Abstract
Tannin acyl hydrolase, commonly known as tannase, is found as an intracellular or extracellular enzyme. Filamentous fungi were reported to dominate among all tannase-producing microorganisms. Despite its long history and numerous publications, tannase is still considered one of the costly industrial enzymes. The production and applications of tannase have been extensively studied; researches related to strain isolation and improvement, process development, and applications of tannases have resulted in a great number of scientific publications. The enzyme has potential uses in the treatment of tannery or industrial effluents. This chapter presents an overview of the natural substrate of tannins, fungal production of tannins, production of tannase by fermentation, and tannase applications in food and other industrial products.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
4.1 Introduction
Tannin acyl hydrolase, commonly known as tannase, is a hydrolase enzyme that is induced in the presence of tannic acid (Belmares et al. 2004). It catalyzes the hydrolysis of ester and depside bonds in hydrolyzable tannins such as tannic acid and gallic acid esters releasing glucose and gallic acid (Sharma et al. 2000). This enzyme was accidentally discovered by Tieghem (1867) in an experiment of formation of gallic acid into an aqueous solution of tannins, where two fungal species grew later, identified as Penicillium glaucum and Aspergillus niger (Lekha and Lonsane 1997).
Tannase enzyme is known to display two different activities. The first one is an esterase activity; by which it can hydrolyze ester bonds of gallic acid esters with glucose (galloyl-glucose) or alcohols (e.g., methyl gallate). The second activity is called depsidase activity; by which it can hydrolyze depside bonds of digallic acid (Saxena and Saxena 2004). Tannase catalyzes the hydrolysis reaction of the ester bonds present in the gallotannins, complex tannins, and gallic acid esters (Vivas et al. 2004). Tannase can be obtained from plants, animals, and microbial sources. Microorganisms are considered the most important and commercial sources of tannase, that is because the produced tannases are more stable than similar ones obtained from the other sources. Moreover, microorganisms can produce tannase in high quantities. Microbial tannase is favored also because the microbes can be subjected to genetic manipulation more readily than plants and animals, resulting in an increase in tannase production (Aguilar and Gutierrez-Sanchez 2001; Purohit et al. 2006). As a result, enzymes of microbial origin are having important applications in many areas of bio-based industries (Barthomeuf et al. 1994). In microbial sources, the Aspergillus and Penicillium genus, and lactic acid bacteria mostly produce tannase (Zakipour-Molkabadi et al. 2013; Abdel-Azeem et al. 2021).
Tannase is an industrially important enzyme and has several applications in various industries such as foods, animal feeds, cosmetic, pharmaceutical, chemical, and leather industries (Aguilar et al. 2007; Jun et al. 2007; Kour et al. 2019). The major commercial applications of the tannases are elaboration of instantaneous tea or acorn liquor, hydrolyze tea cream in the processing of tea in the production of gallic acid (Lekha and Lonsane 1997), manufacturing of wine, beer, coffee, and fruit juices (Aguilar et al. 2001); cleaning up the leather industry effluents containing the pollutant tannin; and in the reduction of antinutritional effects of tannins in animal feed (Mukherjee and Banerjee 2006). Gallic acid (3,4,5-trihydroxybenzoic acid) and related compounds possess many potential therapeutic properties. The major application of gallic acid is the synthesis of a broad-spectrum antibacterial agent. It has always been a molecule of industrial importance because of its applications in different sectors from healthcare and food to dyes, inks, paints, and photography (Mukherjee and Banerjee 2003; Yadav et al. 2019). The present review shows relevant points related to the fungal tannase. The substrates for tannase production, the tannase producing fungi, production of tannase by fermentation using different methods, like submerged fermentation (SmF), solid-state fermentation (SSF) and modified solid-state fermentation (MSSF), and ways to produce it, with the goal to contribute to the knowledge for potential applications of fungal tannase in food and other industrial products.
4.2 Tannins: Natural Substrate
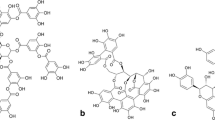
Tannins are naturally occurring polyphenolic compounds with varying molecular weights that occur naturally in the plant kingdom (Aguilar et al. 2007). After lignin, tannins are the second most abundant group of plant phenolics (Aguilar et al. 2001). These phenolic compounds of high molecular weight are usually present in plants with molar masses extending from 500 to over 3000 Da and up to 20,000 Da (Rana et al. 2004). In the plant kingdom, these tannins are found in leaves, bark, and wood. Tannins are considered to be the plants’ secondary metabolic products because they play no direct role in plant metabolism. Tannins are polymeric phenolic compounds with numerous hydroxyl groups and quite diverse in chemical structure (Hagerman 1992). It detected more than 8000 different tannins (Aguilar et al. 2007). They can be found in many different plants and plant residues such as Anacardium occidentale (cashew), Vitis vinifera (grape), Malpighia glabra (Barbados cherry), and Hancornia speciosa (mangaba fruit). One of the best sources of tannins is Acacia species, which belong to the family of Leguminosae in the plant kingdom. Such residues, rich in tannins, can be excellent substrates for the production of tannase (De Lima et al. 2014). Hydrolysis of some of the tannins yields the simple, seven-carbon gallic acid and others give ellagic acid or other phenolic acids (Okuda and Ito 2011).
On the basis of their structural characteristics, it is, therefore, possible to divide the tannins into four major groups: gallotannins, ellagitannins, complex tannins, and condensed tannins (Khanbabaee and van Ree 2001). The term “hydrolyzable tannins” includes both the gallotannins and the ellagitannins. Hydrolyzable tannins (HT) are involved in a monosaccharide core, usually, glucose, esterified with gallic acid, developing the gallotannins, or with hexahydrodiphenoic acid, the precursor of ellagic acid, and gallic acid, developing the ellagitannins (Serrano et al. 2009). Hydrolyzable tannins can be hydrolyzed into smaller compounds, for example, gallic and ellagic acid (Bele et al. 2010). Gallotannins are the simplest hydrolyzable tannins, containing polyphenolic and a polyol residue. The plants Rhus semialata (Chinese galls), Quercus infectoria (Turkish galls), Caesalpinia spinosa (Tara pods), Aver ginnala (Korean maple, Acer tannin leaves), Rhus coriaria (Sumac leaves), Hammelis virginiana (Hamammelis—hazelnuts) were documented to be rich in gallotannins (Bhat et al. 1998). Gallotannins yield gallic acid and glucose or quinic acid on hydrolysis (Khanbabaee and Van Ree 2001).
Ellagitannins yield ellagic acid, gallic acid, and glucose on hydrolysis. Plants such as Caesalpinia coriaria (Divi-Divi), Caesalpinia brevifolia (Algarobillatannin) Terminalia chebula (myrobalan seeds), Castanea sativa (Chestnut), Eucalyptous sieberiana, Schinopsis sp., Quercus velonia, Q. aegilops (Valonea tannin), Quercus coccifera (Garouille) were reported to contain ellagitannins in their bark, nuts or leaves (Bhat et al. 1998; Mukherjee and Banerjee 2003). Complex tannins are tannins in which a catechin unit is bound glycosidically to a gallotannin or an ellagitannin unit forms acyl bond with pro anthocyanadines such as catechin, for example, tea polyphenols (Khanbabaee and Van Ree 2001). Condensed tannins are referred to proanthocyanidins. These are generally found in plants that possess a woody growth and also in plant gums and exudates (Haslam 1996). Common plants containing condensed tannins are Acacia sp. (Wattle tannins), Schinopsis spp. and Loxopterygium sp. (Querbacho wood), Pinus sp. (Pine bark), Quercus sp. (Oakwood), and Aucoumea kleneana (Gaboon wood) (Bhat et al. 1998). Condensed proanthocyanidins degrade in strong acid to give corresponding anthocyanidin (Falcão and Araújo 2018).
4.3 Different Source of Tannase
Tannase is known to be a ubiquitous enzyme of the microbial world (Murugan et al. 2007). Microorganisms such as bacteria, fungi, and yeasts are known as prominent producers, also, a few animals and plants have been found as a source of tannase. The enzymes are present in tannin-rich vegetables, fruits, leaves, and bark, and also a few of them are present in fruits of Terminalia chebula, pods of Caesalpinia coriaria, and bark of Cassia fistula (Madhavakrishna et al. 1960). Although novel researches showed that the colonizing microorganisms of these animals are the actual producers, not the animals (Belur and Mugeraya 2011).
4.3.1 Plant Source
Tannase is present in tannin-plants such as Turkish gall (Quercus infectoria), sumac (Rhus coriaria), tara (Caesalpinia spinosa), chestnut (Castanea sativa, Bhat et al. 1998), gum arabic tree (Acacia nilotica, Lal et al. 2012), red gram (Cajanus cajan, Kuppusamy et al. 2015) and waste testa (Anacardium occidentales, Lenin et al. 2015), English oak (Quercus robur), Pendunculate oak (Quercus rubra), Karee tree (Rhus typhina) leaves. Plants having condensed tannins are babul (Acacia arabica), konnam (Cassia fistula), avaram (Cassia auriculata), and others. Plants synthesize gallic acid, hexahydroxyphenic acid, and chebulinic acid in addition to the amount of sugar. These acids possibly undergo esterification with glucose molecules during the ripening process with the aid of tannase ultimately resulting in the synthesis of tannins.
4.3.2 Animal Source
Microorganisms that live in the gastrointestinal of the cow, goats, sheep, and other animals are known as effective tannase producers such as many species of bacteria isolated from fecal samples of native sheep and goats, which can hydrolyze acorn tannin in the rumen and reduce negative effects of tannin on animals (Mosleh et al. 2014). Examples of Tannase-producing bacteria are Streptococcus pneumonia and Streptococcus.
4.3.3 Bacterial and Fungal Sources
Microorganisms are producing tannase enzymes in large quantities than other organisms. They are the main source of commercial enzyme production. The microbial enzymes present several advantages over other sources since the microbial enzymes are more stable in comparison to similar enzymes from other sources (Jana et al. 2014). Microorganisms can ease of genetic manipulation of microbial enzymes thus; they are easy to manipulate genetically, which increases and improves the enzymatic activity (Aguilar and Gutierrez-Sanchez 2001; Purohit et al. 2006). The suitable tannase-producing microorganisms reported are fungi and few bacteria and yeast. Fungal tannase has high activity titer in the degradation of hydrolyzable tannins. However, yeast tannases relatively disintegrate tannic acid easily and flaunt a relatively lesser affinity on the other hand in the degradation of natural tannins (Kumar et al. 2019).
4.3.3.1 Yeast Tannase
There are few reports about yeast used as tannase production. Among yeast Candida species (Aoki et al. 1976) Mycotorula japonica (Belmares et al. 2004) Pichia sp. (Deschamps et al. 1983) Debaryomyoes hansenii (Deschamps et al. 1983).
4.3.3.2 Bacteria Tannase
Lewis and Starkey (1969) reported first bacterium capable of hydrolyzing gallotannins as a sole energy source was Achomobacter sp., also, Bacillus and Lactobacillus genus represents a group of tannase producing bacteria (Mondal et al. 2001; Sabu et al. 2006; Nishitani et al. 2004). Lactic acid bacteria have an important role in tannin food degradation. Few other bacterial tannase producers comprise Klebsiella (Deschamps et al. 1983), Lonepinella (Goel et al. 2007), Citrobacter (Kumar et al. 1999), Corynebacterium, Pantonea (Pepi et al. 2010), Serratia (Belur et al. 2010), Pseudomonas (Selwal et al. 2010), Selenomonas (Skene and Brooker 1995), and Streptococcus (Jiménez et al. 2014) as shown in Table 4.1.
4.3.3.3 Fungal Tannase
Tannase is now known to be an ever-present enzyme of the microbial world and has widespread occurrence in various fungi. Most of the reported tannase-producing organisms are fungi (Bhat et al. 1998) and only a few are bacteria (Mondal and Pati 2000; Mondal et al. 2001). Fungi have the ability to degrade tannins as a sole carbon source (Aguilar and Gutierrez-Sanchez 2001) as shown in Table 4.2. The genus Aspergillus is considered as the best producer, followed by Penicillium, both standing out as great decomposers of tannins (Sabu et al. 2005). The main genera of fungi known as producers of tannase are Aspergillus, Penicillium, Fusarium, and Trichoderma (De Sena et al. 2014; Rastegari et al. 2020). Other fungi, including Chaetomium, Rhizoctonia, and Cylindrocarpon are capable of degrading tannery waste constituents (Cruz-Hernandez et al. 2005). The common genus used for tannase production either for research purposes or industrial production was Aspergillus and the common Aspergillus species used for tannase production was Aspergillus niger (Manjit et al. 2008). The tannase from Aspergillus has been used widely for the production of gallic acid from tannins (Purohit et al. 2006).
4.3.4 Rich Tannin Plants
Different studies have indicated the presence of hydrolyzable tannins in diverse plant species, especially in their leaves, fruits, bark, and branches. Endophytic microorganisms are fungi and bacteria living inside the aerial parts of the plants without, causing any seeming damage to the host (Abo Nouh 2019). According to Reges de Sena et al. (2014) isolated endophytic fungi isolated from Jamun (Syzygium cumini (L.) Skeels) leaves, and identified as Pestalotiopsis guepinii can produce tannase enzymes. Mahapatra and Banerjee (2009) reported endophytic fungi hyalopus sp. isolated from medicinal plant Ocimum sanctum presented the high production of tannase enzyme. According to a study by Cavalcanti et al. (2017), endophytic fungi from species A. niger and A. fumigatus isolated from barks of angico (Anadenanthera colubrina) and cajueiro (Anacardium occidentale) presented high production of tannase enzyme (Rana et al. 2020).
Cruz-Hernandez et al. (2005) isolated and characterized 11 fungal strains from species of (Penicillium commune, Aspergillus niger, Aspergillus rugulosa, Aspergillus terricola, Aspergillus ornatus, and Aspergillus fumigatus) isolated from soil and tannin-rich plants (damaged tissue from Quercus spp., Carya illinoensis, Larrea tridentata and Pinus sembroides) of the Mexican semidesert. These xerophilic fungi were able to produce tannase and degrade high tannin amounts in low humidity conditions. Zakipour-Molkabadi et al. (2013) reported strains of Penicillium sp. EZ-ZH190 from moldy tea leave samples can produce tannase enzymes.
4.3.5 Soil
Kasieczka-Burnecka et al. (2007) isolated an Antarctic filamentous fungus from the soil of King George Island (South Shetlands). This strain (identified as Verticillium sp.) produced two psychrophilic tannases with an optimal temperature of 20 and 25 °C, respectively. Marco et al. (2009) demonstrated that a novel extracellular tannase from the xerophilic fungus A. niger GHI was produced under solid-state conditions. Nalan and Merih (2009) isolates were selected for gallic acid production by tannase species were isolated from forest soil. Species producing tannase enzyme, for example, A. niger, P. canescens, P. frequentans, P. spinulosum, P. purpurogenum, and P. zacinthae. Rhizopus oryzae was isolated from soil samples of Indian Institute of Technology campus to show ability for tannase production (Abou-Bakr et al. 2013; Yadav 2021). Liu et al. (2016) reported amount of 35 strains of fungi species isolated from the soil of Caatinga were used for qualitative selection of strains with potential for production of tannase, the promising potential fungi Aspergillus spp. UCP1284 is able to produce tannase using cashew bagasse as a substrate. In another study by Ire and Nwanguma (2020) 15 different soil samples were collected within Lagos (Oshodi), Nigeria for the potential of tannase production by Lasiodiplodia plurivora ACN-10 in SmF and SSF using Terminalia cattapa (almond leaves) and Magnifera indica (mango leaves) as substrates.
4.3.6 Mangroves and Caves
The mangrove ecosystem is very particular in that it harbors several tannins-rich plant species, such as Rhizophora apiculata, and R. mucronata (Georgei and Ong 2013). Penicillium digitatum FETLDS1, isolated from the dumping area of tannin-rich barks of Rhizophora apiculata in mangrove areas in Perak, Malaysia can produce extracellular tannase (Sandai et al. 2012). Georgei and Ong (2013) reported Aspergillus niger FBT1, a local extracellular strain for tannase production, was isolated from soil collected from Mangrove Forest. Neethu and Pradeep (2018) reported A. niger, A. japonicus, and A. aculeatus species isolated from the soil and effluent samples were collected from different mangrove areas that have the ability to produce tannase enzymes.
Caves are an underexplored ecosystem that may reveal microorganisms of industrial and biotechnological application (Barton 2006). A study by De Melo et al. (2013) reported isolates belonging to seven different genera: Aspergillus, Penicillium, Fusarium, Rhizopus, Epicoccum, Trichoderma, and Cladosporium. Isolated from the soil of caves, they have the ability for tannase production. The most tannase-producing species are Aspergillus japonicus, A. niger, A. tamarii, A. foetidus, A. tubingensis, A. ochraceus, Penicillium funiculosum, P. oxalicum, P. corylophilum, and P. citrinum.
4.3.7 Marine Habitat
The deep-sea includes the deep-sea hypersaline anoxic basins (DHABs), which are depressions of the seafloor found at more than 2 km below sea level. Aspergillus awamori, A. candidus, A. fumigatus (Panno et al. 2013), Penicillium notatum (Gayen and Ghosh 2013), and several strains from Posidonia oceanica isolated from (DHABs). Aspergillus awamori BTMFW032, isolated from seawater, produced acidophilic tannase as an extracellular enzyme (Beena 2010; Verma et al. 2017). In a study by Panno et al. (2011), 88 fungal species are isolated from seagrass Posidonia oceanica, species identified by morphological and molecular methods, and the most important genera were Penicillium, Cladosporium, and Acremonium. Farag et al. (2018) reported Aspergillus nomius GWA5 was isolated from marine sediment to produce an active tannase for degradation of tannin.
4.3.8 Tannery or Industrial Effluents
Tannery effluent samples were used for isolating tannase-producing fungi and a promising fungus isolated from a tannery soil sample was identified as Aspergillus niger (Murugan et al. 2007). Manjit et al. (2008) reported a tannase yielding fungal culture identified as A. fumigatus MA, which was isolated from the effluent collected from a local small-scale tannery. The fungal culture produced high yields of extracellular tannase under SSF using different agroforest residues. A. tamari 1M138810 (B) a tannic acid degrading funguses was isolated from soil inundated by the effluent of a tannery at Oji River local (Enemuor and Odibo 2009). Hamada et al. (2013) reported tannase production from Aspergillus niger isolated from a tannery soil sample, with a maximum hydrolytic clear zone (53 mm ± 0). Brahmbhatt et al. (2014) reported tannase producing fungi belonging to genera Aspergillus, Mucor, Penicillium, and Rhizopus species from various tea waste dump sites, agro-residue waste sites, and tannery effluents. These soil samples and tannery effluents are rich in tannins and their derivatives (Girdhari and Peshwe 2015). Twenty-nine fungal isolates were screened on the basis of their tannase-producing efficiency under stationary conditions isolated from soil samples of various tea waste dumpsites, agro-residue waste sites, and sites near local tannery industries.
The fungal isolates Aspergillus fumigatus, Aspergillus carbonarius, Penicillium lividum, and Penicillium citrinum exhibit maximum tannase activity. Shajitha and Nisha (2018) isolated a number of mycobiota like Aspergillus niger, A. flavus, A. fumigatus, Rhizopus spp., Rhizopus stolonifer, Geotrichum spp., Penicillium sp., Trichoderma viride, from tea industry waste disposal area soil for tannase production, only four fungal strains Trichoderma viride, Aspergillus flavus, Rhizopus spp., and Aspergillus niger showed the maximum hydrolyzing zone. A significantly highest hydrolyzing zone was shown by Trichoderma viride followed by Aspergillus flavus. Farhaan and Patil (2019) reported Aspergillus spp. and Aspergillus flavus respectively as tannase producers isolated from soil samples of nearby local tannery industries. A study by Al-Mraai et al. (2019) reported fungi of genus Aspergillus are active producers of extracellular tannase, Aspergillus niger isolated from soil and tea waste-producing tannase enzyme.
4.4 Production of Tannase by Fermentation
Several fermentation systems have been developed for the production of tannase from fungi using various production media. These systems can be divided into liquid surface fermentation (LSF), submerged fermentation (SmF), solid-state fermentation (SSF), and modified solid-state fermentation (MSSF). Belmares et al. (2004) mentioned in a comprehensive review that tannase production has been carried out mainly under submerged (SmF) and solid-state fermentation (SSF) techniques, depending on the strain and culture conditions. Several studies have reported interesting advantages of tannase produced by SSF over that produced by SmF. All of these studies showed that the first advantage of SSF technique is the higher enzyme titers than in SmF, when comparing the production of the same strain and fermentation broth (Aguilar et al. 2002). Microorganisms can be utilized using SSF for conversion of plant materials and agro-industrial wastes that contain tannin-rich substrates while using SSF to produce economic products such as tannase and gallic acid production, while overcoming their accumulation problems in the environment (Paranthaman et al. 2009). Filamentous fungi are suitable for the SSF process compared to other fermentation processes (Aguilar et al. 2002). The fungal species of Aspergillus and Penicillium are the most active microorganisms known, capable of producing tannase through submerged and solid-state fermentation (Abdel-Nabey et al. 2011).
The conditions for obtaining the maximal production of the enzyme depend on two factors: the system utilized and the source of the enzyme (Rodriguez-Duran et al. 2011). Medium optimization by single dimensional search is laborious and time-consuming, especially for a large number of variables and it does not ensure desirable conditions. Plackett-Burman design is widely used in screening experiments as the number of experiments run required is very few, leading to saving of time, chemicals, and manpower (Jamal et al. 2009). The powdered plant tannin substrates (leaves, fruits, pods, bark) were usually extracted either with water or with an organic solvent like acetone (Huang et al. 2005). Tannins were documented to be serving as dual purposes as a solid support in SSF and also as a carbon source such as Larrea tridentates, Syzygium cumini, Ziziphus mauritiana (Kumar et al. 2007). Among the various species, A. niger, A. flavus, and A. oryzae were found to be the best tannase producers using tannic acid as a sole source of carbon (Hassan et al. 2018).
4.5 Tannase Applications
Tannase has received a great deal of attention from the discovery, and it is used in a wide range of applications in many industries including in detannification of food (Boadi and Neufeld 2001), preparation of food preservatives, and pharmaceuticals (Belmares et al. 2004). Actually, the main applications of tannase are elaboration of instantaneous tea (Lekha and Lonsane 1997), acorn, and gallic acid production, which is used for the synthesis of trimethoprim (Yu et al. 2004). Also, tannase is used as clarifying agent in fruit juice (Shrivastava and Kar 2009) beer, wines (Bajpai and Patil 2008), and manufacture of coffee flavored (Anwar and Imartika 2007) beverages (Aguilar et al. 2001; Belmares et al. 2004), treatment of green tea to inhibit the carcinogenic and mutagenic effects of N-nitrosamines, stabilization of malt polyphenols (Lekha and Lonsane 1997), improved color stability, and additional organoleptic properties. In animal feeding, tannase is used to reduce the antinutritional effects of tannins and improve animal digestibility.
4.5.1 Food Industries
4.5.1.1 Instantaneous Tea Elaboration
After water, tea is the second most highly consumed beverage worldwide (Venditti et al. 2010). It is an infusion obtained from leaves of Camellia sinensis and is consumed by two-thirds of the world’s population (Łuczaj and Skrzydlewska 2005). Tea drinking is associated with the reduction of serum cholesterol, prevention of low-density lipoprotein oxidation, and decreased risk of cardiovascular disease and cancer (Karak and Bhagat 2010). During the production of tea beverages, hot and clear tea infusions tend to form turbid precipitates after cooling. These precipitates, called tea creams, are formed by a complex mixture of polyphenols. Tea cream formation is a quality problem and may have antinutritional effects (Lu et al. 2009). Tannase can hydrolyze the ester bonds of catechins to release free Gallic acid and water-soluble compounds with lower molecular weight, reducing turbidity, and increasing solubility of tea beverage in cold water at 4 °C (Aguilar-Zárate et al. 2014). Precipitates are formed by the interaction of phenolic compounds and caffeine called “tea cream.” Tea cream formation is a feature that was effected on the storage quality of these products (Li et al. 2017). Thus, tannase has been widely used to hydrolyze tea cream in the processing of tea (Su et al. 2009).
Enzymatic treatment of tea beverages leads to a better color appearance, less cream formation, better taste, mouth feel, and overall acceptance (Lu et al. 2009). Also, the hydrolysis of the main tea phenols epigallocatechin gallate and epicatechin gallate to epigallocatechin and epicatechin, respectively, increases the antioxidant activity of tea beverage (Lu and Chen 2008). Tannase was documented to be used in deesterification of tea polyphenols in non-converted green tea leaves, which enhances the natural levels of gallic acid and epigallochatechin and favors the formation of epitheaflavic acids, which were responsible for bright reddish color and good cold-water solubility (Aguilar and Gutierrez-Sanchez 2001). N-nitrosodimethylamine (NDMA) inhibition by tannase-treated green tea is reported, owing to its antioxidant potential (Lu and Chen 2007). Tea infusions contain four types of catechins such as epicatechin, epigallocatechin, epicatechin gallate, and epigallocatechin gallate (Chen et al. 2014). The taste of tea gets increased with increasing concentration of catechin and the taste palatability gets reduced (Zhang et al. 2016). Tea catechins have another disadvantage in that it has poor bioavailability (Fan et al. 2016). Safety evaluation of tannase enzyme that was from A. oryzae was conducted and found to be safe for its usage in tea preparation (Kumar et al. 2019).
4.5.1.2 Beverage Clarification
New fruit juices (pomegranate, cranberry, raspberry, etc.) have recently been acclaimed for their health benefits, in particular, for their antioxidant properties. However, the presence of high tannin content in those fruits is responsible for haze and sediment formation, as well as for color, bitterness, and astringency of the juice upon storage (Aguilar et al. 2007). Tannase is utilized to reduce and remove the undesirable bitterness present in fruits juices by enzymatic treatment possessing the advantage of lowering the haze and increasing the quality of the beverages (Rout and Banerjee 2006). When the concentration of tannins in fruits increase, such as in blueberry, pomegranate, and raspberry, leads to the formation of sediment, color, and bitter taste during the storage of the beverages. In these cases, an enzymatic treatment with tannase is recommended (Aguilar et al. 2007). Rout and Banerjee (2006) documented a reduction of 25% of tannin content in pomegranate juice by using a treatment with tannase, while by using tannase and gelatin the tannin content diminishes by 49%. Hydrolysis by immobilized tannase removed up to 73.6% of the tannin present in Indian gooseberry (Phyllanthus emblica) juice. This enzymatic treatment reduced the content of tannin but increased the gallic acid concentration with a minimum reduction in vitamin C (only 2%) (Srivastava and Kar 2009, 2010).
Tannase is used as a clarifying agent in refreshing drinks with coffee flavor, and, recently, a process for the enhancement of the antioxidant properties of coffee by tannase and other enzymes has been patented (Bel-Rhlid et al. 2009). Tannase has also been utilized in grape musts and barley worts as a prefermentative treatment, coupled with conventional fining for stabilizing wine and beer (Cantarelli et al. 1989). Tannase is employed for the elaboration of acorn wine. Its use in this process favors the production of a better beverage with an alcoholic content of 10%, reducing sugars content of 7%, and a pH of 4.0. In this process, tannase produced by an Aspergillus strain helps in improving the flavor of the beverage (Aguilar and Gutierrez-Sanchez 2001). Tannase is used to prevent discoloration and haze development during beer storage. It was also used to make acorn wine from Korean acorns (Quercus spp.) and to treat grape must and juice along with lactase to remove phenolic substances for stabilizing and increasing the quality of the wine (Lekha and Lonsane 1997).
4.5.1.3 Nutritional Improvement of Legume Flours
Legumes are of major nutritional importance, especially in developing countries. Seed legumes have high protein contents, and this protein is of good biological value. However, they also have several anti-nutritional factors, affecting the digestibility of nutrients. Different thermal and biological processes have been used to reduce the anti-nutritional factors content, increasing their nutritional value. The flours obtained from the processed legumes can be used as ingredients in food preparations. Several studies mention the uses of tannase alone or in combination with other enzymes for the degradation of some antinutritional factors (tannins) present in legume flours. Duenas et al. (2007) studied the effect of the addition of tannase and other enzymes to a lentil (Lens culinaris ) flour. They found the production of several phenolic compounds after the treatment with tannase, the decrease of other phenolics such as catechin, epicatechin, and catechin 3-O-glucose, and significant increment in the antioxidant activity. On the other hand, the application of tannase on pea (Pisum sativum ) led to a decrease in all the phenolic compounds studied and a reduction in the antioxidant capacity (Duenas et al. 2009). But, in a different experiment, the hydrolysis of pea flour by tannase led to a significant improvement in the daily weight gain of rats (Urbano et al. 2007).
4.5.2 Animal Feeds Industries
It is well known that high levels of dietary tannins have negative effects on animal nutrition. Tannins form a strong complex with enzymes, minerals, and other nutrients. They are also responsible for a bitter taste, which considerably reduces the feed intake (Belmares et al. 2004). The presence of tannins in tannin-rich plants and agro-industrial waste, which are used as animal feed, make them unusable. Tannins are known as anti-nutritional factors because of their interaction with macromolecules such as proteins makes them indigestible (Frutos et al. 2004). Anti-nutritional effect of tannin could be reduced by a treatment with tannase or tannase-producing microorganisms. For example, there are some cultivars of sorghum with high content of tannins. Tannin content could be decreased by enzymatic treatment, and this material could be used as a complement in animal diet (Aguilar and Gutierrez-Sanchez 2001). Nuero and Reyes (2002) reported the production of an enzymatic extract containing tannase from mycelial wastes of penicillin manufacture. This preparation was applied to several flours used as animal feed (barley, bran, maize, oat, rye, soya, and wheat flour). The enzymatic extract from mycelia waste released similar amounts of reducing sugars from all flours when compared with a commercial enzymatic additive used in animal feeding. Tannase is applied for the treatment of tannin-rich plants in the production of animal feed. If they are first treated with tannase, tannin content is decreased and this can then be used as a complement in animal diet. Tannase utilization can be carried out in two ways: direct contact of enzymatic extracts with the material to hydrolyze the polyphenols and avoid their unpleasant polymerization, or growing tannase-producing fungal strains on tannin-rich materials, which are degraded to simpler compounds (De Sena et al. 2014).
4.5.3 Gallic Acid Production
Gallic acid (3,4,5-trihydroxybenzoic acid) is a phenolic compound and the monomeric unit of the gallotannins and complex tannins. Gallic acid and related compounds possess many potential therapeutic properties including anticancer and antimicrobial properties (Ow and Stupans 2003). One of the most important applications of tannase is gallic acid production from hydrolyzable tannins (Kar et al. 2002). Gallic acid is commercially important for its applications in several industries for the synthesis of a variety of chemicals used in the food and pharmaceutical industries. Gallic acid is utilized as a precursor for the synthesis of trimethoprim (2,4, diamino 3,4,5 trimethoxy benzyl pyrimidine) is an antibacterial drug (Sittig 1988). A broad-spectrum antioxidant and antibacterial agent, which is bacteriostatic, since it inhibits folic acid metabolism in pathogenic bacteria (Mukherjee and Banerjee 2003).
In combination with sulfonamide, trimethoprim exerts antibacterial effect at low concentrations against Streptococci and Staphylococci bacteria, Shigella sp., Corynebacterium diphtheriae, E. coli, Vibrio cholerae, Bacillus pertussis, and Clostridium welchii. Choi et al. (2010) reported the potential of gallic acid as an antiviral agent. They have also cytotoxicity against cancer cells (Beniwal et al. 2013). Curiel et al. (2010) reported a process for the enzymatic production of gallic acid. They immobilized a recombinant tannase from L. plantarum expressed then utilized the immobilized enzyme for the hydrolysis of commercial tannic acid. At least 95% of tannic acid was transformed into gallic acid, obtaining an almost pure compound.
Tannase is an enzyme characterized for catalyzing the hydrolysis of gallic acid esters. Weetall (1985) reported the enzymatic synthesis of a variety of gallic acid esters. He applied an immobilized tannase from Aspergillus niger to a solution of gallic acid in different alcohols (C1–C12) and diols (C3–C6). In the chemical industry, gallic acid is used for forming pyrogallol, and gallate esters find their applications in food industries, cosmetic industries, and so on. Pyrogallol has also been used as a photographic film developer (Banerjee et al. 2007; Yu and Li 2008; Beniwal et al. 2013). It is also used in the leather industry, in manufacturing gallic acid esters, such as propyl gallate, a potent antioxidant utilized as an antioxidant in fats and oils, in the manufacture of pyrogallol, and as a photosensitive resin in semiconductor production (Sariozlu and Kivanc 2009).
4.5.4 Bioremediation of Tannin-Contaminated Wastewaters
Tannins occur commonly in the effluents derived from several agro-industries. The treatment of this kind of wastewaters is usually difficult because tannins are highly soluble and inhibit the growth of many microorganisms (He et al. 2007). Industrial tannins when used in the tanning industry can represent a serious environmental problem on a global level, although vegetable tanning agents are natural materials, they are poorly biodegradable and act as growth inhibitors toward many microorganisms, ultimately affecting the receiving ecosystem (Prigione et al. 2018). The tannase enzyme has potential uses in the treatment of tannin-containing effluents and pre-treatment of tannin-containing animal feed (Belur and Mugeraya 2011). Fungal strains capable of performing the biotransformation of polyphenolic substances contained in tannins could have a certain environmental impact as bioremediation agents. Moreover, biotransformed tannins could have several applications in agriculture, in the animal feed and wine industries, and the tanning process, for example, improving tanning yields or leather quality (Prigione et al. 2018).
Several authors have reported the biodegradation of tannin-containing wastewaters using model systems. Kachouri et al. (2005) studied the biodegradation and decolorization of olive-mill wastewater by Aspergillus flavus . The fungi removed 58% of color and 46% of the chemical demand of oxygen of the wastewater after six days of cultivation. More recently, Murugan and Al-Sohaibani (2010) reported the use of immobilized tannase from Aspergillus candidus for the removal of tannin and the associated color from tannery effluent. Enzymatic treatment removed about 42% of the tannin content and 20% of the color of tannery wastewater. These findings suggest that tannase or tannase-producing microorganisms could be utilized for pretreatment of tannin-rich wastewaters.
4.6 Conclusion
Tannin acyl hydrolase (tannase) is one of the important hydrolytic microbial enzymes. Fungi are the most producing tannase in other microorganisms with special to genus Aspergillus, which is considered as the best producer of tannase. Microbial tannase is more stable than others sources of tannase and can be genetically modified so that the enzyme is considered industrially important and has several applications in various industries such as foods, animal feeds, cosmetics, pharmaceutical, chemical, industries, and so on. The most important application of the tannase enzyme is producing gallic acid and also degradation of industrial tannins because it causes a serious problem in the environment.
References
Abdel-Azeem AM, Yadav AN, Yadav N, Usmani Z (2021) Industrially important fungi for sustainable development, Biodiversity and ecological perspective, vol 1. Springer, Cham
Abdel-Nabey MA, Sherief AA, El-Tanash AB (2011) Tannin biodegradation and some factors affecting tannase production by two Aspergillus sp. Biotechnology 10:149–158
Abo Nouh FA (2019) Endophytic fungi for sustainable agriculture. Microb Biosyst 4:31–44
Abou-Bakr HA, El-Sahn MA, El-Banna AA (2013) Screening of tannase-producing fungi isolated from tannin-rich sources. Int J Agric Food Res 2:1–12
Aguilar CN, Gutierrez-Sanchez G (2001) Review: Sources, properties, applications and potential uses of tannin acyl hydrolase. Food Sci Technol Int 7:375–382
Aguilar CN, Augur C, Favela-Torres E, Viniegra-González G (2001) Production of tannase by Aspergillus niger Aa-20 in submerged and solid state fermentation: influence of glucose and tannic acid. J Ind Microbiol Biotechnol 26:296–302
Aguilar CN, Favela-Torres E, Viniegra-Gonzalez G, Augur C (2002) Culture conditions dictate protease and tannase production in submerged and solid-state cultures of Aspergillus niger Aa-20. Appl Biochem Biotechnol 103:407–414
Aguilar CN, Rodriguez R, Gutierrez-Sanchez G, Augur C, Favela-Torres E, Prado-Barragan LA et al (2007) Microbial tannases: advances and perspectives. Appl Microbiol Biotechnol 76:47–59
Aguilar-Zárate P, Cruz-Hernández MA, Montañez JC, Belmares-Cerda RE, Aguilar CN (2014) Bacterial tannase: production, properties and applications. Rev Mex Ing Quím 13:63–67
Al-Mraai STY, Al-Fekaiki DF, Abd Al-Manhel AJ (2019) Purification and characterization of tannase from the local isolate of Aspergillus niger. J App Biol Biotechnol 7:029–034
Anwar YAS, Imartika H (2007) The production of tannin acyl hydrolase from Aspergillus niger. J Microbiol Indonesia 1:91–94
Aoki K, Shinke R, Nishira H (1976) Purification and some properties of yeast tannase. Agric Biol Chem 40:79–85
Bajpai B, Patil S (1997) Introduction of tannin acyl hydrolase (EC 3.1.1.20) activity on some members of fungi imperfecti. Enzym Microb Technol 20:612–614
Bajpai B, Patil S (2008) A new approach to microbial production of gallic acid. Braz J Microbiol 39:708–711
Banerjee D, Mondal K, Bikas R (2001) Production and characterization of extracellular and intracellular tannase from newly isolated Aspergillus aculeatus DBF9. J Basic Microbiol 6:313–318
Banerjee R, Mukherjee G, Patra KC (2005) Microbial transformation of tannin-rich substrate to gallic acid through co-culture method. Bioresour Technol 96:949–953
Banerjee D, Mahapatra S, Pati BR (2007) Gallic acid production by submerged fermentation of Aspergillus aculeatus DBF9. Res J Microbiol 2:462–468
Barthomeuf C, Regerat F, Pourrat H (1994) Production, purification and characterization of a tannase from Aspergillus niger LCF8. J Ferment Bioeng 77:320–323
Barton HA (2006) Introduction to cave geomicrobiology: a review for the nonspecialist. J Cave Karst Stud 68:43–54
Batra A, Saxena RK (2005) Potential tannase producers from the genera Aspergillus and Penicillium. Process Biochem 40:1553–1557
Beena PS (2010) Production, purification, genetic characterization and application studies of tannase enzyme from marine fungus Aspergillus awamori. PhD Thesis, Faculty of Science Cochin University
Bele AA, Jadhav VM, Kadam VJ (2010) Potential of tannins: a review. Asian J Plant Sci 9:209–214
Belmares R, Contreras-Esquivel JC, Rodriguez-Herrera R, Coronel AR, Aguilar CN (2004) Microbial production of tannase: an enzyme with potential use in food industry. Lebensm Wiss Technol 37:857–864
Bel-Rhlid R, Kraehenbuehl K, Cavin C, Raab TW, Page N (2009) Compositions for preparing a coffee beverage comprising hydrolysed chlorogenic acid, World Intellectual Property Organization Patent Application (WO09132887)
Belur PD, Mugeraya G (2011) Microbial production of tannase. Res J Microbiol 6:25–40
Belur PD, Gopal M, Nirmala KR, Basavaraj N (2010) Production of novel cell-associated tannase from newly isolated Serratia ficaria DTC. J Microbiol Biotechnol 20:732–736
Beniwal V, Kumar A, Goel G, Chhokar V (2013) A novel low molecular weight acido-thermophilic tannase from Enterobacter cloacae MTCC 9125. Biocatal Agric Biotechnol 2:132–137
Bhat TK, Singh B, Sharma OP (1998) Microbial degradation of tannins – a current perspective. Biodegradation 9:343–357
Boadi DK, Neufeld RJ (2001) Encapsulation of tannase for the hydrolysis of tea tannins. Enzyme Microb Technol 28:590–595
Bradoo S, Gupta R, Saxena R (1996) Screening of extracellular tannase producing fungi: development of a rapid simple plate assay. J Gen Appl Microbiol 42:325–329
Bradoo S, Gupta R, Saxena RK (1997) Parametric optimization and biochemical regulation of extracellular tannase from Aspergillus japonicus. Process Biochem 32:135–139
Brahmbhatt D, Modi HA, Jain NK (2014) Preliminary isolation and screening of tannase producing bacteria and fungi. Int J Curr Microbiol App Sci 311:193–203
Cantarelli C, Brenna O, Giovanelli G, Rossi M (1989) Beverage stabilization through enzymatic removal of phenolics. Food Biotechnol 3:203–213
Cavalcanti RMF, Ornela PHDO, Jorge JA, Guimarães LHS (2017) Screening, selection and optimization of the culture conditions for tannase production by endophytic fungi isolated from Caatinga. J Appl Biol Biotechnol 5:001–009
Chaitanyakumar A, Anbalagan M (2016) Expression, purification and immobilization of tannase from Staphylococcus lugdunensis MTCC 3614. AMB Express 6:89
Chen GH, Yang CY, Lee SJ, Wu CC, Tzen JT (2014) Catechin content and the degree of its galloylation in oolong tea are inversely correlated with cultivation altitude. J Food Drug Anal 22:303–309
Choi HJ, Song JH, Bhatt LR, Baek SH (2010) Anti-human rhinovirus activity of gallic acid possessing antioxidant capacity. Phytother Res 24:1292–1296
Costa AM, Ribeiro WX, Kato E, Monteiro ARG, Peralta RM (2008) Production of tannase by Aspergillus tamarii in submerged cultures. Braz Arch Biol Technol 51:399–404
Cruz-Hernandez M, Contreras-Esquivel JC, Lara F, Rodrıguez R, Aguilar CN (2005) Isolation and evaluation of tannin-degrading fungal strains from the Mexican desert. Z Naturforsch C J Biosci 60:844–848
Curiel JA, Rodríguez H, Acebrón I, Mancheño JM, De Las RB, Rosario M (2009) Production and physicochemical properties of recombinant Lactobacillus plantarum tannase. J Agric Food Chem 57:6224–6230
Curiel JA, Betancor L, de las Rivas B, Munoz R, Guisan JM, Fernandez-Lorente G (2010) Hydrolysis of tannic acid catalyzed by immobilized-stabilized derivatives of tannase from Lactobacillus plantarum. J Agric Food Chem 58:6403–6409
De Lima JS, Cruz R, Fonseca JC, de Medeiros EV, Maciel MDHC, Moreira KA, Motta CMDS (2014) Production, characterization of tannase from Penicillium montanense URM 6286 under SSF using agroindustrial wastes, and application in the clarification of grape juice (Vitis vinifera L.). ScientificWorldJournal 2014:182025
De Melo AG, Souza PNDC, Maia NDC, Thomas AB, da Silva LBR, Batista LR et al (2013) Screening and identification of tannase-producing fungi isolated from Brazilian caves. Afr J Microbiol Res 7:483–487
De Sena AR, dos Santos ACDB, Gouveia MJ, de Mello MRF, Leite TCC, Moreira KA et al (2014) Production, characterization and application of a thermostable tannase from Pestalotiopsis guepinii URM 7114. Food Technol Biotechnol 52:459–467
Deschamps AM, Otuk G, Lebeault JM (1983) Production of tannase and degradation of chestnut tannin by bacteria. J Ferment Technol 61:55–59
Duenas M, Hernandez T, Estrella I (2007) Influence of the action of exogenous enzymes on the polyphenolic composition of pea: effect on the antioxidant activity. Eur Food Res Technol 225:493–500
Duenas M, Hernandez T, Estrella I (2009) Changes in the content of bioactive polyphenolic compounds of lentils by the action of exogenous enzymes. Effect on their antioxidant activity. Food Chem 101:90–97
Enemuor SCL, Odibo FJC (2009) Culture conditions for the production of a tannase of Aspergillus tamarii IMI388810 (B). Afr J Biotechnol 8:2554–2557
Falcão L, Araújo MEM (2018) Vegetable tannins used in the manufacture of historic leathers. Molecules 23:1–20
Fan FY, Shi M, Nie Y, Zhao Y, Ye JH, Liang YR (2016) Differential behaviors of tea catechins under thermal processing: formation of non-enzymatic oligomers. Food Chem 196:347–354
Farag AM, Hassan SW, El-Says AM, Ghanem KM (2018) Purification, characterization and application of tannase enzyme isolated from marine Aspergillus nomius GWA5. J Pure Appl Microbiol 12:1939–1949
Farhaan MM, Patil RC (2019) Screening and characterisation of tannase producing fungi from soil near tannery industry. Eur J Biotechnol Biosci 7:68–70
Farias GM, Gorbea C, Elkins JR, Griffin GJ (1994) Purification characterization and substrate relationships of the tannase from Cryphonectria parasitica. Physiol Mol Plant Pathol 44:51–63
Frutos P, Hervas G, Giráldez FJ, Mantecón AR (2004) Tannins and ruminant nutrition. Spanish J Agric Res 2:191–202
Gayen S, Ghosh U (2013) Purification and characterization of tannin acyl hydrolase produced by mixed solid-state fermentation of wheat bran and marigold flower by Penicillium notatum NCIM 923. Bio Med Res Int 2013:1–6
Georgei DS, Ong CB (2013) Improvement of tannase production under submerged fermentation by Aspergillus niger FBT1 isolated from a mangrove forest. Bio Technol 94:45–456
Girdhari SN, Peshwe SA (2015) Isolation and screening of tannase producing fungi. Int J Curr Microbiol App Sci 4:765–774
Goel G, Puniya AK, Singh K (2007) Phenotypic characterization of tannin–protein complex degrading bacteria from faeces of goat. Small Ruminant Res 69:217–220
Goel G, Kumar A, Beniwal V, Raghav M, Puniya AK, Singh K (2011) Degradation of tannic acid and purification and characterization of tannase from Enterococcus faecalis. Int Biodeterior Biodegradation 65:1061–1065
Hagerman AE (1992) Tannin-protein interactions. ACS Symp Ser 506:236–247
Hamada A, Abou-Bakr, Malak A, El-Sahn, Amr A, El-Banna (2013) Screening of tannase-producing fungi isolated from tannin-rich sources. Int J Agric Food Res 2:1–12
Haslam E (1996) Natural polyphenols (vegetable tannins) as drugs: possible modes of action. J Nat Prod 59:205–215
Hassan SWM, Ghanem KM, El-Shahat AF, El-Says AMI (2018) Production of tannase by free and immobilized marine Aspergillus nomius under optimized culture conditions. RJPBCS 9:30–44
He Q, Yao K, Sun D, Shi B (2007) Biodegradability of tannin-containing wastewater from leather industry. Biodegradation 18(4):465–472
Huang W, Ni J, Borthwick AGL (2005) Biosynthesis of valonia tannin hydrolase and hydrolysis of valonia tannin to ellagic acid by Aspergillus SHL 6. Process Biochem 40:245–1249
Ire SF, Nwanguma AC (2020) Comparative evaluation on tannase production by Lasiodiplodia plurivora ACN-10 under submerged fermentation (SmF) and solid state fermentation (SSF). Asian J Biotechnol and Bioresource Technol 6:39–49. Article no. AJB2T.55864
Iwamoto K, Tsuruta H, Nishitaini Y, Osawa R (2008) Identification and cloning of a gene encoding tannase (tannin acylhydrolase) from Lactobacillus plantarum ATCC 14917T. Syst Appl Microbiol 31:269–277
Jamal P, Tompang MF, Alam MZ (2009) Optimization of media composition for the production of bioprotein from pineapple skins by liquid-state bioconversion. J Appl Sci 9:3104–3109
Jana A, Halder SK, Banerjee A, Paul T, Pati BR, Mondal KC, Mohapatra PKD (2014) Biosynthesis, structural architecture and biotechnological potential of bacterial tannase: a molecular advancement. Bioresour Technol 157:327–340
Jiménez N, Barcenilla JM, de Felipe FL, de las Rivas B, Muñoz R (2014) Characterization of a bacterial tannase from Streptococcus gallolyticus UCN34 suitable for tannin biodegradation. Appl Microbiol Biotechnol 98:6329–6337
Jun C, Yoo M, Lee W, Kwak K, Bae M, Hwang W, Son D, Chai K (2007) Ester derivatives from tannase-treated prunioside and their anti-inflammatory activities. Bull Kor Chem Soc 28:73–76
Kachouri S, Halaouli S, Lomascolo A, Asther M, Hamdi M (2005) Decolourization of black oxidized olive-mill wastewater by a new tannase-producing Aspergillus flavus strain isolated from soil. World J Microbiol Biotechnol 21:1465–1470
Kar B, Banerjee R, Bhattacharyya BC (2002) Optimization of physicochemical parameters of gallic acid production by evolutionary operation-factorial design technique. Process Biochem 37:1395–1140
Karak T, Bhagat RM (2010) Trace elements in tea leaves, made tea and tea infusion: a review. Food Res Int 43:2234–2252
Kasieczka-Burnecka M, Kuc, Kalinowska KH, Knap M, Turkiewicz M (2007) Purification and characterization of two cold-adapted extracellular tannin acyl hydrolases from an Antarctic strain Verticillium sp. P9. Appl Microbiol Biotechnol 77:77–89
Khanbabaee K, van Ree T (2001) Tannins: classification and definition. Nat Prod Rep 18:641–649
Kour D, Rana KL, Yadav N, Yadav AN, Singh J, Rastegari AA et al (2019) Agriculturally and industrially important fungi: current developments and potential biotechnological applications. In: Yadav AN, Singh S, Mishra S, Gupta A (eds) Recent advancement in white biotechnology through fungi, Perspective for value-added products and environments, vol 2. Springer International Publishing, Cham, pp 1–64. https://doi.org/10.1007/978-3-030-14846-1_1
Kumar RA, Gunasekaran P, Lakshmanan M (1999) Biodegradation of tannic acid by Citrobacter freundii isolated from a tannery effluent. J Basic Microbiol Int J Biochem Physiol Genet Morphol Ecol Microorg 39(3):161–168
Kumar R, Sharma J, Singh R (2007) Production of tannase from Aspergillus ruber under solid-state fermentation using jamun (Syzygium cumini) leaves. Microbiol Res 162:384–390
Kumar SS, Sreekumar R, Sabu A (2019) Tannase and its applications in food processing. In: Parameswaran B, Varjani S, Raveendran S (eds) Green bio-processes. Energy, environment, and sustainability. Springer, Singapore
Kuppusamy M, Thangavelu V, Kumar A, Chockalingam (2015) Kinetics and modeling of tannase production using Aspergillus foetidus in batch fermentation. Int J Pharm Pharm Sci 7:64–67
Lal D, Divya Shrivastava D, Verma HN, Gardne JJ (2012) Production of tannin acyl hydrolase (E.C. 3.1.1.20) from Aspergillus niger isolated from bark of Acacia nilotica. J Microbiol Biotechnol Res 4:566–572
Lekha PK, Lonsane BK (1997) Production and application of tannin acyl hydrolase: state of the art. Adv Appl Microbiol 44:215–260
Lenin B, Lokeswari N, Reddy D (2015) Separation and optimization of phenolic component from Anacardium occidentales testa by solvent extraction method. World J Pharm Res 4:870–874
Lewis JA, Starkey RL (1969) Decomposition of plant tannins by some soil microorganisms. Soil Sci 107(4):235–241
Li J, Xiao Q, Huang Y, Ni H, Wu C, Xiao A (2017) Tannase application in secondary enzymatic processing of inferior Tieguanyin oolong tea. Electron J Biotechnol 28:87–94
Liu TPSL, Porto TS, Moreira KA, Takaki GMC, Brandão-Costa R, Herculano PN et al (2016) Tannase production by Aspergillus spp. UCP1284 using cashew bagasse under solid state fermentation. Afr J Microbiol Res 10(16):565–571
Lu MJ, Chen C (2007) Enzymatic tannase treatment of green tea increases in vitro inhibitory activity against N-nitrosation of dimethylamine. Process Biochem 42:1285–1290
Lu MJ, Chen C (2008) Enzymatic modification by tannase increases the antioxidant activity of green tea. Food Res Int 41:130–137
Lu MJ, Chu SC, Yan L, Chen C (2009) Effect of tannase treatment on protein-tannin aggregation and sensory attributes of green tea infusion. LWT—Food Sci Technol 42:338–342
Łuczaj W, Skrzydlewska E (2005) Antioxidative properties of black tea. Prev Med 40(6):910–918
Madhavakrishna W, Bose S, Nayudamma Y (1960) Estimation of tannase and certain oxidizing enzymes in Indian vegetable tanstuffs. Bull Cent Leath Res Inst 7(1):11
Mahapatra S, Banerjee D (2009) Extracellular tannase production by endophytic hyalopus sp. J Appl Microbiol 55:255–259
Mahendran B, Raman N, Kim D (2005) Purification and characterization of tannase from Paecilomyces variotii: hydrolysis of tannic acid using immobilized tannase. Appl Microbiol Biotechnol 70:445–451
Manjit YA, Aggarwal NK, Kumar K, Kumar A (2008) Tannase production by Aspergillus fumigatus MA under solid-state fermentation. World J Microbiol Biotechnol 24:3023–3030
Marco MG, Luis VR, Erika LR, Jacqueline R, Mario ACH, Raúl R, Juan C, Cristóbal NA (2009) Anovel tannase from the xerophilic fungus Aspergillus niger GH1. J Microbiol Biotechnol 1:1–10
Mondal KC, Pati BR (2000) Studies on the extracellular tannase from newly isolated Bacillus licheniformis KBR 6. J Basic Microbiol 40:223–232
Mondal KC, Samanta S, Giri S, Pati BR (2001) Distribution of tannic acid degrading microorganisms in the soil and comparative study of tannase from two fungal strains. Acta Microbiol Pol 50:75–82
Mosleh H, Naghiha A, Keshtkaran AN, Khajavi M (2014) Isolation and identification of tannin-degrading bacteria from native sheep and goat feces in Kohgiloye and Boyer-Ahmad Province. Int J Adv Biol Biomed Res 2:176–180
Mukherjee G, Banerjee R (2003) Production of gallic acid. Biotechnological routes (Part 1). Chim Oggi 21:59–62
Mukherjee G, Banerjee R (2006) Effects of temperature, pH and additives on the activity of tannase produced by a co-culture of Rhizopus oryzae and Aspergillus foetidus. World J Microbiol Biotechnol 22:207–212
Murugan K, Al-Sohaibani SA (2010) Biocompatible removal of tannin and associated color from tannery effluent using the biomass and tannin acyl hydrolase (E.C.3.1.1.20) enzymes of mango industry solid waste isolate Aspergillus candidus MTTC 9628. Res J Microbiol 5:262–271
Murugan K, Saravanababu S, Arunachalam M (2007) Screening of tannin acyl hydrolase (E.C.3.1.1.20) producing tannery effluent fungal isolates using simple agar plate and SmF process. Bioresour Technol 98:946–949
Nalan YS, Merih K (2009) Isolation of gallic acid producing microorganisms and their use in the production of gallic acid from gall nuts and sumac. Afr J Biotechnol 8:1110–1115, 20
Neethu RS, Pradeep S (2018) Isolation and characterization of potential tannase producing fungi from mangroves and tanneries. Indian J Appl Microbiol 21:1–13
Nishitani Y, Sasaki E, Fujisawa T, Osawa R (2004) Genotypic analyses of lactobacilli with a range of tannase activities isolated from human feces and fermented foods. Syst Appl Microbiol 27:109–117
Nuero OM, Reyes F (2002) Enzymes for animal feeding from Penicillium chrysogenum mycelial wastes from penicillin manufacture. Lett Appl Microbiol 34(6):413–416
Okuda T, Ito H (2011) Tannins of constant structure in medicinal and food plants-hydrolyzable tannins and polyphenols related to tannins. Molecules 16(3):2191–2217
Ordonez RM, Colombo I, Alberto MR, Isla MI (2011) Production of tannase from wood-degrading fungus using as substrate plant residues: purification and characterization. World J Microbiol Biotechnol 27:2325–2333
Ow YY, Stupans I (2003) Gallic acid and gallic acid derivatives: effects on drug metabolizing enzymes. Curr Drug Metab 4:241–248
Panno L, Voyron S, Anastasi A, MussatSartor R, Varese GC (2011) Biodiversity of marine fungi associated with the seagrass Posidonia oceanica: an ecological and biotechnological perspective. Biol Mar Mediterr 18:85–88
Panno L, Bruno M, Voyron S, Anastasi A, Gnavi G, Miserere L et al (2013) Diversity, ecological role and potential biotechnological applications of marine fungi associated to the seagrass Posidonia oceanica. New Biotechnol 30:685–694
Paranthaman R, Vidyalakshmi R, Murugesh S, Singaravadivel K (2009) Effects of fungal coculture for the biosynthesis of tannase and gallic acid from grape wastes under solid state fermentation. Glob J Biotechnol Biochem 4:29–36
Pepi M, Lampariello LR, Altieri R, Esposito A, Perra G, Renzi M et al (2010) Tannic acid degradation by bacterial strains Serratia spp. and Pantoea sp. isolated from olive mill waste mixtures. Int Biodeterior Biodegradation 64(1):73–80
Prigione V, Spina F, Tigini V, Giovando S, Varese GC (2018) Biotransformation of industrial tannins by filamentous fungi. Appl Microbiol Biotechnol 102:10361–10375
Purohit JS, Dutta JR, Nanda RK, Banerjee R (2006) Strain improvement for tannase production from co-culture of Aspergillus foetidus and Rhizopus oryzae. Bioresour Technol 97:795–801
Raghuwanshi S, Dutt K, Gupta P, Misra S, Saxena RK (2011) Bacillus sphaericus: the highest bacterial tannase producer with potential for gallic acid synthesis. J Biosci Bioeng 111(6):635–640
Rana N, Bhat T (2005) Effect of fermentation system on the production and properties of tannase of Aspergillus niger van Tieghem MTCC 2425. J Gen Appl Microbiol 51:203–212
Rana G, Katerji N, Introna M, Hammami A (2004) Microclimate and plant water relationship of the “overhead” table grape vineyard managed with three different covering techniques. Sci Hortic 102:105–120
Rana KL, Kour D, Kaur T, Devi R, Yadav AN, Yadav N et al (2020) Endophytic microbes: biodiversity, plant growth-promoting mechanisms and potential applications for agricultural sustainability. Antonie Van Leeuwenhoek 113:1075–1107. https://doi.org/10.1007/s10482-020-01429-y
Rastegari AA, Yadav AN, Yadav N (2020) New and future developments in microbial biotechnology and bioengineering: trends of microbial biotechnology for sustainable agriculture and biomedicine systems: perspectives for human health. Elsevier, Amsterdam
Reges de Sena A, Santos ACB, Gouveia MJ, Mello MRFM, Leite TCC et al (2014) Production of tannase by Pestalotiopsis guepinii. Food Technol Biotechnol 52(4):459–467
Rodríguez H, de las Rivas B, Gómez-Cordovés C, Muñoz R (2008) Characterization of tannase activity in cell-free extracts of Lactobacillus plantarum CECT 748T. Int J Food Microbiol 121:92–98
Rodriguez-Duran VL, Valdivia-Urdiales B, Contreras-Esquivel JC, Rodriguez-Herrera R, Aguilar CN (2011) Novel strategies for upstream and downstream processing of tannin acyl hydrolase. Enzyme Res 2011:1–20
Rout S, Banerjee R (2006) Production of tannase under MSSF and its application in fruit juice debittering. Indian J Biotechnol 5:346–350
Sabu A, Pandey A, Daud MJ, Szakacs G (2005) Tamarind seed powder and palm kernel cake: two novel agro residues for the production of tannase under solid state fermentation by Aspergillus niger ATCC 16620. Bioresour Technol 96:1223–1228
Sabu A, Augur C, Swati C, Pandey A (2006) Tannase production by Lactobacillus sp. ASR-S1 under solid-state fermentation. Process Biochem 41(3):575–580
Sandai D, Ibrahim D, Kassim J (2012) Calcium alginate entrapped cells of Penicillium digitatum FETL DS1 for the improvement of tannase production. BTAIJ 6:27–34
Sariozlu NY, Kivanc M (2009) Isolation of gallic acid producing microorganisms and their use in the production of gallic acid from gall nuts and sumac. Afr J Biotechnol 8:1110–1115
Saxena S, Saxena RK (2004) Statistical optimization of tannase production from Penicillium variable using fruits (chebulic myrobalan) of Terminalia chebula. Biotechnol Appl Biochem 39:99–106
Selwal MK, Yadav A, Selwal KK, Aggarwal NK, Gupta R, Gautam SK (2010) Optimization of cultural conditions for tannase production by Pseudomonas aeruginosa IIIB 8914 under submerged fermentation. World J Microbiol Biotechnol 26:599–605
Serrano J, Puupponen-Pimiä R, Dauer A, Aura AM, Saura-Calixto F (2009) Tannins: current knowledge of food sources, intake, bioavailability and biological effects. Mol Nutr Food Res 53:S310–S329
Shajitha G, Nisha MK (2018) Tannase production from agro-wastes as substrate by Trichoderma viride. Int J Curr Res Life Sci 07(05):1994–1997
Sharma KP, John PJ (2011) Purification and characterization of tannase and tannase gene from Enterobacter sp. Process Biochem 46(1):240–244
Sharma S, Bhat TK, Dawra RK (2000) A spectrophotometric method for assay of tannase using rhodamine. Anal Biochem 279:85–89
Shrivastava A, Kar K (2009) Characterization and application of tannase produced by Aspergillus niger ITCC 6514.07 on pomegranate rind. Brazil J Microbiol 40:782–789
Sittig M (1988) Trimethoprim. In: Sittig M (ed) Pharmaceutical manufacturing encylopedia. Noyes Publication, New Jersey, pp 282–284
Skene IK, Brooker JD (1995) Characterization of tannin acylhydrolase activity in the ruminal bacterium Selenomonas ruminantium. Anaerobe 1:321–327
Srivastava A, Kar R (2009) Characterization and application of tannase produced by Aspergillus niger ITCC 6514.07 on pomegranate rind. Braz J Microbiol 40:782–789
Srivastava A, Kar R (2010) Application of immobilized tannase from Aspergillus niger for the removal of tannin from myrobalan juice. Indian J Microbiol 50:46–51
Su E, Xia T, Gao L, Dai Q, Zhang Z (2009) Immobilization and characterization of tannase and its haze-removing. Food Sci Technol Int 15:545–552
Tieghem P (1867) Sur la fermentation gallique. CR Acad Sci Paris 65:1091–1094
Urbano G, Lopez-Jurado M, Porres JM (2007) Effect of treatment with α-galactosidase, tannase or a cell-wall degrading enzyme complex on the nutritive utilisation of protein and carbohydrates from pea (Pisum sativum L.) flour. J Sci Food Agric 87:1356–1363
Van de Lagemaat J, Pyle DL (2005) Modelling the uptake and growth kinetics of Penicillium glabrum in a tannic acid-containing solid state fermentation for tannase production. Process Biochem 40:1773–1782
Vattem DA, Shetty K (2002) Solid-state production of phenolic antioxidants from cranberry pomace by Rhizopus oligosporum. Food Biotechnol 16:189–210
Vattem DA, Shetty K (2003) Ellagic acid production and phenolic antioxidants activity in cranberry pomace (Vaccinium macrocarpo) mediated by Lentinus edodes using a solid-state system. Process Biochem 39:367–379
Venditti E, Bacchetti T, Tiano L, Carloni P, Greci L, Damiani E (2010) Hot vs. cold water steeping of different teas: do they affect antioxidant activity? Food Chem 119(4):1597–1604
Verma P, Yadav AN, Kumar V, Singh DP, Saxena AK (2017) Beneficial plant-microbes interactions: biodiversity of microbes from diverse extreme environments and its impact for crop improvement. In: Singh DP, Singh HB, Prabha R (eds) Plant-microbe interactions in agro-ecological perspectives, Microbial interactions and agro-ecological impacts, vol 2. Springer, Singapore, pp 543–580. https://doi.org/10.1007/978-981-10-6593-4_22
Vivas N, Laguerrre M, Pianet de Boissel I, Vivas de Gaulejac N, Nonier MF (2004) Conformational interpretation of vascalagin and castalagin physicochemical properties. J Agric Food Chem 52:2073–2078
Weetall H (1985) Enzymatic synthesis of gallic acid esters. Appl Biochem Biotechnol 11(1):25–28
Yadav AN (2021) Soil microbiomes for sustainable agriculture, Functional annotation, vol 2. Springer, Cham
Yadav AN, Kour D, Rana KL, Yadav N, Singh B, Chauhan VS et al (2019) Metabolic engineering to synthetic biology of secondary metabolites production. In: Gupta VK, Pandey A (eds) New and future developments in microbial biotechnology and bioengineering. Elsevier, Amsterdam, pp 279–320. https://doi.org/10.1016/B978-0-444-63504-4.00020-7
Yu XW, Li YQ (2008) Expression of Aspergillus oryzae tannase in Pichia pastoris and its application in the synthesis of propyl gallate in organic solvent. Food Technol nd Biotechnol 46:80–85
Yu X, Li Y, Wang C, Wu D (2004) Immobilization of Aspergillus niger tannase by microencapsulation and its kinetic characteristics. Biotechnol Appl Biochem 40:151–155
Zakipour-Molkabadi E, Hamidi-Esfahani Z, Sahari MA, Azizi MH (2013) A new native source of Tannase producer, Penicillium sp. EZ-ZH190: characterization of the enzyme. Iran J Biotechnol 11(4):244–250
Zhang YN, Yin JF, Chen JX, Wang F, Du QZ, Jiang YW, Xu YQ (2016) Improving the sweet aftertaste of green tea infusion with tannase. Food Chem 192:470–476
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Gezaf, S.A., Abo Nouh, F.A., Abdel-Azeem, A.M. (2021). Fungal Communities from Different Habitats for Tannins in Industry. In: Abdel-Azeem, A.M., Yadav, A.N., Yadav, N., Sharma, M. (eds) Industrially Important Fungi for Sustainable Development. Fungal Biology. Springer, Cham. https://doi.org/10.1007/978-3-030-85603-8_4
Download citation
DOI: https://doi.org/10.1007/978-3-030-85603-8_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-85602-1
Online ISBN: 978-3-030-85603-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)