Abstract
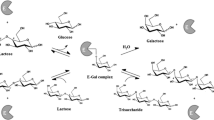
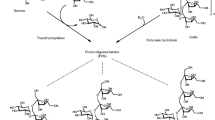
Prebiotics are important components in functional foods. Galacto-oligosaccharides (GOS) are nondigestible oligosaccharides whose prebiotic condition has been conclusively proven. GOS are produced from lactose, which is a cheap commodity, usually in surplus from the cheese manufacturing industry. GOS synthesis is catalyzed by β-galactosidases that are cheap enzymes with a long record of use in the food industry for the production of low-lactose milk and dairies, so its use for GOS synthesis represents a remarkable technological opportunity. GOS have salient features that make them ideal for their incorporation into functional foods, especially in dairy products, so that a scheme of circular economy can be envisaged. Even though GOS production is well established at industrial level, several challenges remain that are related to the rather low yield of the kinetically controlled reaction of synthesis from lactose. This results in poor substrate utilization and complex downstream operations, so major efforts are devoted to developing better suited enzymes and cheaper and simpler downstream operations. This chapter focuses on GOS production within the framework of functional foods, with special emphasis on technological perspectives of their production, but basic aspects, like reaction mechanisms and mode of prebiotic action, are also revised.
Similar content being viewed by others
References
Ambrogi V, Bottacini F, Cao L, Kuipers B, Schoterman M, van Sinderen D. Galacto-oligosaccharides as infant prebiotics: production, application, bioactive activities and future perspectives. Crit Rev Food Sci Nutr [Internet]. 2021;0(0):1–14. https://doi.org/10.1080/10408398.2021.1953437.
Balthazar CF, Silva HLA, Celeguini RMS, Santos R, Pastore GM, Conte Junior CA, et al. Effect of galactooligosaccharide addition on the physical, optical, and sensory acceptance of vanilla ice cream. J Dairy Sci. 2015;98(7):4266–72.
Benešová E, Lipovová P, Dvořáková H, Králová B. β-D-Galactosidase from Paenibacillus thiaminolyticus catalyzing transfucosylation reactions. Glycobiology. 2010;20(4):442–51.
Buitron DI, Sepulveda L, Martinez TKM, Aguilar CN, Medina DD, Rodriguez-Herrera R, et al. Biotechnological approach for the production of prebiotics and search for new probiotics and their application in the food industry. Appl Food Biotechnol [Internet]. 2018;5(4):185–92. Available from: http://journals.sbmu.ac.ir/afb/article/view/20174
Business-wire. Global galacto-oligosaccharide (GOS) market research report 202 [Internet]. online. 2021 [cited 2021 Nov 11]. p. 1. Available from: https://www.businesswire.com/news/home/20210623005487/en/Global-Galacto-oligosaccharide-GOS-Market-Research-Report-2021%2D%2D-ResearchAndMarkets.com
Catenza KF, Donkor KK. Recent approaches for the quantitative analysis of functional oligosaccharides used in the food industry: a review. Food Chem [Internet]. 2021;355(March):129416. Available from: https://doi.org/10.1016/j.foodchem.2021.129416.
Chen XY, Gänzle MG. Lactose and lactose-derived oligosaccharides: more than prebiotics? Int Dairy J. 2017;67:61–72.
Chen Q, Xiao Y, Zhang W, Zhang T, Jiang B, Stressler T, et al. Current research on cellobiose 2-epimerase: enzymatic properties, mechanistic insights, and potential applications in the dairy industry. Trends Food Sci Technol [Internet]. 2018;82:167–76. https://doi.org/10.1016/j.tifs.2018.09.009.
Clasado Inc. GRAS Notice 484. GRAS Exemption Claim for Galacto-oligosaccharides [Internet]. 2013. Available from: http://wayback.archive-it.org/7993/20171031042742/https://www.fda.gov/downloads/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/UCM379675.pdf
Córdova A, Astudillo C, Vera C, Guerrero C, Illanes A. Performance of an ultrafiltration membrane bioreactor (UF-MBR) as a processing strategy for the synthesis of galacto-oligosaccharides at high substrate concentrations. J Biotechnol. 2016;223:26–35
Costabile A, Walton GE, Tzortzis G, Vulevic J, Charalampopoulos D, Gibson GR. Effects of orange juice formulation on prebiotic functionality using an in vitro colonic model system. PLoS One. 2015;10(3):1–12.
Dekker P, Koenders D, Bruins M. Lactose-free dairy products: market developments, production, nutrition and health benefits. Nutrients [Internet] 2019;11(3):551. Available from: https://www.mdpi.com/2072-6643/11/3/551
Ding H, Zhou L, Zeng Q, Yu Y, Chen B. Heterologous expression of a thermostable β-1,3-galactosidase and its potential in synthesis of galactooligosaccharides. Mar Drugs. 2018;16(11):1–12.
Fara A, Sabater C, Palacios J, Requena T, Montilla A, Zárate G. Prebiotic galactooligosaccharides production from lactose and lactulose by Lactobacillus delbrueckii subsp. bulgaricus CRL450. Food Funct. 2020;11(7):5875–86.
Fischer C, Kleinschmidt T. Effect of glucose depletion during the synthesis of galactooligosaccharides using a trienzymatic system. Enzyme Microb Technol [Internet]. 2019;121(October 2018):45–50. https://doi.org/10.1016/j.enzmictec.2018.10.009.
Fischer C, Kleinschmidt T. Synthesis of galactooligosaccharides by Cryptococcus laurentii and Aspergillus oryzae using different kinds of acid whey. Int Dairy J. 2021;112:104867
Fox PF. Lactose: chemistry and properties. In: McSweeney P, Fox P, editors. Advanced dairy chemistry [Internet]. New York: Springer New York; 2009. p. 1–15. https://doi.org/10.1007/978-0-387-84865-5_1.
Frenzel M, Zerge K, Clawin-Rädecker I, Lorenzen PC. Comparison of the galacto-oligosaccharide forming activity of different β-galactosidases. LWT – Food Sci Technol. 2015;60(2):1068–71.
Friesland Foods Domo. Gras notice 236. GRAS exemption claim for galacto-oligosaccharides (GOS). 2007.
Füreder V, Rodriguez-Colinas B, Cervantes FV, Fernandez-Arrojo L, Poveda A, Jimenez-Barbero J, et al. Selective synthesis of galactooligosaccharides containing β(1→3) linkages with β-galactosidase from Bifidobacterium bifidum (Saphera). J Agric Food Chem. 2020;68(17):4930–8.
Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, et al. Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol [Internet]. 2017;14(8):491–502. Available from: http://www.nature.com/doifinder/10.1038/nrgastro.2017.75
González-Delgado I, López-Muñoz MJ, Morales G, Segura Y. Optimisation of the synthesis of high galacto-oligosaccharides (GOS) from lactose with β-galactosidase from Kluyveromyces lactis. Int Dairy J. 2016;61:211–9.
GRAS Associates. Gras notice 729. Galactooligosaccharides (GOS), food usage conditions for general gecognition of safety [Internet]. 2017. Available from: https://www.fda.gov/media/111860/download
Guerrero C, Vera C, Illanes A. Optimisation of synthesis of oligosaccharides derived from lactulose (fructosyl-galacto-oligosaccharides) with β-galactosidases of different origin. Food Chem. 2013 Jun;138(4):2225–32.
Guerrero C, Vera C, Serna N, Illanes A. Immobilization of Aspergillus oryzae β-galactosidase in an agarose matrix functionalized by four different methods and application to the synthesis of lactulose. Bioresour Technol. 2017;232:53–63
Guerrero C, Aburto C, Suárez S, Vera C, Illanes A. Effect of the type of immobilization of β-galactosidase on the yield and selectivity of synthesis of transgalactosylated oligosaccharides. Biocatal Agric Biotechnol [Internet]. 2018;16(August):353–63. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1878818118304250
Guimarães JT, Balthazar CF, Silva R, Rocha RS, Graça JS, Esmerino EA, et al. Impact of probiotics and prebiotics on food texture. Curr Opin Food Sci [Internet]. 2020;33:38–44. Available from: https://linkinghub.elsevier.com/retrieve/pii/S2214799319301316
Hernández O, Ruiz-Matute AI, Olano A, Moreno FJ, Sanz ML. Comparison of fractionation techniques to obtain prebiotic galactooligosaccharides. Int Dairy J. 2009;19(9):531–6.
Huerta LM, Vera C, Guerrero C, Wilson L, Illanes A. Synthesis of galacto-oligosaccharides at very high lactose concentrations with immobilized β-galactosidases from Aspergillus oryzae. Process Biochem [Internet]. 2011;46(1):245–52. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1359511310003296
Huh KT, Toba T, Adachi S. Oligosaccharide structures formed during acid hydrolysis of lactose. Food Chem. 1991;39(1):39–49.
Ibrahim OO. Functional oligosaccharides: chemicals structure, manufacturing, health benefits, applications and regulations. J Food Chem Nanotechnol. 2018:65–76.
Illanes A, Vera C, Wilson L. Enzymatic production of galacto-oligosaccharides. In: Illanes A, Guerrero C, Vera C, Wilson L, Raúl Conejeros FS, editors. Lactose-derived prebiotics [Internet]. London: Academic; 2016. p. 111–89. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780128027240000044.
Iqbal S, Nguyen T-H, Nguyen TT, Maischberger T, Haltrich D. β-Galactosidase from Lactobacillus plantarum WCFS1: biochemical characterization and formation of prebiotic galacto-oligosaccharides. Carbohydr Res [Internet]. 2010 Jul 2 [cited 2019 Apr 29];345(10):1408–16. Available from: https://www.sciencedirect.com/science/article/pii/S0008621510001242
Iwatani S, Yamamoto N. Functional food products in Japan: a review. Food Sci Hum Wellness [Internet]. 2019 Jun;8(2):96–101. Available from: https://doi.org/10.1016/j.fshw.2019.03.011.
Jenab E, Omidghane M, Mussone P, Armada DH, Cartmell J, Montemagno C. Enzymatic conversion of lactose into galacto-oligosaccharides: the effect of process parameters, kinetics, foam architecture, and product characterization. J Food Eng [Internet]. 2018;222:63–72. https://doi.org/10.1016/j.jfoodeng.2017.11.015.
Johnstone N, Milesi C, Burn O, van den Bogert B, Nauta A, Hart K, et al. Anxiolytic effects of a galacto-oligosaccharides prebiotic in healthy females (18–25 years) with corresponding changes in gut bacterial composition. Sci Rep 2021;11(1):1–11.
Jurado E, Camacho F, Luzón G, Vicaria JMM. Kinetic models of activity for β-galactosidases: influence of pH, ionic concentration and temperature. Enzyme Microb Technol [Internet]. 2004;34(1):33–40. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0141022903002771
Kasche V. Mechanism and yields in enzyme catalysed equilibrium and kinetically controlled synthesis of β-lactam antibiotics, peptides and other condensation products. Enzyme Microb Technol [Internet]. 1986;8(1):4–16. Available from: https://linkinghub.elsevier.com/retrieve/pii/0141022986900037
Kim CS, Ji ES, Oh DK. A new kinetic model of recombinant β-galactosidase from Kluyveromyces lactis for both hydrolysis and transgalactosylation reactions. Biochem Biophys Res Commun. 2004;316(3):738–43.
Kötzler MP, Hancock SM, Withers SG. Glycosidases: functions, families and folds. In: eLS [Internet]. Chichester: Wiley; 2014. p. 1–14. https://doi.org/10.1002/9780470015902.a0020548.pub2
Kruschitz A, Nidetzky B. Downstream processing technologies in the biocatalytic production of oligosaccharides. Biotechnol Adv [Internet]. 2020;43(November 2019):107568. Available from: https://doi.org/10.1016/j.biotechadv.2020.107568.
Lu L, Guo L, Wang K, Liu Y, Xiao M. β-Galactosidases: a great tool for synthesizing galactose-containing carbohydrates. Biotechnol Adv [Internet]. 2020;39(November 2019):107465. https://doi.org/10.1016/j.biotechadv.2019.107465.
Martínez-Villaluenga C, Cardelle-Cobas A, Olano A, Corzo N, Villamiel M, Jimeno ML. Enzymatic synthesis and identification of two trisaccharides produced from lactulose by transgalactosylation. J Agric Food Chem [Internet]. 2008 Jan;56(2):557–63. https://doi.org/10.1021/jf0721343.
Martins GN, Ureta MM, Tymczyszyn EE, Castilho PC, Gomez-Zavaglia A. Technological aspects of the production of fructo and galacto-oligosaccharides. Enzymatic synthesis and hydrolysis. Front Nutr. 2019;6(May):78
Matharu AS, de Melo EM, Houghton JA. Opportunity for high value-added chemicals from food supply chain wastes. Bioresour Technol [Internet]. 2016;215(4):123–30. Available from: https://linkinghub.elsevier.com/retrieve/pii/S096085241630325X
Neri DFM, Balcão VM, Costa RS, Rocha IC a P, EMFC F, DPM T, et al. Galacto-oligosaccharides production during lactose hydrolysis by free Aspergillus oryzae β-galactosidase and immobilized on magnetic polysiloxane-polyvinyl alcohol. Food Chem. 2009;115(1):92–9.
Neri-Numa IA, Arruda HS, Geraldi MV, Maróstica Júnior MR, Pastore GM. Natural prebiotic carbohydrates, carotenoids and flavonoids as ingredients in food systems. Curr Opin Food Sci. 2020;33:98–107.
Nestle Nutritión U.S. GRAS notice 620. GRAS exemption claim for galacto-oligosaccharides [Internet]. 2016. Available from: https://wayback.archive-it.org/7993/20180124043912/https://www.fda.gov/downloads/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/UCM504605.pdf
Ohtsuka K, Tsuji K, Nakagawa Y, Vera H, Ozawa O, Uchida T, et al. Availability of 4’galactosyllactose (O-.BETA.-D-galactopyranosyl-(1.RAR.4)-O-.BETA.-D-galactopyranosyl-(1.RAR.4)-D-glucopyranose) in rat. J Nutr Sci Vitaminol (Tokyo) [Internet]. 1990;36(3):265–76. Available from: http://www.jstage.jst.go.jp/article/jnsv1973/36/3/36_3_265/_article
Palai T, Mitra S, Bhattacharya PK. Kinetics and design relation for enzymatic conversion of lactose into galacto-oligosaccharides using commercial grade β-galactosidase. J Biosci Bioeng. 2012;114(4):418–23.
Panesar PS, Kaur R, Singh RS, Kennedy JF. Biocatalytic strategies in the production of galacto-oligosaccharides and its global status. Int J Biol Macromol [Internet]. 2018;111:667–79. https://doi.org/10.1016/j.ijbiomac.2018.01.062.
Petuely F. Bifidusflora bei Flaschenkindern durch bifidogene Substanzen (Bifidusfaktor). Z Kinderheilkd [Internet]. 1957;79(2):174–9. https://doi.org/10.1007/BF00440162.
Rajagopalan G, Krishnan C. Functional oligosaccharides: production and action. ACS Symp Ser. 2019;1329:155–80.
Rather M, Mishra S. β-Glycosidases: an alternative enzyme based method for synthesis of alkyl-glycosides. Sustain Chem Process [Internet] 2013;1(1):7. Available from: http://sustainablechemicalprocesses.springeropen.com/articles/10.1186/2043-7129-1-7
Rodriguez-Colinas B, De Abreu MA, Fernandez-Arrojo L, De Beer R, Poveda A, Jimenez-Barbero J, et al. Production of galacto-oligosaccharides by the β-galactosidase from Kluyveromyces lactis: comparative analysis of permeabilized cells versus soluble enzyme. J Agric Food Chem. 2011;59(19):10477–84.
Rodriguez-Colinas B, Fernandez-Arrojo L, Ballesteros AO, Plou FJ. Galactooligosaccharides formation during enzymatic hydrolysis of lactose: towards a prebiotic-enriched milk. Food Chem. 2014;145:388–94.
Rodriguez-Colinas B, Fernandez-Arrojo L, Santos-Moriano P, Ballesteros AO, Plou FJ. Continuous packed bed reactor with immobilized β-galactosidase for production of galactooligosaccharides (GOS). Catalysts [Internet] 2016 Nov 30;6(12):189. Available from: http://www.mdpi.com/2073-4344/6/12/189
Rosa MC, Carmo MRS, Balthazar CF, Guimarães JT, Esmerino EA, Freitas MQ, et al. Dairy products with prebiotics: an overview of the health benefits, technological and sensory properties. Int Dairy J. 2021;117:105009
Sangwan V, Tomar SK, Singh RRB, Singh AK, Ali B. Galactooligosaccharides: novel components of designer foods. J Food Sci. 2011;76(4):R103–11.
Santibáñez L, Fernández-Arrojo L, Guerrero C, Plou FJ, Illanes A. Removal of lactose in crude galacto-oligosaccharides by β-galactosidase from Kluyveromyces lactis. J Mol Catal B Enzym. 2016;133:85–91.
Scott F, Vera C, Conejeros R. Technical and economic analysis of industrial production of lactose-derived prebiotics with focus on galacto-oligosaccharides. In: Lactose-derived prebiotics [Internet]. Elsevier; 2016. p. 261–84. Available from: https://linkinghub.elsevier.com/retrieve/pii/B978012802724000007X
Seijo M, Bonanno MS, Vénica CI, Marotte C, Martín P, de Portela ML, Bergamini CV, et al. A yoghurt containing galactooligosaccharides and having low-lactose level improves calcium absorption and retention during growth: experimental study. Int J Food Sci Technol. 2021:1–9.
Sneha HP, Beulah KC, Murthy PS. Enzyme immobilization methods and applications in the food industry. In: Enzymes in food biotechnology [Internet]. Elsevier; 2019. p. 645–58. https://doi.org/10.1016/B978-0-12-813280-7.00037-2.
Soni & Associates Inc. GRAS notice 569. GRAS notification for galacto-oligosaccharide (infant formula use) [Internet] 2015. Available from: http://www.fda.gov/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/default.htm
Spherix Consulting Inc. Gras notice 334. Generally recognized as safe (GRAS) determination for the use of galacto-oligosaccharides (GOS) in foods and infant formulas [Internet]. 2010. Available from: http://wayback.archive-it.org/7993/20171031050145/https://www.fda.gov/downloads/Food/IngredientsPackagingLabeling/GRAS/NoticeInventory/UCM269519.pdf
Spherix Consulting Inc. GRAS notice 721. Generally recognized as safe determination for the use of VITAGOS in infant formula and selected conventional foods [Internet]. 2016. Available from: https://www.fda.gov/media/110690/download
Splechtna B, Nguyen T-H, Haltrich D. Comparison between is dcontinuous and continuous lactose conversion processes for the production of prebiotic galacto-oligosaccharides using β-galactosidase from Lactobacillus reuteri. J Agric Food Chem. 2007;55(16):6772–7.
Suh MG, Hong YH, Jung EY, Suh HJ. Inhibitory effect of galactooligosaccharide on skin pigmentation. Prev Nutr Food Sci. 2019;24(3):321–6.
Swennen K, Courtin CM, Delcour JA. Non-digestible oligosaccharides with prebiotic properties. Crit Rev Food Sci Nutr [Internet]. 2006;46(6):459–71. https://doi.org/10.1080/10408390500215746.
The CAZypedia Consortium. Ten years of CAZypedia: a living encyclopedia of carbohydrate-active enzymes. Glycobiology [Internet]. 2018;28(1):3–8. Available from: https://academic.oup.com/glycob/article/28/1/3/4430919
Ugidos-Rodríguez S, Matallana-González MC, Sánchez-Mata MC. Lactose malabsorption and intolerance: a review. Food Funct. 2018;9(8):4056–68.
Urrutia P, Rodriguez-Colinas B, Fernandez-Arrojo L, Ballesteros AO, Wilson L, Illanes A, et al. Detailed analysis of galactooligosaccharides synthesis with β-galactosidase from Aspergillus oryzae. J Agric Food Chem. 2013a;61(5):1081–7.
Urrutia P, Mateo C, Guisan JM, Wilson L, Illanes A. Immobilization of Bacillus circulans β-galactosidase and its application in the synthesis of galacto-oligosaccharides under repeated-batch operation. Biochem Eng J [Internet]. 2013b 77:41–8. https://doi.org/10.1016/j.bej.2013.04.015.
van den Heuvel EGHM, Schoterman MHC, Muijs T. Transgalactooligosaccharides stimulate calcium absorption in postmenopausal women. J Nutr [Internet]. 2000;130(12):2938–42. Available from: https://academic.oup.com/jn/article/130/12/2938/4686280
Vera C. Optimización de la síntesis de galacto-oligosacaridos en modalidad de lote alimentado [Internet]. Pontificia Universidad Católica de Valparaíso; 2012 [cited 2019 Dec 8]. Available from: http://repositorio.conicyt.cl/handle/10533/184354
Vera C, Guerrero C, Illanes A. Determination of the transgalactosylation activity of Aspergillus oryzae β-galactosidase: effect of pH, temperature, and galactose and glucose concentrations. Carbohydr Res [Internet]. 2011a;346(6):745–52. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0008621511000541
Vera C, Guerrero C, Illanes A, Conejeros R. A pseudo steady-state model for galacto-oligosaccharides synthesis with β-galactosidase from Aspergillus oryzae. Biotechnol Bioeng [Internet]. 2011b;108(10):2270–9. https://doi.org/10.1002/bit.23201.
Vera C, Guerrero C, Conejeros R, Illanes A. Synthesis of galacto-oligosaccharides by β-galactosidase from Aspergillus oryzae using partially dissolved and supersaturated solution of lactose. Enzyme Microb Technol [Internet]. 2012;50(3):188–94. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0141022911002535
Vera C, Guerrero C, Illanes A, Conejeros R. Fed-batch synthesis of galacto-oligosaccharides with Aspergillus oryzae β-galactosidase using optimal control strategy. Biotechnol Prog. 2014;30(1):59–67.
Vera C, Guerrero C, Aburto C, Cordova A, Illanes A. Conventional and non-conventional applications of β-galactosidases. Biochim Biophys Acta - Proteins Proteomics [Internet]. 2020;1868(1):140271. https://doi.org/10.1016/j.bbapap.2019.140271.
Vera C, Illanes A, Guerrero C. Enzymatic production of prebiotic oligosaccharides. Curr Opin Food Sci. 2021;37:160–70.
Vulevic J, Juric A, Walton GE, Claus SP, Tzortzis G, Toward RE, et al. Influence of galacto-oligosaccharide mixture (B-GOS) on gut microbiota, immune parameters and metabonomics in elderly persons. Br J Nutr [Internet]. 2015;114(4):586–95. Available from: https://www.cambridge.org/core/product/identifier/S0007114515001889/type/journal_article
Warmerdam A, Zisopoulos FK, Boom RM, Janssen AEM. Kinetic characterization of galacto-oligosaccharide (GOS) synthesis by three commercially important β-galactosidases. Biotechnol Prog. 2014;30(1):38–47.
Yañez-Ñeco CV, Cervantes FV, Amaya-Delgado L, Ballesteros AO, Plou FJ, Arrizon J. Synthesis of β(1 → 3) and β(1 → 6) galactooligosaccharides from lactose and whey using a recombinant β-galactosidase from Pantoea anthophila. Electron J Biotechnol. 2021;49:14–21.
Yang J, Xu Y. Functional carbohydrate polymers: prebiotics. Polymers for food applications 2018. 651–691 p.
Yazawa K, Imai K, Tamura Z. Oligosaccharides and polysaccharides specifically utilizable by Bifidobacteria. Chem Pharm Bull (Tokyo) [Internet]. 1978;26(11):3306–11. Available from: https://www.jstage.jst.go.jp/article/cpb1958/26/11/26_11_3306/_article
Zhu ZY, Cui D, Gao H, Dong FY, Liu XC, Liu F, et al. Efficient synthesis and activity of beneficial intestinal flora of two lactulose-derived oligosaccharides. Eur J Med Chem. 2016;114:8–13.
Ziegler C, Suss E, Lanciki A. Improving on AOAC 2001.02: GOS determination in foods using HPAEC–PAD [Internet]. 21-02-2021. 2021 [cited 2021 Nov 15]. p. 1. Available from: https://www.chromatographyonline.com/view/improving-on-aoac-2001-02-gos-determination-in-foods-using-hpaec-pad
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Vera, C., Guerrero, C., Illanes, A. (2022). Galacto-oligosaccharides. In: Jafari, S.M., Rashidinejad, A., Simal-Gandara, J. (eds) Handbook of Food Bioactive Ingredients. Springer, Cham. https://doi.org/10.1007/978-3-030-81404-5_33-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-81404-5_33-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-81404-5
Online ISBN: 978-3-030-81404-5
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences