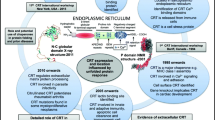
Abstract
Calreticulin (Calr) is an endoplasmic reticulum (ER) chaperone involved in protein quality control, Ca2+ regulation and other cellular processes. The structure of Calr is unusual, reflecting different functions of the protein: a proline-rich β-hairpin arm and an acidic C-terminal tail protrude from a globular core, composed of a β-sheet sandwich and an α-helix. The arm and tail interact in the presence of Ca2+ and cover the upper β-sheet, where a carbohydrate-binding site gives the chaperone glycoprotein affinity. At the edge of the carbohydrate-binding site is a conserved, strained disulphide bridge, formed between C106 and C137 of human Calr, which lies in a polypeptide-binding site. The lower β-sheet has several conserved residues, comprised of a characteristic triad, D166-H170-D187, Tyr172 and the free C163. In addition to its role in the ER, Calr translocates to the cell surface upon stress and functions as an immune surveillance marker. In some myeloproliferative neoplasms, the acidic Ca2+-binding C-terminal tail is transformed into a polybasic sequence.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
2.1 Introduction
In eukaryotic cells, ER is the site for synthesis, maturation and quality control of secreted, membrane-bound and ER-resident proteins (Halperin et al. 2014; Kepp and Galluzzi 2020). This process involves translation of mRNAs on ER membrane-bound ribosomes, translocation of the nascent polypeptide across the ER membrane and co-translational folding to the native three-dimensional structure. To assist in these processes, the ER contains elaborate systems involved in post-translational modification and quality control of newly synthesized proteins (Vincenz-Donnelly L and Hipp MS 2017; Halperin et al. 2014). An important protein quality control system in the ER is constituted by the chaperones calreticulin (Calr) and calnexin (Canx), which act in concert with protein disulfide isomerases (e.g. Pdia3/ERp57) to facilitate the synthesis and maturation of proteins, and to prevent accumulation of misfolded proteins (Bergeron et al. 1994; Michalak et al. 1998; Coe and Michalak 2010; Halperin et al. 2014; Matsusaki et al. 2020). Canx is a transmembrane protein, with its major part in the ER lumen, while Calr is an ER luminal protein (Bergeron et al. 1994; Wada et al. 1995; Michalak et al. 1998; Danilczyk et al. 2000). In humans, these proteins are encoded by separate genes with 9 (Calr) or 20 (Canx) exons (McCauliffe et al. 1992; Tjoelker et al. 1994). In mammals, Calr and Canx furthermore play important roles in the loading of peptides on MHC class I (Blees et al. 2017). This chapter reviews current knowledge on the structure of human Calr (hCalr).
2.2 Primary Structures of Calr and Related Proteins
Analyses of Canx and Calr amino acid sequences have revealed that they are evolutionarily conserved proteins encoded by single genes. In the testicles a Canx homologue, calmegin (Clgn) and a Calr homologue, calsperin (Calr3) are also present (Ohsako et al. 1994; Watanabe et al. 1994; Ikawa et al. 2011). In plants, several Calr isoforms are encoded by separate genes (Jia et al. 2009; Del Bem 2011; Wasąg et al. 2019). In yeasts, a Canx homologue is present (de Virgilio et al. 1993; Parlati et al. 1995).
Figure 2.1 shows an alignment of amino acid sequences of selected Calr/Canx family members. Common to both Canx and Calr is an N-terminal domain of approximately 200 amino acid residues, followed by a proline-rich domain of approximately 100 (Calr and Calr3) or 145 (Canx and Clgn) amino acid residues, which is followed by a C-terminal domain. In Calr (and Calr3) the C-terminal (C) domain is highly acidic and consists of approximately 100 amino acids ending in an ER retrieval sequence of four residues (KDEL in hCalr). In Canx (and Clgn) the C-terminal domain has a transmembrane stretch of 20 amino acids and a cytoplasmic “tail” of approximately 90 amino acids.
Alignment of Calrs and related proteins from various species. (a) Calr E. histolytica XP_655241; (b) Calr D. discoideum Q23858; (c) Calr M. musculus P14211; (d) Calr R. norvegicus P18418; (e) Calr H. sapiens P27797; (f) Calr C. elegans P27798; (g) Calr3 H. sapiens Q96L12; (h) Calr T. cruzi XP_812571; (i) Canx C. lupus P24643; (j) Canx H. sapiens P27824; (k) Clgn H. sapiens O14967. The alignment was carried out using Clustal 2.1 and default parameters. Coloring: core region—blue, P-domain—yellow. C-domain—green
Relatively few residues are invariant among all Calr and related proteins (Fig. 2.1) and these mainly reside in the N-terminal (N) domain and the middle proline-rich (P) domain. Conserved stretches/clusters of amino acids in the N-domain are found in the vicinity of the conserved disulfide bridge (C106-C137 in Calr) and in the P-domain in the neighborhood of conserved W residues, being part of characteristic repeat sequences. A third interesting residue (C163 in hCalr) is conserved in all known mammalian and some other Calrs.
2.3 Higher Order Structures of Calr and Related Proteins
Table 2.1 shows a list of Calr and Canx proteins from different species with solved three dimensional (3D) structures. Figure 2.2 shows two partial structures for human Calr, plus one view of its full-length elongated structure in the context of the MHC-I peptide-loading complex. The 3D structures of Canx and Calr show close similarity but also some important differences. Both proteins have a globular core consisting of a β-sheet sandwich, where the strands are arranged in a “jelly roll” fold, and an α-helix. The sandwich is composed of two β-sheets, where the N-terminal 200 amino acids contribute 6 strands to the first, “upper” β-sheet and 6 strands to the second “lower” β-sheet. Intertwined in the two sheets are two β-strands derived from the C-terminal domain of the molecules, the first constituting the Calr β19 strand of the upper β-sheet and the second constituting the Calr β20a/b strand of the lower β-sheet (Fig. 2.2a). From the core extend the proline-rich stretches as β-hairpins (P-domain, Fig. 2.2c), stabilised by hydrogen bonds between four (Canx) or three (Calr) sets of repeat sequences. The α-helix of the globular core is longer in Calr, where it continues into the acidic tail (Fig. 2.2c), while in Canx the α-helix is followed by a transmembrane sequence and the cytoplasmic domain.
H. Sapiens (Hs) Calr structures. (a) XRC core structure (Chouquet et al. 2011). The structural Ca2+ ion is shown in yellow. (b) XL-MS structure (Boelt et al. 2016). (c) HsCalr (red) in complex with MHC-I (yellow), Pdia3 (blue), tapasin (cyan) and ß2-microglobulin (green) (Blees et al. 2017). (d) Zoom on the cluster2 residues common to Calr (cyan) and Canx (green) proteins
There are several interesting characteristic features of the conserved structure of Calr. A carbohydrate-binding site on the upper concave β-sheet capable of accommodating a tetrasaccharide and having affinity for terminal glucose residues. This site contains several conserved residues (cluster 1) of importance for carbohydrate binding (Kozlov et al. 2010a, b; Chouquet et al. 2011; Moreau et al. 2016).
An exposed disulphide bridge (C106–C137 in hCalr) is located on the edge of the carbohydrate-binding site. This bridge is unusual due to its surface location and its geometry, which reveals an almost parallel, strained conformation with a sulphur atom distance of 2.8–3 Å (in contrast to normal unstrained disulphide bonds, which have a torsion angle of approximately 90° and a sulphur atom distance of 2 Å (Thornton 1981). The disulphide bridge is important for thermodynamic stability and reduceable by DTT but is not affected by alkylating agents under conditions, where the free Cys163 in hCalr can be alkylated (100 mM sodium phosphate, pH 7.2, room temperature), showing that it is stable under near-to-physiological conditions (Houen and Koch 1994; Højrup et al. 2001; Jørgensen et al. 2005), in accordance with its surface-exposed localisation at the edge of the carbohydrate-binding site. Interestingly, the disulphide bridge seems to be important for lectin activity (Kozlov et al. 2010a).
The lower β-sheet has a conserved structural Ca2+ ion-binding site (Fig. 2.2a) and also contains several residues conserved in published structures of Calr and Caln (cluster 2) and in primary sequences of Calr and related proteins (Fig. 2.1), including a triad composed of D166, H170 and D187 (DHD triad). Finally, the C-terminal α-helix contains two conserved residues including W347, close to the DHD triad (Fig. 2.2d) (Kozlov et al. 2010a, b; Chouquet et al. 2011; Moreau et al. 2016).
Chemical cross-linking in solution in combination with mass spectrometry (MS) of human Calr has confirmed the overall structure of the globular domain and has added some structural information on the parts of Calr absent in the crystallised “amputated” Calr (Fig. 2.2b). In the presence of Ca2+, the P-domain hairpin folds back on itself and associates with the C-domain, which itself forms an α-helix extending “upwards” to meet the P-domain. Together, they cover the lectin site, however, in the absence of Ca2+, they are more flexible and do not sterically limit access to the lectin site to the same extent. X-ray studies of Trypanosoma cruzi and Entamoeba histolytica Calrs have also shown for the first time a possible hinge motion at the basis of the Calr P-domain, as a switch between an open and a more compact conformation. This part of the Calr molecule differs in Canx (Moreau et al. 2016).
Structural studies of full-length Calr proteins have been hindered by its flexibility. However, its overall ordered structure could be trapped by cryo-electron microscopy in the context of the MHC-I peptide-loading complex (Blees et al. 2017). In this complex, Calr still shows substantial flexibility, as compared to its structurally stable binding partners. This flexibility remains despite the fact that it interacts with an MHC-class I glycan through its carbohydrate-binding site, with Pdia3 through the tip of its P-arm and with tapasin through its acidic tail.
The overall structure of Calr and Canx indicated by the structural data for various amputated forms of the molecules has been confirmed by several studies with other methods including electron cryo-microscopy (ECM), small angle X-ray scattering (SAXS) and nuclear magnetic resonance (NMR) spectroscopy (Table 2.1). Both molecules have a stable globular beta sandwich core with an α helix at the bottom. From the core protrudes a β-hairpin P-domain and a C-domain, which in the presence of Ca2+ make contacts and cover the lectin site.
2.4 Calr Structure-Function Relationships
The thermodynamic stability of Calr is moderate and depends on the general packing of the core region, the conserved, surface-exposed disulphide bridge and Ca2+ association (Jørgensen et al. 2005; Duus et al. 2013). Calr and Canx are chaperones for glycoprotein synthesis and several of the conserved residues in cluster 1, including the disulphide bridge, have been shown to be involved in lectin activity of Calr/Canx (Kozlov et al. 2010a, b). These residues are also strikingly conserved in the parasite Calrs (T. cruzi, E. histolytica) (Moreau et al. 2016). A putative polypeptide binding site has been located at the edge of the carbohydrate-binding upper β-sheet and Calr has been shown to interact with several non-glycosylated proteins (Duus et al. 2009; Pocanschi et al. 2011; Chouquet et al. 2011; Møllegaard et al. 2011; Moreau et al. 2016).
Calr has been implicated in several diseases including cancers, autoimmune diseases, and others (Tesniere et al. 2008; Gold et al. 2010; Zamanian et al. 2013; Wiersma et al. 2015; Eggleton et al. 2016; Schcolnik-Cabrera et al. 2019; Houen 2019). Most evident in relation to structure are somatic alterations seen in myeloproliferative neoplasms (MPNs), including polycythemia vera, myelofibrosis and essential thrombocythemia (Klampfl et al. 2013; Nangalia et al. 2013; Pietra et al. 2016; Imai et al. 2017). These alterations are mainly insertions and deletions (INDELS) in exon 9 leading to a common polybasic stretch of amino acids in the C-terminus, in close conjunction to the α-helix in the C-domain. Since these INDELS have only been detected in MPNs, it can be speculated that the frame-shifted Calr forms allow the affected myeloid precursor cells to remain viable, to grow and differentiate to some degree and to exhibit neoplastic properties. The structural polybasic alterations of the C-terminus can be speculated to mimic the binding of Ca2+ to the acidic C-terminus of wild type Calr, allowing the protein to remain stable and viable, but imparting some new properties on the molecule (i.e. an oncogenic alteration). In agreement with this, the frameshifted Calr has been shown to interact with the thrombopoietin receptor, which itself can cause MPN upon mutation and which signals through a pathway involving the Janus kinase (JAK), mutations of which is the most prominent cause of MPN (Vainchenker et al. 2019).
In relation to cancer in general, Calr has been shown to translocate to the cell surface in response to various forms of cellular stress, where it may serve as a signal for immune recognition (Tesniere et al. 2008; Wiersma et al. 2015; Eggleton et al. 2016; Schcolnik-Cabrera et al. 2019). However, it is presently unclear, whether the various involvements of Calr in cell transformation and immune surveillance are related to structural alterations or to changes in concentration and subcellular localization.
2.5 Summary Points
-
Calr and Canx are conserved Ca2+-binding molecular chaperones in eucaryotes.
-
Both Calr and Canx have a globular β-sandwich/α-helix core with a structural Ca2+ ion. From the core extends a characteristic β-hairpin P-domain and a C-domain. In Calr, the C-domain is acidic and has a large capacity for Ca2+ binding. In Canx, the C-domain has a transmembrane stretch and a cytoplasmic domain.
-
The upper β-sheet of the globular core sandwich has a carbohydrate-binding site and a disulphide bridge at the edge, which both are involved in carbohydrate interactions. A polypeptide binding site is located at the edge of the carbohydrate-binding site but has not been precisely mapped yet.
-
The lower β-sheet of the globular sandwich has a conserved D166-H170-D187 triad, a free C163 and several other conserved residues, including Y172 (hCalr numbering) (Fig. 2.2d).
-
The C-terminal α-helix at the bottom of the α-helix core has a conserved W347 residue facing the D166-H170-D187 triad (hCalr numbering) (Fig. 2.2d).
-
The structure of Calr/Canx proteins reflects their multiple functions and interaction partners, including nascent (glycosylated) proteins, cyclophilin, Pdia3, and others, as illustrated by the role of Calr and Canx in the peptide loading complex.
-
Characteristic mutations in the exon coding for the extreme C-terminus of hCalr changes the polyacidic Ca2+-binding sequence to a polybasic sequence.
2.6 Future Issues
-
Detailed structural analysis of complete Calr/Canx proteins in complex with substrates and binding partners.
-
Detailed analysis of the role of individual amino acids in the thermodynamic stability and functions of Calr and Canx.
-
Detailed understanding of the role of the polybasic sequence in frameshifted Calr variants involved in myeloproliferative neoplasms.
-
Detailed understanding of cell surface functions of Calr.
Abbreviations
- Calr:
-
Calreticulin
- Calr3:
-
Calsperin
- Canx:
-
Calnexin
- CDS:
-
Circular dichroism spectroscopy
- Clgn:
-
Calmegin
- DTT:
-
Dithiothreitol
- ECM:
-
Electron cryo-microscopy
- ER:
-
Endoplasmic reticulum
- INDELS:
-
Insertions and deletions
- JAK:
-
Janus kinase
- MHC:
-
Major histocompatibility complex
- MM:
-
Molecular modelling
- MPN:
-
Myeloproliferative neoplasms
- NMRS:
-
Nuclear magnetic resonance spectroscopy
- SAXS:
-
Small angle X-ray scattering
- XL-MS:
-
Cross-linking/mass spectrometry
- XRC:
-
X-ray crystallography
References
Bergeron JJ, Brenner MB, Thomas DY, Williams DB (1994) Calnexin: a membrane-bound chaperone of the endoplasmic reticulum. Trends Biochem Sci 19:124–128. https://doi.org/10.1016/0968-0004(94)90205-4
Blees A, Januliene D, Hofmann T, Koller N, Schmidt C, Trowitzsch S, Moeller A, Tampé R (2017) Structure of the human MHC-I peptide-loading complex. Nature 551:525–528. https://doi.org/10.1038/nature24627
Boelt SG, Norn C, Rasmussen MI, André I, Čiplys E, Slibinskas R, Houen G, Højrup P (2016) Mapping the Ca2+ induced structural change in calreticulin. J Proteome 142:138–148. https://doi.org/10.1016/j.jprot.2016.05.015
Chouquet A, Païdassi H, Ling WL, Frachet P, Houen G, Arlaud GJ, Gaboriaud C (2011) X-ray structure of the human calreticulin globular domain reveals a peptide-binding area and suggests a multi-molecular mechanism. PLoS One 6:e17886. https://doi.org/10.1371/journal.pone.0017886
Coe H, Michalak M (2010) ERp57, a multifunctional endoplasmic reticulum resident oxidoreductase. Int J Biochem Cell Biol 42:796–799. https://doi.org/10.1016/j.biocel.2010.01.009
Danilczyk UG, Cohen-Doyle MF, Williams DB (2000) Functional relationship between calreticulin, calnexin, and the endoplasmic reticulum luminal domain of calnexin. J Biol Chem 275:13089–13097. https://doi.org/10.1074/jbc.275.17.13089
de Virgilio C, Bürckert N, Neuhaus JM, Boller T, Wiemken A (1993) CNE1, a Saccharomyces cerevisiae homologue of the genes encoding mammalian calnexin and calreticulin. Yeast 9:185–188. https://doi.org/10.1002/yea.320090209
Del Bem LE (2011) The evolutionary history of calreticulin and calnexin genes in green plants. Genetica 139:255–259. https://doi.org/10.1007/s10709-010-9544-y
Duus K, Sandhu N, Jørgensen CS, Hansen PR, Steinø A, Thaysen-Andersen M, Højrup P, Houen G (2009) Interaction of the chaperone calreticulin with proteins and peptides of different structural classes. Protein Pept Lett 16:1414–1423. https://doi.org/10.2174/092986609789353772
Duus K, Larsen N, Tran TA, Güven E, Skov LK, Jespersgaard C, Gajhede M, Houen G (2013) Chemical and thermal unfolding of calreticulin. Protein Pept Lett 20:562–568. https://doi.org/10.2174/0929866511320050009
Eggleton P, Bremer E, Dudek E, Michalak M (2016) Calreticulin, a therapeutic target? Expert Opin Ther Targets 20:1137–1147. https://doi.org/10.1517/14728222.2016.1164695
Ellgaard L, Riek R, Herrmann T, Güntert P, Braun D, Helenius A, Wüthrich K (2001a) NMR structure of the calreticulin P-domain. Proc Natl Acad Sci U S A 98:3133–3138. https://doi.org/10.1073/pnas.051630098
Ellgaard L, Riek R, Braun D, Herrmann T, Helenius A, Wüthrich K (2001b) Three-dimensional structure topology of the calreticulin P-domain based on NMR assignment. FEBS Lett 488:69–73. https://doi.org/10.1016/s0014-5793(00)02382-6
Ellgaard L, Bettendorff P, Braun D, Herrmann T, Fiorito F, Jelesarov I, Güntert P, Helenius A, Wüthrich K (2002) NMR structures of 36 and 73-residue fragments of the calreticulin P-domain. J Mol Biol 322:773–784. https://doi.org/10.1016/s0022-2836(02)00812-4
Gold LI, Eggleton P, Sweetwyne MT, Van Duyn LB, Greives MR, Naylor SM, Michalak M, Murphy-Ullrich JE (2010) Calreticulin: non-endoplasmic reticulum functions in physiology and disease. FASEB J 24:665–683. https://doi.org/10.1096/fj.09-145482
Hahn M, Borisova S, Schrag JD, Tessier DC, Zapun A, Tom R, Kamen AA, Bergeron JJ, Thomas DY, Cygler M (1998) Identification and crystallization of a protease-resistant core of calnexin that retains biological activity. J Struct Biol 123:260–264. https://doi.org/10.1006/jsbi.1998.4032
Halperin L, Jung J, Michalak M (2014) The many functions of the endoplasmic reticulum chaperones and folding enzymes. IUBMB Life 66:318–326. https://doi.org/10.1002/iub.1272
Højrup P, Roepstorff P, Houen G (2001) Human placental calreticulin characterization of domain structure and post-translational modifications. Eur J Biochem 268:2558–2565. https://doi.org/10.1046/j.1432-1327.2001.02138.x
Houen G (2019) COMMENTARY: calreticulin - oncogene, anti-oncogene, or both? Curr Protein Pept Sci 20:111–112. https://doi.org/10.2174/138920372001181031111149
Houen G, Koch C (1994) Human placental calreticulin: purification, characterization and association with other proteins. Acta Chem Scand 48:905–911. https://doi.org/10.3891/acta.chem.scand.48-0905
Ikawa M, Tokuhiro K, Yamaguchi R, Benham AM, Tamura T, Wada I, Satouh Y, Inoue N, Okabe M (2011) Calsperin is a testis-specific chaperone required for sperm fertility. J Biol Chem 286:5639–5646. https://doi.org/10.1074/jbc.M110.140152
Imai M, Araki M, Komatsu N (2017) Somatic mutations of calreticulin in myeloproliferative neoplasms. Int J Hematol 105:743–747. https://doi.org/10.1007/s12185-017-2246-9
Jia XY, He LH, Jing RL, Li RZ (2009) Calreticulin: conserved protein and diverse functions in plants. Physiol Plant 136:127–138. https://doi.org/10.1111/j.1399-3054.2009.1223.x
Jørgensen CS, Trandum C, Larsen N, Ryder LR, Gajhede M, Skov LK, Højrup P, Barkholt V, Houen G (2005) Conformational stability of calreticulin. Protein Pept Lett 12:687–693. https://doi.org/10.2174/0929866054696082
Kepp O, Galluzzi L (eds) (2020) Biology of the endoplasmic reticulum. Int Rev Cell Mol Biol 350:1–325. https://doi.org/10.1016/S1937-6448(20)30020-4
Klampfl T, Gisslinger H, Harutyunyan AS, Nivarthi H, Rumi E, Milosevic JD, Them NC, Berg T, Gisslinger B, Pietra D, Chen D, Vladimer GI, Bagienski K, Milanesi C, Casetti IC, Sant'Antonio E, Ferretti V, Elena C, Schischlik F, Cleary C, Six M, Schalling M, Schönegger A, Bock C, Malcovati L, Pascutto C, Superti-Furga G, Cazzola M, Kralovics R (2013) Somatic mutations of calreticulin in myeloproliferative neoplasms. N Engl J Med 369:2379–2390. https://doi.org/10.1056/NEJMoa1311347
Kozlov G, Pocanschi CL, Rosenauer A, Bastos-Aristizabal S, Gorelik A, Williams DB, Gehring K (2010a) Structural basis of carbohydrate recognition by calreticulin. J Biol Chem 285:38612–38620. https://doi.org/10.1074/jbc.M110.168294
Kozlov G, Bastos-Aristizabal S, Määttänen P, Rosenauer A, Zheng F, Killikelly A, Trempe JF, Thomas DY, Gehring K (2010b) Structural basis of cyclophilin B binding by the calnexin/calreticulin P-domain. J Biol Chem 285:35551–35557. https://doi.org/10.1074/jbc.M110.160101
Kozlov G, Muñoz-Escobar J, Castro K, Gehring K (2017) Mapping the ER interactome: the P domains of calnexin and calreticulin as plurivalent adapters for foldases and chaperones. Structure 25:1415–1422. https://doi.org/10.1016/j.str.2017.07.010
Matsusaki M, Kanemura S, Kinoshita M, Lee YH, Inaba K, Okumura M (2020) The protein disulfide isomerase family: from proteostasis to pathogenesis. Biochim Biophys Acta 1864:129338. https://doi.org/10.1016/j.bbagen.2019.04.003
McCauliffe DP, Yang YS, Wilson J, Sontheimer RD, Capra JD (1992) The 5'-flanking region of the human calreticulin gene shares homology with the human GRP78, GRP94, and protein disulfide isomerase promoters. J Biol Chem 267:2557–2562
Michalak M, Mariani P, Opas M (1998) Calreticulin, a multifunctional Ca2+ binding chaperone of the endoplasmic reticulum. Biochem Cell Biol 76:779–785. https://doi.org/10.1139/bcb-76-5-779
Møllegaard KM, Duus K, Træholt SD, Thaysen-Andersen M, Liu Y, Palma AS, Feizi T, Hansen PR, Højrup P, Houen G (2011) The interactions of calreticulin with immunoglobulin G and immunoglobulin Y. Biochim Biophys Acta 1814:889–899. https://doi.org/10.1016/j.bbapap.2011.03.015
Moreau C, Cioci G, Iannello M, Laffly E, Chouquet A, Ferreira A, Thielens NM, Gaboriaud C (2016) Structures of parasite calreticulins provide insights into their flexibility and dual carbohydrate/peptide-binding properties. IUCrJ 3:408–419. https://doi.org/10.1107/S2052252516012847
Nangalia J, Massie CE, Baxter EJ, Nice FL, Gundem G, Wedge DC, Avezov E, Li J, Kollmann K, Kent DG, Aziz A, Godfrey AL, Hinton J, Martincorena I, Van Loo P, Jones AV, Guglielmelli P, Tarpey P, Harding HP, Fitzpatrick JD, Goudie CT, Ortmann CA, Loughran SJ, Raine K, Jones DR, Butler AP, Teague JW, O'Meara S, McLaren S, Bianchi M, Silber Y, Dimitropoulou D, Bloxham D, Mudie L, Maddison M, Robinson B, Keohane C, Maclean C, Hill K, Orchard K, Tauro S, Du MQ, Greaves M, Bowen D, BJP H, Harrison CN, NCP C, Ron D, Vannucchi AM, Papaemmanuil E, Campbell PJ, Green AR (2013) Somatic CALR mutations in myeloproliferative neoplasms with nonmutated JAK2. N Engl J Med 369:2391–2405. https://doi.org/10.1056/NEJMoa1312542
Nørgaard Toft K, Larsen N, Steen Jørgensen F, Højrup P, Houen G, Vestergaard B (2008) Small angle X-ray scattering study of calreticulin reveals conformational plasticity. Biochim Biophys Acta 1784:1265–1270. https://doi.org/10.1016/j.bbapap.2008.05.005
Ohsako S, Hayashi Y, Bunick D (1994) Molecular cloning and sequencing of calnexin-t. An abundant male germ cell-specific calcium-binding protein of the endoplasmic reticulum. J Biol Chem 269:14140–14148
Parlati F, Dignard D, Bergeron JJ, Thomas DY (1995) The calnexin homologue cnx1+ in Schizosaccharomyces pombe, is an essential gene which can be complemented by its soluble ER domain. EMBO J 14:3064–3072
Pietra D, Rumi E, Ferretti VV, Di Buduo CA, Milanesi C, Cavalloni C, Sant'Antonio E, Abbonante V, Moccia F, Casetti IC, Bellini M, Renna MC, Roncoroni E, Fugazza E, Astori C, Boveri E, Rosti V, Barosi G, Balduini A, Cazzola M (2016) Differential clinical effects of different mutation subtypes in CALR-mutant myeloproliferative neoplasms. Leukemia 30:431–438. https://doi.org/10.1038/leu.2015.277
Pocanschi CL, Kozlov G, Brockmeier U, Brockmeier A, Williams DB, Gehring K (2011) Structural and functional relationships between the lectin and arm domains of calreticulin. J Biol Chem 286:27266–27277. https://doi.org/10.1074/jbc.M111.258467
Pollock S, Kozlov G, Pelletier MF, Trempe JF, Jansen G, Sitnikov D, Bergeron JJ, Gehring K, Ekiel I, Thomas DY (2004) Specific interaction of ERp57 and calnexin determined by NMR spectroscopy and an ER two-hybrid system. EMBO J 23:1020–1029. https://doi.org/10.1038/sj.emboj.7600119
Schcolnik-Cabrera A, Oldak B, Juárez M, Cruz-Rivera M, Flisser A, Mendlovic F (2019) Calreticulin in phagocytosis and cancer: opposite roles in immune response outcomes. Apoptosis 24:245–255. https://doi.org/10.1007/s10495-019-01532-0
Schrag JD, Bergeron JJ, Li Y, Borisova S, Hahn M, Thomas DY, Cygler M (2001) The structure of calnexin, an ER chaperone involved in quality control of protein folding. Mol Cell 8:633–446. https://doi.org/10.1016/s1097-2765(01)00318-5
Tesniere A, Panaretakis T, Kepp O, Apetoh L, Ghiringhelli F, Zitvogel L, Kroemer G (2008) Molecular characteristics of immunogenic cancer cell death. Cell Death Differ 15:3–12. https://doi.org/10.1038/sj.cdd.4402269
Thielmann Y, Weiergräber OH, Mohrlüder J, Willbold D (2009) Structural framework of the GABARAP-calreticulin interface--implications for substrate binding to endoplasmic reticulum chaperones. FEBS J 276:1140–1152. https://doi.org/10.1111/j.1742-4658.2008.06857.x
Thornton JM (1981) Disulphide bridges in globular proteins. J Mol Biol 151:261–287
Tjoelker LW, Seyfried CE, Eddy RL Jr, Byers MG, Shows TB, Calderon J, Schreiber RB, Gray PW (1994) Human, mouse, and rat calnexin cDNA cloning: identification of potential calcium binding motifs and gene localization to human chromosome 5. Biochemistry 33:3229–3236. https://doi.org/10.1021/bi00177a013
Vainchenker W, Plo I, Marty C, Varghese LN, Constantinescu SN (2019) The role of the thrombopoietin receptor MPL in myeloproliferative neoplasms: recent findings and potential therapeutic applications. Expert Rev Hematol 12:437–448. https://doi.org/10.1080/17474086.2019.1617129
Villamil Giraldo AM, Lopez Medus M, Gonzalez Lebrero M, Pagano RS, Labriola CA, Landolfo L, Delfino JM, Parodi AJ, Caramelo JJ (2010) The structure of calreticulin C-terminal domain is modulated by physiological variations of calcium concentration. J Biol Chem 285:4544–4553. https://doi.org/10.1074/jbc.M109.034512
Vincenz-Donnelly L, Hipp MS (2017) The endoplasmic reticulum: a hub of protein quality control in health and disease. Free Radic Biol Med 108:383–393. https://doi.org/10.1016/j.freeradbiomed.2017.03.031
Wada I, Imai S, Kai M, Sakane F, Kanoh H (1995) Chaperone function of calreticulin when expressed in the endoplasmic reticulum as the membrane-anchored and soluble forms. J Biol Chem 270:20298–20304. https://doi.org/10.1074/jbc.270.35.20298
Wasąg P, Grajkowski T, Suwińska A, Lenartowska M, Lenartowski R (2019) Phylogenetic analysis of plant calreticulin homologs. Mol Phylogenet Evol 134:99–110. https://doi.org/10.1016/j.ympev.2019.01.014
Watanabe D, Yamada K, Nishina Y, Tajima Y, Koshimizu U, Nagata A, Nishimune Y (1994) Molecular cloning of a novel Ca2+-binding protein (calmegin) specifically expressed during male meiotic germ cell development. J Biol Chem 269:7744–7749
Wiersma VR, Michalak M, Abdullah TM, Bremer E, Eggleton P (2015) Mechanisms of translocation of ER chaperones to the cell surface and immunomodulatory roles in cancer and autoimmunity. Front Oncol 5:7. https://doi.org/10.3389/fonc.2015.00007
Zamanian M, Veerakumarasivam A, Abdullah S, Rosli R (2013) Calreticulin and cancer. Pathol Oncol Res 19:149–154. https://doi.org/10.1007/s12253-012-9600-2
Conflicts-of-Interest Disclosure Statement
GH, PHP, EC, CG, RS declare no conflicts-of-interest.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Houen, G., Højrup, P., Ciplys, E., Gaboriaud, C., Slibinskas, R. (2021). Structural Analysis of Calreticulin, an Endoplasmic Reticulum-Resident Molecular Chaperone. In: Agellon, L.B., Michalak, M. (eds) Cellular Biology of the Endoplasmic Reticulum . Progress in Molecular and Subcellular Biology, vol 59. Springer, Cham. https://doi.org/10.1007/978-3-030-67696-4_2
Download citation
DOI: https://doi.org/10.1007/978-3-030-67696-4_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-67695-7
Online ISBN: 978-3-030-67696-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)