Abstract
Autonomic nervous system (ANS) regulates many important systems in the human body, such as blood pressure, heart rate, digestion, respiratory system and many others. Dysfunction of the ANS is related to disorders in different systems in the human body, and it can be recognizable in the ANS biosignals. Digital signal processing (DSP) of the ANS biosignals provides information about the functioning of the autonomic nervous system, which is very useful in everyday clinical practice. There is a large variety of different methods for ANS signals analysis, with different features extracted. Due to the increase in the collectible amount of data, there is a growing demand for the automatization of the signal analysis process, which could reduce human error, save time and improve the quality and the accuracy of the calculated results.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
- Autonomic nervous system
- Signal processing
- Heart rate variability (HRV)
- Baroreflex sensitivity index (BRS)
1 Introduction
The autonomic nervous system (ANS) regulates many important systems in the human body, such as blood pressure, heart rate, digestion, respiratory system and many others. These systems have in common that they are mostly independent of human will. According to its function, the ANS could be divided into the sympathetic nervous system (SNS) and the parasympathetic nervous system (PNS). SNS is activated in situations where the body needs to prepare for the possible threat, “fight or flight” situations.
In these situations, the main purpose is to optimize body functions for possible danger, such as pupil dilatation and more light entering the eyes and an increase in blood pressure and heart rate with bronchial dilatation leading to greater blood oxygenation. As opposite, PNS is responsible for “rest and digest” situations, where the body needs to recover and the only purpose is to optimize body functions for basic maintenance, such as a decrease in blood pressure and heart rate which enables conservation of energy [1].
Dysfunction of the ANS is related to disorders in different systems in the human body, and it can be recognizable in the ANS biosignals. Digital signal processing (DSP) of the ANS biosignals provides information about the functioning of the ANS, which is very useful in everyday clinical practice. The purpose of this manuscript is to present possible methods for the analysis of the ANS biosignals. In Sect. 2 ANS testing and data acquisiton will be briefly described. Section 3 presents time and frequency domain characteristics of the ANS biosignals and Sect. 4 presents additional methods of the ANS biosignals analysis.
2 ANS Testing and Data Acquisition
The basis of the testing of the ANS is the response of different systems that are under its influence (like heart rate and blood pressure) to different well-defined challenges. Because of this, there is no unique test, and usually, a battery of different autonomic tests, that combines different approaches, is used in clinical practice [2].
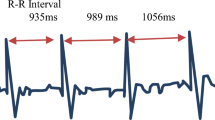
One of the most frequently examined parts of the ANS is the cardiovascular autonomic nervous system, because it has easily recordable biosignals – heart rate (HR) and blood pressure (BP). Basic cardiovascular biosignals are presented in Fig. 1.
2.1 Types of ANS Testing
ANS recordings are usually performed in laboratory conditions, however, they can also be performed bedside. A typical laboratory for ANS recordings is presented in Fig. 2. Continuously monitoring of blood pressure and heart rate is performed with pressure cuff and ECG electrodes, with a patient in the supine position. Different measurement procedures are usually performed: heart rate and blood pressure response to Valsalva maneuver – a measure of parasympathetic and sympathetic function; heart rate response to deep breathing test – measure of parasympathetic function; and blood pressure response to passive tilt or active standing – a measure of sympathetic function [3, 4]. Additionally, a sudomotor function can be tested with the quantitative sudomotor axon reflex sweat test (QSART) or with the sympathetic skin response (SSR), which presents the momentary change of the electrical potential of the skin.
2.2 Duration of Recordings
Different clinical procedures require a different amount of information, and because of this, recordings of different duration can be performed. Usually, recordings are divided into short-term and long-term recordings. Short-term recordings usually have a duration from 0.5 to 5 min and are related to specific conditions [5]. Long-term recordings are usually defined as 24-h Holter recordings, performed with wearable sensors in subjects freely moving under daily conditions. Different durations of recordings acquire different interpretations, and additional information is necessary for detailed analysis. Some of the variables important for long-term recordings interpretation are physical activity and posture [6].
2.3 Characteristics of the Specific Types of Recordings
When recordings are performed in laboratory conditions, all variables can be well controlled, such as patient’s activity and environmental conditions (temperature…).
Analysis of the data can be performed in real time and possible difficulties, that could influence the quality of recorded material, can be corrected in real time through active cooperation with the patients. But, this type of examination is time consuming and it enables recording only a limited number of patients with a limited amount of data.
Because of that, various possibilities are examined, to see how a bigger amount of the ANS data could be collected. One of the examples is presented in the work of Komazawa et al., where they collected information from more than 27 000 subjects [7]. With the smartphone camera, that detects the pulse waveform from luminance changes of blood flow on the tip of the finger, the information for HRV analysis were collected. The application enables the collection of a large amount of data at any time, it acquires data about the brightness of the skin and derives a pulse wave based on luminance change. Acquired data is analyzed and the information about the sympathetic and parasympathetic activity is presented on the user’s mobile phone.
When performing recordings in non-laboratory conditions (such as a mobile application), it is more convenient to record a bigger number of patients and collect a large amount of data. Also, recording in non-laboratory conditions could provide information about the functioning of the ANS through daily activities. But there are also some disadvantages to this type of recordings. There is no active cooperation with the patients and no control over recording environmental conditions. The quality of data depends on the patient’s cooperability and responsibility, and only off-line analysis is available, with no insight into the data quality during the recordings. Data acquisition is dependent on the relationship between the sensor and the skin, and this relationship can vary through daily activities. The smartphone and similar applications have a much lower sampling rate than the laboratory devices (30 Hz vs 1000 Hz), and this also influences the quality of the recorded signal [8].
The quality of the recorded signal can be improved with the measurement of the quality information feedback for the participants, user can discard corrupted data and repeat the measurement, but in most cases, this part of the application is not included [9].
Differences in the collection of the data can provide different insights into the ANS activity, and regarding the type of study design, different characteristics of the signal are appropriate.
3 Time and Frequency Domain Characteristics
Recorded signals can be analyzed through different procedures. Basic cardiovascular biosignals, heart rate and blood pressure, have different time and frequency domain characteristics.
3.1 Heart Rate
The highest emphasis in everyday clinical practice is the analysis of the heart rate (HR). The analysis is usually performed in the time domain in the form of heart rate variability (HRV): standard deviation of RR interval (SDRR), a standard deviation of NN intervals (SDNN), root mean square for standard deviation (RMSSD); and in the frequency domain in the form of spectral analysis: Power Spectral Density (PSD), low frequency (LF, 0.04–0.15 Hz), high frequency (HF, 0.16–0.4 Hz) and LF/HF ratio [10]. There is a large number of time-domain and frequency-domain measures, but the most important question is which of these variables have clinical meaning, and according to that the variables for analysis should be selected [11].
HR signals by their nature can be non-stationary and non-linear, and because of that, the non-linear analysis is also useful in quantification of the structure and complexity of the HR signals [12]. Different metrics, such as detrended fluctuation analysis (DFA), the approximate entropy and correlation dimension, are variables of interest for this type of analysis.
3.2 Blood Pressure
The other important ANS variable is blood pressure (BP). It can be analyzed in the time domain in the form of systolic blood pressure (sBP), diastolic blood pressure (dBP), pulse pressure (PP) or mean arterial pressure [13]. Also, analysis can be performed in the frequency domain in the form of power spectral density (Fast Fourier Transform – FFT), mean and total power, LF band (0.04 to 0.15 Hz), HF band (0.4 to 1.5 Hz) [14].
4 Additional Methods of ANS Signal Analysis
With the growth of the amount of data and different recording procedures, there is a need for additional methods for DSP of ANS biosignals.
4.1 Heart Rate Variability (HRV)
One of the most commonly used methods is heart rate variability (HRV) analysis. The method has well defined protocol and clear mathematical definitions [15]. The most frequently used variable is SDNN (standard deviation of the normal-to-normal interval), and it presents a marker of overall HRV with the contribution of both, sympathetic and parasympathetic nervous system activity. The type of HRV analysis is dependent on the duration of the recordings, which can be divided into short-term (5 min) and long term (24 h) recordings [11]. Data for analysis can be collected with different recording devices (hardwired/wireless photoplethysmography; ring, hand, or chest-based sensors) and analysis can be performed on-line or on a server [16]. Longitudinal HRV monitoring is suitable for “big data” analysis and there are machine learning models for risk prediction based on HRV [17]. The disadvantage of the method is that there are different types of software for its analysis and there can be disagreement between different methods for estimation of HRV [18].
4.2 Baroreflex Sensitivity Indices (BRS)
The baroreflex sensitivity (BRS) index is an important cardiovascular indicator that allows the quantification and analysis of the body's adaptability to hemodynamic changes [19]. Newly introduced BRS indices are BRSa1, α-BRSa, β-BRSa and vagal index BRSv. BRS indices are calculated from the systolic blood pressure (sBP) curve during the Valsalva maneuver [20, 21]. Indices are usually calculated manually, which is a time-consuming process and dependent on the subjective assessment, which improved the risk of human error. To reduce the subjective human error, reduce the necessary time and improve the precision of the calculated indices, the semi-automated application for calculation was introduced [22]. Semi-automated results showed a statistically significant high association with manually calculated indices, and the results of the application are presented in Fig. 3. Subjective assessment and the likelihood of human error are considerably reduced by using semi-automated calculation, and the application is time saving so it enables analysis of a large amount of data.
4.3 Blood Pressure and Pulse Rate Variability
Similar to HRV, blood pressure (BP) variability can also be calculated and it varies depending on the duration of BP recordings [23]. Very short-term BP variability presents standard deviation or fluctuation of BP in various frequency bands, mostly recorded in laboratory conditions. Short-term BPV is expressed in the form of standard deviation, coefficient of variation (CoV), 24-h weighted SD, and average real variability (ARV), mostly recorded through the 24 h ambulatory BP monitoring (ABPM). Long-term BPV is related to ABPM over 48 h or home BP monitoring.
(HBPM) over several days or weeks, but it has questionable reliability due to the long duration of the recordings.
Pulse rate variability (PRV) is calculated by pulse oximeter photoplethysmography (PPG), a method that detects blood volume variations in the body tissue, and it provides an estimation of the heart rate. PRV is calculated from the peak to peak time intervals of the PPG signal [24]. PRV could substitute HRV in some non-stationary situations with the usage of wearable and portable medical devices, and because of its robust signal in some situations, PRV has more practical value than HRV signal [25]. A disadvantage of the method is that PRV shows considerable variations regarding the measurement sites [26].
5 Conclusion
Autonomic nervous system testing can be performed in different conditions and there is a large variety of different types of signal analysis, regarding the parameters of the signals and type of signal acquisition. Due to the increase in the collectible amount of data, there is a growing demand for the automatization of the signal analysis process, which could reduce human error, save time and improve the quality and the accuracy of the calculated results.
References
Goldstein, D.S.: Principles of autonomic medicine (2017). https://neuroscience.nih.gov/publications
Ziemssen, T., Siepmann, T.: The investigation of the cardiovascular and sudomotor autonomic nervous system-a review. Front Neurol. 10, 53 (2019). https://doi.org/10.3389/fneur.2019.00053
Freeman, R.: Assessment of cardiovascular autonomic function. Clin. Neurophysiol. 117(4), 716–730 (2006). ISSN 1388-2457
Novak, P.: Quantitative autonomic testing. J. Vis. Exp. (53), 2502 (2011). https://doi.org/10.3791/2502. PMID: 21788940; PMCID: PMC3196175
Sztajzel, J.: Heart rate variability: a noninvasive electrocardiographic method to measure the autonomic nervous system. Swiss Med. Wkly. 134(35–36), 514–522 (2004)
Hayano, J., Yuda, E.: Pitfalls of assessment of autonomic function by heart rate variability. J. Physiol. Anthropol. 38, 3 (2019). https://doi.org/10.1186/s40101-019-0193-2
Komazawa, M., Itao, K., Kobayashi, H., Luo, Z.: On human autonomic nervous activity related to behavior, daily and regional changes based on big data measurement via smartphone. Health 8, 827–845 (2016). https://doi.org/10.4236/health.2016.89088
Peng, R.C., Zhou, X.L., Lin, W.H., Zhang, Y.T.: Extraction of heart rate variability from smartphone photoplethysmograms. Comput. Math. Methods Med. 2015, 516826 (2015). https://doi.org/10.1155/2015/516826. Epub 2015 Jan 12
Liu, I., Ni, S., Peng, K.: Enhancing the robustness of smartphone photoplethysmography: a signal quality index approach. Sensors 20(7), E1923 (2020). https://doi.org/10.3390/s20071923
Bong, S.Z., Murugappan, M., Yaacob, S.: Analysis of electrocardiogram (ECG) signals for human emotional stress classification. In: Ponnambalam, S.G., Parkkinen, J., Ramanathan, K.C. (eds.) Trends in Intelligent Robotics, Automation, and Manufacturing. IRAM 2012. Communications in Computer and Information Science, vol. 330. Springer, Heidelberg (2012)
Shaffer, F., Ginsberg, J.P.: An overview of heart rate variability metrics and norms. Front. Public Health 5, 258 (2017). https://doi.org/10.3389/fpubh.2017.00258
Germán-Sallóa, Z., Germán-Sallób, M.: Non-linear methods in HRV analysis. Procedia Technol. 22, 645–651 (2016). https://doi.org/10.1016/j.protcy.2016.01.134
Kengne, A.P., Czernichow, S., Huxley, R., Grobbee, D., Woodward, M., Neal, B., Zoungas, S., Cooper, M., Glasziou, P., Hamet, P., Harrap, S.B., Mancia, G., Poulter, N., Williams, B., Chalmers, J.: ADVANCE Collaborative Group. Blood pressure variables and cardiovascular risk: new findings from ADVANCE. Hypertension. 54(2), 399–404 (2009). https://doi.org/10.1161/HYPERTENSIONAHA.109.133041. EPUB 2009 May 26
Andriessen, P., Schoffelen, R.L., Berendsen, R.C., de Beer, N.A., Oei, S.G., Wijn, P.F., Blanco, C.E.: Noninvasive assessment of blood pressure variability in preterm infants. Pediatr. Res. 55(2), 220–3 (2004). Epub 2003 Nov 19
Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Eur. Heart J. 17(3), 354–381 (1996)
Drury, R.L.: Heart rate variability in an integrated hardware/software system using artificial intelligence to provide assessment, intervention and performance optimization support. In: Theodoros, A. (ed.) Autonomic Nervous System Monitoring. IntechOpen Limited, London (2019)
Chiew, C.J., Liu, N., Tagami, T., Wong, T.H., Koh, Z.X., Ong, M.E.H.: Heart rate variability based machine learning models for risk prediction of suspected sepsis patients in the emergency department. Medicine 98(6), e14197 (2019). https://doi.org/10.1097/MD.0000000000014197
Vest, A.N., Da Poian, G., Li, Q., Liu, C., Nemati, S., Shah, A.J., Clifford, G.D.: An open source benchmarked toolbox for cardiovascular waveform and interval analysis. Physiol. Meas. 39(10), 105004 (2018). https://doi.org/10.1088/1361-6579/aae021
Schrezenmaier, C., Singer, W., Swift, N.M., Sletten, D., Tanabe, J., Low, P.A.: Adrenergic and vagal baroreflex sensitivity in autonomic failure. Arch. Neurol. 64, 381–6 (2007). https://doi.org/10.1001/archneur.64.3.381
Palamarchuk, I.S., Baker, J., Kimpinski, K.: Non-invasive measurement of baroreflex during Valsalva maneuver: evaluation of alpha and beta-adrenergic components. Clin. Neurophysiol. 127, 1645–51 (2016). https://doi.org/10.1016/j.clinph.2015.10.047
Palamarchuk, I.S., Baker, J., Kimpinski, K.: Non-invasive measurement of adrenergic baroreflex during Valsalva maneuver reveals three distinct patterns in healthy subjects. Clin. Neurophysiol. 127, 858–63 (2016). https://doi.org/10.1016/j.clinph.2015.04.057
Višnjić, N., Moštak, I., Mutak, T., Pucić, D., Junaković, A., Adamec, I., Krbot Skorić, M., Habek, M.: Automated calculation of baroreflex sensitivity (BRS) indices. Eur. J. Neurol. 27, 266 (2020). https://doi.org/10.1111/ene.14307
Chadachan, V.M., Ye, M.T., Tay, J.C., Subramaniam, K., Setia, S.: Understanding short-term bloodpressure-variability phenotypes: from concept to clinical practice. Int. J. Gen. Med. 11, 241–254 (2018). https://doi.org/10.2147/IJGM.S164903. eCollection 2018.
Dehkordi, P., Garde, A., Karlen, W., Wensley, D., Ansermino, J.M., Dumont, G.A.: Pulse rate variability compared with heart rate variability in children with and without sleep disordered breathing. In: Conference Proceedings IEEE Engineering in Medicine and Biology Society, pp. 6563–6566 (2013). https://doi.org/10.1109/EMBC.2013.6611059
Chou, Y., Zhang, R., Feng, Y., Lu, M., Lu, Z., Xu, B.: A real-time analysis method for pulse rate variability based on improved basic scale entropy. J. Healthc. Eng. (2017). https://doi.org/10.1155/2017/7406896
Yuda, E., Yamamoto, K., Yoshida, Y., Hayano, J.: Differences in pulse rate variability with measurement site. J. Physiol. Anthropol. 39(1), 4 (2020). https://doi.org/10.1186/s40101-020-0214-1
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Ethics declarations
MKS has nothing to disclose.
IA has nothing to disclose.
MC has nothing to disclose.
MH has nothing to disclose.
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Krbot Skorić, M., Adamec, I., Cifrek, M., Habek, M. (2021). Analysis of Autonomic Nervous System Biosignals. In: Badnjevic, A., Gurbeta Pokvić, L. (eds) CMBEBIH 2021. CMBEBIH 2021. IFMBE Proceedings, vol 84. Springer, Cham. https://doi.org/10.1007/978-3-030-73909-6_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-73909-6_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-73908-9
Online ISBN: 978-3-030-73909-6
eBook Packages: EngineeringEngineering (R0)