Abstract
Naked mole-rats are extremely tolerant to low concentrations of oxygen (hypoxia) and high concentrations of carbon dioxide (hypercapnia), which is consistent with the environment that they inhabit. Naked mole-rats combine subterranean living with living in very densely populated colonies where oxygen becomes depleted and carbon dioxide accumulates. In the laboratory, naked mole-rats fully recover from 5 h exposure to 5% O2 and 5 h exposure to 80% CO2, whereas both conditions are rapidly lethal to similarly sized laboratory mice. During anoxia (0% O2) naked mole-rats enter a suspended animation-like state and switch from aerobic metabolism of glucose to anaerobic metabolism of fructose. Additional fascinating characteristics include that naked mole-rats show intrinsic brain tolerance to anoxia; a complete lack of hypoxia-induced and CO2-induced pulmonary edema; and reduced aversion to high concentrations of CO2 and acidic fumes. Here we outline a constellation of physiological and molecular adaptations that correlate with the naked mole-rat’s hypoxic/hypercapnic tolerance and which offer potential targets for ameliorating pathological conditions in humans, such as the damage caused during cerebral ischemia.

Photo Credit: Thomas Park
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
9.1 Tolerance to Hypoxia and Anoxia
The naked mole-rat (Heterocephalus glaber) shows robust tolerance to both low concentrations of oxygen (O2) and high concentrations of carbon dioxide (CO2) (Larson and Park 2009; Park et al. 2017). We suggest that these tolerances and their underlying mechanisms, which are detailed below, are adaptations to living in a chronically hypoxic/hypercapnic environment where many individuals share the same limited air supply resulting in low concentrations of O2 and high concentrations of CO2 (Bennett and Faulkes 2000). Figure 9.1 provides the highlights of the findings presented in this chapter.
In the laboratory, we found that naked mole-rats tolerate severe hypoxia (5% O2) for at least 5 h with no notable effect, whereas 5% O2 is lethal to similarly sized laboratory mice (Mus musculus) in less than 15 min (Fig. 9.2a). Similarly, we also tested naked mole-rats and mice with an even more extreme challenge: 0% O2 (anoxia). The procedure we followed was to monitor breathing and terminate the exposure when 60 seconds passed without a breath for the naked mole-rats, and when 20 seconds passed for mice. Both species became unconscious in less than a minute. However, the last breath for naked mole-rats was, on average, 250 seconds after entering 0% O2 whereas the last breath for mice was, on average, a much shorter 46 seconds (Fig. 9.2b). Another striking species difference was that all of the naked mole-rats recovered and were returned to their colonies, whereas none of the mice recovered.
Extreme hypoxia (5% O2) and anoxia (0% O2) resistance in naked mole-rats. (a) Animals were placed into an atmosphere chamber that was pre-filled with 5% O2 balanced with nitrogen. The bars indicate the average duration in hours until breathing ceased or until the experiment was arbitrarily terminated after 5 h. N = 4 animals for each group. Time to cessation of breathing was significantly shorter for mice compared to naked mole-rats (t-test, t = 563 (df = 6), p < .0001). Also, after re-exposure to room air, all naked mole-rats recovered, but none of the mice recovered. (b) Animals were placed into an atmosphere chamber that was pre-filled with 0% O2 (100% nitrogen). The bars indicate the average duration in minutes until breathing ceased. N = 4 animals for each group. Time to cessation of breathing was significantly shorter for mice compared to naked mole-rats (t-test, t = 112.53 (df = 6), p < .0001). Also, after re-exposure to room air, all naked mole-rats recovered, but no mice recovered. Photos on the right depict a naked mole-rat and a mouse. NMR = naked mole-rat. (These data were originally published in Park et al. 2017)
In a different experimental protocol, we exposed naked mole-rats and mice to 0% O2 for a variety of fixed durations (6, 10, 18, and 30 min) while we monitored breathing and heart rates. The groups of naked mole-rats exposed for 6, 10, and 18 min recovered, whereas the group exposed for 30 min did not. Data from the group exposed for 18 min are shown in Fig. 9.3. The atmosphere chamber that we used to test the animals is shown in Fig. 9.3a. The naked mole-rats showed a rapid and dramatic decrease in both breathing rate (Fig. 9.3b) and heart rate (Fig. 9.3c), with heart rate remaining remarkably invariant for the duration of the exposure. In contrast, the mice showed a steady decline in heart rate which became undetectable after 6 min (black curve in Fig. 9.3c) and breathing ceased in less than a minute.
Effects of anoxia on respiration rate and heart rate in mice and naked mole-rats. (a). Photograph showing a naked mole-rat standing in a chamber where gas mixtures were delivered. (b and c). During 18 min exposure to anoxia, respiration rate and heart rate were drastically reduced in naked mole-rats. The red bar on the x axis of b and c indicates when the animals were exposed to anoxia. During anoxia, the heart of the naked mole-rats continued to beat (pink curve in c), while the heart rate of the mice (black curve in c) declined rapidly to the level of noise. N = 3 naked mole-rats and 4 mice. After re-exposure to room air, all of the naked mole-rats recovered, but none of the mice did. (These data were originally published in Park et al. 2017)
Next, we used electroencephalography (EEG) to measure brain activity before, during, and after a 10-min bout of anoxia. We found that anoxia resulted in a severe reduction in the average amplitude of the EEG recordings (Fig. 9.4). The dramatic reduction in brain activity, in addition to the robust reduction in respiration rate and heart rate (Fig. 9.3), suggests that naked mole-rats enter a suspended animation-like state (Blackstone et al. 2005; Blackstone and Roth 2007) during anoxia, which would greatly reduce energy demand. This hypothesis is supported by Pamenter et al. (2018) who demonstrated that naked mole-rats can suppress their metabolism by up to 85% in acute severe hypoxia.
Effects of anoxia on EEG recordings from naked mole-rats. The trace at the top is an example from one animal. The vertical calibration bar corresponds to 600 microvolts. The graph below shows the average integral over time. Anoxia began at time 0. The inset expands the data for the 10 min of anoxia. n = 3. For each animal, two recording electrodes were stereotaxically positioned into the caudate putamen. Recordings were acquired using a 4 channel Pinnacle Technology EEG system
In the in vivo experiments described above, the naked mole-rats showed robust tolerance to oxygen deprivation compared to mice. Another hypoxia-related phenomenon displayed by naked mole-rats is intrinsic brain tolerance to hypoxia in vitro (Larson and Park 2009; Larson et al. 2014). To assess intrinsic brain tolerance, we used the hippocampal brain slice paradigm. We measured time to anoxic depolarization (loss of all electrical activity) after switching from 95% O2 to nominal 0% O2 in the slice recording chamber . Slices from naked mole-rats maintained synaptic function during anoxia for significantly longer than slices from mice (Fig. 9.5).
Naked mole-rat hippocampal neurons are resistant to anoxic depolarization. The gas mixture supplying the slice chamber was switched from 95% O2/ 5% CO2 to 95% nitrogen/5% CO2. The graph indicates the average duration in minutes to anoxic depolarization (AD). N = 11 slices from naked mole-rats and 36 slices from mice. Slices from mice showed AD significantly faster than slices from naked mole-rats (t-test, t = 8.85 (DF = 45), p < .0001). These data were originally published in Larson and Park 2009, but this graph includes additional data collected since the original publication. The photograph shows slices in the recording chamber . (Photograph by Thomas Park)
9.2 Tolerance to Hypercapnia
In their home environment, naked mole-rats may not only be challenged by hypoxic conditions, but also by hypercapnic conditions. To assess global, in vivo tolerance to chronic hypercapnia in naked mole-rats and mice, we exposed them to a mixture of 80% CO2 and 20% O2 for 5 h (hypercapnia without hypoxia). The naked mole-rats tolerated and recovered from this exposure with no overt issue, whereas the mice died, on average, in under 5 min (Fig. 9.6).
Naked mole-rats are resistant to hypercapnia. Animals were placed into an atmosphere chamber that was pre-filled with 80% CO2 and 20% O2. The bars indicate the average duration in hours until breathing ceased or until the experiment was arbitrarily terminated at 5 h. N = 4 animals for each group. Time to cessation of breathing was significantly shorter for mice compared to naked mole-rats (t-test, t = 980.73 (df = 6), p < .0001). Also, after re-exposure to room air, all naked mole-rats recovered, but none of the mice recovered. (These data were originally published in Park et al. 2017)
9.3 Lack of Pulmonary Edema from Hypoxia or Hypercapnia
In humans and laboratory mice and rats, both hypoxia and hypercapnia cause pulmonary edema, (Bärtsch et al. 2005; Sitkovsky et al. 2004; Guais et al. 2011; Pritchett et al. 2005). In contrast, naked mole-rats do develop neither hypoxia- nor hypercapnia-induced pulmonary edema (Park et al. 2017). To assess hypoxia-induced pulmonary edema, we exposed naked mole-rats and mice to low O2 concentrations for 15 min and then determined the wet-to-dry ratio of the lungs post-mortem at the end of the experiment. Naked mole-rats showed a complete absence of pulmonary edema following exposure to low O2, whereas the mice showed severe pulmonary edema for O2 concentrations below 10% (Fig. 9.7a). To assess CO2-induced pulmonary edema, we measured lung wet-to-dry ratios after exposing animals to a high concentration of CO2 for 15 min. As for hypoxia, naked mole-rats showed no pulmonary edema following exposure to any CO2 concentrations they were exposed to, whereas mice showed robust pulmonary edema from exposure to all concentrations of CO2 over 15% (Fig. 9.7b).
Naked mole-rats do not develop hypoxia-induced or hypercapnia-induced pulmonary edema. Edema was assessed by calculating the wet lung weight-to-dry lung weight ratio. The curves represent percent increase from baseline. N = 3–5 animals for each concentration. (a) Hypoxia-induced pulmonary edema. For 7.5% O2, mouse lung tissue showed significantly higher wet-to-dry ratio values than naked mole-rat lung tissue (t = 4.84 (df = 5), p < .001). This was also the case for 5% O2 (t = 2.94 (df = 6), p < .05). (b) CO2-induced pulmonary edema. For 20% CO2, mouse lung tissue showed significantly higher wet-to-dry ratio values than that of naked mole-rats (t = 12.03 (df = 5), P < .0001). This was also the case for 33% CO2 (t = 8.35 (df = 5), P < .001) and for 50% CO2 (t = 11.05 (df = 4), P < .001). (These data were originally published in Park et al. 2017)
9.4 Putative Adaptations for Hypoxia Tolerance
Naked mole-rats display a number of biological features that may be adaptations that contribute to this species’ hypoxia tolerance. The resting metabolic rate of naked mole-rats is about two-thirds that of similarly sized mammals (Buffenstein and Yahav 1991), meaning that they require less O2-derived energy to maintain normal function. Much of that saving is due to lack of thermogenesis in this species (Buffenstein and Yahav 1991; see Buffenstein and Craft 2021). In addition, hemoglobin in the blood of naked mole-rats has an unusually high affinity for oxygen (Johansen et al. 1976; Hacker et al. 2020), meaning that naked mole-rat hemoglobin can achieve a higher percentage of saturation in low O2 concentrations compared to most other mammals.
As shown in Fig. 9.5, the brain of the naked mole-rat shows intrinsic hypoxia tolerance (Larson and Park 2009). Hippocampal brain slices from naked mole-rats maintain synaptic function under hypoxia and anoxia for much longer than other mammals that have been tested (Larson et al. 2014). Part of the intrinsic tolerance is likely associated with retention of the glutamate N-methyl-D-aspartate (NMDA) receptor subunit GluN2D in adulthood. In contrast to other glutamate receptors, NMDA receptors are highly Ca2+ permeable and during periods of hypoxia, a lack of oxygen results in decreased ATP production and a concomitant membrane depolarization causing glutamate release (and impaired glutamate reuptake), as well as relieving NMDA receptors from magnesium ion block. Consequently, NMDA receptors are a key source of neuronal excitotoxicity during hypoxic events, such as during stroke (Wu and Tymianski 2018). NMDA receptors are tetrameric ion channels and the GluN2D subunit is inhibited by hypoxia (Bickler et al. 2003; Peterson et al. 2012b), which can thus suppress NMDA-mediated excitotoxicity. In most mammals, the GluN2D subunit is prevalent in neonates, but its expression decreases precipitously during maturation (Laurie et al. 1997). Indeed, we observed that adult mouse brains only retain about 13% of neonatal levels of GluN2D, whereas adult naked mole-rat brains retain about 66% of neonate levels (Fig. 9.8a). Therefore, we propose that the maintained high level of GluN2D in naked mole-rat brain provides resistance to hypoxia-mediated, NMDA receptor-induced excitotoxicity.
Retention of the GluN2D NMDA receptor subunit in adult naked mole-rat brains. (a). Graph showing the percentage of GluN2D retained in adult for mice and naked mole-rats compared to neonatal levels. The raw data was optical density of immunoreactive bands in hippocampal slices from neonates and adults. N = 4 neonates and 4 adults for each species. The graph is from Peterson et al. (2012b). (b). Peak intracellular Ca2+ accumulation from a 10-min exposure to hypoxia in hippocampal slices from p6 and p20 mice and naked mole-rats. The raw data were measurements of relative internal cytosolic calcium indicated by fura-2 fluorescence emission. N = 10–14 slices from 3 to 6 animals for each group. (c). Example fluorescence images from a mouse before hypoxia (top) and during hypoxia (bottom) Quenching in the bottom image indicates an increase in intracellular Ca2+. (b and c) are from Peterson et al. (2012a)
Consistent with retention of high levels of GluN2D in adult naked mole-rat brain, hippocampal brain slices from adult naked mole-rats show substantially less intracellular calcium accumulation during hypoxia compared to laboratory mice (Peterson et al. 2012a). Hypoxia-induced intracellular calcium accumulation is associated with neuronal injury and cell death (Deshpande et al. 1987; Lee et al. 1991; Bickler 2004). We observed that slices from both mice and naked mole-rats responded to hypoxia with a decrease in fluorescence, corresponding to an increase in intracellular Ca2+ (Fig. 9.8b). Also, whereas in both mice and naked mole-rats, more Ca2+ accumulated in slices from older animals, slices from naked mole-rats still accumulated significantly less Ca2+ than the slices from mice regardless of age.
During hypoxia, oxygen depletion forces the brain into generating ATP from anaerobic glycolysis, which produces lactic acid as a by-product. Consequently, alongside glutamate-mediated excitotoxicity, a process called acidotoxicity occurs due to the activation of Ca2+-permeable acid-sensing ion channel 1a (ASICa) (Xiong et al. 2004). There are 6 different ASIC subunits, which show similar distribution throughout the brain of naked mole-rats and mice (with the exception of a virtual absence of the acid-insensitive ASIC4 subunit) (Schuhmacher and Smith 2016). However, hippocampal and cortical neurons from naked mole-rats have much smaller ASIC-mediated currents compared to mice and cortical neurons show resistance to acidotoxicity (Husson and Smith 2018), i.e. reduced ASIC function provides resistance to the impact of acidosis during periods of hypoxia.
An additional adaptation that contributes to the naked mole-rat’s anoxia tolerance involves a switch from aerobic metabolism of glucose to anaerobic metabolism of fructose (Park et al. 2017; Reznick et al. 2021). The evidence for this remarkable adaptation is as follows. Under anoxia, concentrations of fructose increased dramatically in the blood and organs of naked mole-rats, but not mice (Fig. 9.9). Importantly, cells in naked mole-rat organs have higher expression levels of the fructose transporter, GLUT5 (Fig. 9.10a) which transports fructose into cells, and significantly higher levels of the enzyme ketohexokinase (Fig. 9.10b), which converts fructose into fructose-1-phosphate that can enter glycolysis. Thus, under anoxia, it appears that naked mole-rats release fructose into their blood and organs, and they have the molecular machinery in place to pump the fructose into their cells and convert it into an intermediate that can be used to make energy.
Concentrations of fructose in the blood and organs of mice and naked mole-rats. Exposed to normoxia versus 10 min of anoxia. (These data were originally published in Park et al. 2017)
Naked mole-rat cells show multiple adaptations for utilizing fructose. (a), the fructose transporter, GLUT5 is expressed at higher levels in all naked mole-rat organs compared to mice, other than in the kidney. (b), ketohexokinase (KHK-A) can metabolize fructose into fructose-1-phosphate and is expressed at significantly higher levels in multiple organs of naked mole-rats compared to mice. Mouse data is depicted with white bars and naked mole-rat data is depicted with pink bars. (These data were originally published in Park et al. 2017)
The advantage of the fructose pathway is that it can drive glycolysis in the absence of oxygen. In contrast, the glucose pathway is blocked during anoxia due to the generation of protons as a by-product of glycolysis inhibiting phosphofructokinase, which is essential for converting fructose-6-phosphate to fructose-1,6-bisphosphate (Fig. 9.11a). However, fructose metabolism in the absence of oxygen generates much less ATP compared to glucose metabolism in the presence of oxygen. And this may be the driving force behind the naked mole-rat entering a suspended animation-like state during anoxia (Figs. 9.3 and 9.4): to reduce and match energy use with reduced energy production. Presumably, only a small amount of energy is needed to maintain this suspended animation state under anoxia.
Utilization of fructose by naked mole-rat brains. (a) Model of the glycolysis pathway showing how fructose enters cells via the GLUT5 transporter and is converted into fructose-1-phosphate by ketohexokinase (KHK) which enters the glycolysis pathway; note the end-production-inhibition of phosphofructokinase by protons, which prevents continuous glucose fueled glycolysis. (b, c) Data from brain slices that were incubated with carbon 13 labeled fructose and then exposed to 5% O2 for 5, 15, or 30 min. For the two example intermediates (DHAP and Pyruvate) there was significantly more 13carbon accumulation during hypoxia in slices from naked mole-rats compared to slices from mice, indicating that naked mole-rat cells can incorporate more fructose-derived carbons compared to mice. (These data were originally published in Park et al. 2017)
To demonstrate that naked mole-rat brain cells could actually use fructose, we incubated brain slices in a solution that included 13carbon-labeled fructose. Then we exposed the slices to hypoxia and measured the quantity of 13carbon in several glycolysis intermediates. Most of the intermediates that we sampled showed significantly larger increases in 13carbon in slices from naked mole-rats compared to slices from mice (Fig. 9.11b, c).
Next, we tested the ability of hippocampal brain slices and isolated hearts from naked mole-rats and mice to function when we switch from glucose to fructose in the bath solutions. Brain slices from naked mole-rats showed less decline and better recovery from application of fructose compared to slices from mice (Fig. 9.12a). Isolated hearts from naked mole-rats showed virtually no decline in function, whereas hearts from mice showed a substantial decline, particularly during a second application of fructose (Fig. 9.12b). These data support the hypothesis that naked mole-rat cells are adapted to utilize fructose as an energy source to maintain cellular function during periods of hypoxia.
Brain slice and isolated heart function when glucose was replaced with fructose in naked mole-rats, but not in mice. (a) Drawing of the hippocampal slice preparation (left) and evoked potential data from naked mole-rats and mice before, during, and after switching from glucose to fructose in the bath solution. (b) Cardiac function data from isolated hearts from naked mole-rats and mice before, during, and after switching from glucose to fructose in the bath solution twice in series. Abbreviations: fEPSP field Excitatory Postsynaptic Potentials, LVDP Left Ventricular Developed Pressure. (These data were originally published in Park et al. 2017)
Intriguingly, Farhat et al. (2020) showed that, under chronic hypoxia (11% O2 for 4 weeks), naked mole-rat brain tissue showed a significant and substantial decrease in metabolic enzyme activity and brain sodium/potassium-ATPase activity. These results suggest a strategy for energy conservation during oxygen deprivation: naked mole-rat cells can make adjustments to match metabolic need with energy production.
9.5 Putative Adaptations for Hypercapnia Tolerance
There are also several underlying features that have been identified as potential adaptations that contribute to this species’ hypercapnia tolerance. A major pathological issue associated with breathing high concentrations of CO2 is the resulting acidosis that triggers pulmonary edema (Lee and Pisarri 2001; Russell et al. 1984; Gourine 2005; Park et al. 2017). The blood of naked mole-rats is however exceptionally efficient at buffering acid (Johansen et al. 1976; Park et al. 2017).
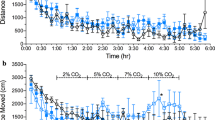
Also, there are several interesting features of the sensory cells in naked mole-rats that normally detect and respond to acidosis. These unmyelinated peripheral nerve fibers are called C fibers. Firstly, naked mole-rats have far fewer cutaneous C fibers than many other mammals, including several other African mole-rat species (Smith et al. 2012). Secondly, the C fibers of naked mole-rats lack the neuropeptides Substance P and calcitonin gene related peptide (Park et al. 2003, 2008), which, in other mammals are released during acidosis of the lungs and are thought to contribute to pulmonary edema (Germonpré et al. 1995). Third, the voltage-gated sodium channel NaV1.7 on the C fibers of naked mole-rats has an amino acid variation that results in enhanced NaV1.7 inhibition preventing action potential generation in the presence of acid , even though acid sensors (e.g. TRPV1 and ASICs) are functional, i.e. acid acts like an anesthetic of sensory neurones (Smith et al. 2011). As a result, naked mole-rats not only show a lack of CO2-induced pulmonary edema (Fig. 9.7b), but they also show reduced avoidance to CO2 (Fig. 9.13a) and air borne acetic acid fumes (Fig. 9.13b) and virtually no response to foot injection of acidic saline (Fig. 9.13c) compared to mice (Park et al. 2008; Eigenbrod et al. 2019). See Lewin et al.(2021) for details on the pain biology of the naked mole-rat.
Naked mole-rats show reduced sensitivity to CO2 and complete insensitivity to acetic acid fumes and injection of acetic saline. (a) Avoidance to CO2 was tested in a chamber where one end was infused with room air and the other end with CO2 (either 2.5%, 5%, or 10%). We recorded the time spent near each end as a metric of avoidance. Mice spent relatively little time near the CO2 end of the chamber compared to the room air end for each concentration of CO2. Naked mole-rats only avoided the highest concentration. (These data were originally published in Park et al. 2017). (b) A similar behavioral test using 20% acetic acid fumes showed strong avoidance in laboratory rats and mice but not naked mole-rats. (These data were originally published in LaVinka and Park 2012). (c) Mice injected in the foot with acidic saline show robust licking at the injection site indicative of pain but naked mole-rats show virtually no licking. (These data were originally published in Park et al. 2008)
9.6 Discussion and Summary
In summary, naked mole-rats have evolved to thrive in an environment that would be deadly to many other mammals: an environment that may be characterized by sporadically low concentrations of O2 and high concentrations of CO2, for example during communal sleeping in cramped chambers. In laboratory experiments, naked mole-rats show remarkable tolerance to both hypoxia and hypercapnia. A constellation of putative adaptations has been identified in association with their tolerances. For hypoxia tolerance: intrinsic brain tolerance to hypoxia, the ability to enter a low-energy, suspended animation-like state, and the ability to use anaerobic fructose metabolism. Hypercapnia tolerance is provided for by peripheral nerve fibers that do not respond to acidosis from CO2, which would usually be associated with pain and pulmonary edema.
We previously proposed that naked mole-rats achieve at least part of their tolerance by retaining some neonatal characteristics (Larson and Park 2009; Peterson et al. 2012a, b; Penz et al. 2015; Orr et al. 2016). It has been known for some time that neonate mammals are tolerant to hypoxia and hypercapnia (Bickler et al. 2003; Kirschbaum and DeHaven 1968; Pritchett et al. 2005). Naked mole-rats and neonatal mice share a common brain characteristic associated with intrinsic hypoxia tolerance in the form of elevated levels of the NMDA GluN2D subunit (Peterson et al. 2012b), which is switched off by hypoxia and thus limits Ca2+ entry (Peterson et al. 2012a) and calcium toxicity (Bickler et al. 2003). In addition, naked mole-rat hippocampal and cortical neurons also display smaller acid-gated currents mediated by ASICs, which provides resistance to acidotoxicity (Husson and Smith 2018) in response to hypoxia generated acidosis.
Tolerance to CO2-induced pulmonary edema (Fig. 9.7) is likely related to differential functioning of the sensory nerves in the lungs that normally detect acidosis and trigger edema. This population of sensory nerves are C fibers that come from the spinal nerves and the vagus nerve. Normally these nerve fibers respond to acidosis by releasing neuropeptides that trigger vasodilation and bronchoconstriction, resulting in pulmonary edema (Germonpré et al. 1995; Lee and Pisarri 2001; Russell et al. 1984). This process does not happen in the naked mole-rats. Naked mole-rats do not fire action potentials in response to acidosis because they express a variant of NaV1.7 that inhibits the channel (Smith et al. 2011). Neonatal mice also appear to have a non-functional neurokinin pathway. However, in the neonates this appears to be a developmental issue, not a gene variant (King and Barr 2003; Pritchett et al. 2005).
Fructose metabolism in the naked mole rat is particularly interesting as it is an adaptation of preexisting pathways present in other mammalian models. In naked mole-rat cells, the fructose transporter GLUT5 is upregulated compared to the mouse, as is ketohexokinase, a key enzyme that converts fructose to a metabolite that can be utilized in glycolysis to produce energy in the absence of oxygen. This secondary energy source can fuel the brain and heart during anoxic challenges and is important in our understanding of metabolism under stressors as experienced in ischemic stroke, heart attack, metabolic syndrome and cancer.
It seems likely that the extreme tolerance of the naked mole-rat will prove to involve numerous adaptations in addition to those that have been published and discussed in this chapter. One avenue to discovery will undoubtedly involve using the published genome of the naked mole-rat, which is proving to be a tremendously powerful tool for exploring the relationship between genetic sequence and the extreme phenotypic characteristics of this species (Kim et al. 2011; Fang et al. 2014). For example, naked mole-rats display a unique point mutation in hypoxia-inducible factor 1α (HIF1α), a master regulator of gene transcription that activates over 40 downstream genes in response to low oxygen conditions. This mutation in the VHL-binding domain is highly consequential because under normoxic conditions, VHL controls ubiquitin-dependent degradation of HIF1α. The naked mole-rat’s T407I mutation is consistent with a relaxation of HIF1α degradation and resultant higher gene expression of this critically important regulator of hypoxic signal transduction (Kim et al. 2011). Indeed, there is now some evidence for a contribution of HIF1α to hypoxia resistance in the naked mole-rat (Xiao et al. 2017).
In summary, it is becoming clear that there is great value in the study of animals such as the naked mole-rat, which can complement the traits held by conventional laboratory animal models. The naked mole-rat exhibits a constellation of features that map to unique scenarios similar to human pathologies and provide insights into mechanisms and therapies that could prove translational.
References
Bärtsch P, Mairbäurl H, Maggiorini M, Swenson ER (2005) Physiological aspects of high-altitude pulmonary edema. J Appl Physiol 98:1101–1110
Bennett NC, Faulkes CG (2000) African Mole-Rats: Ecology and Eusociality. Cambridge University Press, Cambridge
Bickler PE (2004) Clinical perspectives: neuroprotection lessons from hypoxia-tolerant organisms. J Exp Biol 207:3243–3249
Bickler PE, Fahlman CS, Taylor DM (2003) Oxygen sensitivity of NMDA receptors: relationship to NR2 subunit composition and hypoxia tolerance of neonatal neurons. Neuroscience 118:25–35
Blackstone E, Roth MB (2007) Suspended animation-like state protects mice from lethal hypoxia. Shock 27:370–372
Blackstone E, Morrison M, Roth MB (2005) H2S induces a suspended animation-like state in mice. Science 308(5721):518
Buffenstein R, Craft W (2021) The idiosyncratic physiological traits of the naked mole-rat; a resilient animal model of aging, longevity, and healthspan. In: Buffenstein R, Park TJ, Holmes MM (eds) The Extraordinary Biology of the Naked Mole-Rat. Springer, New York, pp 221–254
Buffenstein R, Yahav S (1991) Is the naked mole-rat Heterocephalus glaber an endothermic yet poikilothermic mammal? J Therm Biol 16:227–232
Deshpande JK, Siesjo BK, Wieloch T (1987) Calcium accumulation and neuronal damage in the rat hippocampus following cerebral ischemia. J Cereb Blood Flow Metab 7:89–95
Eigenbrod O, Debus KY, Reznick J, Bennett NC, Sánchez-Carranza O, Omerbašić D, Hart DW, Barker AJ, Zhong W, Lutermann H, Katandukila JV, Mgode G, Park TJ, Lewin GR (2019) Rapid molecular evolution of pain insensitivity in multiple African rodents. Science 364(6443):852–859
Fang X, Seim I, Huang Z, Gerashchenko MV, Xiong Z, Turanov AA, Zhu Y, Lobanov AV, Fan D, Yim SH, Yao X, Ma S, Yang L, Lee SG, Kim EB, Bronson RT, Šumbera R, Buffenstein R, Zhou X, Krogh A, Park TJ, Zhang G, Wang J, Gladyshev VN (2014) Adaptations to a subterranean environment and longevity revealed by the analysis of mole rat genomes. Cell Rep 8:1354–1364
Farhat E, Devereaux MEM, Pamenter ME, Weber J-M (2020) Naked mole-rats suppress energy metabolism and modulate membrane cholesterol in chronic hypoxia. Am J Physiol Regul Integr Comp Physiol 319:R148–R155
Germonpré PR, Joos GF, Pauwels RA (1995) Characterization of the neurogenic plasma extravasation in the airways. Arch Int Pharmacodyn Ther 329:185–203
Gourine AV (2005) On the peripheral and central chemoreception and control of breathing: an emerging role of ATP. J Physiol 568:715–724
Guais A, Brand G, Jacquot L, Karrer M, Dukan S, Grévillot G, Molina TJ, Bonte J, Regnier M, Schwartz L (2011) Toxicity of carbon dioxide: a review. Chem Res Toxicol 24:2061–2070
Hacker L, Brunker J, Smith ESJ, Quiros-Gonzalez I, Bohndiek SE (2020) Photoacoustics resolves species-specific differences in hemoglobin concentration and oxygenation. J Biomed Opt 25:095002
Husson Z, Smith ESJ (2018) Naked mole-rat cortical neurons are resistant to acid-induced cell death. Mol Brain 11:26
Johansen K, Lykkeboe G, Weber RE, Maloiy GM (1976) Blood respiratory properties in the naked mole rat Heterocephalus glaber, a mammal of low body temperature. Respir Physiol 28(3):303–314
Kim EB, Fang X, Fushan AA, Huang Z, Lobanov AV, Han L, Marino SM, Sun X, Turanov AA, Yang P, Yim SH, Zhao X, Kasaikina MV, Stoletzki N, Peng C, Polak P, Xiong Z, Kiezun A, Zhu Y, Chen Y, Kryukov GV, Zhang Q, Peshkin L, Yang L, Bronson RT, Buffenstein R, Wang B, Han C, Li Q, Chen L, Zhao W, Sunyaev SR, Park TJ, Zhang G, Wang J, Gladyshev VN (2011) Genome sequencing reveals insights into physiology and longevity of the naked mole rat. Nature 479(7372):223–227
King TE, Barr GA (2003) Functional development of neurokinin peptides substance P and neurokinin a in nociception. Neuroreport 14:1603–1607
Kirschbaum TH, DeHaven JC (1968) Maternal and fetal blood constituents. In: Assali NS (ed) The Fetus and Neonate. Academic, New York, pp 143–187
Larson J, Park TJ (2009) Extreme hypoxia tolerance of naked mole-rat brain. NeuroReport 20:1634–1637
Larson J, Drew KL, Folkow LP, Milton SL, Park TJ (2014) No oxygen? No problem! Intrinsic brain tolerance to hypoxia in vertebrates. J Exp Biol 217:1024–1039
Laurie DJ, Bartke I, Schoepfer R, Naujoks K, Seeburg PH (1997) Regional, developmental and interspecies expression of the four NMDAR2 subunits, examined using monoclonal antibodies. Brain Res Mol Brain Res 51:23–32
LaVinka PC, Park TJ (2012) Blunted behavioral and c Fos responses to acidic fumes in the African naked mole-rat. PLoS One 7(9):e45060
Lee LY, Pisarri TE (2001) Afferent properties and reflex functions of bronchopulmonary C-fibers. Respir Physiol 125:47–65
Lee KS, Frank S, Vaderklish P, Arai A, Lynch G (1991) Inhibition of proteolysis protects hippocampal neurons from ischemia. Proc Natl Acad Sci U S A 88:7233–7237
Lewin GR, Smith ESJ, Reznick J, Debus K, Barker A, Park TJ (2021) The somatosensory world of the African naked mole-rat. In: Buffenstein R, Park TJ, Holmes MM (eds) The Extraordinary Biology of the Naked Mole-Rat. Springer, New York, pp 197–220
Orr ME, Garbarino VR, Salinas A, Buffenstein R (2016) Extended postnatal brain development in the longest-lived rodent: prolonged maintenance of Neotenous traits in the naked mole-rat brain. Front Neurosci 10:504
Pamenter ME, Dzal YA, Thompson WA, Milsom WK (2018) Do naked mole rats accumulate a metabolic acidosis or an oxygen debt in severe hypoxia? J Exp Biol 222: jeb191197
Park TJ, Comer C, Carol A, Lu Y, Hong HS, Rice FL (2003) Somatosensory organization and behavior in naked mole-rats: II. Peripheral structures, innervation, and selective lack of neuropeptides associated with thermoregulation and pain. J Comp Neurol 465:104–120
Park TJ, Lu Y, Jüttner R, Smith ES, Hu J, Brand A, Wetzel C, Milenkovic N, Erdmann B, Heppenstall PA, Laurito CE, Wilson SP, Lewin GR (2008) Selective inflammatory pain insensitivity in the African naked mole-rat (Heterocephalus glaber). PLoS Biol 6(1):e13
Park TJ, Reznick J, Peterson BL, Blass G, Omerbašić D, Bennett NC, Kuich PHJL, Zasada C, Browe BM, Hamann W, Applegate DT, Radke MH, Kosten T, Lutermann H, Gavaghan V, Eigenbrod O, Bégay V, Amoroso VG, Govind V, Minshall RD, Smith ESJ, Larson J, Gotthardt M, Kempa S, Lewin GR (2017) Fructose-driven glycolysis supports anoxia resistance in the naked mole-rat. Science 356(6335):307–311
Penz OK, Fuzik J, Kurek AB, Romanov R, Larson J, Park TJ, Harkany T, Keimpema E (2015) Protracted brain development in a rodent model of extreme longevity. Sci Rep 5:11592
Peterson BL, Larson J, Buffenstein R, Park TJ, Fall CP (2012a) Blunted neuronal calcium response to hypoxia in naked mole-rat hippocampus. PLoS One 7:e31568
Peterson BL, Park TJ, Larson J (2012b) Adult naked mole-rat brain retains the NMDA receptor subunit GluN2D associated with hypoxia tolerance in neonatal mammals. Neurosci Lett 506:342–345
Pritchett K, Corrow D, Stockwell J, Smith A (2005) Euthanasia of neonatal mice with carbon dioxide. Comp Med 55:275–281
Reznick J, Park TJ, Lewin GR (2021) A sweet story of metabolic innovation in the naked mole-rat. In: Buffenstein R, Park TJ, Holmes MM (eds) The Extraordinary Biology of the Naked Mole-Rat. Springer, New York, pp 271–286
Russell NJ, Raybould HE, Trenchard D (1984) Role of vagal C-fiber afferents in respiratory response to hypercapnia. J Appl Physiol 56:1550–1558
Schuhmacher LN, Smith ES (2016) Expression of acid-sensing ion channels and selection of reference genes in mouse and naked mole rat. Mol Brain 9:97
Sitkovsky MV, Lukashev D, Apasov S, Kojima H, Koshiba M, Caldwell C, Ohta A, Thiel M (2004) Physiological control of immune response and inflammatory tissue damage by hypoxia-inducible factors and adenosine A2A receptors. Annu Rev Immunol 22:657–682
Smith ESJ, Omerbašić D, Lechner SG, Anirudhan G, Lapatsina L, Lewin GR (2011) The molecular basis of acid insensitivity in the African naked mole-rat. Science 334(6062):1557–1560
Smith ESJ, Purfürst B, Grigoryan T, Park TJ, Bennett NC, Lewin GR (2012) A specific paucity of unmyelinated C-fibers in cutaneous peripheral nerves of the African naked-mole rat: a comparative analysis using six species of bathyergidae. J Comp Neurol 520:2785–2803
Wu QJ, Tymianski M (2018) Targeting NMDA receptors in stroke: new hope in neuroprotection. Mol Brain 11:15
Xiao B, Wang S, Yang G, Sun X, Zhao S, Lin L, Cheng J, Yang W, Cong W, Sun W, Kan G, Cui S (2017) HIF-1α contributes to hypoxia adaptation of the naked mole rat. Oncotarget 8:109941–109951
Xiong ZG, Zhu XM, Chu XP, Minami M, Hey J, Wei WL, MacDonald JF, Wemmie JA, Price MP, Welsh MJ, Simon RP (2004) Neuroprotection in ischemia: blocking calcium-permeable acid-sensing ion channels. Cell 118:687–698
Acknowledgements
We are grateful for the extremely helpful suggestions of two reviewers. We are also grateful for grant support from the National Science Foundation (grant number 0744979 to TJP), a Cancer Research UK/RCUK Multidisciplinary Project Award (C56829/A22053 to ESJS) and ERC advanced grants (AdG 789128 and AdG 294678 to GRL).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Park, T.J. et al. (2021). African Naked Mole-Rats Demonstrate Extreme Tolerance to Hypoxia and Hypercapnia. In: Buffenstein, R., Park, T.J., Holmes, M.M. (eds) The Extraordinary Biology of the Naked Mole-Rat. Advances in Experimental Medicine and Biology, vol 1319. Springer, Cham. https://doi.org/10.1007/978-3-030-65943-1_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-65943-1_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-65942-4
Online ISBN: 978-3-030-65943-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)