessment tools. Ann Rheum Dis. 2010;69:417–9.
Hammer HB, Kvien TK. Comparisons of 7- to 78-joint ultrasonography scores: all different joint combinations show equal response to adalimumab treatment in patients with rheumatoid arthritis. Arthritis Res Ther. 2011;13:R78.
Hurnakova J, Filippucci E, Cipolletta E, Di Matteo A, Salaffi F, Carotti M, Draghessi A, Di Donato E, Di Carlo M, Lato V, Horvath R, Komarc M, Pavelka K, Grassi W. Prevalence and distribution of cartilage damage at the metacarpal head level in rheumatoid arthritis and osteoarthritis: an ultrasound study. Rheumatology (Oxford). 2019;58(7):1206–13. https://doi.org/10.1093/rheumatology/key443.
Kaiser MJ, Hauzeur JP, Blacher S, Foidart JM, Deprez M, Rossknecht A, Malaise MG. Contrast-enhanced coded phase-inversion harmonic sonography of knee synovitis correlates with histological vessel density: 2 automated digital quantifications. J Rheumatol. 2009;36:391–400.
Klauser A, Demharter J, De Marchi A, et al. The IACUS study group. Contrast enhanced gray-scale sonography in assessment of joint vascularity in rheumatoid arthritis: results from the IACUS study group. Euro Radial. 2005;15(12):2404–10.
Klauser A, Halpern EJ, Frauscher F, Gvozdic D, Duftner C, Springer P, Schirmer M. Inflammatory low back pain: high negative predictive value of contrast-enhanced color Doppler ultrasound in the detection of inflamed sacroiliac joints. Arthritis Rheum. 2005;53:440–4.
Löffler C, Sattler H, Uppenkamp M, Bergner R. Contrast-enhanced ultrasound in coxitis. Joint Bone Spine. 2016;83:669–74. https://doi.org/10.1016/j.jbspin.2015.10.012.
Luz KR, Pinheiro MM, Petterle GS, Dos Santos MF, Fernandes AR, Natour J, Furtado RN. A new musculoskeletal ultrasound scoring system (US10) of the hands and wrist joints for evaluation of early rheumatoid arthritis patients. Rev Bras Rheumatol Engl Ed. 2016;56(5):421–31.
Mouterde G, Aegerter P, Correas JM, Breban M, D'Agostino MA. Value of contrast-enhanced ultrasonography for the detection and quantification of enthesitis vascularization in patients with spondyloarthritis. Arthritis Care Res (Hoboken). 2014;66:131–8.
Mouterde G, Carotti M, D'Agostino MA. Contrast-enhanced ultrasound in musculoskeletal diseases. J Radiol. 2009;90(1 Pt 2):148–55.
Naredo E, D'Agostino MA, Wakefield RJ, Möller I, Balint PV, Filippucci E, Iagnocco A, Karim Z, Terslev L, Bong DA, Garrido J, Martínez-Hernández D, Bruyn GA, OMERACT Ultrasound Task Force*. Reliability of a consensus-based ultrasound score for tenosynovitis in rheumatoid arthritis. Ann Rheum Dis. 2013;72(8):1328–34. https://doi.org/10.1136/annrheumdis-2012-202092. Epub 2012 Sep 14
Naredo E, Gamero F, Bonilla G, et al. Ultrasonographic assessment of inflammatory activity in rheumatoid arthritis: comparison of extended versus reduced joint evaluation. Clin Exp Rheumatol. 2005;23:881–4.
Naredo E, Monteagudo I. Applications of Doppler techniques in rheumatology. Clin Exp Rheumatol. 2014;32(Suppl. 80):S12–9.
Ohrndorf S, Hensch A, Naumann L, Hermann KG, Scheurig-Münkler C, Meier S, Burmester GR, Backhaus M. Contrast-enhanced ultrasonography is more sensitive than grayscale and power Doppler ultrasonography compared to MRI in therapy monitoring of rheumatoid arthritis patients. Ultraschall Med. 2011;32(Suppl 2):E38–44.
Piscaglia F, Nolsøe C, Dietrich CF, Cosgrove DO, Gilja OH, Bachmann Nielsen M, Albrecht T, Barozzi L, Bertolotto M, Catalano O, Claudon M, Clevert DA, Correas JM, D'Onofrio M, Drudi FM, Eyding J, Giovannini M, Hocke M, Ignee A, Jung EM, Klauser AS, Lassau N, Leen E, Mathis G, Saftoiu A, Seidel G, Sidhu PS, ter Haar G, Timmerman D, Weskott HP. The EFSUMB guidelines and recommendations on the clinical practice of contrast enhanced ultrasound (CEUS): update 2011 on non-hepatic applications. Ultraschall Med. 2012;33(1):33–59.
Reece RJ, Canete JD, Parsons WJ, Emery P, Veale DJ. Distinct vascular patterns of early synovitis in psoriatic, reactive, and rheumatoid arthritis. Arthritis Rheum. 1999;42:1481–4.
Ribbens C, Andre B, Marcelis S, et al. Rheumatoid hand joint synovitis: gray-scale and power Doppler US quantifications following anti-tumor necrosis factor-a treatment: pilot study. Radiology. 2003;229:562–9.
Salaffi F, Carotti M, Manganelli P, Filippucci E, Giuseppetti GM, Grassi W. Contrast-enhanced power Doppler sonography of knee synovitis in rheumatoid arthritis: assessment of therapeutic response. Clin Rheumatol. 2004;23:285–90.
Salaffi F, Ciapetti A, Gasparini S, Carotti M, Filippucci E, Grassi W. A clinical prediction rule combining routine assessment and power Doppler ultrasonography for predicting progression to rheumatoid arthritis from early-onset undifferentiated arthritis. Clin Exp Rheumatol. 2010;28(5):686–94.
Sarzi-Puttini P, Filippucci E, Adami S, et al. Clinical, ultrasound, and predictability outcomes following Certolizumab Pegol treatment (with methotrexate) in patients with moderate-to-severe rheumatoid arthritis: 52-week results from the CZP-SPEED study. Adv Ther. 2018;35:1153–68.
Scheel AK, Hermann KG, Kahler E, et al. A novel ultrasonographic synovitis scoring system suitable for analyzing finger joint inflammation in rheumatoid arthritis. Arthritis Rheum. 2005;52:733–43.
Simpson E, Hock E, Stevenson M, Wong R, Dracup N, Wailoo A, Conaghan P, Estrach C, Edwards C, Wakefield R. What is the added value of ultrasound joint examination for monitoring synovitis in rheumatoid arthritis and can it be used to guide treatment decisions? A systematic review and cost-effectiveness analysis. Health Technol Assess. 2018;22:1–258.
Stramare R, Coran A, Faccinetto A, Costantini G, Bernardi L, Botsios C, Perissinotto E, Grisan E, Beltrame V, Raffeiner B. MR and CEUS monitoring of patients with severe rheumatoid arthritis treated with biological agents: a preliminary study. Radiol Med. 2014;119:422–31.
Sun X, Deng X, Geng Y, Ji L, Xie W, Zhang X, Zhang Z. A simplified and validated ultrasound scoring system to evaluate synovitis of bilateral wrists and hands in patients with rheumatoid arthritis. Clin Rheumatol. 2018;37(1):185–91.
Szkudlarek M, Court-Payen M, Jacobsen S, et al. Interobserver agreement in ultrasonography of the finger and toe joints in rheumatoid arthritis. Arthritis Rheum. 2003;48:955–62.
Taylor PC, Steuer A, Gruber J, Cosgrove DO, Blomley MJ, Marsters PA, et al. Comparison of ultrasonographic assessment of synovitis and joint vascularity with radiographic evaluation in a randomized, placebo-controlled study of infliximab therapy in early rheumatoid arthritis. Arthritis Rheum. 2004;50:1107–16.
Terslev L, Naredo E, Aegerter P, Wakefield RJ, Backhaus M, Balint P, Bruyn GAW, Iagnocco A, Jousse-Joulin S, Schmidt WA, Szkudlarek M, Conaghan PG, Filippucci E, D'Agostino MA. Scoring ultrasound synovitis in rheumatoid arthritis: a EULAR-OMERACT ultrasound taskforce-part 2: reliability and application to multiple joints of a standardised consensus-based scoring system. RMD Open. 2017;3(1):e000427. https://doi.org/10.1136/rmdopen-2016-000427. eCollection 2017
Torp-Pedersen S, et al. Power and color Doppler ultrasound settings for inflammatory flow. Impact on scoring of disease activity in patients with rheumatoid arthritis. Arthritis Rheumatol. 2015;67(2):386–95.
Torp-Pedersen ST, Terslev L. Settings and artefacts relevant in colour/power Doppler ultrasound in rheumatology. Ann Rheum Dis. 2008;67(2):143–9.
Wakefield RJ, Gibbon WW, Conaghan PG, et al. The value of sonography in the detection of bone erosions in patients with rheumatoid arthritis: a comparison with conventional radiography. Arthritis Rheum. 2000;43:2762–70.
Zhao CY, Jiang YX, Li JC, Xu ZH, Zhang Q, Su N, Yang M. Role of contrast-enhanced ultrasound in the evaluation of inflammatory arthritis. Chin Med J. 2017;130:1722–30.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Carotti, M., Filippucci, E., Salaffi, F., Martino, F. (2022). Therapy Efficacy Evaluation in Synovitis. In: Martino, F., Silvestri, E., Orlandi, D. (eds) Musculoskeletal Ultrasound in Orthopedic and Rheumatic disease in Adults. Springer, Cham. https://doi.org/10.1007/978-3-030-91202-4_26
Download citation
DOI: https://doi.org/10.1007/978-3-030-91202-4_26
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-91201-7
Online ISBN: 978-3-030-91202-4
eBook Packages: MedicineMedicine (R0)