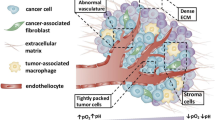
Introduction
Molecular imaging encompasses all in vivo imaging techniques that enable the visualization, characterization, and measurement of biological processes at the molecular and cellular levels in humans and other living systems (Mankoff 2007). It permits assessment of biochemical processes and targets localization patterns that are invisible at the anatomical imaging level (Rowe and Pomper 2021).
The basic principle of in vivo imaging relies on the visualization of tissue contrast. In most cases, this contrast is generated by an imaging agent capable of interacting specifically with a target. Hence, imaging agents serve only as “tracer” molecules creating or enhancing the contrast on the image to help the visualization and that do not induce any pharmacological effect, nor any molecular perturbation of the cell, its microenvironment, or the biological process studied. There is a wide range of contrast agents, suitable for each imaging modality. These agents can be molecules or...
Similar content being viewed by others
References
Agard NJ, Prescher JA, Bertozzi CR, et al (2004) A Strain-Promoted [3 + 2] Azide-Alkyne Cycloaddition for Covalent Modification of Biomolecules in Living Systems. J Am Chem Soc 126:15046–15047.
Anger HO (1952) NATURE August 2, 1952. Nature 170:200–201
Bailly C, Cléry P-F, Faivre-Chauvet A, Bourgeois M, Guérard F, Haddad F, Barbet J, et al (2017) Molecular sciences immuno-PET for clinical theranostic approaches. https://doi.org/10.3390/ijms18010057
Baldwin WM, Valujskikh A, Fairchild RL (2019) The neonatal fc receptor: key to Homeostasic control of IgG and IgG-related biopharmaceuticals. Am J Transplant 19(7):1881–1887. https://doi.org/10.1111/ajt.15366
Beylergil V, Morris PG, Smith-Jones PM, Modi S, Solit D, Hudis CA, Yang L et al (2013) Pilot Study of 68 Ga-DOTA-F(Ab 0) 2-Trastuzumab in Patients with Breast Cancer. Nucl Med Commun 34:1157–1165. https://doi.org/10.1097/MNM.0b013e328365d99b
Boros E, Holland JP (2018) Chemical aspects of metal ion chelation in the synthesis and application antibody-based radiotracers HHS public access. J Labelled Comp Radiopharm 61(9):652–671. https://doi.org/10.1002/jlcr.3590
Carrasquillo JA, Pandit-Taskar N, O’Donoghue JA, Humm JL, Zanzonico P, Smith-Jones PM, Divgi CR et al (2011) 124I-HuA33 antibody PET of colorectal cancer. J Nucl Med 52(8):1173–1180. https://doi.org/10.2967/jnumed.110.086165
Dijkers ECF, Kosterink JGW, Rademaker AP, Perk LR, Van Dongen GAMS, Bart J, De Jong JR, De Vries EGE, Lub-de MN, Hooge. (2009) Development and characterization of Clinical-Grade89Zr-Trastuzumab for HER2/Neu ImmunoPET imaging. J Nucl Med 50(6):974–981. https://doi.org/10.2967/jnumed.108.060392
Divgi CR, Pandit-Taskar N, Jungbluth AA, Reuter VE, Gönen M, Ruan S, Pierre C et al (2007) Preoperative characterisation of clear-cell renal carcinoma using Iodine-124-labelled antibody chimeric G250 (124I-CG250) and PET in patients with renal masses: a phase I trial. Lancet Oncol 8(4):304–310. https://doi.org/10.1016/S1470-2045(07)70044-X
Divgi CR, Uzzo RG, Gatsonis C, Bartz R, Treutner S, Jian Qin Y, Chen D et al (2013) Positron emission tomography/computed tomography identification of clear cell renal cell carcinoma: results from the REDECT trial. J Clin Oncol 31(2):187–194. https://doi.org/10.1200/JCO.2011.41.2445
Fehm T, Jäger W, Kraemer S, Sohn C, Solomayer-Meyberg G, Solomayer EF, Kurek R, Wallwiener D, Gebauer G (2004) Changes of serum HER2 status during clinical course of metastatic breast cancer patients. Anticancer Res 24(6):4205–4210
Gebhart G, Lamberts LE, Wimana Z, Garcia C, Emonts P, Ameye L, Stroobants S et al (2016) Molecular imaging as a tool to investigate heterogeneity of advanced HER2-positive breast cancer and to predict patient outcome under Trastuzumab Emtansine (T-DM1): the ZEPHIR trial. Ann Oncol 27(4):619–624. https://doi.org/10.1093/annonc/mdv577
Goldenberg DM, Nabi HA (1999) Breast cancer imaging with radiolabeled antibodies. Semin Nucl Med 29(1):41–48. https://doi.org/10.1016/S0001-2998(99)80028-2
Grillo-Lopez A, White C, Dallaire B, Varns C, Shen C, Wei A, Leonard J et al (2005) Rituximab the first monoclonal antibody approved for the treatment of lymphoma. Curr Pharm Biotechnol 1(1):1–9. https://doi.org/10.2174/1389201003379059
Herrmann K, Larson SM, Weber WA (2017) Theranostic concepts: more than just a fashion trend-introduction and overview. J Nucl Med 58:1S–2S. https://doi.org/10.2967/jnumed.117.199570
Herzog H, Rosch F, Stocklin G, Lueders C, Qaim SM, Feinendegen LE (1993) Measurement of pharmacokinetics of Yttrium-86 radiopharmaceuticals with PET and radiation dose calculation of analogous Yttrium-90 radiotherapeutics. J Nucl Med 34(12):2222–2226
Hnatowich DJ, Virzi F, Rusckowski M (1987) Investigations of Avidin and biotin for imaging applications
Keinänen O, Brennan JM, Membreno R, Fung K, Gangangari K, Dayts EJ, Williams CJ, Zeglis BM (2019) Dual radionuclide theranostic pretargeting. Mol Pharm 16(10):4416–4421. https://doi.org/10.1021/acs.molpharmaceut.9b00746
Keinänen O, Fung K, Brennan JM, Zia N, Harris M, van Dam E, Biggin C et al (2020) Harnessing 64Cu/67Cu for a Theranostic approach to Pretargeted Radioimmunotherapy. Proc Natl Acad Sci U S A 117(45):28316–28327. https://doi.org/10.1073/pnas.2009960117
Kelly VJ, Wu ST, Gottumukkala V, Coelho R, Palmer K, Nair S, Erick T, Puri R, Ilovich O, Mukherjee P (2020) Preclinical evaluation of an 111In/225Ac Theranostic targeting transformed MUC1 for triple negative breast cancer. Theranostics 10(15):6946–6958. https://doi.org/10.7150/thno.38236
Kiess AP, Banerjee SR, Mease RC, Rowe SP, Rao A, Foss CA, Chen Y et al (2015) Prostate-specific membrane antigen as a target for cancer imaging and therapy HHS public access. Q J Nucl Med Mol Imaging 59
Köhler G, Milstein C (1975) Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256(Mopc 21):495–497. http://www.nature.com/articles/256495a0%0Apapers3://publication/doi/10.1038/256495a0
Lai Y, Hsiao P, Yanev S, Liu G (2018) A revisit to the Pretargeting concept-a target conversion. Front Pharmacol 9:1476. https://doi.org/10.3389/fphar.2018.01476. WwwFrontiersinOrg
Lambin, Philippe, Emmanuel Rios-Velazquez, Ralph Leijenaar, Sara Carvalho, Ruud G.P.M. Van Stiphout, Patrick Granton, Catharina M.L. Zegers, et al. 2012. “Radiomics: extracting more information from medical images using advanced feature analysis.” Eur J Cancer 48 (4): 441–446. https://doi.org/10.1016/j.ejca.2011.11.036
Larimer BM, Wehrenberg-Klee E, Caraballo A, Mahmood U (2016) Quantitative CD3 PET imaging predicts tumor growth response to anti-CTLA-4 therapy. J Nucl Med 57(10):1607–1611. https://doi.org/10.2967/jnumed.116.173930
Lee I, Lim I, Byun BH, Il Kim B, Choi CW, Woo S-K, Il Kim K, et al (2021) A preliminary clinical trial to evaluate 64 Cu-NOTA-Trastuzumab as a positron emission tomography imaging agent in patients with breast cancer. https://doi.org/10.1186/s13550-021-00746-1.
Mankoff (2007) A definition of molecular imaging. www.snm.org/llsap
Mease RC, Foss CA, Pomper MG, Morgan RH. (2013) PET imaging in prostate cancer: focus on prostate-specific membrane antigen. https://doi.org/10.2174/092986712
Menke-Van Der Houven Van Oordt, Willemien C, Gootjes EC, Huisman MC, Vugts DJ, Roth C, Luik AM, Mulder ER et al (2015) 89Zr-Cetuximab PET imaging in patients with advanced colorectal cancer. Oncotarget 6(30). www.impactjournals.com/oncotarget/:30384
Meyer J-P, Adumeau P, Lewis JS, Zeglis BM (2016) Click chemistry and radiochemistry: the first 10 years graphical abstract HHS public access. Bioconjug Chem 27(12):2791–2807. https://doi.org/10.1021/acs.bioconjchem.6b00561
Mortimer JE, Bading JR, Park JM, Frankel PH, Carroll MI, Tran TT, Poku EK et al (2018) Tumor uptake of 64 Cu-DOTA-Trastuzumab in patients with metastatic breast cancer. J Nucl Med 59(1):38–43. https://doi.org/10.2967/jnumed.117.193888
O’donoghue JA, Smith-Jones PM, Humm JL, Ruan S, Pryma DA, Jungbluth AA, Divgi CR et al (2011) 24 I-HuA33 antibody uptake is driven by A33 antigen concentration in tissues from colorectal cancer patients imaged by Immuno-PET. J Nucl Med 52:1878–1885. https://doi.org/10.2967/jnumed.111.095596
Osborne JR, Green DA, Spratt DE, Lyashchenko S, Fareedy SB, Robinson BD, Beattie BJ et al (2014) A prospective pilot study of 89Zr-J591/prostate specific membrane antigen positron emission tomography in men with localized prostate cancer undergoing radical prostatectomy. J Urol 191(5):1439–1445. https://doi.org/10.1016/j.juro.2013.10.041
Pandit-Taskar N, O’donoghue JA, Durack JC, Lyashchenko SK, Cheal SM, Beylergil V, Lefkowitz RA, et al (2015) Personalized Medicine and Imaging A Phase I/II Study for Analytic Validation of Zr-J591 ImmunoPET as a Molecular Imaging Agent for Metastatic Prostate Cancer. https://doi.org/10.1158/1078-0432.CCR-15-0552
Pandit-Taskar N, O’Donoghue JA, Ruan S, Lyashchenko SK, Carrasquillo JA, Heller G, Martinez DF et al (2016) First-in-human imaging with 89Zr-Df-IAB2M anti-PSMA Minibody in patients with metastatic prostate cancer: pharmacokinetics, biodistribution, dosimetry, and lesion uptake. J Nucl Med 57(12):1858–1864. https://doi.org/10.2967/jnumed.116.176206
Phelps ME (2000) PET: the merging of biology and imaging into molecular imaging. J Nucl Med 41(4):661–681
Phelps ME, Hoffman EJ, Mullani NA, Ter-Pogossian MM, et al (1975) Journal of Nuclear Medicine 16(3):210–224.
Philpott GW, Schwarz SW, Anderson CJ, Dehdashti F, Connett JM, Zinn KR, Meares CF, Cutler PD, Welch M, Barry A, Siegel. (1995) Radioimmuno PET_64Cu.PDF. J Nucl Med 36(10):1818–1824
Pinto JT, Suffoletto BP, Berzin TM, Qiao CH, Lin S, Tong WP, May F, Mukherjee B, Heston WDW (1996) Prostate-specific membrane antigen: a novel folate hydrolase in human prostatic carcinoma cells. Clin Cancer Res 2(9):1445–1451
Reardan DT, Meares CF, Goodwin DA, McTigue M, David GS, Stone MR, Leung JP, Bartholomew RM, Frincke JM (1985) Antibodies against metal chelates. Nature 316(6025):265–268. https://doi.org/10.1038/316265a0
Reivich M, Kuhl D, Wolf A, Greenberg J, Phelps M, Ido T, Casella V et al (1979) The [18 F]Fluorodeoxyglucose method for the measurement of local cerebral glucose utilization in man. Ore Res 44. http://ahajournals.org:127
Rondon A, Ty N, Bequignat JB, Quintana M, Briat A, Witkowski T, Bouchon B et al (2017) Antibody PEGylation in Bioorthogonal Pretargeting with Trans-Cyclooctene/Tetrazine Cycloaddition: in vitro and in vivo evaluation in colorectal cancer models. Sci Rep 7(1):1–11. https://doi.org/10.1038/s41598-017-15051-y
Rösch F, Herzog H, Qaim SM (2017) The beginning and development of the Theranostic approach in nuclear medicine, as exemplified by the radionuclide pair 86Y and 90Y. Pharmaceuticals 10(2):1–28. https://doi.org/10.3390/ph10020056
Rousseau C, Goldenberg DM, Colombie M, Sebille JC, Meingan P, Ferrer L, Baumgartner P et al (2020) Initial clinical results of a novel Immuno-pet Theranostic probe in human epidermal growth factor receptor 2-negative breast cancer. J Nucl Med 61(8):1205–1211. https://doi.org/10.2967/jnumed.119.236000
Rowe SP, Pomper MG (2021) Molecular imaging in oncology: current impact and future directions. CA Cancer J Clin 0(0):1–20. https://doi.org/10.3322/caac.21713
Schoffelen R, Boerman OC, Goldenberg DM, Sharkey RM, Van Herpen ML, Franssen GM, Mcbride WJ, et al (2013) Development of an imaging-guided CEA-Pretargeted radionuclide treatment of advanced colorectal cancer: first clinical results. https://doi.org/10.1038/bjc.2013.376
Seidlin SM, Marinelli LD, Oshry E (1946) Radioactive iodine therapy: effect on functioning metastases of adenocarcinoma of the thyroid. J Am Med Assoc 132(14):838–847. https://doi.org/10.1001/jama.1946.02870490016004
Slamon DJ, Leyland-Jones B, Shak S et al (2001) Use of Chemotherapy Plus a Monoclonal Antibody Against Her2. New England J Med 344(11):783–792
Tamura K, Kurihara H, Yonemori K, Tsuda H, Suzuki J, Kono Y, Honda N et al (2013) 64Cu-DOTA-Trastuzumab PET imaging in patients with HER2-positive breast cancer. J Nucl Med 54(11):1869–1875. https://doi.org/10.2967/jnumed.112.118612
Tavaré R, Escuin-Ordinas H, Mok S, McCracken MN, Zettlitz KA, Salazar FB, Witte ON, Ribas A, Anna MW (2016) An effective Immuno-PET imaging method to monitor CD8-dependent responses to immunotherapy. Cancer Res 76(1):73–82. https://doi.org/10.1158/0008-5472.CAN-15-1707
Ter-Pogossian MM, Eichling JO, Davis DO, Welch MJ (1970) The measure in vivo of regional cerebral oxygen utilization by means of oxyhemoglobin labeled with radioactive Oxygen-15. J Clin Invest 49(2):381–391. https://doi.org/10.1172/JCI106247
Thompson AM, Jordan LB, Quinlan P, Anderson E, Skene A, Dewar JA, Purdie CA (2010) Prospective comparison of switches in biomarker status between primary and recurrent breast cancer: the breast recurrence in tissues study (BRITS). Breast Cancer Res 12(6):R92. https://doi.org/10.1186/bcr2771
Thorpe SJ, Turner C, Heath A, Feavers I, Vatn I, Natvig JB, Thompson KM (2003) Clonal analysis of a human Antimouse antibody (HAMA) response. Scand J Immunol 57(1):85–92. https://doi.org/10.1046/j.1365-3083.2003.01189.x
Tolmachev V, Vorobyeva A (2022) Cancers editorial radionuclides in diagnostics and therapy of malignant tumors: new development. https://doi.org/10.3390/cancers14020297
Touchefeu Y, Bailly C, Frampas E, Eugène T, Rousseau C, Bourgeois M, Bossard C et al (2021) Promising clinical performance of Pretargeted Immuno-PET with anti-CEA bispecific antibody and Gallium-68-labelled IMP-288 peptide for imaging colorectal cancer metastases: a pilot study. Eur J Nucl Med Mol Imaging 48(3):874–882. https://doi.org/10.1007/s00259-020-04989-3
Ulaner GA, Lyashchenko SK, Riedl C, Ruan S, Zanzonico PB, Lake D, Jhaveri K, Zeglis B, Lewis JS, O’Donoghue JA (2018) First-in-human human epidermal growth factor receptor 2–targeted imaging using 89 Zr-Pertuzumab PET/CT: dosimetry and clinical application in patients with breast cancer. J Nucl Med 59(6):900–906. https://doi.org/10.2967/jnumed.117.202010
Van Helden EJ, Elias SG, Gerritse SL, Van Es SC, Boon E, Huisman MC, Van Grieken NCT et al (2020) [89 Zr]Zr-Cetuximab PET/CT as biomarker for cetuximab monotherapy in patients with RAS wild-type advanced colorectal cancer. Eur J Nucl Med Mol Imaging 47:849–859. https://doi.org/10.1007/s00259-019-04555-6
Van Wauwe JP, De Mey JR, Goossens JG (1980) OKT3: a monoclonal anti-human T Lymphocyte antibody with potent mitogenic properties. J Immunol (Baltimore, Md.: 1950) 124(6):2708–2713. http://www.ncbi.nlm.nih.gov/pubmed/6966296
Verel I, Visser GWM, Boellaard R, Boerman OC, Van Eerd J, Snow GB, Lammertsma AA, Van Dongen. GAMS (2003) Quantitative 89Zr Immuno-PET for in vivo scouting of 90Y-labeled monoclonal antibodies in xenograft-bearing nude mice. J Nucl Med 44(10):1663–1670
Wei W, Ni D, Ehlerding EB, Luo QY, Cai W (2018) PET imaging of receptor tyrosine kinases in cancer. Mol Cancer Ther. American Association for Cancer Research Inc. https://doi.org/10.1158/1535-7163.MCT-18-0087
Zeglis BM, Lewis JS (2011) A practical guide to the construction of Radiometallated bioconjugates for positron emission tomography. Dalton Trans 40(23):6168–6195. https://doi.org/10.1039/c0dt01595d.A
Zeglis BM, Sevak KK, Reiner T, Mohindra P, Carlin SD, Zanzonico P, Weissleder R, Lewis JS (2013) A Pretargeted PET imaging strategy based on bioorthogonal Diels-Alder click chemistry. J Nucl Med 54(8):1389–1396. https://doi.org/10.2967/jnumed.112.115840
Acknowledgments
This work is financially supported by the NExT (Nantes Excellence Trajectory) initiative (call for I-SITE projects), an action of the second Programme d’Investissements d’Avenir (PIA2) launched by the French government and implemented by the ANR (reference ANR-16-IDEX-0007). This initiative takes place in the Pays de la Loire region, in Nantes Métropole.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Nature Switzerland AG
About this entry
Cite this entry
Rbah-Vidal, L. (2023). Immuno-PET as a Tool for Cancer Detection and Monitoring Response to Treatments. In: Rezaei, N. (eds) Handbook of Cancer and Immunology. Springer, Cham. https://doi.org/10.1007/978-3-030-80962-1_359-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-80962-1_359-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-80962-1
Online ISBN: 978-3-030-80962-1
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences