Abstract
Sarcopenia is a term describing the age-related loss of muscle mass, muscle strength, and/or physical function. It is a progressive, and generalized disorder that is multifactorial in origin, and associated with a considerable burden of morbidity, in terms of falls and fractures, cognitive impairment, frailty, disability, and hospitalizations. The health economic burden of sarcopenia is considerable and looks set to continue to increase as the proportion of older persons in the population rises. In this chapter, we briefly describe some of the most commonly used existing definitions of sarcopenia and review the prevalence of sarcopenia in various settings (community-dwelling adults and hospitalized individuals). We review the modifiable and non-modifiable risk factors, which include age, gender, nutrition, exercise, smoking, or alcohol consumption. Finally, we review the burden of sarcopenia on healthcare costs in the hospital setting, from a societal perspective. Further high-quality research in the field of sarcopenia is warranted, using standardized operational definitions of sarcopenia, to identify factors that could be leveraged to prevent or slow the onset or progression of sarcopenia. Systematic screening of older individuals is also warranted to identify persons with decreasing muscle strength, who may be at risk of sarcopenia and its associated negative consequences.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1.1 Introduction
The term sarcopenia derives from the Greek “sarx” meaning flesh and “penia” meaning loss or poverty. It was first introduced by Rosenberg [1] in 1997 to describe the age-related loss of skeletal muscle mass and function, after seminal publications by Evans and Campbell regarding the declining functional status observed in older individuals with changes in body composition [2,3,4]. Initially, it was thought that only muscle wasting occurred in elderly individuals, but sarcopenia is now recognized as a complex concept that involves not only loss of muscle mass but also decreased muscle strength, and a resulting decline in functional capacity. Functional parameters came to be included in the definition because they have consistently been shown to be a stronger predictor of outcomes than muscle mass alone [5]. Indeed, sarcopenia is a progressive and generalized skeletal disorder that is not necessarily synonymous with leanness. It may also be present in overweight and obese individuals, a condition now termed “sarcopenic obesity” (see Chap. 14). A distinction may also be made between primary, or age-related sarcopenia, and secondary sarcopenia, which is more disease-related.
1.2 Definitions
Over the years since its first description, there has been a steady increase in research and publications about sarcopenia, and a number of groups and societies have published operational definitions of sarcopenia for use in clinical practice and in research settings. Following a meeting of a group of geriatricians and scientists from academia and industry in 2009, the International Working Group on Sarcopenia published a definition of sarcopenia, namely the “age-associated loss of skeletal muscle mass and function” [6]. In 2010, the European Working Group on Sarcopenia in Older People (EWGSOP) developed a clinical definition together with consensus diagnostic criteria for age-related sarcopenia, which recommended that both low muscle mass and low muscle function (i.e., strength or performance) be considered [7]. The EWGSOP further defined stages of severity, with a gradual scale from pre-sarcopenia to sarcopenia to severe sarcopenia. The EWGSOP consensus was updated in 2019 to reflect the growing body of evidence that has emerged since its first publication [8]. In parallel, professional societies in Asia also worked to prepare consensus definitions on sarcopenia, due to the fact that the cultural, lifestyle, and anthropometric differences call for specific considerations when diagnosing sarcopenia in people of Asian descent. In this regard, the Asian Working Group on Sarcopenia published a diagnostic algorithm for sarcopenia using cut-offs and reference levels derived from Asian populations [9], which was updated in 2019, revising some of the component criteria and cut-offs [10]. In 2011, the Society on Sarcopenia, Cachexia and Wasting Disorders Trialist Workshop convened a consensus conference, which concluded that “sarcopenia, i.e., reduced muscle mass, with limited mobility should be considered an important clinical entity” [11]. Their definition was based on walk speed on the 6-min walk test and lean appendicular mass corrected for height. Finally, the Foundation for the National Institutes of Health (FNIH) Sarcopenia Project also developed an operational definition of sarcopenia based on data from 9 sources totalling over 26,000 individuals to identify clinically relevant and independently validated thresholds that could be used to identify participants for clinical trials, and individuals with significant functional limitation [12].
The abundance of research in the field of sarcopenia culminated in the recognition of sarcopenia as a distinct disease, with its inclusion in the International Classification of Diseases tenth Revision (ICD-10) in 2016 (under the ICD code M62.84) [13, 14]. This important step meant that the condition could be cited on medical records, death certificates, and other data sources, which can help harmonize practices, compare data, and promote research. The recognition of sarcopenia as a disease entity also provides additional stimulus for pharmaceutical companies to invest in research and development in this area, by allowing for billing and reimbursement possibilities.
The various definitions of sarcopenia developed by professional societies, as well as other combinations of criteria used in the literature to define sarcopenia, will be discussed in greater detail in Chap. 3. Suffice to say, however, that there are wide variations across all these definitions in the components included, the methods used to measure these components, and the cut-off values used to distinguish pathological states. For the measurement of muscle mass, dual-energy X-ray absorptiometry (DXA) is a widely available, noninvasive method for determining muscle quantity (i.e., total body lean tissue mass or appendicular skeletal muscle mass) and is considered by many as the gold standard. However, inconsistencies may exist across measurements performed with different machines, rendering comparison difficult [15]. Other methods used to assess muscle mass include bioelectrical impedance analysis (BIA), computed tomography (CT), or magnetic resonance imaging (MRI), as well as simple anthropometric measures. Muscle strength can be measured easily and inexpensively by assessing hand-grip strength with a calibrated handheld dynamometer. For patients in whom disability of any type precludes measurement of hand-grip strength, leg strength can be used as a proxy, for example, via the chair stand test, timed chair stand test, or the timed up-and-go test. Other measures of mobility, such as gait speed, or walk test performance are also widely used. The heterogeneity of definitions, criteria, measurement methods, and cut-off values makes it extremely difficult to compare estimates of sarcopenia prevalence between studies. Estimates are also affected by the populations used to define the normal range reference values and the setting in which those cut-offs are applied (e.g., community-dwelling adults, versus nursing home residents, versus acute hospital care) [16]. This underlines the need for a consensual definition, to enable comparison of the burden of disease worldwide, as the lack of agreement between definitions hampers the integration of sarcopenia into clinical practice.
1.3 Prevalence of Sarcopenia
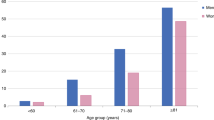
The prevalence of sarcopenia is notoriously difficult to compare across studies, in view of this heterogeneity of definitions and measurement possibilities. Nevertheless, recent years have seen a striking increase in the number of publications investigating sarcopenia prevalence, risk factors, and outcomes. Many of these are now using established definitions, thus allowing for some comparison of rates across studies as the body of evidence grows. Table 1.1 displays a selection of prevalence estimates from recent publications in various populations. It can be seen that there are wide variations in reported rates between studies, and even within studies, when different criteria are used to define sarcopenia. The burgeoning volume of publications on the prevalence of sarcopenia has led to ever more precise estimates, and reports providing pooled estimates from systematic reviews and/or meta-analysis. For example, in a systematic review and meta-analysis, Mayhew et al. examined 109 studies using 8 different sarcopenia definitions (including the EWGSOP, AWGS, FNIH, and IWGS definitions), with a total of 227 individual prevalence estimates in community-dwelling older adults (>60 years) without specific health conditions [24]. Overall, estimated prevalence ranged from 9.9 to 40.4% and was lowest with the EWGSOP/AWGS (12.9%, 95% CI: 9.9, 15.9%), IWGS (9.9%, 95% CI: 3.2, 16.6%), and FNIH (18.6%, 95% CI: 11.8, 25.5%) definitions [24]. In another systematic review and meta-analysis, Shafiee et al. included 35 population-based studies reporting the prevalence of sarcopenia in healthy adults aged ≥60 years from different regions of the world, using the EWGSOP, IWGS, and AWGS definitions [25]. They reported an overall prevalence of 10% in both men and women, although estimates ranged from 0.35 to 36.6% across studies, depending on the definition used. There was significant heterogeneity between men and women in Shafiee’s meta-analysis [25]. Furthermore, analysis by region showed that individuals in non-Asian countries were more likely to have sarcopenia than those from Asian countries, in both genders (11% vs. 10% in men, 13% vs. 9% in women) [25].
Even though it is almost impossible to pinpoint an actual rate of prevalence of sarcopenia, projections indicate that the rate is rising and looks set to continue increasing in the future, as worldwide population ageing adds growing numbers of older people to the pool of potentially sarcopenic individuals. In a study using the various diagnostic cut-offs proposed by the EWGSOP for lean mass, muscle strength, and gait speed, Ethgen et al. applied interpolated age- and gender-specific estimates of sarcopenia prevalence to the Eurostat population projections for Europe up to 2045 [36]. From a previous publication comparing prevalence rates at different cut-offs [37], Ethgen et al. chose first the definition yielding the lowest prevalence estimates, applied it to projected population estimates for Europe, and found that it would correspond to a 72.4% increase in overall prevalence of sarcopenia in the elderly, rising from 11.1% in 2016 to 12.9% in 2045. Applying the definition yielding the highest prevalence estimates, overall prevalence rates were projected to increase from 20.2% in 2016 to 22.3% in 2045 [36]. These projections portend a substantial burden of sarcopenia in coming decades, which will have important repercussions for society in terms of healthcare delivery and costs.
1.4 Risk Factors
Numerous risk factors for sarcopenia have been reported in the literature, some of which are non-modifiable, such as age and gender; others are modifiable and exert their influence across the life course. Among the non-modifiable risk factors, the most consistent body of evidence supports an increasing risk of sarcopenia with older age [5, 18, 22, 27, 29, 38]. Indeed, muscle mass begins to decline around the fifth decade of life, with an annual decline rate of 1–2% [39,40,41], accelerating in the sixth and subsequent decades to reach a loss of around 15% per decade beyond the age of 70 [42, 43]. Regarding gender, conflicting results have been reported regarding the difference in risk in men and women, but consensus seems to be emerging in favor of an increased risk of sarcopenia in men. Landi et al. reported a 13-fold increased risk in male nursing home residents (odds ratio (OR) 13.39; 95% CI 3.51–50.63) [44], while Nguyen et al. reported a twofold increase in risk of sarcopenia among male outpatients at a geriatric hospital (OR 2.03, 95% CI 1.29–3.21) [22]. Despite the existence of differences in baseline strength between the sexes, with men having greater baseline strength than women, it has been reported that muscle strength declines to a greater degree in men, thus potentially contributing to their higher risk of sarcopenia [44,45,47].
Concerning modifiable risk factors, nutrition and lifestyle behaviors (notably exercise) appear to be associated with muscle mass and strength in older age [48, 49]. Older people experience a natural decline in energy requirements [50], which may be accompanied by declining appetite, impaired taste or smell, and changes in gastrointestinal motility and digestion [51]. If also compounded by functional impairment reducing the ability to prepare food, or social isolation, which may reduce the desire to eat or enjoyment of mealtimes, all these features come together in a vicious circle that may lead to loss of weight and muscle mass and strength, putting older individuals at risk of malnutrition and in turn, sarcopenia and/or frailty [48, 49]. The contribution of adequate nutrition to healthy aging has long been established [52], and there have been a number of studies examining the effects of various dietary components and patterns on sarcopenia and its constituent elements. However, apart from the obvious need to ensure that all older adults have adequate nutrition both in terms of quantity and quality, the potential of individual dietary patterns to affect outcome remains unclear. Indeed, there have been conflicting findings regarding the association between protein intake and muscle strength, for example, although observational evidence tends to suggest that both strength and function are improved with increased protein intake [48]. In the same way, it is difficult to distinguish the effects of individual nutrients, such as antioxidants and omega-3 fatty acids, although overall, the best evidence supports the benefits of the Mediterranean dietary pattern in terms of functional status and incident disability [48].
The effect of exercise in reducing the negative impact of sarcopenia has been demonstrated by several studies [52,53,55]. In a systematic umbrella review, Beckwee et al. investigated the efficacy of different exercise interventions to counter sarcopenia in older adults [53]. They found high-quality evidence in favor of a positive and significant effect of resistance training on muscle mass, muscle strength, and physical performance from a total of 14 systematic reviews, of which 7 performed meta-analysis. Based on the evidence from their review, these authors suggest that benefits in terms of muscle mass, muscle strength, and gait speed can be expected with high-intensity resistance training, which they recommend for at least 6–12 weeks, in order to achieve these levels of improvement [53]. Similarly, Lai et al. compared the effects of exercise interventions on lean body mass, muscle strength, and physical performance in a network meta-analysis and found that resistance training (of a minimum 6 weeks duration) was the most effective intervention in improving muscle strength in older individuals [54].
Other risk factors have been less extensively investigated. Nonetheless, a meta-analysis of 12 studies totalling 22,515 participants found smoking to be an independent risk factor for sarcopenia (OR 1.20 (95% CI 1.06–1.35) in men and 1.21 (95% CI 0.92–1.59) in women) [56]. The same group also performed a meta-analysis of 13 studies including 13,155 participants to investigate the effect of alcohol on sarcopenia, but their findings did not support the hypothesis that alcohol consumption could be a risk factor for sarcopenia [57].
Other factors that have been shown to be associated with sarcopenia include age-related loss of motor-neuron end plates [58], loss of anabolic hormones and insulin resistance [42, 59], diabetes [60], obesity/waist circumference [46], level of education [29], and dependency [27, 44].
1.5 The Health Economic Burden of Sarcopenia
Sarcopenia is associated with an increased risk of falls and fractures [61, 62], frailty [63], disability [64], and cognitive impairment [65]. Low grip strength has been shown to be associated with increased morbidity and mortality [66, 67], and a meta-analysis of 11 studies investigating the impact of EWGSOP-defined sarcopenia on mortality found a more than threefold increase in the risk of mortality among sarcopenic subjects (pooled OR 3.596 (95% CI 2.96–4.37)) [68]. These deleterious outcomes can in turn translate into extended recovery time, longer length of hospital stay, and increased medical costs [68, 69].
In community-dwelling adults in the Netherlands, Mijnarends et al. reported that the mean healthcare costs of individuals with sarcopenia were significantly higher than those of non-sarcopenic subjects (€ 4325, 95% CI € 3198–€5471 vs. €1533, 95% CI €1153–€1912, respectively), mainly driven by the living situation (i.e., residential care) [70]. In the hospital setting, two studies from Portugal investigated the costs of hospitalization associated with sarcopenia. Sousa et al. assessed the hospitalization costs in 656 medical and surgical patients (24.2% sarcopenic) using diagnosis-related group codes at discharge [71]. They found that sarcopenic patients were generally older and had a longer length of stay, resulting in a median (interquartile) cost of € 3151 (€ 4175) per sarcopenic patient, compared to non-sarcopenic patients (median (IQR) € 2170 (€ 2515), p < 0.001) [71]. After adjustment for confounders, the economic impact of sarcopenia on hospitalization cost, i.e., the incremental cost per patient, in the overall sample was estimated at € 1117 (95% CI €644–1588), and sarcopenia was estimated to increase hospitalization costs by 39.2% in those with no comorbidities and by 54.3% in those with comorbidities [71]. In the second Portuguese study, Antunes et al. assessed hospitalization costs among 201 hospitalized older adults in a general hospital [72]. After adjustment, both sarcopenia (OR = 5.70, 95% CI 1.57–20.71) and low muscle strength alone (OR = 2.40, 95% CI 1.12–5.15) were associated with increased hospital costs. From a societal perspective, Janssen et al. evaluated the costs of sarcopenia in a representative sample of US adults aged 60 years and older, from the NHANES III and National Medical Care Utilization and Expenditures Survey (NMCUES) datasets [73]. They estimated that the direct healthcare cost attributable to sarcopenia in the USA in 2000 was $18.5 billion ($10.8 billion in men, $7.7 billion in women), representing 1.5% of total healthcare expenditure, with sensitivity analyses indicating that the cost could be as low as $11.8 billion and as high as $26.2 billion. They further estimated that a 10% reduction in the prevalence of sarcopenia would result in savings of $1.1 billion (dollar value in the year 2000) per year in US healthcare costs [73]. A more recent study from the USA updates this information and shows that costs are already on the rise; Goates et al. performed a retrospective economic burden study among 4011 adults aged 40 years and over from the NHANES dataset [74], of whom 15.1% were sarcopenic. They reported an annual total cost of hospitalization for individuals with sarcopenia of $40.4 billion, with an average estimated marginal cost increase in annual hospital spending of $2315 per sarcopenic individual, compared to those with normal muscle mass and function. In addition, they reported that individuals with sarcopenia had an almost twofold increase in the risk of hospitalization (OR 1.94, p < 0.001) and more hospital stays on average, compared to those without sarcopenia [74]. There is wide heterogeneity among studies on the economic burden of sarcopenia, with different approaches used to estimate costs, different time horizons for measurements, and different definitions of sarcopenia. With populating ageing continuing its onward march around the world, there is a compelling need to continue providing up-to-date estimates of sarcopenia-associated healthcare costs, particularly using standardized definitions and cost analysis parameters, not least to prepare for the substantial burden that this will represent on healthcare systems in the coming decades. The alarming estimates of the burden that sarcopenia represents on healthcare also underscores the need to focus preventive measures on preserving muscle mass, strength, and function as long as possible into older age.
1.6 Conclusion
Sarcopenia is characterized by an age-related loss of muscle mass, muscle strength, and/or physical function. It is associated with a high risk of morbidity and mortality, poor clinical outcomes, and increased events, such as falls, fractures, and hospitalizations. It represents a significant burden on healthcare systems worldwide, which looks set to increase in the coming decades. A strong research agenda is warranted to expand our knowledge of the etiological factors involved in the development of sarcopenia, and which could be leveraged to prevent or slow the onset of sarcopenia, or its progression to more severe forms. Systematic screening of older individuals is warranted to detect those with lower muscle strength, with a view to initiating early interventions to retard sarcopenia. Resistance training, of a minimum 6 weeks duration, has been shown to be most effective in achieving improvements in muscle strength in older adults. The benefit of adequate nutrition in contributing to healthy ageing has also been well established. Other interventions to promote healthy ageing and preserve muscle mass, strength and function into older age are warranted, to counter the effects of ageing and maintain functional capacity as long as possible. The considerable economic burden of sarcopenia on healthcare costs justifies the implementation of preventive measures, perhaps over the life course and almost certainly warranted from midlife onwards, in order to stem the tide of negative consequences that flows from the presence of sarcopenia.
References
Rosenberg IH. Sarcopenia: origins and clinical relevance. J Nutr. 1997;127:990S–1S. https://doi.org/10.1093/jn/127.5.990S.
Evans WJ, Campbell WW. Sarcopenia and age-related changes in body composition and functional capacity. J Nutr. 1993;123:465–8. https://doi.org/10.1093/jn/123.suppl_2.465.
Evans WJ. What is sarcopenia? J Gerontol A Biol Sci Med Sci. 1995;50 Spec No:5–8. https://doi.org/10.1093/gerona/50a.special_issue.5.
Evans WJ. Skeletal muscle loss: cachexia, sarcopenia, and inactivity. Am J Clin Nutr. 2010;91:1123S–7S. https://doi.org/10.3945/ajcn.2010.28608A.
Cruz-Jentoft AJ, Sayer AA. Sarcopenia. Lancet. 2019;393:2636–46. https://doi.org/10.1016/S0140-6736(19)31138-9.
Fielding RA, Vellas B, Evans WJ, Bhasin S, Morley JE, Newman AB, Abellan van Kan G, Andrieu S, Bauer J, Breuille D, Cederholm T, Chandler J, De Meynard C, Donini L, Harris T, Kannt A, Keime Guibert F, Onder G, Papanicolaou D, Rolland Y, Rooks D, Sieber C, Souhami E, Verlaan S, Zamboni M. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International Working Group on Sarcopenia. J Am Med Dir Assoc. 2011;12:249–56. https://doi.org/10.1016/j.jamda.2011.01.003.
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinkova E, Vandewoude M, Zamboni M, European Working Group on Sarcopenia in Older P. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in older people. Age Ageing. 2010;39:412–23. https://doi.org/10.1093/ageing/afq034.
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyere O, Cederholm T, Cooper C, Landi F, Rolland Y, Sayer AA, Schneider SM, Sieber CC, Topinkova E, Vandewoude M, Visser M, Zamboni M, Writing Group for the European Working Group on Sarcopenia in Older P, the Extended Group for E. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48:16–31. https://doi.org/10.1093/ageing/afy169.
Chen LK, Liu LK, Woo J, Assantachai P, Auyeung TW, Bahyah KS, Chou MY, Chen LY, Hsu PS, Krairit O, Lee JS, Lee WJ, Lee Y, Liang CK, Limpawattana P, Lin CS, Peng LN, Satake S, Suzuki T, Won CW, Wu CH, Wu SN, Zhang T, Zeng P, Akishita M, Arai H. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc. 2014;15:95–101. https://doi.org/10.1016/j.jamda.2013.11.025.
Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, Jang HC, Kang L, Kim M, Kim S, Kojima T, Kuzuya M, Lee JSW, Lee SY, Lee WJ, Lee Y, Liang CK, Lim JY, Lim WS, Peng LN, Sugimoto K, Tanaka T, Won CW, Yamada M, Zhang T, Akishita M, Arai H. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. 2020;21:300–307.e2. https://doi.org/10.1016/j.jamda.2019.12.012.
Morley JE, Abbatecola AM, Argiles JM, Baracos V, Bauer J, Bhasin S, Cederholm T, Coats AJ, Cummings SR, Evans WJ, Fearon K, Ferrucci L, Fielding RA, Guralnik JM, Harris TB, Inui A, Kalantar-Zadeh K, Kirwan BA, Mantovani G, Muscaritoli M, Newman AB, Rossi-Fanelli F, Rosano GM, Roubenoff R, Schambelan M, Sokol GH, Storer TW, Vellas B, von Haehling S, Yeh SS, Anker SD, Society on Sarcopenia C, Wasting Disorders Trialist W. Sarcopenia with limited mobility: an international consensus. J Am Med Dir Assoc. 2011;12:403–9. https://doi.org/10.1016/j.jamda.2011.04.014.
Studenski SA, Peters KW, Alley DE, Cawthon PM, McLean RR, Harris TB, Ferrucci L, Guralnik JM, Fragala MS, Kenny AM, Kiel DP, Kritchevsky SB, Shardell MD, Dam TT, Vassileva MT. The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. J Gerontol A Biol Sci Med Sci. 2014;69:547–58. https://doi.org/10.1093/gerona/glu010.
Anker SD, Morley JE, von Haehling S. Welcome to the ICD-10 code for sarcopenia. J Cachexia Sarcopenia Muscle. 2016;7:512–4. https://doi.org/10.1002/jcsm.12147.
Cao L, Morley JE. Sarcopenia is recognized as an independent condition by an international classification of disease, tenth revision, clinical modification (ICD-10-CM) code. J Am Med Dir Assoc. 2016;17:675–7. https://doi.org/10.1016/j.jamda.2016.06.001.
Buckinx F, Landi F, Cesari M, Fielding RA, Visser M, Engelke K, Maggi S, Dennison E, Al-Daghri NM, Allepaerts S, Bauer J, Bautmans I, Brandi ML, Bruyere O, Cederholm T, Cerreta F, Cherubini A, Cooper C, Cruz-Jentoft A, McCloskey E, Dawson-Hughes B, Kaufman JM, Laslop A, Petermans J, Reginster JY, Rizzoli R, Robinson S, Rolland Y, Rueda R, Vellas B, Kanis JA. Pitfalls in the measurement of muscle mass: a need for a reference standard. J Cachexia Sarcopenia Muscle. 2018;9:269–78. https://doi.org/10.1002/jcsm.12268.
Churilov I, Churilov L, MacIsaac RJ, Ekinci EI. Systematic review and meta-analysis of prevalence of sarcopenia in post acute inpatient rehabilitation. Osteoporos Int. 2018;29:805–12. https://doi.org/10.1007/s00198-018-4381-4.
Purcell SA, MacKenzie M, Barbosa-Silva TG, Dionne IJ, Ghosh S, Olobatuyi OV, Siervo M, Ye M, Prado CM. Sarcopenia prevalence using different definitions in older community-dwelling Canadians. J Nutr Health Aging. 2020;24:783–90. https://doi.org/10.1007/s12603-020-1427-z.
Martone AM, Marzetti E, Salini S, Zazzara MB, Santoro L, Tosato M, Picca A, Calvani R, Landi F. Sarcopenia identified according to the EWGSOP2 definition in community-living people: prevalence and clinical features. J Am Med Dir Assoc. 2020;21:1470–4. https://doi.org/10.1016/j.jamda.2020.03.007.
Ligthart-Melis GC, Luiking YC, Kakourou A, Cederholm T, Maier AB, de van der Schueren MAE. Frailty, sarcopenia, and malnutrition frequently (co-)occur in hospitalized older adults: a systematic review and meta-analysis. J Am Med Dir Assoc. 2020;21:1216–28. https://doi.org/10.1016/j.jamda.2020.03.006.
Pang BWJ, Wee SL, Lau LK, Jabbar KA, Seah WT, Ng DHM, Ling Tan QL, Chen KK, Jagadish MU, Ng TP. Prevalence and associated factors of sarcopenia in Singaporean adults-the Yishun study. J Am Med Dir Assoc. 2021;22(4):885.e1–10. https://doi.org/10.1016/j.jamda.2020.05.029.
Wearing J, Konings P, de Bie RA, Stokes M, de Bruin ED. Prevalence of probable sarcopenia in community-dwelling older Swiss people—a cross-sectional study. BMC Geriatr. 2020;20:307. https://doi.org/10.1186/s12877-020-01718-1.
Nguyen TN, Nguyen TN, Nguyen AT, Nguyen TX, Nguyen HTT, Nguyen TTH, Pham T, Vu HTT. Prevalence of sarcopenia and its associated factors in patients attending geriatric clinics in Vietnam: a cross-sectional study. BMJ Open. 2020;10:e037630. https://doi.org/10.1136/bmjopen-2020-037630.
Makizako H, Nakai Y, Tomioka K, Taniguchi Y. Prevalence of sarcopenia defined using the Asia Working Group for Sarcopenia criteria in Japanese community-dwelling older adults: a systematic review and meta-analysis. Phys Ther Res. 2019;22:53–7. https://doi.org/10.1298/ptr.R0005.
Mayhew AJ, Amog K, Phillips S, Parise G, McNicholas PD, de Souza RJ, Thabane L, Raina P. The prevalence of sarcopenia in community-dwelling older adults, an exploration of differences between studies and within definitions: a systematic review and meta-analyses. Age Ageing. 2019;48:48–56. https://doi.org/10.1093/ageing/afy106.
Shafiee G, Keshtkar A, Soltani A, Ahadi Z, Larijani B, Heshmat R. Prevalence of sarcopenia in the world: a systematic review and meta- analysis of general population studies. J Diabetes Metab Disord. 2017;16:21. https://doi.org/10.1186/s40200-017-0302-x.
Kim H, Hirano H, Edahiro A, Ohara Y, Watanabe Y, Kojima N, Kim M, Hosoi E, Yoshida Y, Yoshida H, Shinkai S. Sarcopenia: prevalence and associated factors based on different suggested definitions in community-dwelling older adults. Geriatr Gerontol Int. 2016;16(Suppl 1):110–22. https://doi.org/10.1111/ggi.12723.
Sousa AS, Guerra RS, Fonseca I, Pichel F, Amaral TF. Sarcopenia among hospitalized patients—a cross-sectional study. Clin Nutr. 2015;34:1239–44. https://doi.org/10.1016/j.clnu.2014.12.015.
Cruz-Jentoft AJ, Landi F, Schneider SM, Zuniga C, Arai H, Boirie Y, Chen LK, Fielding RA, Martin FC, Michel JP, Sieber C, Stout JR, Studenski SA, Vellas B, Woo J, Zamboni M, Cederholm T. Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing. 2014;43:748–59. https://doi.org/10.1093/ageing/afu115.
Volpato S, Bianchi L, Cherubini A, Landi F, Maggio M, Savino E, Bandinelli S, Ceda GP, Guralnik JM, Zuliani G, Ferrucci L. Prevalence and clinical correlates of sarcopenia in community-dwelling older people: application of the EWGSOP definition and diagnostic algorithm. J Gerontol A Biol Sci Med Sci. 2014;69:438–46. https://doi.org/10.1093/gerona/glt149.
Lee WJ, Liu LK, Peng LN, Lin MH, Chen LK, Group IR. Comparisons of sarcopenia defined by IWGS and EWGSOP criteria among older people: results from the I-Lan longitudinal aging study. J Am Med Dir Assoc. 2013;14(528):e521–7. https://doi.org/10.1016/j.jamda.2013.03.019.
Pongchaiyakul C, Limpawattana P, Kotruchin P, Rajatanavin R. Prevalence of sarcopenia and associated factors among Thai population. J Bone Miner Metab. 2013;31:346–50. https://doi.org/10.1007/s00774-013-0422-4.
Janssen I. Influence of sarcopenia on the development of physical disability: the cardiovascular health study. J Am Geriatr Soc. 2006;54:56–62. https://doi.org/10.1111/j.1532-5415.2005.00540.x.
Rolland Y, Lauwers-Cances V, Cournot M, Nourhashemi F, Reynish W, Riviere D, Vellas B, Grandjean H. Sarcopenia, calf circumference, and physical function of elderly women: a cross-sectional study. J Am Geriatr Soc. 2003;51:1120–4. https://doi.org/10.1046/j.1532-5415.2003.51362.x.
Lauretani F, Russo CR, Bandinelli S, Bartali B, Cavazzini C, Di Iorio A, Corsi AM, Rantanen T, Guralnik JM, Ferrucci L. Age-associated changes in skeletal muscles and their effect on mobility: an operational diagnosis of sarcopenia. J Appl Physiol (1985). 2003;95:1851–60. https://doi.org/10.1152/japplphysiol.00246.2003.
Baumgartner RN, Koehler KM, Gallagher D, Romero L, Heymsfield SB, Ross RR, Garry PJ, Lindeman RD. Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol. 1998;147:755–63. https://doi.org/10.1093/oxfordjournals.aje.a009520.
Ethgen O, Beaudart C, Buckinx F, Bruyere O, Reginster JY. The future prevalence of sarcopenia in Europe: a claim for public health action. Calcif Tissue Int. 2017;100:229–34. https://doi.org/10.1007/s00223-016-0220-9.
Beaudart C, Reginster JY, Slomian J, Buckinx F, Locquet M, Bruyere O. Prevalence of sarcopenia: the impact of different diagnostic cut-off limits. J Musculoskelet Neuronal Interact. 2014;14:425–31.
Iannuzzi-Sucich M, Prestwood KM, Kenny AM. Prevalence of sarcopenia and predictors of skeletal muscle mass in healthy, older men and women. J Gerontol A Biol Sci Med Sci. 2002;57:M772–7. https://doi.org/10.1093/gerona/57.12.m772.
von Haehling S, Morley JE, Anker SD. An overview of sarcopenia: facts and numbers on prevalence and clinical impact. J Cachexia Sarcopenia Muscle. 2010;1:129–33. https://doi.org/10.1007/s13539-010-0014-2.
Abellan van Kan G. Epidemiology and consequences of sarcopenia. J Nutr Health Aging. 2009;13:708–12. https://doi.org/10.1007/s12603-009-0201-z.
Doherty TJ. Invited review: aging and sarcopenia. J Appl Physiol (1985). 2003;95:1717–27. https://doi.org/10.1152/japplphysiol.00347.2003.
Kim TN, Choi KM. Sarcopenia: definition, epidemiology, and pathophysiology. J Bone Metab. 2013;20:1–10. https://doi.org/10.11005/jbm.2013.20.1.1.
Grimby G, Saltin B. The ageing muscle. Clin Physiol. 1983;3:209–18. https://doi.org/10.1111/j.1475-097x.1983.tb00704.x.
Landi F, Liperoti R, Fusco D, Mastropaolo S, Quattrociocchi D, Proia A, Russo A, Bernabei R, Onder G. Prevalence and risk factors of sarcopenia among nursing home older residents. J Gerontol A Biol Sci Med Sci. 2012;67:48–55. https://doi.org/10.1093/gerona/glr035.
Fuggle N, Shaw S, Dennison E, Cooper C. Sarcopenia. Best Pract Res Clin Rheumatol. 2017;31:218–42. https://doi.org/10.1016/j.berh.2017.11.007.
Shaw SC, Dennison EM, Cooper C. Epidemiology of sarcopenia: determinants throughout the lifecourse. Calcif Tissue Int. 2017;101:229–47. https://doi.org/10.1007/s00223-017-0277-0.
Goodpaster BH, Park SW, Harris TB, Kritchevsky SB, Nevitt M, Schwartz AV, Simonsick EM, Tylavsky FA, Visser M, Newman AB. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci. 2006;61:1059–64. https://doi.org/10.1093/gerona/61.10.1059.
Robinson SM, Reginster JY, Rizzoli R, Shaw SC, Kanis JA, Bautmans I, Bischoff-Ferrari H, Bruyere O, Cesari M, Dawson-Hughes B, Fielding RA, Kaufman JM, Landi F, Malafarina V, Rolland Y, van Loon LJ, Vellas B, Visser M, Cooper C, ESCEO working group. Does nutrition play a role in the prevention and management of sarcopenia? Clin Nutr. 2018;37:1121–32. https://doi.org/10.1016/j.clnu.2017.08.016.
Welch AA. Nutritional influences on age-related skeletal muscle loss. Proc Nutr Soc. 2014;73:16–33. https://doi.org/10.1017/S0029665113003698.
Wakimoto P, Block G. Dietary intake, dietary patterns, and changes with age: an epidemiological perspective. J Gerontol A Biol Sci Med Sci. 2001;56 Spec No 2:65–80. https://doi.org/10.1093/gerona/56.suppl_2.65.
Malafarina V, Uriz-Otano F, Gil-Guerrero L, Iniesta R. The anorexia of ageing: physiopathology, prevalence, associated comorbidity and mortality. A systematic review. Maturitas. 2013;74:293–302. https://doi.org/10.1016/j.maturitas.2013.01.016.
Kadoch MA. The power of nutrition as medicine. Prev Med. 2012;55:80. https://doi.org/10.1016/j.ypmed.2012.04.013.
Beckwee D, Delaere A, Aelbrecht S, Baert V, Beaudart C, Bruyere O, de Saint-Hubert M, Bautmans I. Exercise interventions for the prevention and treatment of sarcopenia. A systematic umbrella review. J Nutr Health Aging. 2019;23:494–502. https://doi.org/10.1007/s12603-019-1196-8.
Lai CC, Tu YK, Wang TG, Huang YT, Chien KL. Effects of resistance training, endurance training and whole-body vibration on lean body mass, muscle strength and physical performance in older people: a systematic review and network meta-analysis. Age Ageing. 2018;47:367–73. https://doi.org/10.1093/ageing/afy009.
Law TD, Clark LA, Clark BC. Resistance exercise to prevent and manage sarcopenia and dynapenia. Annu Rev Gerontol Geriatr. 2016;36:205–28. https://doi.org/10.1891/0198-8794.36.205.
Steffl M, Bohannon RW, Petr M, Kohlikova E, Holmerova I. Relation between cigarette smoking and sarcopenia: meta-analysis. Physiol Res. 2015;64:419–26. https://doi.org/10.33549/physiolres.932802.
Steffl M, Bohannon RW, Petr M, Kohlikova E, Holmerova I. Alcohol consumption as a risk factor for sarcopenia—a meta-analysis. BMC Geriatr. 2016;16:99. https://doi.org/10.1186/s12877-016-0270-x.
Drey M, Krieger B, Sieber CC, Bauer JM, Hettwer S, Bertsch T, Group DS. Motoneuron loss is associated with sarcopenia. J Am Med Dir Assoc. 2014;15:435–9. https://doi.org/10.1016/j.jamda.2014.02.002.
Morley JE, Anker SD, von Haehling S. Prevalence, incidence, and clinical impact of sarcopenia: facts, numbers, and epidemiology-update 2014. J Cachexia Sarcopenia Muscle. 2014;5:253–9. https://doi.org/10.1007/s13539-014-0161-y.
Kim TN, Park MS, Yang SJ, Yoo HJ, Kang HJ, Song W, Seo JA, Kim SG, Kim NH, Baik SH, Choi DS, Choi KM. Prevalence and determinant factors of sarcopenia in patients with type 2 diabetes: the Korean Sarcopenic Obesity Study (KSOS). Diabetes Care. 2010;33:1497–9. https://doi.org/10.2337/dc09-2310.
Schaap LA, van Schoor NM, Lips P, Visser M. Associations of sarcopenia definitions, and their components, with the incidence of recurrent falling and fractures: the longitudinal aging study Amsterdam. J Gerontol A Biol Sci Med Sci. 2018;73:1199–204. https://doi.org/10.1093/gerona/glx245.
Zhang Y, Hao Q, Ge M, Dong B. Association of sarcopenia and fractures in community-dwelling older adults: a systematic review and meta-analysis of cohort studies. Osteoporos Int. 2018;29:1253–62. https://doi.org/10.1007/s00198-018-4429-5.
Syddall H, Cooper C, Martin F, Briggs R, Aihie Sayer A. Is grip strength a useful single marker of frailty? Age Ageing. 2003;32:650–6. https://doi.org/10.1093/ageing/afg111.
Xu W, Chen T, Cai Y, Hu Y, Fan L, Wu C. Sarcopenia in community-dwelling oldest old is associated with disability and poor physical function. J Nutr Health Aging. 2020;24:339–45. https://doi.org/10.1007/s12603-020-1325-4.
Peng TC, Chen WL, Wu LW, Chang YW, Kao TW. Sarcopenia and cognitive impairment: a systematic review and meta-analysis. Clin Nutr. 2020;39:2695–701. https://doi.org/10.1016/j.clnu.2019.12.014.
Cooper R, Kuh D, Cooper C, Gale CR, Lawlor DA, Matthews F, Hardy R, Falcon THAS. Objective measures of physical capability and subsequent health: a systematic review. Age Ageing. 2011;40:14–23. https://doi.org/10.1093/ageing/afq117.
Cooper R, Kuh D, Hardy R, Mortality Review G, Falcon THAS. Objectively measured physical capability levels and mortality: systematic review and meta-analysis. BMJ. 2010;341:c4467. https://doi.org/10.1136/bmj.c4467.
Beaudart C, Zaaria M, Pasleau F, Reginster JY, Bruyere O. Health outcomes of sarcopenia: a systematic review and meta-analysis. PLoS One. 2017;12:e0169548. https://doi.org/10.1371/journal.pone.0169548.
Norman K, Otten L. Financial impact of sarcopenia or low muscle mass—a short review. Clin Nutr. 2019;38:1489–95. https://doi.org/10.1016/j.clnu.2018.09.026.
Mijnarends DM, Schols JMGA, Halfens RJG, Meijers JMM, Luiking YC, Verlaan S, Evers SMAA. Burden-of-illness of Dutch community-dwelling older adults with sarcopenia: health related outcomes and costs. Eur Geriatr Med. 2016;7:276–84. https://doi.org/10.1016/j.eurger.2015.12.011.
Sousa AS, Guerra RS, Fonseca I, Pichel F, Ferreira S, Amaral TF. Financial impact of sarcopenia on hospitalization costs. Eur J Clin Nutr. 2016;70:1046–51. https://doi.org/10.1038/ejcn.2016.73.
Antunes AC, Araujo DA, Verissimo MT, Amaral TF. Sarcopenia and hospitalisation costs in older adults: a cross-sectional study. Nutr Diet. 2017;74:46–50. https://doi.org/10.1111/1747-0080.12287.
Janssen I, Shepard DS, Katzmarzyk PT, Roubenoff R. The healthcare costs of sarcopenia in the United States. J Am Geriatr Soc. 2004;52:80–5. https://doi.org/10.1111/j.1532-5415.2004.52014.x.
Goates S, Du K, Arensberg MB, Gaillard T, Guralnik J, Pereira SL. Economic impact of hospitalizations in US adults with sarcopenia. J Frailty Aging. 2019;8:93–9. https://doi.org/10.14283/jfa.2019.10.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Ecarnot, F., Rogoli, D., Maggi, S. (2021). Epidemiology of Sarcopenia. In: Veronese, N., Beaudart, C., Sabico, S. (eds) Sarcopenia. Practical Issues in Geriatrics. Springer, Cham. https://doi.org/10.1007/978-3-030-80038-3_1
Download citation
DOI: https://doi.org/10.1007/978-3-030-80038-3_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-80037-6
Online ISBN: 978-3-030-80038-3
eBook Packages: MedicineMedicine (R0)