Abstract
Stroke is a neurological syndrome that may affect severely lower-limb movements and the normal gait. The complete or partial restoration may be achieved through alternative rehabilitation therapies, such as Motor Imagery (MI)-based Brain Computer Interfaces (BCIs). Although these systems have shown promising results on post-stroke patients with severe disability, their performance recognizing MI may be reduced for people executing MI tasks with high difficult or producing weak brain activation. This study presents a proposal to improve the calibration stage of a low-cost electroencephalographic (EEG) based MI BCI with pedal end-effector, which integrally aims to activate continuously the central and peripheral mechanisms related to lower-limbs, and obtain the best feature vectors for MI recognition. This setup enables users to perform pedaling MI and receive passive pedaling into a Calibration phase. Consequently users can produce related EEG signals useful to obtain those more discriminant MI feature vectors through a probability analysis combining patterns from pedaling MI and passive pedaling. Here, Riemannian geometry and Common Spatials Patterns (CSP) for feature extraction were used independently or combined in our approach. Preliminary results show that the proposed method may improve the BCI performance. For healthy subjects, the approach using CSP achieved accuracy (ACC) up to 98.43%, whereas for PS1 and PS2 obtained ACC of 71.07% and 79.24%, respectively. However, Riemannian geometry plus CSP using LDA reached better results for healthy subjects and patients (mean ACC of 73.84%).
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Stroke is the leading cause of adult long-term disability [1, 2], and may cause severe cognitive and motor impairments for survives. Loss of independence in their daily life, such as mobility, balance and walking are often affected [1, 3]. Gait rehabilitation is the major goal for post-stroke people [3].
Significant neurological improvements of post-stroke patients may take place up to 6 months, when receiving motor rehabilitation, which has great importance and benefits to recover their independence. Evidences have demonstrated that chronic stroke patients maintain neuroplasticity, which increases the chance for recovery [1]. Generally, these patients receive traditional rehabilitation to improve their motor function [4]. For instance, the mirror therapy is a traditional method in which the hemiplegic patients are able to see uniquely the reflex-response of the non-affected limb [5], therefore they receive visual feedback. In contrast, BCIs based on robotic devices have been successfully used for motor rehabilitation after stroke, providing physical feedback. Thus, this technology can be used on post-stroke patients that cannot voluntarily initiate a movement [6], as BCIs are communication systems that translate brain waves into artificial output to control the external world [1].
Some BCIs have been proposed to recognize motor imagery using different approaches for feature extraction, being Riemannian geometry [4, 7, 8] and CSP [9] very promising methods. In [4, 7], Riemannian geometry was used successfully in a BCI to recognize pedaling motor imagery for lower-limb rehabilitation of post-stroke patients. In [9], a CSP-based BCI approach was proposed to select MI features. The performance extracting features using independently each one of these methods from EEG related to MI may be negatively affected due to several factors: stroke episode severity, making difficult MI execution or producing weak brain activation; uncertainty labelling feature vectors; EEG variability, etc. We hypothesize that using feature vectors from passive movements may increase class discrimination of a MI-based BCI, as similar brain regions are activated by executing a same imagined or real movement. Furthermore, new approaches also combining Riemannian geometry and CSP may be explored to discriminate better MI tasks.
This study aims to advance designing feature extraction methods for a motor imagery BCI based on pedal end-effector that continuously promote activation on central and peripheral mechanisms linked to lower-limbs, using pedaling MI and passive pedaling tasks in the Calibration phase. Riemannian geometry, CSP and Riemannian plus CSP are evaluated for feature extraction, into a new approach-based on probability for obtaining those feature vectors from pedaling MI more related to patterns of passive pedaling movements. This type of systems for providing pedaling exercises have shown effectiveness in both gait and lower-limb rehabilitation, and can be easily extended into developing countries, due to their relative low-cost [7].
This paper is structured into three sections as follows. Section 2 describes first the dataset used, followed by the proposed system to recognize pedaling motor imagery in our approach. Afterwards, the results and discussions are presented in Sect. 3, in which the performance of our approaches is analyzed. Finally, the conclusion about the proposed method are given in Sect. 4.
2 Materials and Methods
2.1 Data Description
An EEG acquisition system with a sampling rate 250 Hz using the low-cost open-source OpenBCI Cyton Board was used to collect the EEG database in our previous study [4]. EEG data from eight healthy subjects (one female and seven males) and two post-stroke patients (females) was collected over FC1, FC2, C3, Cz, C4, CP1, CP2, and Pz locations. The reference (A1) and ground (A2) were placed on the left and right earlobes, respectively [4].
For each participant, the database was divided into a calibration set and an evaluation set. The former was composed of seven sessions (12 trials per sessions), completing a balanced set with 168 periods of 5 s for both resting and pedaling MI classes. Data from the first six sessions was used for training (144 periods), whereas last session (24 periods) was employed for validation. On the other hand, the evaluation set was formed by 2 sessions composed of 12 trials each one, resulting in a total of 24 trials containing rest state (5 s in length), pedaling MI (up to 5 s), and passive pedaling movements (5 s in length). It is worth mentioning that each trial was defined for a period of 20 s (5 s “resting" and 5 s “MI") capturing the calibration set, whereas each trial was designed for a period up to 25 s (5 s “resting", 5 s “MI", and 5 s receiving passive movements) collecting evaluation set in an on-line condition. In on-line, each participant was encouraged to trigger a motorized pedal by executing pedaling MI tasks, and he/she received passive movements for a period of 5 s as a feedback after successfully turning on the pedal [4].
In this study, we exploit this database for obtaining robust methods for feature extraction, enhancing the discrimination between rest state and pedaling MI. As novelty, we aim to find the best feature vectors from pedaling MI by analyzing their proximity to equivalent patterns from real movements. Then, we used those periods of 5 s from the evaluation set containing cortical activity generated while participants received passive movements.
2.2 Proposed BCI
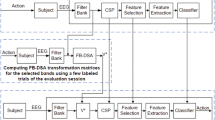
Figure 1 shows our EEG-based MI BCI proposal, which Calibration phase (see Fig. 1a) is designed here for participants to perform pedaling MI task for a period of 5 s, and receive successively passive movements for other period of 5 s. In practice, this approach should promote cortical and physical motor rehabilitation since the Calibration phase, targeting one of the main goal of patients and physiotherapists, more motor training during rehabilitation session. Next subsections our proposed BCI is in detail, including a new approach for robust feature extraction.
2.2.1 Pre-processing
Similar to [4] EEG data from both Calibration and On-line phases is band-pass filtered over epochs of 1s each 64 ms, using in sequence the Fast Fourier Transform (FFT) and the inverse FFT. Specifically, EEG signals are transformed into the frequency domain for removing all frequency components outside the range of interest, for after getting back the EEG filtered into the time domain through an inverse transformation [4]. In our study, two frequency ranges from 0.1 to 30 Hz [4, 10] and from 8 to 30 Hz [7, 11] for band-pass filtering were evaluated on each participant.
2.2.2 Discriminative Feature Extraction
In our approach, we assume that the collected EEG data from each participant on the Calibration phase has several trials containing the same real and imagery motor tasks. Thus, in order to obtain more discriminative feature vectors for both rest state and pedaling MI during Calibration phase, we propose to include patterns from passive pedaling movements in the feature analysis. This approach is composed of the following stages: 1) Feature extraction using filtered EEG epochs of 1 s with rest state, MI and passive (real) movements motor tasks; 2) Finding feature vectors of MI closer to passive movements patterns; 3) Composing a new balanced calibration set with the best feature vectors from MI and rest state to finally calibrate the MI-based BCI.
The CSP method [9] and the Riemannian geometry [4, 7, 8] were used independently or combined in our study for feature extraction, which operate on a set of covariance matrices, computed in our case from EEG epochs of 1 s. The former is a supervised spatial filter by Linear Discriminat Analysis (LDA), widely used to extract the most discriminative features X, which are obtained after projecting the set of covariance matrices through a matrix P that only retains the first and last m rows [11]. Similarly the set of covariance matrices can be analyzed by Riemmanian geometry to calculate a projection matrix P onto the tangential space, and obtain the spatial feature set X. This implementation using Riemmanian geometry can be performed by the functions covariances, meancovariances, Tangentspace, available at.Footnote 1 It is worth mentioning that the projection matrix P is computed here in both methods, only considering the covariance matrices from rest state and pedaling MI.
The second stage aims to select the feature vectors from pedaling MI which are more related to passive movements. Let X \(\in \) \(\mathbb {R}^{N \times f}\) be a balanced feature vector set containing N objects x\(_i\) \(\in \) \(\mathbb {R}^{1 \times f}\) linked to pedaling MI (first N/3 objects), passive movements (next N/3 objects), and rest state (last N/3 objects). The probability of each MI pattern to come from the rest state is given by the Equations (1)-(3).
where i ranges from 1 to N, j takes values from 1 to 2N/3, \(d_{ij}\) is the Euclidean distance between x\(_i\) and x\(_j\), \(p_{ij}\) is the probability of x\(_i\) and x\(_j\) to correspond to a same class, \(p_j\) is the probability of x\(_{j}\) be a pattern coming from the rest state. Then, \(p_j\) values is used as a threshold \((p_j > p_{th})\) in order to select those MI patterns closer to rest state, as shown in Equation (4). For further analysis, we denote M the total non-rejected MI patterns.
where \(p_{mi}\) is a set containing the probability \(p_j\) of MI patterns (\(j = 1\) to N/3), and \(p_{rm}\) is a set containing the probability \(p_j\) of real motor patterns (\(j = N/3+1\) to 2N/3).
Equations (1)-(3) are also used to find those M patterns from rest state with less probability of be associated to those non-rejected MI feature vectors. Let X contains first feature vectors linked to rest state (N/3 objects), followed by the best feature vectors from pedaling MI (next M objects), i ranges from 1 to \(N/3+M\) and j takes values from 1 to N/3 applying now Equations (1)-(2), while i ranges from \(N/3+1\) to \(N/3+M\) for Equation (3) and each j value. Afterwards, a balanced new calibration set is formed by EEG epochs containing more probable MI tasks \((p_j \le p_{th})\) and rest state.
Finally, the covariance matrices are again computed on the new balanced set (with M EEG epochs per class), updating their projection matrix P for obtaining the spatial features X. Notice that P was also used for feature extraction over covariance matrices of EEG epochs from the testing set (calibration phase) and evaluation set (from the on-line phase). Also notice that P can be computed again by CSP or Riemannian geometry.
2.2.3 Feature Selection and Classification
Here, the new calibration set X from Riemmanian geometry containing feature vectors from MI tasks and rest state are analyzed through the Pair-Wise Feature Proximity (PWFP) method to remove redundant features, using Fisher score computation and feature similarity [4, 12]. The best feature vectors are used to compute the classification model, as shown in Fig. 1(a). Three different classifiers based on discriminant analysis were tested, such as LDA using diagonal or full matrix, and Regularized Discriminant Analysis (RDA). These classifiers have been successfully applied for EEG based-BCIs and achieved promising results to discriminate both resting state and pedaling MI classes [4, 7, 13].
2.3 Statistical Evaluation
Both calibration and validation sets were employed to evaluate the EEG-based MI BCI, using our approach for robust feature extraction through Riemannian geometry and/or CSP. Here, the metrics Accuracy (ACC) and Kappa were used for comparison.
3 Results and Discussion
As aforementioned, two frequency ranges were evaluated. In addition, the BCI performance using our proposed method was compared with the Baseline Method (without probability analysis).
Figures 2 and 3 show the performance of the proposed BCI (pBCI) in both Calibration and On-line phases, respectively, over eight healthy subjects (P01 to P08) and two post-stroke patients (PS1 and PS2) during both rest state and pedaling MI recognition. On Calibration phase, our proposed BCI (pBCI) achieved ACC values up to 76.98% (Kappa = 0.54) and 75.93% (Kappa = 0.52) for healthy subjects and post-stroke patients groups, respectively, using only Riemannian geometry. On the other hand, when Riemannian geometry was combined with CSP, ACC up to 68.00% (Kappa = 0.36) and 73.45% (Kappa = 0.45) were achieved for each groups, respectively. Finally, our approach, using only CSP, achieved in each group ACC up to 73.21% (Kappa = 0.46) and 72.14% (Kappa = 0.44), respectively.
The calibrated BCI was used on the evaluation set (from the Online phase) to recognize epochs of 1 s each 64 ms throughout periods that each participant was performing pedaling MI tasks to trigger the motorized pedal. As shown in Fig. 3, the proposed BCI (pBCI) using only Riemannian geometry presented high accuracy or outperformed the Baseline Method (BM) for almost all participants (except for Patient PS2). Particularly, our approach based on LDA using diagonal matrix achieved on healthy subjects better performance (ACC up to 95.29%) than RDA or LDA using full matrix (for both ACC up to 90.91%). Also, our approach reached low values (ACC up to 15.09%) for PS1, increasing its performance for PS2 without outperforming BM.
In addition, Fig. 3 shows the results using Riemannian geometry combined with CSP. For seven participants (P01, P02, P03, P04, P06, P08 and PS1), pBCI achieved high accuracy or outperformed BM, similar when applying only Riemannian geometry. For healthy subjects, LDA based on full matrix presented better performance (ACC up to 98.18%) than LDA using diagonal matrix and RDA. Furthermore, for PS1 the performance using pBCI increased notably (ACC up to 71.07%) compared to BM (ACC up to 0.16%).
Finally, the proposed system using only CSP presented high accuracy or outperformed the BM both for the six healthy subjects (P01, P03, P04, P05, P06 and P08) and all post-stroke subjects, as shown in Fig. 3. Similar to our approach using Riemannian geometry, CSP plus LDA based on diagonal matrix achieved better performance for healthy subjects (ACC up to 98.43%) and PS1 (ACC up to 65.41%) than applying LDA with full matrix or RDA. Furthermore, PS2 achieved high values for the pBCI (ACC up to 79.24%) using LDA with full matrix and RDA, compared to BM (ACC up to 25.85%).
Notice that our approach using Riemannian geometry achieved better results on the Calibration phase than applying CSP or Riemannian geometry plus CSP (mean ACC of 68.81%, 61.08% and 63.38%, respectively). Also notice in the Online phase, Riemannian geometry plus CSP reached better performance than using only Riemannian geometry or CSP (mean ACC of 73.84%, 65.25% and 70.08%, respectively).
3.1 Further Discussion
BCIs based on MI may play an important role to motor rehabilitation in post-stroke patients, in cases where post-stroke patients may need assistance to voluntarily initiate a movement [4, 7]. Then, our EEG-based MI system may improve their lower-limb rehabilitation through triggering the motorized pedal using MI. In [10], the authors presented a study based on pedaling MI using 32 EEG channels to apply the CAR filter and they explored different setups of electrode configuration (between 4 and 9) and feature extraction methods in frequency domain (mean band power from the power spectral density) to evaluate their system. Five healthy subjects took part in the experiment and an accuracy of 55.1% was obtained. In [7] a recognition system of pedaling motor imagery using 19 EEG channels for lower-limb rehabilitation was proposed, which used Riemannian geometry to improve the feature extraction. Ten healthy subjects were selected for the tests, which consisted of two-stages in order to obtain EEG dataset for calibration and validation. The recognition system based on subject-specific bands achieved mean accuracy of 96.43%. Despite of the high accuracy, only ten healthy subjects evaluated this approach, and a subject-specific band location was not performed [7]. In [4], a low-cost BCI based on 8 EEG channels for lower-limb motor recovery of post-stroke patients was proposed using Riemannian geometry, in which two post-stroke subjects obtained accuracies of 41.67% and 91.67%, and healthy subjects reached 100%. Although high accuracy was reached, patients felt tiredness due to the long time of Calibration phase [4].
As a highlight, our proposed system encourages the patient to execute MI tasks and provide passive movements during BCI calibration step. We hypothesize that our approach of BCI intervention may be more effective for post-stroke rehabilitation, reducing patient’s boredom. In addition, the use of real movements for obtaining the best features vectors from MI and rest state may improve the BCI calibration, which can make the BCI intervention more effective.
4 Conclusion
This work introduced an EEG-based MI BCI to recognize pedaling motor imagery for post-stroke patient rehabilitation, which improved its performance by applying methods to select a better calibration set using probability analysis and exploring different methods for feature extraction. Our approach using Riemannian geometry plus CSP achieved better result (mean ACC of 73.84%) when compared to other methods. As a limitation of our study, we performed the analysis using a database. In future works, an experimental protocol will be carried out on healthy patients, using the proposed system. Also, methods for subject-specific bands will be explored.
References
Vourvopoulos A, Jorge C, Abreu R, Figueiredo P, Fernandes J, Badia SB (2019) Efficacy and brain imaging correlates of an immersive motor imagery BCI-driven VR system for upper limb motor rehabilitation: a clinical case report. Front Hum Neurosci 13
About stroke at https://www.stroke.org/en/about-stroke
Tamburella F, Moreno JC, Valenzuela DSH et al (2019) Influences of the biofeedback content on robotic post-stroke gait rehabilitation: electromyographic vs joint torque biofeedback. J NeuroEng Rehabil 16
Romero-Laiseca MA, Delisle-Rodriguez D, Cardoso V et al (2020) A low-cost lower-limb brain-machine interface triggered by pedaling motor imagery for post-stroke patients rehabilitation. IEEE Trans Neural Syst Rehabil Eng 28:988–996
Nagai H, Tanaka T (2019) Action observation of own hand movement enhances event-related desynchronization. IEEE Trans Neural Syst Rehabil Eng 27:1407–1415
Ang KK, Chua KSG, Phua KS et al (2014) A randomized controlled trial of EEG-based motor imagery brain-computer interface robotic rehabilitation for stroke. Clin EEG Neurosci 46:310–320
Delisle-Rodriguez D, Cardoso V, Gurve D et al (2019) System based on subject-specific bands to recognize pedaling motor imagery: towards a BCI for lower-limb rehabilitation. J Neural Eng 16:056005
Yger F, Berar M, Lotte F (2017) Riemannian approaches in brain-computer interfaces: a review. IEEE Trans Neural Syst Rehabil Eng 25:1753–1762
Molla MKI, Shiam AA, Islam MR, Tanaka T (2020) Discriminative feature selection-based motor imagery classification using EEG signal. IEEE Access 8:98255–98265
Rodríguez-Ugarte M, Iáñez E, Ortíz M, Azorín JM (2017) Personalized offline and pseudo-online BCI models to detect pedaling intent. Front Neuroinformatics 11:45
Müller-Gerking J, Pfurtscheller G, Flyvbjerg H (1999) Designing optimal spatial filters for single-trial EEG classification in a movement task. Clin Neurophysology 110:787–798
Happy SL, Mohanty R, Routray A (2017) An effective feature selection method based on pair-wise feature proximity for high dimensional low sample size data. In: 2017 25th European signal processing conference (EUSIPCO)
Lotte F, Bougrain L, Cichocki A et al (2018) A review of classification algorithms for EEG-based brain–computer interfaces: a 10 year update. J Neural Eng 15:031005
Acknowledgements
This study was financed in part by FAPES (Finance Code: 33361.503.19197.11092017). Authors would like to thank CNPq and FAPES/CAPES (88887.198558/2018-00) from Brazil.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this paper
Cite this paper
Silva, L.A., Delisle-Rodriguez, D., Bastos-Filho, T. (2022). Finding Discriminant Lower-Limb Motor Imagery Features Highly Linked to Real Movements for a BCI Based on Riemannian Geometry and CSP. In: Bastos-Filho, T.F., de Oliveira Caldeira, E.M., Frizera-Neto, A. (eds) XXVII Brazilian Congress on Biomedical Engineering. CBEB 2020. IFMBE Proceedings, vol 83. Springer, Cham. https://doi.org/10.1007/978-3-030-70601-2_337
Download citation
DOI: https://doi.org/10.1007/978-3-030-70601-2_337
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-70600-5
Online ISBN: 978-3-030-70601-2
eBook Packages: EngineeringEngineering (R0)






