Abstract
Virtual Reality (VR) is an emerging neuroergonomics tool for stroke rehabilitation. It can be employed to promote post-stroke recovery during rehabilitation as a result of its neuroplasticity enhancing effects. This study systematically reviewed and meta-synthesised evidence on the effectiveness of virtual reality on selected markers of neuroplasticity among stroke survivors (SSv). The databases searched were PEDro, CINHAL, the Cochrane Library, and PUBMed using combinations of Medical subject heading (MeSH) terms and keywords in the titles, abstracts and text for the population, intervention and major outcome (PICO format). The studies included were randomized clinical trials that compared the effects VR among adult SSv. The PEDro scale was used for quality appraisal of the included studies. Forest plot (RevMan version 5.3) was used for the metasynthesis of the results, level of significance was set at α = 0.05. A total of 6 studies were included in the meta-analysis (involving 441 stroke survivors). The pooled effects on the improvement in motor function (SMD = −1.05; CI = −1.53, −0.56, Z = 4.22, p < 0.0001, I2 = 93%) and balance performance (SMD = −3.06; CI = −3.80, −2.32, Z = 8.11, p < 0.0001, I2 = 94%) was significantly in the favour of VR. There is evidence that virtual reality is an effective neuroergonomics modality for encouraging neuroplasticity through its effects on the motor function, balance and muscle strength of stroke survivors.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Stroke is the major cause of disability worldwide, with a high social-economic impact [1, 2]. One out of every four stroke cases is fatal and between 25 to 50% of the survivors requires a rehabilitative treatment [3, 4]. The World Health Organization reported that 15 million people globally experience a stroke annually [5]. Of these, 5 million die and another 5 million are left permanently disabled, placing a burden on family and community. Stroke affects about 62 million people worldwide [6], and is the second leading cause of death and the third leading contributor to burden of disease globally [6,7,8].
Stroke rehabilitation is complex, long lasting and expensive and its functional outcome is influenced not only by the brain lesion site and extension, but also by medical, demographic and neuropsychologic factors. Neurorehabilitation after a stroke is valued highly by patients, and studies have shown a strong evidence for its effectiveness [2, 9,10,11]. There are various models of neurorehabilitation techniques available for the management of stroke patients. The two conventional models commonly described are rehabilitation through facilitation like Bobath technique and the motor re-learning model [8]. There are other specific neurorehabilitation techniques for which systematic reviews are available, they include constraint induced movement therapy (in which the unaffected arm is immobilised for few hours each day in order to encourage learned use of the affected arm), body-weight supported treadmill training and other aerobic exercise training [7]. Stroke recovery and management requires neurorehabilitation techniques that enhances neuroplasticity. Current trend and studies have shown a transitioning from these conventional therapies to neuro-engineering models. Such emerging approaches to stroke rehabilitation include virtual reality, motor imagery and robotics [8].
Virtual reality is a new technology that simulates a three-dimensional virtual world on a computer and enables the generation of visual, audio, and haptic feedback for the full immersion of users [12]. Users of virtual reality can interact with and observe objects in three-dimensional visual space without limitation. Virtual reality is a neuroergonomic tool [13], capable of enhancing neuroplasticity/learning [14], thus supporting its use in neurorehabilitation. At present, virtual reality training has been widely used in rehabilitation of balance dysfunction [15]. When patients perform virtual reality training, the prefrontal, parietal cortical areas and other motor cortical networks are activated [16]. Growing evidence from clinical studies reveals that virtual reality training improves the neurological function of patients with spinal cord injury [17], cerebral palsy [18], and other neurological impairments [19,20,21]. These findings suggest that virtual reality training can activate the cerebral cortex and improve the spatial orientation capacity of patients, thus facilitating the cortical control on balance and improved motor functioning in stroke patients.
Literature appears unsettled with regards to the effects of virtual reality on the health outcomes of stroke survivors. While the study by Wang et al. reported significant beneficial effects of virtual reality in improving motor function of stroke survivors [22], the study by Brunner et al. reported a non-significant effect [23]. When literature becomes shrouded with conflicting reports from primary studies, systematic reviews can be used to provide superior evidence [24]. This study therefore systematically reviewed the evidences from randomised clinical trials on the effects of virtual reality in the rehabilitation of post-stroke patients.
2 Methods
2.1 Design
A systematic review with meta-analysis of randomized controlled trials on the effects of virtual reality on functional outcome of stroke survivors.
2.2 Inclusion Criteria
Types of Studies:
Original research manuscripts in peer-reviewed journals published in English Language were included. Only randomized control trials that evaluated the effects of virtual reality on functional outcomes of stroke survivors were included.
Types of Participants:
The participants in the primary studies were adults of any gender with a clinically diagnosed incidence of stroke.
Types of Intervention:
Only studies whose primary aim was to determine the effects of virtual reality as an intervention for stroke rehabilitation were included.
Types of Outcome Measures:
Studies involving any of post stroke functional outcome measures such as barthel index, functional independence measure, Fugl-Meyer assessment scale etc.
2.3 Information Sources
An extensive search strategy to recognize studies that can be used for the review was grouped into the search of bibliographic database and grey literature and eligibility criteria system of study inclusion. This procedure was created in accordance with the rules of the Cochrane Handbook of systematic reviews of intervention [25]. And advice for Healthcare review by the centre for reviews and dissemination [26].
Search Strategy:
An extensive study strategy created to search bibliographic databases and grey literature that involved several combinations of search terms from Medical subject Heading (MeSH) terms and keywords in the titles, abstracts and text for the population, intervention and major outcome measures first in a pilot search to establish sensitivity and specificity of the search strategy. A host of commands which included the use of Boolean logic and search truncations was employed for the searches. There were modification of the strategy to suit the syntax and subject heading of the databases. The databases for the search were PEDro, CINHAL, the Cochrane Library, and PUBMed. Trial register and directory of open-access repository websites including https://www.clinicaltrial.gov, https://www.opendor.org and the web of science conference proceedings were also searched. Additionally, hand search was done from the reference list of identified studies and suggested articles.
Study Record and Data Management:
Search results were exported to Ref works to check for duplication of studies. Bibliographic records were exported from Ref works into Microsoft Excel (Microsoft 2010) to facilitate articles inclusion and exclusion. On the basis of inclusion criteria, eligibility review questions and structures for the studies, considerations to the two levels of eligibility assessment were produced, piloted and refined when appropriate.
Selection Process:
The eligibility criteria were liberally applied at the beginning to ensure that relevant studies were included and that no study was excluded without thorough evaluation. At the outset, studies were only excluded if they clearly met one or more of the exclusion criteria. Screening was conducted online simultaneously on the title and abstract by two reviewers to identify potentially relevant studies. Each reviewer crosschecked the initial screening results of the other. The two reviewers then read through the full text of selected studies for further screening (using the prior eligibility criteria). Differences of opinions occurring at any stage regarding inclusion or exclusion were resolved by discussion and reflection, in consultation with a third reviewer if warranted. When decision could not be made based on available information, study authors were contacted (to the maximum of three email attempts) to clarify issues of selection of any study. Studies were excluded and the reasons for exclusion were recorded when authors fail to respond to requests for clarifications on unclear issues regarding their reports. Details of the flow of studies throughout the process of assessment of eligibility and study selection were presented, along with the reasons for exclusion in a flow chart (PRISMA diagram).
2.4 Data Collection Processes
Quality Appraisal for Included Studies:
The quality of the selected studies were assessed using the Physiotherapy Evidence Database (PEDro) quality appraisal tool. The PEDro is an eleven-item scale in which the first item relates to external validity and the other ten items assess the internal validity of a clinical trial. One point is given for each satisfied criterion (except for the first item) yielding a maximum score of 10. The higher the score, the better the quality of the study and the following grades were used: 9–10 (excellent); 6–8 (good); 4–5 (fair); <4 (poor). A point for a particular criterion was awarded only if the article explicitly reported that the criterion was met. A score of one was given for each yes answer and zero for no, unclear and not applicable (N/A) answers. The overall score was reported as a tally of all yes answers out of 10 based on the applicable answers for each study. Scores of individual items from the critical appraisal tool were added to present the total score.
Data Synthesis and Assessment of Heterogeneity:
The Research question on the overall effects of virtual reality on the functional outcomes of stroke survivors were asked and answers attempted and appropriate statistical method was used. Given that the variables were on the ratio scale (continuous variable), weighted mean difference was used when outcomes were consistent or standard mean differences when there was the existence of variation in outcomes with a confidence interval of 95%. Meta-analysis was done whenever two or more studies existed that assessed similar outcomes using similar intervention. This was done to determine the pooled effect sizes across studies using a random effect model and relying on the level of heterogeneity of the outcomes. Assessment of heterogeneity was done via the Cochrane Chi-square test (10% significant level) and Higgins I2 for which values of 25%, 50% and 75% were interpreted as low, medium and high heterogeneity respectively as stipulated by the guidance on the Cochrane Handbook for Systematic Reviews of interventions [25].
2.5 Data Analysis
Investigation and presentation of outcomes were made using the main outcome. Studies that were homogenous in study design, intervention and control were pooled together for meta-analysis using a random effect model [25]. Appropriate statistical techniques were used for each type of continuous (weighted mean differences if outcomes are consistent or standard mean difference if different outcomes were used, with 95% CI). Interpretation of studies that are heterogeneous was done by narrative synthesis following the guideline of the Centre for Reviews and Dissemination to investigate the relationship and findings within and between the included studies [26]. Data analysis (Meta-analysis) was done using RevMan 5.3 software.
3 Results
3.1 Flow of Studies through the Review
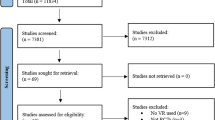
The initial searches identified a number of potential relevant papers. The flow of papers through the process of assessment of eligibility is represented with reasons for exclusion of papers at each stage of the process as in Fig. 1.
3.2 Characteristics of Included Trials
A total of 5,496 articles were generated from the aforementioned search strategy (Fig. 1) while 5,490 articles were eliminated after reading the abstracts and titles. Only six studies that contributed data for 441 stroke survivors were finally included in this review (see Table 1). All and none of the studies had random and concealed allocations respectively as shown in Tables 2 and 3. Considering both the PEDro ratings and sample size used, one study provided level-1 evidence whereas the others were considered as level 2 studies as shown in Table 3.
3.3 Methodological Quality Appraisal
The methodological quality of the included trials ranged from fair to good, with a average PEDro score of 7.9. Two trials had methodologically good quality with scores ≥6. The individual PEDro items satisfied by almost all the trials were random allocation to groups and point estimates and variability data as shown in Table 2.
3.4 Interventions
The major intervention used was exercise based virtual training. The most common exercise frequency and duration of time used was 3–5 days per week and 40–60 min per day, respectively. The most commonly prescribed treatment duration of the programme was ≥4weeks as shown in Tables 1 and 4.
3.5 Outcome Measures
The Pedoscan, Biorescue, Footscan and Balancia Software were used to assess balance. Action reach arm test (ARAT) and Wolf motor function test (WMFT) were used in assessing motor function.
3.6 The Effect of Virtual Reality on Motor Function
The meta-analysis incorporated three trials that assessed motor function resulting in a total of 93 participants. There was a significant pooled effect (Z = 4.22, p < 0.0001) on motor function in favour of virtual reality group (SMD = −1.05; CI = −1.53, −0.56). The included studies were weakly homogenous (X2 = 27.63, I2 = 93%) and a moderate risk of bias (42.8%). All the studies were however in favour of the experimental group as shown in Fig. 2.
3.7 The Effect of Virtual Reality on Balance Performance
The meta-analysis incorporated three trials that assessed balance performance resulting in a total of 87 participants. There was a significant pooled effect (Z = 8.11, p < 0.0001) on balance performance in favour of the virtual reality group (SMD = −3.06; CI = −3.80, −2.32). The included studies were strongly homogenous (X2 = 35.57, I2 = 94%) and had a moderate risk of bias (57.1%). All the studies were in favour of virtual reality as shown in Fig. 3.
4 Discussion
Virtual reality is an approach to user-computer interface that involves real-time simulation of an environment, scenario or activity that allows for user interaction via multiple sensory channels [31]. It creates sensory illusions that produce a more or less believable simulation of reality with the aim of fostering brain and behavioural responses in the virtual world that are analogous to those that occur in the real world [32]. VR simulations can be highly engaging, which provides crucial motivation for rehabilitative applications that require consistent, repetitive practice. Following damage to the brain as seen in stroke survivors, their ability to interact with the physical environment is diminished, thus compounding their disability. Virtual reality may potentially help reduce the burden of such physical limitations by providing an alternative, favourable environment in which to practice motor skills. It can be used to deliver meaningful and relevant stimulation to an individual’s nervous system and thereby capitalise on the plasticity of the brain to promote motor learning and rehabilitation [33].
In this review, the use of virtual reality was found to be effective in promoting motor functional recovery among stroke survivors. It may be argued that motor plan is represented by the two premovement components [Negative Slope (NS) and Bereitschaft Potential (BP)] of the Motor Related Cortical Potential (MRCP) from an electroencephalogram (EEG) [34]. While the NS-wave (activity in the premotor area) which starts about 500 ms before the movement is believed to represent the urge to act, the BP (seen 1–3 s before the movement) is thought to reflect the early motor preparation (motor programme) in the supplementary motor (SM) area, as well as the superior and inferior parietal lobe [34,35,36,37]. Similar cerebral motor plans in the motor and pre-motor areas have been reported for real and virtual tasks actions [34]. It is therefore possible that virtual reality rehabilitation mimics the neural mechanisms of actual neurorehabilitation viz-a-viz the neuroplastic effects.
Virtual reality faccilitates the motor functional recovery of the paretic upper limb through neural reorganization. This can be clinically revealed by a functional magnetic resonance imaging (fMRI) scan that is capable of measuring the blood oxygen level dependent (BOLD) signal. Changes in both the location and level of the BOLD signal can reveal evidence of neuroplasticity [38]. In an RCT on the effects of Leap-Motion based virtual reality of motor functional recovery and cortical reorganization of subacute stroke survivors, Wang and his colleagues using an fMRI reported a shift in the activated motor area from the ipsilateral to contralateral motor area that was more obvious in the experimental groups [22]. This led to a significantly improved motor function compared with the control group that received conventional therapy. This change may be attributed to increased practice-induced neuroplasticity as a result of repetitive practice associated with virtual reality training and/or imitation-dependent neuroplasticity initiated in the virtual environment and carried out by the patient in the real world through mechanisms such as synaptic pruning, Hebbian mechanism, or long term potentiation (LTP) [37].
There was also a pooled significant improvement in the balance performance of stroke survivors in favour of virtual reality training [39]. The control of human balance is a comprehensive process relying on the integration of visual, vestibular and somatosensory inputs to the central nervous system. Balance performance can be therefore be affected by a dysfunction in the proprioceptors, muscle weakness, joint immobility and instability, pain or visual deficits; these impairments characterizes post-stroke morbidity. Balance as an outcome measure has been identified to be one of the key areas to be considered during stroke rehabilitation. About 70–80% of stroke patients experience a fall as a result of balance dysfunctions [30]. Virtual reality can be used to encourage long term potentiation of the vertibular cortext and its pathways for balance functioning through the visual feedback enhanced in a virtual environment; thus, “pathways that fire together, wire together”. In an RCT to determine the effects of a community based virtual reality training on the balance performance of chronic stroke survivors, Kim et al. found that virtual reality significantly decreased the anterioposterior and total postural sway path lengths as well as the postural sway speed [27].
A major advantage of virtual reality training over conventional neurorehabilitation approach is adherence. Virtual reality is an entertaining, motivating and fun-therapy and thus encourages patient-participation, repetition, attention and enjoyment which are recipes for neuroplasticity. However, virtual reality is not without its own demerits. These include problems of availability, affordability, acceptability and adaptability especially in low and middle income countries where stroke morbidity and mortality is greatest.
5 Conclusion
Virtual reality is an effective neuroergonomic tool for the neurorehabilitation of stroke survivors by harnessing its neuroplastic effects.
References
Avan, A., Digaleh, H., Di Napoli, M., Stranges, S., Behrouz, R., Shojaeianbabaei, G., Amiri, A., Tabrizi, R., Mokhber, N., Spence, J.D., Azarpazhooh, M.R.: Socioeconomic status and stroke incidence, prevalence, mortality, and worldwide burden: an ecological analysis from the Global Burden of Disease Study 2017. BMC Med. 17(1), 1–30 (2019)
Ekechukwu, E.N.D., Omotosho, I.O., Hamzat, T.K.: Comparative effects of interval and continuous aerobic training on haematological variables post-stroke–a randomized clinical trial. Afr. J. Physiother. Rehabil. Sci. 9(1–2), 1–8 (2017)
Thrift, A.G., Thayabaranathan, T., Howard, G., Howard, V.J., Rothwell, P.M., Feigin, V.L., Norrving, B., Donnan, G.A., Cadilhac, D.A.: Global stroke statistics. Int. J. Stroke 12(1), 13–32 (2017)
Ru, X., Dai, H., Jiang, B., Li, N., Zhao, X., Hong, Z., He, L., Wang, W.: Community-based rehabilitation to improve stroke survivors’ rehabilitation participation and functional recovery. Am. J. Phys. Med. Rehabil. 96(7), e123-9 (2017)
Someeh, N., Shamshirgaran, S.M., Farzipoor, F., Asghari-Jafarabadi, M.: The moderating role of underlying predictors of survival in patients with brain stroke: a statistical modeling. Sci. Rep. 10(1), 1–9 (2020)
Queensland Brain Institute Stroke facts. https://qbi.uq.edu.au/brain/brain-diseases/stroke/stroke-facts. Accessed 04 Feb 2021
Ekechukwu, E.N.D., Olowoyo, P., Nwankwo, K.O., Olaleye, O.A., Ogbodo, V.E., Hamzat, T.K., Owolabi, M.O.: Pragmatic solutions for stroke recovery and improved quality of life in low-and middle-income countries—a systematic review. Front. Neurol. 25(11), 337 (2020)
Owolabi, M.O., Platz, T., Good, D., Dobkin, B.H., Ekechukwu, E.N.D., Li, L.: Translating innovations in stroke rehabilitation to improve recovery and quality of life across the globe. Front. Neurol. 11 (2020)
Moradi, V., Hossein, M.A., Shariat, A., Cleland, J.A., Ansari, N.N., Savari, S.: Neurorehabilitation, the practical method of returning to work after stroke. Iranian J. Public Health (2021)
Lieshout, E.C.: Neurorehabilitation for upper limb recovery after stroke: the use of non-invasive brain stimulation (Doctoral dissertation, Utrecht University)
Anaya, M.A., Branscheidt, M.: Neurorehabilitation after stroke: from bedside to the laboratory and back. Stroke 50(7), e180-2 (2019)
Gandhi, R.D., Patel, D.S.: Virtual reality–opportunities and challenges. Virtual Real. 5(01) (2018)
Gramann, K., Fairclough, S.H., Zander, T.O., Ayaz, H.: Trends in neuroergonomics. Front. Hum. Neurosci. 5(11), 165 (2017)
Eng, C.M., Calkosz, D.M., Yang, S.Y., Williams, N.C., Thiessen, E.D., Fisher, A.V.: Doctoral colloquium—enhancing brain plasticity and cognition utilizing immersive technology and virtual reality contexts for gameplay. In: 2020 6th International Conference of the Immersive Learning Research Network (iLRN), pp. 395–398. IEEE, 21 June 2020
Lei, C., Sunzi, K., Dai, F., Liu, X., Wang, Y., Zhang, B., He, L., Ju, M.: Effects of virtual reality rehabilitation training on gait and balance in patients with Parkinson’s disease: a systematic review. PloS One 14(11), e0224819 (2019)
Calabrò, R.S., Naro, A., Russo, M., Leo, A., De Luca, R., Balletta, T., Buda, A., La Rosa, G., Bramanti, A., Bramanti, P.: The role of virtual reality in improving motor performance as revealed by EEG: a randomized clinical trial. J. Neuroeng. Rehabil. 14(1), 1–6 (2017)
Miguel-Rubio, D., Rubio, M.D., Salazar, A., Camacho, R., Lucena-Anton, D.: Effectiveness of virtual reality on functional performance after spinal cord injury: a systematic review and meta-analysis of randomized controlled trials. J. Clin. Med. 9(7), 2065 (2020)
Ghai, S., Ghai, I.: Virtual reality enhances gait in cerebral palsy: a training dose-response meta-analysis. Front Neurol. 26(10), 236 (2019)
Wang, B., Shen, M., Wang, Y.X., He, Z.W., Chi, S.Q., Yang, Z.H.: Effect of virtual reality on balance and gait ability in patients with Parkinson’s disease: a systematic review and meta-analysis. Clin. Rehabil. 33(7), 1130–1138 (2019)
Norouzi, E., Gerber, M., Pühse, U., Vaezmosavi, M., Brand, S.: Combined virtual reality and physical training improved the bimanual coordination of women with multiple sclerosis. Neuropsychol. Rehabil. 18, 1–8 (2020)
Reynolds, L., Rodiek, S., Lininger, M., McCulley, M.A.: Can a virtual nature experience reduce anxiety and agitation in people with dementia? J. Housing Elderly 32(2), 176–193 (2018)
Wang, Z.R., Wang, P., Xing, L., Mei, L.P., Zhao, J., Zhang, T.: Leap Motion-based virtual reality training for improving motor functional recovery of upper limbs and neural reorganization in subacute stroke patients. Neural Regeneration Res. 12(11), 1823 (2017)
Brunner, I., Skouen, J.S., Hofstad, H., Aßmus, J., Becker, F., Sanders, A.M., Pallesen, H., Kristensen, L.Q., Michielsen, M., Thijs, L., Verheyden, G.: Virtual reality training for upper extremity in subacute stroke (VIRTUES): a multicenter RCT. Neurology 89(24), 2413–2421 (2017)
Sylvester, R.J., Canfield, S.E., Lam, T.B., Marconi, L., MacLennan, S., Yuan, Y., MacLennan, G., Norrie, J., Omar, M.I., Bruins, H.M., Hernandez, V.: Conflict of evidence: resolving discrepancies when findings from randomized controlled trials and meta-analyses disagree. Eur. Urol. 71(5), 811–819 (2017)
Higgins, J.P., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A. (eds.) Cochrane Handbook for Systematic Reviews of Interventions. Wiley (2019)
Tacconelli, E.: Systematic reviews: CRD’s guidance for undertaking reviews in health care. Lancet Infectious Dis. 10(4), 226 (2010)
Kim, N., Park, Y., Lee, B.H.: Effects of community-based virtual reality treadmill training on balance ability in patients with chronic stroke. J. Phys. Ther. Sci. 27(3), 655–658 (2015)
Bang, Y.S., Son, K.H., Kim, H.J.: Effects of virtual reality training using Nintendo Wii and treadmill walking exercise on balance and walking for stroke patients. J. Phys. Ther. Sci. 28(11), 3112–3115 (2016)
Park, S.K., Yang, D.J., Uhm, Y.H., Heo, J.W., Kim, J.H.: The effect of virtual reality-based eccentric training on lower extremity muscle activation and balance in stroke patients. J. Phys. Ther. Sci. 28(7), 2055–2058 (2016)
Yang, S., Hwang, W.H., Tsai, Y.C., Liu, F.K., Hsieh, L.F., Chern, J.S.: Improving balance skills in patients who had stroke through virtual reality treadmill training. Am. J. Phys. Med. Rehabil. 90(12), 969–978 (2011)
Adamovich, S.V., Fluet, G.G., Tunik, E., Merians, A.S.: Sensorimotor training in virtual reality: a review. NeuroRehabilitation 25(1), 29–44 (2009)
Bohil, C.J., Alicea, B., Biocca, F.A.: Virtual reality in neuroscience research and therapy. Nat. Rev. Neurosci. 12(12), 752–762 (2011)
Weiss, P.L., Keshner, E.A., Levin, M.F. (eds.): Virtual Reality for Physical and Motor Rehabilitation. Springer, New York (2014)
Bozzacchi, C., Giusti, M.A., Pitzalis, S., Spinelli, D., Di Russo, F.: Similar cerebral motor plans for real and virtual actions. PLoS One 7(10), e47783 (2012)
Bozzacchi, C., Giusti, M.A., Pitzalis, S., Spinelli, D., Di Russo, F.: Awareness affects motor planning for goal-oriented actions. Biol .Psychol. 89(2), 503–514 (2012)
Shibasaki, H., Hallett, M.: What is the Bereitschaftspotential? Clin. Neurophysiol. 117(11), 2341–2356 (2006)
Libet, B., Gleason, C.A., Wright, E.W., Pearl, D.K.: Time of conscious intention to act in relation to onset of cerebral activity (readiness-potential). In: Neurophysiology of Consciousness, pp. 249–268. Birkhäuser, Boston (1993)
Cheung, K.L., Tunik, E., Adamovich, S.V., Boyd, L.A.: Neuroplasticity and virtual reality. In: Virtual Reality for Physical and Motor Rehabilitation, pp. 5–24. Springer, New York (2014)
Gaerlan, M.G.: The role of visual, vestibular, and somatosensory systems in postural balance. UNLV theses, dissertations, Professional Papers, and Capstones, p. 357 (2010)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Ekechukwu, E.N.D. et al. (2022). Virtual Reality, a Neuroergonomic and Neurorehabilitation Tool for Promoting Neuroplasticity in Stroke Survivors: A Systematic Review with Meta-analysis. In: Black, N.L., Neumann, W.P., Noy, I. (eds) Proceedings of the 21st Congress of the International Ergonomics Association (IEA 2021). IEA 2021. Lecture Notes in Networks and Systems, vol 223. Springer, Cham. https://doi.org/10.1007/978-3-030-74614-8_64
Download citation
DOI: https://doi.org/10.1007/978-3-030-74614-8_64
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-74613-1
Online ISBN: 978-3-030-74614-8
eBook Packages: EngineeringEngineering (R0)