Abstract
This chapter focuses on the role of upper airway (UA) evaluation by imaging modalities in pediatric obstructive sleep apnea (OSA). This chapter emphasizes upon studies using imaging techniques that may further enhance our understanding about the mechanisms of UA obstruction leading to pediatric OSA and could assist in treatment selection.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Adenotonsillar hypertrophy is the most important predisposing factor in children with obstructive sleep apnea (OSA) [1,2,3]. The association between the subjective evaluation of tonsillar size and OSA severity as determined by polysomnography is weak. High-quality studies suggest no association [4,5,6]. Additionally, the pathogenesis of upper airway (UA) narrowing is more complex in children with risk factors such as obesity, craniofacial malformations, Down syndrome (DS), or neuromuscular disorders [7].
The first line treatment of pediatric OSA is adenotonsillectomy (ATE) [7, 8]. There are other non-surgical and surgical options for treatment that are used in residual OSA or complex OSA (children with risk factors) [7]. Treatment outcomes for ATE have been extensively studied and there is a high incidence of residual OSA after ATE, especially in children with risk factors such as obesity (up to 59%) and Down syndrome (up to 87%) [9,10,11,12,13,14,15,16,17,18]. Commonly described anatomical causes of persistent disease include mandibular deficiency, glossoptosis, macroglossia, soft palate enlargement, lymphoid hypertrophy surrounding the UA, and inferior displacement of the hyoid bone or abnormal neuromuscular control [3, 17, 19, 20].
Polysomnography gives the diagnosis of OSA but not the anatomical level of obstruction in the UA. In a time of personalized medicine, it seems crucial to couple the exact individual anatomical risk factor with the most appropriate treatment to avoid unnecessary risks and ineffective surgeries. Several studies investigated the role of UA imaging as noninvasive evaluation of UA obstruction. These techniques include lateral neck radiographs, cephalometry, computerized tomography (CT), cone beam CT, magnetic resonance imaging (MRI) and post-processing of these images using computational fluid dynamics (CFD) for functional respiratory imaging (FRI), long-range optical coherence tomography (LR-OCT), and drug induced sleep (sedation) endoscopy (DISE). Every technique has advantages and disadvantages, while an overview is presented in Table 16.1 [21].
This chapter reviews studies using imaging techniques that may further enhance our understanding about the mechanisms of UA obstruction leading to pediatric OSA and could assist in the selection of treatment.
Lateral Neck Radiography
Lateral neck radiography is relatively simple, is widely accessible, and has a low cost. However, the images are taken in an upright position in awake patients. One study in adults investigated the difference between supine and upright lateral cephalograms and observed no additional anatomical differences [22]. Another study in five adults with OSA measured the total volume and cross-sectional area (CSA) change in supine versus upright position by computerized tomography (CT). They concluded that the airway was significantly smaller when patients were in a supine position [23]. There are currently no such studies available in children. Furthermore, the airway is depicted in a two-dimensional (2D) view, resulting in a possible loss of information.
Another disadvantage is that this technique utilizes ionizing radiation. Effective dose is expressed in Sieverts which is a single dose parameter that reflects the risk of exposure in terms of whole-body exposure. An annual effective dose from natural background radiation is about 2.5–3 millisievert (mSv), the worldwide average natural dose is about 2.4 mSv per year. A lateral neck X-ray requires minimal radiation with an average of 0.2 millisievert (mSv) (reported variation in the literature ranges between 0.07 and 0.3 mSv) [24,25,26].
The obtained lateral neck radiography shows the vertebrae, the oral and nasal airways, the nasopharynx, part of the trachea, the epiglottis, the soft tissue in front of the vertebrae, and the adenoids and tonsils. Some ratios can be determined using different methods, the most frequently analyzed radiographic parameter in studies is the adenoidal-nasopharyngeal (AN) ratio (Fig. 16.1) [2, 3, 27,28,29,30,31,32,33,34,35]. However, a systematic review concluded that there are conflicting outcomes because of methodological problems that limit the applicability of these results to clinical decision-making [36]. A cohort study evaluated the ability of lateral radiography to assess adenoid hypertrophy in 72 children using four different methods [37]. Twenty-six children (36.1%) without OSA and only nine children (12.5%) with severe OSA were studied. The best correlations between OSA severity and radiography were found using the anterior airway measurement (the distance from the anterior adenoid to the post-maxillary antrum; method by Hibbert et al.) [33]. Using the definition of obstructive apnea/hypoapnea index (oAHI) >1 with adjustment for sex, age, body- mass index (BMI), the method of e (presented in Fig. 16.1) provided the largest receiver operating characteristic (ROC) area (0.775) for predicting OSA, with a sensitivity of 86.7% and a specificity of 55.6%.
(a–e) Lateral X- ray. (a) Hibbert’s method: the distance from the anterior adenoid to the post-maxillary antrum. (b) The distance along a perpendicular line from the pharyngeal tubercle on the base of the skull to the adenoidal convexity (maximal adenoidal thickness). (c) Distance measured along a line from the posterior–superior edge of the hard palate to the spheno- occipital synchondrosis on the base of the skull. (d) Minimal width of the airway immediately behind the soft palate at (e). (e) Width of the supero-anterior soft palate 1 cm below the hard palate (half a centimeter in children younger than 3 years old). (From Waters et al. [37]. Reprinted with permission from John Wiley and Sons)
Additionally, there are a few more studies that show correlations between lateral X-ray findings and OSA severity while one study failed to show a correlation [2, 3, 28, 29, 35]. Brooks et al. reported a correlation between obstructive apneas and AN ratio (according to the method described by Fujioka) in a study of 33 children. Children with OSA had a larger AN ratio (0.83 ± 0.3 vs. 0.69 ± 0.03; p < 0.003), resulting in reasonable, although suboptimal, positive and negative predictive values of 71% and 75%, respectively [28]. Li et al. investigated the value of tonsillar–pharyngeal (TP) ratio as a measure of tonsillar enlargement in 35 children (mean age of 6.2 (4–10) years old) with suspected OSA. There was a significant correlation between the TP ratio and the AHI (r = 0.8; p < 0.001) and oxygen desaturation index (r = 0.51; p = 0.002). However, there was no correlation between tonsil size and the TP ratio. The ROC curve analysis revealed that a TP ratio cut off of 0.5 was optimal for predicting severe OSA, with the area under the curve being 1.0. The corresponding sensitivity and specificity were 95.8% and 81.8%, respectively, while the positive and negative predictive values were 92.0% and 90.0%, respectively [3]. Xu et al. determined whether parents’ observation, clinical examination, and lateral upper airway radio- graph were useful in diagnosing OSA in 50 children (OSA group; n = 31, mean age 7.8 ± 3.2 years old and primary snoring group; n = 19, mean age 8.1 ± 3.7 years old). There was a sensitivity of 81% and specificity of 58% for UA narrowing on lateral X-ray by AN ratio for predicting OSA. Secondly, combining UA narrowing with mouth breathing, nocturnal enuresis, observed apnea during sleep, intrusive naps, mouth breathing, enlarged tonsils, and radiologic features of narrowing increased the positive predictive value to 73% and the negative predictive value to 80% [2].
Currently, there are no studies that have investigated whether lateral neck radiography can predict the effect of treatment.
In summary, this technique has relatively good predictive values for the diagnosis of OSA. In view of the advantages associated with this technique, further studies in other and larger populations are warranted. Furthermore, the predictive value can be improved by incorporating certain clinical predictors such as obesity, mouth breathing, nocturnal enuresis, observed apnea during sleep, intrusive naps, and enlarged tonsils on clinical examination.
Cephalometry
Cephalometry involves a standardized lateral radiographic view of the head and neck with more calculations of markers, distances, and ratios (Fig. 16.2). Therefore, there is a need for an experienced radiologist. The method shows skeletal (including mandibular and hyoid position) and soft-tissue (tongue and soft palate) UA structures. The advantages and disadvantages are comparable to lateral neck X-ray. Due to several measurements, the position of the head is critical. The following angles are commonly described in cephalometric studies: maxillary protrusion is expressed by the sella, nasion, and A-point (SNA) angle, the mandibular protrusion is expressed by the sella, nasion, and B-point (SNB) and point A nasion to B (ANB) angle is the difference of the SNA and SNB angles. In sagittal direction, an increase of the ANB angle describes a skeletal Class II malocclusion and in the vertical direction it shows a mandibular clockward rotation [38,39,40].
Cephalometric references and landmarks used in the meta-analysis. S Sella, N nasion, Ba basion, ANS anterior nasal spine, PNS posterior nasal spine, PP palatal plane, A A-point, B B-point, MP mandibular plane (gonion-menton), PNS-AD1 distance from PNS to the nearest adenoid tissue measured along the line PNS- Ba, PNS-AD2 distance from PNS to the nearest adenoid tissue measured along the line perpendicular to S-Ba, LI, long axis of the mandibular incisor. (From Katyal et al. [48]. Copyright © 2013 American Association of Orthodontists. Published by Mosby, Inc. All rights reserved. Reprinted with permission from Elsevier)
Some case-control studies in adults investigated the usefulness of cephalometric measurements in OSA patients. A meta-analysis in adults has shown a strong correlation between OSA severity and mandibular plane hyperdivergence. However, this correlation was not strong enough to indicate that craniofacial morphology had a direct causal effect in the development of OSA in adults [41].
Several case series investigated the influence of skeletal abnormality in children and suggested that children with mouth breathing, adenotonsillar hypertrophy, or sleep-disordered breathing have a retropositioned mandible, narrow maxilla, increased lower anterior face height, increased mandibular plane angle, inferior position of the hyoid bone, and smaller airway space [42,43,44,45,46]. Several studies have also investigated whether cephalometry could predict the diagnosis of Obstructive Sleep Apnea Syndrome (OSAS). Two systematic reviews investigated the association between maxillomandibular discrepancy and OSA. They suggested that children with OSA have more skeletal Class II mandibular growth direction compared to normal children. However, an increased ANB angle of less than 2° in children with OSA and primary snoring, compared with the controls, might not be clinically significant. Evidence for a direct causal relationship between craniofacial structure and OSA could not be supported by these meta-analyses [47, 48].
Galeotti et al. analyzed the correlation between cephalometric variables and OSA severity in 62 children. They reported a correlation between increased oAHI and skeletal discrepancy expressed by ANB angle. Therefore, one could hypothesize that more severe OSA leads to an increased mouth-breathing pattern that in turn strongly affects skeletal growth with Class II skeletal malocclusion and hyperdivergent growth pattern in children [49]. Pirila-Parkkinen et al. investigated the capability of 2D lateral cephalography in recognizing pharyngeal obstruction compared to 3D MRI and clinical observation in 36 children with OSA. The study showed an association between the cephalometric nasopharyngeal and retropalatal airway measurements and MRI findings. However, retroglossal pharyngeal measurements by cephalography did not correlate with MRI variables. Palatal tonsils mainly are situated in the retroglossal region and because of their lateral position; enlarged tonsils can cause the transversal narrowing of the retroglossal airway, which is not detectable on the anteroposterior view of the cephalogram [50].
Similar as for the lateral neck radiography, there are currently no studies which used cephalometry to predict the effect of treatment in pediatric OSA.
In summary, it remains unclear if cephalometry can be a valid diagnostic tool and there are no studies using cephalometry to predict treatment outcome.
Magnetic Resonance Imaging (MRI)
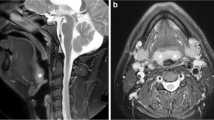
MRI is another method to localize and diagnose the site of UA obstruction (Fig. 16.3). It is accurate, reproducible, and there is no need for radiation. Furthermore, it is possible to have moving images during sleep by cine MRI. However, MRI is expensive, and requires a long examination time, resulting in a higher probability of motion artifacts. That is why it often requires sedation that puts the patient in a state of mimicking physiological sleep as closely as possible, but this also makes it more invasive. Children with OSA are sensitive to the respiratory depressant effects of sedative and hypnotic drugs that affect the UA dynamics, including the possibility of increasing UA obstruction. Dexmedetomidine provides an acceptable level of sedation and less need for airway support during MRI studies [51,52,53,54,55].
Normal-Weight Children
A case-control study investigated anatomical risk factors involved in the development of pediatric OSA in 40 normal-weight children (20 with OSA and 20 controls) by MRI. MRI performed in supine position, without sedation. Volumetric measurements were made from T1-weighted images and T2-weigthed images and were used to evaluate the lymphoid tissues. This study showed a significantly smaller UA volume (1.4 ± 0.7 cm3 versus 1.6 ± 0.7 cm3) and midsagittal nasopharyngeal airway (0.7 ± 0.2 cm3 versus 1.2 ± 0.4 cm3) in OSA patients. The axial cross-sectional area of the oropharyngeal airway was also smaller (0.5 ± 0.3 cm3 versus 0.8 ± 0.5 cm3). Thereby, children with OSA also had a significantly lager soft palate volume and midsagittal palate volume. Additionally, the adenoids and tonsils were considerably larger in the OSA group. Concerning skeletal structure, the study showed that the OSA group had a smaller mandibular volume and a lower vertical position of the hyoid bone [56]. Another study investigated the UA in sedated children with OSA by MRI. In a first study, they compared the UA structure in 18 young children with OSA (mean age 4.8 years) and 18 controls (mean age 4.9 years). They showed a correlation between increased tonsil and adenoid volume and AHI in sedated children [57]. Another study investigated 10 children with adenotonsillar hypertrophy and OSA and 10 controls. Children with OSA had a smaller UA volume, particularly during inspiration, whereas dilatation occurred during expiration. The OSA group had larger adenoid and tonsils in comparison with the control group. The volumes of both mandible and tongue were similar in both groups. The study concluded that the adenoid and tonsils in children with moderate OSA restrict the UA. Additionally, the study found that further UA restriction in OSA patients could be caused by enlargement of the soft palate [58].
Obese Children
Arens et al. investigated the body fat composition and UA structure in 44 obese children (22 with OSA and 20 controls). UA lymphoid hypertrophy, parapharyngeal fat pads, and abdominal visceral fat were significantly increased in obese children with OSA. In regression analysis, only lymphoid tissue correlated with OSA severity [20]. Another study evaluated the frequency of enlarged lingual tonsils in 71 obese children with sagittal fast spin-echo inversion recovery imaging. They concluded that obese children had a high frequency of enlargement of the lingual tonsils with a significantly higher prevalence in those who had previously undergone tonsillectomy. Enlarged lingual tonsils may thus play a role in the pathogenesis of persistent obstructive sleep apnea in obese children [59].
Nandalike et al. evaluated how ATE affects anatomical factors of airway obstruction in 27 obese children with OSA. All underwent polysomnography (PSG) and MRI during wakefulness before and after ATE. ATE was associated with a significant increase in soft palate volume, tongue size, and head and neck subcutaneous fat tissue. A complete resolution of OSA only occurred in 44% of cases and complete resolution was achieved in only 22% of children with severe OSA. Residual OSA was associated with substantial residual adenoid tissue, an increase in the volume of the soft palate and, to a lesser extent, the increased volume of the tongue [60].
Children with DS
Children with DS have certain anatomical factors that predispose for a higher risk of OSA: underdeveloped midface and mandibular hypoplasia, macroglossia, posterior-placed tongue, hypotonia, smaller upper airway, lymphoid hyperplasia, and obesity [51, 61,62,63].
Two studies used imaging in children with DS to investigate UA morphology. Uong et al. investigated the anatomical differences by MRI (size and shape of the upper airway in relation to surrounding tissue) between 11 children with DS without OSA and 14 controls without DS and OSA (mean age of 3 years old). Adenoid and tonsil volume were significantly smaller in the DS children. However, tongue, soft-palate, pterygoid, and parapharyngeal fat pads were similar. There was a smaller upper airway size in DS children, the authors suggested that this was caused by soft tissue crowding within a smaller mid- and lower face skeleton [63]. Another imaging study investigated the stiffness of the airway wall from MRI by a noninvasive method in 21 children with DS and OSA. Airway changes were evaluated by continuous positive airway pressure (CPAP). The localized airway and tissue elasticity were found to increase with increasing OSA severity. They concluded that elasticity-based patient phenotyping could potentially assist clinicians in decision making concerning the needed CPAP pressure [64].
Donnelly et al. concluded that persistent OSA in DS has multiple causes. The most common causes include macroglossia, glossoptosis, recurrent enlargement of the adenoids, and enlarged lingual tonsils [17].
Cine MRI
Cine MRI is a high-resolution imaging method that captures the dynamic movement of the UA in a sedated child. These dynamic images can show both the degree and the direction of airway collapse (anterior-posterior or circumferential). Thereby, it can sometimes show compensating of the tongue or jaw thrusting during the period of obstruction. It is performed for approximately 30 seconds at each level with a rate of imaging of about three images per second [65,66,67,68,69].
Donnelly et al. investigated 16 young patients with OSA and 16 without airway problems or airway diseases. These researchers showed several differences concerning dynamic airway motion [67]. OSA patients were much more likely to demonstrate intermittent collapses of the nasopharynx and exclusively demonstrated intermittent collapse of the hypopharynx. The mean change in diameter of the nasopharynx and hypopharynx, implicating a more compliant UA, was also significantly greater in the OSA group. Abbott et al. investigated 31 children (mean age of 11.3 years) with OSA and 21 control children (mean age of 3.5 years). They included OSA subjects with the following predispositions to obstruction: craniofacial anomalies, DS, persistent OSA, and pre-operative evaluation to complex airway surgery. All children underwent transverse phase gradient-echo cine MRI imaging of the hypopharynx with sedation. The airway volumes were obtained by a k-means clustering algorithm and the airway wall motion was described. The study showed airway distention and airway collapse in children with OSA. Airway volume oscillated in both groups, but the amplitude was much larger in the OSA group. A clear limitation of this study is the fact that the mean age of the control group was much lower than the mean age of the OSA patients. A concern about the large difference in age is that UA size may increase by age. Interestingly, the size of UA volume in some of the youngest subjects with OSA had volumes as large as some of the oldest in the control group. In this study, airway volume did not correlate significantly with age in either the control group or the children with OSA [70].
In summary, MRI gives a better insight into UA anatomy because it provides a detailed assessment of the UA as the pharyngeal size and soft tissue anatomy (including adipose tissue). Several studies involving MRI have shown detailed correlations with OSA severity or the identification of critical anatomical sites. However, more research is needed for sensitivity or specificity for diagnosis and prediction of treatment outcome.
Computerized Tomography (CT)
CT is a fast, noninvasive technique to visualize the UA and is available in the majority of institutions (Fig. 16.4). CT images can be taken during wakefulness and sleep. A major disadvantage of CT is radiation. The radiation dose for a neck CT is approximately 3 mSv for adults [24, 71, 72]. Recently, cine CT or ultra-fast CT has been used more frequently and can obtain multiple images with a lower radiation dose. Fleck et al. evaluated the cine (dynamic 3D) CT technique for the UA in six children with OSA and compared the required radiation dose between these children. The radiation from a low-dose CT scan was between 0.08 and 0.27 mSv [73].
The UA from nares to trachea can be reconstructed into three-dimensional (3D) models and subdivided into five zones for measuring volumes of different parts in the UA and CSA (Figs. 16.5 and 16.6).
Reconstructed into 3D model of the upper airway. (From Slaats et al. [75]. Reprinted with permission from John Wiley and Sons)
3D model of the upper airway divided into five zones. Zone 1 = nostril to bottom of inferior turbinate; Zone 2 = bottom of inferior turbinate to choanae; Zone 3 = choanae to tip of uvula; Zone 4 = uvula to epiglottis; Zone 5 = epiglottis to the first vertebra. (From Slaats et al. [75]. Reprinted with permission from John Wiley and Sons)
Only three studies investigated the utility of CT in pediatric OSA. Van Holsbeke et al. investigated whether anatomical and functional properties of the airway were correlated with OSA severity in 33 children with OSA. The study concluded that children with OSA had a lower volume of UA zone 3, the overlap region of adenoids and tonsils, and a lower mean CSA of the UA. No correlation was found between the clinical scores of UA patency (Brodsky and Mallampati scoring system) and OSA severity indicating that imaging might be more powerful in the assessment of UA patency [74]. Slaats et al. further investigated whether functional respiratory imaging (FRI) by CT could provide more information about UA characteristics in 91 normal-weight children with OSA mainly to predict treatment outcome. They concluded, comparable with Van Holsbeke et al., which a smaller overlap region and a more concave shape of the UA correlated with more severe OSA. There was also no correlation with clinical examination and OSA severity. UA volumes could not predict treatment outcome in this study [75].
Another study about CT images and pediatric OSA was performed in children with DS. Slaats et al. characterized treatment outcome after ATE in 33 young children with DS and OSA by UA imaging (CT images). At baseline, children with more severe OSA had a smaller minimal passage through the upper airway. After treatment, persistent OSA was seen in 79% of the children; however, 79% had a decrease of >50% in oAHI after treatment. Children with less favorable response had a smaller volume of the zones below the tonsils, which is probably due to enlargement of the lingual tonsils, glossoptosis, or macroglossia that is not treated by ATE (Fig. 16.7). In conclusion, this study suggests that UA imaging could have an influence in treatment selection in children with DS and OSA. Exact cut-off values are needed to be confirmed by larger studies [76].
Left: image of a child’s UA with a decrease of more than 50% in oAHI after ATE Right: decrease of less than 50% in oAHI after ATE. (From Slaats et al. [76]. Copyright © 2018 American Academy of Sleep Medicine, reprinted with permission)
In conclusion, CT also provides a detailed analysis of the anatomy. CT studies have shown detailed correlations with OSA severity or the identification of critical anatomical sites without sensitivity or specificity reported. One study indicates that UA imaging could have a role in treatment selection in children with DS and OSA. However, this was a very small study group and exact cut-off values are needed to be confirmed by larger studies. More research is needed in this group because DS is an important risk factor for OSA with a high incidence of residual OSA.
Cone Beam CT (CBCT)
Cone beam computed tomography (CBCT) is a low-dose radiation 3D imaging technique. A disadvantage is that it shows limited differentiation of soft tissue. CBCT is not available in all hospitals. However, it is widely available in dental and oral medicine. For example, it has been used for many maxillofacial applications such as for implant site imaging and treatment planning for craniofacial surgery and orthodontics [77].
The radiation dose is lower compared to normal CT and is more like that of low dose protocol CT. When used in maxillofacial imaging, CBCT produces an eight- to tenfold lower effective dose than a conventional CT examination using standard protocols [78,79,80].
Some studies in adults have demonstrated that CBCT produces extremely accurate anatomical representations [81,82,83]. A review of the use of CBCT in adult patients with OSA concluded that there is a need for more research, but that the use of CBCT for both pre-operative planning and for postoperative evaluation of therapeutic interventions is likely to become increasingly important [84]. There are only a few studies in pediatric OSA. Eimar et al. investigated in 96 children diagnosed with or at risk of SDB, particularly OSA, whether these children had a reduced bone density estimated by mandibular cortical width (MCW) on CBCT images [85]. MCW demonstrates the highest sensitivity and specificity for detection of reduced bone density [86]. It represents the distance, in millimeters, between the lower borders of the mandible to the superior margin of the mandible cortex. MCW values were significantly lower in OSA children (MCW = 2.9 ± 0.6 mm) compared to control children (MCW = 3.5 ± 0.6 mm; P = 0.002). This finding may reflect alterations in bone homeostasis [85].
A case–control study verified the differences in the volume and areas of the UA between 27 children with persistent OSA after AT and a 20 sex-age matched healthy control group by CBCT. Children with OSA had a significant narrowing in the nasopharynx and in the lower portion of the UA. This result demonstrated that other factors than adenotonsillar hypertrophy, such as craniofacial abnormalities, could play a role in the pathogenesis of OSA [87]. Alsufyani et al. evaluated with a clinical pilot study the anatomical changes that occur in the UA before and after AT using CBCT in 12 children. Secondly, they evaluated whether changes in airway reflects in the quality of life [88]. Quality of life was tested by the OSA-18 questionnaire. Only UA constriction and patency correlated with changes in OSA-18. Airway patency gained by at least 150% and constriction relief by at least 15% showed marked improvement in OSA-18 by 40–55%. A limitation of this study was the method for diagnosis of OSA. This was based on history of nocturnal symptoms, physical examination, overnight pulse oximetry, and pediatric sleep questionnaire (PSQ-22) instead of the gold-standard polysomnography [7, 89]. Another limitation of this study was that the analysis did not include other OSA parameters such as saturation and a sleep study after treatment [89].
In conclusion, CBCT provides a detailed analysis of the anatomy by low dose protocol. Unfortunately, there are only a few studies in children that make it unclear if CBCT can be a diagnostic tool or predict treatment outcome.
Functional Respiratory Imaging (FRI)
FRI is a relatively new method that can simulate airflow dynamics and the resulting pressure distribution by anatomical narrowing by computational fluid dynamics (CFD). The 3D model (Fig. 16.6) is used for analysis of anatomical parameters, volume meshing (representation of interior volume), and CFD [74,75,76, 90]. Exact details about CFD are described in the article of Slaats et al. [75]. A few studies investigated the utility of CFD in children with OSA. Preliminary data showed that FRI could identify differences in the UA of children with residual OSA and correlates well with OSA severity than parameters obtained from physical examination [74].
Normal-Weight Children
One study compared CFD data in three children with OSA and three controls. The results suggested that pharyngeal airway shape in children with OSA significantly affects internal pressure distribution compared to nasal resistance [91]. A recent study investigated whether FRI by CT images could provide more information about UA characteristics in 91 normal-weight children with OSA without risk factors, mainly to predict treatment outcome. Imaging parameters correlated with OSA severity at baseline more than the tonsil score obtained by clinical assessment: a smaller overlap region of tonsils/adenoids, a higher resistance, and a more concave shape of the UA correlated with more severe OSA. Thereby, a less constricted airway, as characterized by both a higher conductance and a lower tonsil score, was associated with a less favorable response to (adeno)tonsillectomy. In conclusion, both UA conductance and the tonsil score predicted treatment response [75].
Obese Children
Mihaescu et al. evaluated computational simulation of pre- and post-AT by MRI in an obese child. A significant pressure drop was observed at the site of minimum CSA. There was an increase in airway CSA of the retropalatal pharynx. These findings indicate that ATE is associated with changes in flow characteristics [92]. A retrospective cohort study investigated whether CFD model endpoints correlated with treatment response of ATE in 10 obese children. MRI and CFD data before and after surgery were utilized to calculate the velocity and air pressure distribution. They reported more significant correlations between decreased OSA severity and CFD than with UA anatomical parameters [93].
Children with DS
Only one study investigated whether UA imaging combined with CFD could characterize treatment outcome in children with DS and OSA. They concluded that there was no extra value of CFD in prediction of treatment outcome. However, this was a relatively small study and further studies including larger samples of patients before treatment are needed to validate a model to predict treatment outcome [76].
In conclusion, FRI is a relatively new promising method that may can help in treatment selection. There are only a few studies in children; most of the studies are small. A prospective cohort study concluded that FRI and clinical assessment predicted treatment outcome in children without risk factors. More research is needed to investigate the predictive value in children with DS and obese children because the percentage of residual OSA is higher in these populations.
(Long-Range) Optical Coherence Tomography
Optical coherence tomography (OCT) is another new noninvasive imaging modality to image the UA [94]. This is a diagnostic evaluation without radiation and sedation. This tool utilizes a broadband light source to produce high-resolution cross-sectional images of tissue components with a resolution of 10 μm. The UA can be scanned in approximately 20–40 seconds. The produced images are similar in resolution to those of conventional microscopy. Ridgway et al. investigated the utility of OCT in characterizing the microanatomy of the UA in 15 children in vivo. They concluded that this technique identifies the epithelium and lamina propria [95]. Long-range OCT (LR-OCT) puts the emphasis on a longer range capture of airway wall location by a rotary fiber optic for helical scan to quantify size and shape of hollow organs such as the UA to generate accurate 3D reconstructions (Fig. 16.8). CFD simulations can be performed on these models [96,97,98].
Images converted to 3D model using Mimics software. (From Lazarow et al. [97]. Reprinted with permission from Elsevier)
Two studies investigated the value of OCT in children with OSA. Lazarow et al. investigated the feasibility of LR-OCT to identify airway narrowing in children who underwent ATE. They demonstrated that CSAs were measurably larger in 46 children after ATE [97]. Another study presented the first, airflow simulations by CFD in pediatric airways. They assessed the effect of three realistic airway curvatures in eight children on predicted airway resistance by CFD (before and after ATE). The LR-OCT imaging was incorporated into airway endoscopy exams conducted as part of the standard care before and after surgery. The imaging selection was based on the best signal-to-noise ratio, the best contrast between air and tissues, and the least loss of structure. The airway surface was subjected to a bending algorithm implemented in the software to cure the vertical reconstruction. The algorithm was based on planar curves to sagittal CT and MRI imaged of three normal children. CSA values were obtained by using the area calculation function in Mimics™. Minimal CSA (mCSA) values were calculated by averaging the areas of five consecutive CSAs corresponding to a 1- or 2- mm thick slab of the OCT data of the smallest CSA value. Steady-state, inspiratory airflow simulations were conducted under laminar conditions, along with turbulent simulations by CFD. In this study, CFD findings corroborated that postoperative airway resistance was significantly less compared to pre-operative data. The individual resistances did not vary for different curvatures. This suggests that airway curvature may not be predictive of surgical effects on airway resistance [98].
In summary, this is a novel noninvasive technique to create 3D reconstructions of the UA using OCT images from pre- and postoperative cases. These 3D models provide insight into its structure and shape and can help identify regions of obstruction without radiation. It can be feasibly obtained intraoperatively. Furthermore, more research needs to investigate the predictive value of these reconstructions for surgery selection and compare data in awake and sedated patients.
Drug-Induced Sleep (Sedation) Endoscopy (DISE)
DISE is another method to evaluate the level and degree of UA obstruction (Fig. 16.9). It allows UA visualization by flexible endoscopy or bronchoscope while the child is sedated. It is a promising technique in selecting the type of UA surgery because it provides live video of UA obstruction. There is no need for radiation; it is a simple, safe, and cost-effective technique [99]. However, its interpretation requires experience and the use of a standard protocol is recommended [99,100,101,102,103,104,105,106,107,108,109,110,111]. There are over 21 scoring methods described, of which six are in pediatric OSA. Only two studies reported a correlation between the scoring system and OSA severity [101, 102]. Another disadvantage is the utility of sedation during DISE as described earlier in MRI. A recent systematic review suggested that dexmedetomidine and ketamine do not lead to respiratory depression and are associated with less muscular relaxation, with a more sustained respiratory effort [53, 54]. Thereby, a disadvantage of DISE over cine MRI is that it is not possible to visualize multiple levels of obstruction at the same time. Besides, it is not possible to visualize the depth and thickness of abnormal tissues, such as enlarged lingual tonsils, what could be the cause of tongue base collapse [99].
The potential role for DISE prior to ATE and the effect on treatment outcome is not well defined and is subject of controversy. A review recommended not to systematically use DISE in otherwise healthy infants and children with OSA without prior UA surgery [105]. This recommendation contrasts with previous studies which have shown that DISE prior to ATE may change the surgical decision making, also in surgically naïve children [101, 104, 106,107,108,109].
In conclusion, pre-operative assessment combining DISE could have better outcomes than routine Ear Nose and Throat (ENT) examinations in pediatric OSA. Currently, there is no universally accepted standard score system, this will be necessary in order to provide a more consistent method. More research is needed in all groups of risk factors to determine the role for predicting treatment outcome.
Future Developments
Virtual surgery based on 3D constructions would be the next logical research opportunity in choosing an effective surgical strategy [112]. Children with DS or obese children should be prioritized because the percentage of residual OSA is much higher in these populations. A recent pilot study of Mylavarapu et al. investigated the use of virtual surgery and compared virtual surgery with actual surgery. Virtual surgeries were performed on 10 patients with moderate-to-severe OSA. Changes in oAHI and upper airway resistance, as calculated by computational fluid dynamics for pre- and postoperative modeling, matched well for 8 of 10 patients. Limitations of this study were that the authors did not describe the influence of age and did not compare the virtual surgery with surgical changes in anatomy by MRI after treatment [113, 114]. More research is needed to confirm the clinical usability of this technique in surgical planning and studies that investigate the prediction of treatment outcome. Additionally, there is need for more research using imaging or DISE in children with persistent OSA after treatment. The aforementioned use of virtual surgery modeling could also be expanded beyond ATE surgery.
Summary
Many (surgical) options exist for treatment of pediatric (persistent) OSA. In the present era of personalized medicine, progress has been made in identifying the exact cause of OSA in children.
There are limited data on the usefulness of the different imaging techniques as a diagnostic tool in pediatric OSA and predictive value for treatment outcome. Most of the studies had small sample sizes with different inclusion and exclusion criteria. These simple techniques can already assist in predicting the severity of OSA and are minimally invasive. Thereby, there is a suggestion that functional imaging or virtual surgery could be of extra value in predicting treatment outcome. However, more research is needed to confirm the clinical utility of these techniques. Thereby, a comparison of the different imaging methods, including cost-effectiveness analysis, is warranted.
Abbreviations
- 2D:
-
2-dimensional
- 3D:
-
3-dimensional
- AHI :
-
Apnea hypopnea index
- AN:
-
Adenoidal-nasopharyngeal
- ATE:
-
Adenotonsillectomy
- BMI:
-
Body-mass index
- CAD:
-
Computer aided design
- CBCT:
-
Cone beam computed tomography
- CFD:
-
Computational fluid dynamics
- CI:
-
Confidence interval
- CSA:
-
Cross-sectional area
- CT:
-
Computed tomography
- DS:
-
Down syndrome
- LR-OCT:
-
Long-range optical coherence tomography
- mCSA:
-
Minimal cross-sectional area
- MCW:
-
Mandibular cortical width
- MDCT:
-
Multiple detector computed tomography MRI magnetic resonance imaging
- mSv:
-
Milisievert
- oAHI:
-
Obstructive apnea hypopnea index
- OCT:
-
Optical coherence tomography
- OSA:
-
Obstructive sleep apnea
- PSG:
-
Polysomnography
- RDI:
-
Respiratory disturbance index
- SNA:
-
Sella, nasion, and A-point
- SNB:
-
Sella, nasion, and B-point
- T1:
-
Longitudinal relaxation time
- T2:
-
Transverse relaxation time
- TP:
-
Tonsillar-pharyngeal
- UA:
-
Upper airway
References
Potsic WP, Wetmore RF. Sleep disorders and airway obstruction in children. Otolaryngol Clin N Am. 1990;23:651e3.
Xu Z, Cheuk DK, Lee SL. Clinical evaluation in predicting childhood obstructive sleep apnea. Chest. 2006;130:1765e71.
Li AM, Wong E, Kew J, Hui S, Fok TF. Use of tonsil size in the evaluation of obstructive sleep apnoea. Arch Dis Child. 2002;87:156e9.
Mitchell RB, Garetz S, Moore RH, et al. The use of clinical parameters to predict obstructive sleep apnea syndrome severity in children: the childhood adenotonsillectomy (CHAT) study randomized clinical trial. Otolaryngol Head Neck Surg. 2015;141:130–6.
Brietzke SE, Katz ES, Roberson DW. Can history and physical examination reliably diagnose pediatric obstructive sleep apnea/hypopnea syndrome? A systematic review of the literature. Otolaryngol Head Neck Surg. 2004;131:827e32.
Nolan J, Brietzke SE. Systematic review of pediatric tonsil size and polysomnogram-measured obstructive sleep apnea severity. Otolaryngol Head Neck Surg. 2011;144:844e50.
Kaditis AG, Alonso Alvarez ML, Boudewyns A, Alexopoulos EI, Ersu R, Joosten K, Larramona H, Miano S, Narang I, Trang H, Tsaoussoglou M, Vandenbussche N, Villa MP, Van Waardenburg D, Weber S, Verhulst S. Obstructive sleep disordered breathing in 2- to 18-year-old children: diagnosis and management. Eur Respir J. 2016;47(1):69–94.
Marcus CL, Brooks LJ, Draper KA, et al. American Academy of Pediatrics. Diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics. 2012;130(3):576–84.
Lee CH, Hsu WC, Chang WH, Lin MT, Kang KT. Polysomnographic findings after adenotonsillectomy for obstructive sleep apnoea in obese and non-obese children: a systematic review and meta-analysis. Clin Otolaryngol. 2016;41:498–510.
Tauman R, Gulliver TE, Krishna J, et al. Persistence of obstructive sleep apnea syndrome in children after adenotonsillectomy. J Pediatr. 2006;149(6):803–8.
Bhattacharjee R, Kheirandish-Gozal L, Spruyt K, Mitchell RB, Promchiarak J, Simakajornboon N, et al. Adenotonsillectomy outcomes in treatment of obstructive sleep apnea in children: a multicenter retrospective study. Am J Respir Crit Care Med. 2010;182:676e83.
Friedman M, Wilson M, Lin HC, Chang HW. Updated systematic review of tonsillectomy and adenoidectomy for treatment of pediatric obstructive sleep apnea/hypopnea syndrome. Otolaryngol Head Neck Surg. 2009;140:800e8.
Mitchell RB, Kelly J. Outcome of adenotonsillectomy for obstructive sleep apnea in obese and normal-weight children. Otolaryngol Neck Surg. 2007;137(1):43–8.
Brietzke SE, Gallagher D. The effectiveness of tonsillectomy and adenoidectomy in the treatment of pediatric obstructive sleep apnea/hypopnea syndrome: a meta-analysis. Otolaryngol Head Neck Surg. 2006;134:979e84.
Socarras MA, Landau BP, Durr ML. Diagnostic techniques and surgical outcomes for persistent pediatric obstructive sleep apnea after adenotonsillectomy: a systematic review and meta-analysis. Int J Pediatr Otorhinolaryngol. 2019;121:179–87.
Costa DJ, Mitchell R. Adenotonsillectomy for obstructive sleep apnea in obese children: a meta-analysis. Otolaryngol Head Neck Surg. 2009;140:455e60.
Donnelly LF, Shott SR, LaRose CR, Chini BA, Amin RS. Causes of persistent obstructive sleep apnea despite previous tonsillectomy and adenoidectomy in children with down syndrome as depicted on static and dynamic cine MRI. AJR Am J Roentgenol. 2004;183(1):175–81.
Maris M, Verhulst S, Wojciechowski M, Van de Heyning P, Boudewyns A. Outcome of adenotonsillectomy in children with Down syndrome and obstructive sleep apnoea. Arch Dis Child. 2017;102(4):331–6.
Bacon WH, Krieger J, Turlot JC, Stierle JL. Craniofacial characteristics in patients with sleep apneas syndrome. Cleft Palate J. 1988;25:374e8.
Arens R, Sin S, Nandalike K, Rieder J, Khan UI, Freeman K, et al. Upper airway structure and body fat composition in obese children with obstructive sleep apnea syndrome. Am J Respir Crit Care Med. 2011;183:782e7.
Slaats MA, Van Hoorenbeeck K, Van Eyck A, Vos WG, De Backer JW, Boudewyns A, De Backer W, Verhulst SL. Upper airway imaging in pediatric obstructive sleep apnea syndrome. Sleep Med Rev. 2015;21:59–71.
Pracharktam N, Hans MG, Strohl KP, Redline S. Upright and supine cephalometric evaluation of obstructive sleep apnea syndrome and snoring subjects. Angle Orthod. 1994;64:63e73.
Camacho M, Capasso R, Schendel S. Airway changes in obstructive sleep apnoea patients associated with a supine versus an upright position examined using cone beam computed tomography. J Laryngol Otol. 2014;128(9):824–30.
Mettler FA Jr, Huda W, Yoshizumi TT, Mahesh M. Effective doses in radiology and diagnostic nuclear medicine: a catalog. Radiology. 2008;248:254e63.
Huda W. Effective doses to adult and pediatric patients. Pediatr Radiol. 2002;32(4):272–9.
United Nations Scientific Committee on the Effects of Atomic Radiation. Sources and effects of ionizing radiation. New York: United Nations; 2008. (published 2010). p. 4. ISBN 978-92-1-142274-0. Retrieved 9 Nov 2012.
Shintani T, Asakura K, Kataura A. Evaluation of the role of adenotonsillar hypertrophy and facial morphology in children with obstructive sleep apnea. ORL J Otorhinolaryngol Relat Spec. 1997;59:286e91.
Brooks LJ, Stephens BM, Bacevice AM. Adenoid size is related to severity but not the number of episodes of obstructive apnea in children. J Pediatr. 1998;132:682e6.
Jain A, Sahni JK. Polysomnographic studies in children undergoing adenoidectomy and/or tonsillectomy. J Laryngol Otol. 2002;116:711e5.
Johannesson S. Roentgenologic investigation of the nasopharyngeal tonsil in children of different ages. Acta Radiol. 1968;7:299e304.
Cohen D, Konak S. The evaluation of radiographs of the nasopharynx. Clin Otolanryngol. 1985;10:73e8.
Fujioka M, Young LW, Girdany BR. Radiographic evaluation of adenoidal size in children: adenoidal-nasopharyngeal ratio. AJR Am J Roentgenol. 1979;133:401e4.
Hibbert J, Stell PM. A radiological study of the adenoid in normal children. Clin Otolaryngol Allied Sci. 1979;4(5):321–7.
Sedaghat AR, Flax-Goldenberg RB, Gayler BW, Capone GT, Ishman SL. A case-control comparison of lingual tonsillar size in children with and without Down syndrome. Laryngoscope. 2012;122:1165e9.
Kitamura T, Miyazaki S, Kobayashi R, Kadotani H, Kanemura T, Komada I, Nishikawa M, Okawa M, Koizumi H, Takeuchi S, Suzuki H. Obstructive sleep apnea in preschool-aged Japanese children – efficiency of screening using lateral neck radiography. Acta Otolaryngol. 2016;136(6):606–12.
Feres MF, Hermann JS, Cappellette M Jr, Pignatari SS. Lateral X-ray view of the skull for the diagnosis of adenoid hypertrophy: a systematic review. Int J Pediatr Otorhinolaryngol. 2011;75(1):1–11.
Waters K, Kol-Castro C, Varghese A, Lam LT, Prelog K, Cheng A. Correlations between polysomnographic and lateral airway radiograph measurements in paediatric obstructive sleep apnoea. J Paediatr Child Health. 2013;49(6):445–51.
PaoliJ R, Lauwers F, Lacassagne L, Tiberge M, Dodart L, Boutault F. Craniofacial differences according to the body mass index of patients with obstructive sleep apnoea syndrome: cephalometric study in 85 patients. Br J Oral Maxillofac Surg. 2001;39:40e5.
Kim SJ, Kim YS, Park JH, Kim SW. Cephalometric predictors of therapeutic response to multilevel surgery in patients with obstructive sleep apnea. J Oral Maxillofac Surg. 2012;70:1404e12.
Shintani T, Asakura K, Kataura A. Adenotonsillar hypertrophy and skeletal morphology of children with obstructive sleep apnea syndrome. Acta Otolaryngol Suppl. 1996;523:222e4.
Miles PG, Vig PS, Weyant RJ, Forrest TD, Rockette HE Jr. Cranio-facial structure and obstructive sleep apnea syndrome—a qualitative analysis and meta-analysis of the literature. Am J Orthod Dentofac Orthop. 1996;109:163–72.
Ozdemir H, Altin R, Söğüt A, Cinar F, Mahmutyaziciogu K, Kart L, et al. Craniofacial differences according to AHI scores of children with obstructive sleep apnoea syndrome: cephalometric study in 39 patients. Pediatr Radiol. 2004;34:393e9.
Juliano ML, Machado MA, de Carvalho LB, Zancanella E, Santos GM, do Prado LB, et al. Polysomnographic findings are associated with cephalometric measurements in mouth-breathing children. J Clin Sleep Med. 2009;5(6):554e61.
Linder-Aronson S. Adenoids. Their effect on mode of breathing and nasal airflow and their relationship to characteristics of the facial skeleton and the denition. A biometric, rhino-manometric and cephalometro-radiographic study on children with and without adenoids. Acta Otolaryngol Suppl. 1970;265:1e132.
Guilleminault C, Partinen M, Praud JP, Quera-Salva MA, Powell N, Riley R. Morphometric facial changes and obstructive sleep apnea in adolescents. J Pediatr. 1989;114:997e9.
Vieira BB, Itikawa CE, de Almeida LA, Sander HH, Aragon DC, Anselmo-Lima WT, Valera FCP. Facial features and hyoid bone position in preschool children with obstructive sleep apnea syndrome. Eur Arch Otorhinolaryngol. 2014;271(5):1305–9.
Flores-Mir C, Korayem M, Heo G, Witmans M, Major MP, Major PW. Craniofacial morphological characteristics in children with obstructive sleep apnea syndrome: a systematic review and meta-analysis. J Am Dent Assoc. 2013;144:269–77.
Katyal V, Pamula Y, Martin AJ, Daynes CN, Kennedy JD, Sampson WJ. Craniofacial and upper airway morphology in pediatric sleep-disordered breathing: systematic review and meta-analysis. Am J Orthod Dentofac Orthop. 2013;143:20–30.
Galeotti A, Festa P, Viarani V, Pavone M, Sitzia E, Piga S, Cutrera R, De Vincentiis GC, D'Antò V. Correlation between cephalometric variables and obstructive sleep apnoea severity in children. Eur J Paediatr Dent. 2019;20(1):43–7.
Pirila-Parkkinen K, Lopponen H, Nieminen P, Tolonen U, Paakko E, Pirttiniemi P. Validity of upper airway assessment in children: a clinical, cephalometric, and MRI study. Angle Orthod. 2011;81:433e9.
Patini R, Arrica M, Di Stasio E, Gallenzi P, Cordaro M. The use of magnetic resonance imaging in the evaluation of upper airway structures in paediatric obstructive sleep apnoea syndrome: a systematic review and meta-analysis. Dentomaxillofac Radiol. 2016;45(7):20160136.
Fricke BL, Donnelly LF, Shott SR, Kalra M, Poe SA, Chini BA, et al. Comparison of lingual tonsil size as depicted on MR imaging between children with obstructive sleep apnea despite previous tonsillectomy and adenoidectomy and normal controls. Pediatr Radiol. 2006;36:518e23.
Chatterjee D, Friedman N, Shott S, Mahmoud M. Anesthetic dilemmas for dynamic evaluation of the pediatric upper airway. Semin Cardiothorac Vasc Anesth. 2014;18(4):371–8.
Ehsan Z, Mahmoud M, Shott SR, Amin RS, Ishman SL. The effects of anesthesia and opioids on the upper airway: a systematic review. Laryngoscope. 2016;126(1):270–84.
Mahmoud M, Gunter J, Donnelly LF, Wang Y, Nick TG, Sadhasivam S. A comparison of dexmedetomidine with propofol for magnetic resonance imaging sleep studies in children. Anesth Analg. 2009;109(3):745–53.
Cappabianca S, Iaselli F, Negro A, Basile A, Reginelli A, Grassi R, Rotondo A. Magnetic resonance imaging in the evaluation of anatomical risk factors for pediatric obstructive sleep apnoea-hypopnoea: a pilot study. Int J Pediatr Otorhinolaryngol. 2013;77:69e75.
Arens R, McDonough JM, Costarino AT, Mahboubi S, Tayag-Kier CE, Maislin G, Schwab RJ, Pack AI. Magnetic resonance imaging of the upper airway structure of children with obstructive sleep apnea syndrome. Am J Respir Crit Care Med. 2001;164(4):698–703.
Arens R, Sin S, McDonough JM, Palmer JM, Dominguez T, Meyer H, et al. Changes in upper airway size during tidal breathing in children with obstructive sleep apnea syndrome. Am J Respir Crit Care Med. 2005;171:1298e304.
Guimaraes CV, Kalra M, Donnelly LF, Shott SR, Fitz K, Singla S, Amin RS. The frequency of lingual tonsil enlargement in obese children. AJR Am J Roentgenol. 2008;190:973–5.
Nandalike K, Shifteh K, Sin S, Strauss T, Stakofsky A, Gonik N, et al. Adeno-tonsillectomy in obese children with obstructive sleep apnea syndrome: magnetic resonance imaging findings and considerations. Sleep. 2013;36:841e7.
Fink GB, Madaus WK, Walker GF. A quantitative study of the face in Down’s syndrome. Am J Orthod. 1975;67(5):540–53.
Shapiro BL, Gorlin RJ, Redman RS, Bruhl HH. The palate and Down’s syndrome. N Engl J Med. 1967;276(26):1460–3.
Uong EC, McDonough JM, Tayag-Kier CE, Zhao H, Haselgrove J, Mahboubi S, Schwab RJ, Pack AI, Arens R. Magnetic resonance imaging of the upper airway in children with Down syndrome. Am J Respir Crit Care Med. 2001;163(3 Pt 1):731–6.
Subramaniam DR, Mylavarapu G, Fleck RJ, Amin RS, Shott SR, Gutmark EJ. Effect of airflow and material models on tissue displacement for surgical planning of pharyngeal airways in pediatric down syndrome patients. J Mech Behav Biomed Mater. 2017;71:122–35.
Sharma K, Shrivastav S, Hotwani K. Volumetric MRI evaluation of airway, tongue, and mandible in different skeletal patterns: does a link to obstructive sleep apnea exist (OSA)? Int J Orthod Milwaukee. 2016;27(3):39–48.
Fleck RJ, Shott SR, Mahmoud M, Ishman SL, Amin RS, Donnelly LF. Magnetic resonance imaging of obstructive sleep apnea in children. Pediatr Radiol. 2018;48(9):1223–33.
Donnelly LF, Surdulescu V, Chini BA, Casper KA, Poe SA, Amin RS. Upper airway motion depicted at cine MR imaging performed during sleep: comparison between young patients with and those without obstructive sleep apnea. Radiology. 2003;227(1):239–45.
Nayak KS, Fleck RJ. Seeing sleep: dynamic imaging of upper airway collapse and collapsibility in children. IEEE Pulse. 2014;5(5):40–4.
Shott S, Donnelly LF. Cine magnetic resonance imaging: evaluation of persistent airway obstruction after tonsil and adenoidectomy in children with Down syndrome. Laryngoscope. 2004;114:1724e9.
Abbott MB, Donnelly LF, Dardzinski BJ, Poe SA, Chini BA, Amin RS. Obstructive sleep apnea: MR imaging volume segmentation analysis. Radiology. 2004;232:889e95.
International Commission on Radiological Protection. Publication 87. Managing patient dose in CT. Ann ICRP. 2000;30:1e45.
Frederiksen NL, Benson BW, Sokolowski TW. Effective dose and risk assessment from computed tomography of the maxillofacial complex. Dentomaxillofac Radiol. 1995;24:55e8.
Fleck RJ, Ishman SL, Shott SR, Gutmark EJ, McConnell KB, Mahmoud M, Mylavarapu G, Subramaniam DR, Szczesniak R, Amin RS. Dynamic volume computed tomography imaging of the upper airway in obstructive sleep apnea. J Clin Sleep Med. 2017;13(2):189–96.
Van Holsbeke C, Vos W, Van Hoorenbeeck K, Boudewyns A, Salgado R, Verdonck PR, Ramet J, De Backer J, De Backer W, Verhulst SL. Functional respiratory imaging as a tool to assess upper airway patency in children with obstructive sleep apnea. Sleep Med. 2013;14(5):433–9.
Slaats M, Vos W, Van Holsbeke C, De Backer J, Loterman D, De Backer W, Boudewyns A, Verhulst S. Predicting the effect of treatment in paediatric OSA by clinical examination and functional respiratory imaging. Pediatr Pulmonol. 2017;52(6):799–805.
Slaats MALJ, Loterman D, van Holsbeke C, Vos W, Van Hoorenbeeck K, de Backer J, de Backer W, Wojciechowski M, Boudewyns A, Verhulst S. The role of functional respiratory imaging in treatment selection of children with obstructive sleep apnea and down syndrome. J Clin Sleep Med. 2018;14(4):651–9.
Hatcher DC, Dial C, Mayorga C. Cone beam CT for pre-surgical assessment of implant sites. J Calif Dent Assoc. 2003;31:825e33.
Tsiklakis K, Donta C, Gavala S, Karayianni K, Kamenopoulou V, Hourdakis CJ. Dose reduction in maxillofacial imaging using low dose Cone Beam CT. Eur J Radiol. 2005;56:413e7.
Mozzo P, Procacci C, Tacconi A, Martini PT, Andreis IA. A new volumetric CT machine for dental imaging based on the cone-beam technique: preliminary results. Eur Radiol. 1998;8:1558e64.
Loubele M, Bogaerts R, Van Dijck E, Pauwels R, Vanheusden S, Suetens P, et al. Comparison between effective radiation dose of CBCT and MSCT scanners for dentomaxillofacial applications. Eur J Radiol. 2009;71:461e8.
Alwadei AH, Galang-Boquiren MTS, Kusnoto B, Costa Viana MG, Lin EY, Obrez A, Evans CA, Masoud AI. Computerized measurement of the location and value of the minimum sagittal linear dimension of the upper airway on reconstructed lateral cephalograms compared with 3-dimensional values. Am J Orthod Dentofac Orthop. 2018;154(6):780–7.
Ludlow JB, Gubler M, Cevidanes L, Mol A. Precision of cephalometric landmark identification: cone-beam computed tomography vs conventional cephalometric views. Am J Orthod Dentofacial Orthop. 2009;136:312e1–10.
Chung RR, Lagravere MO, Flores-Mir C, Heo G, Carey JP, Major PW. A comparative analysis of angular cephalometric values between CBCT generated lateral cephalograms versus digitized conventional lateral cephalograms. Int Orthod. 2009;7(4):308–21.
Strauss RA, Wang N. Cone beam computed tomography and obstructive sleep apnoea. Aust Dent J. 2012;57:61e71.
Eimar H, Al-Saleh MAQ, Cortes ARG, Gozal D, Graf D, Flores-Mir C. Sleep-disordered breathing is associated with reduced mandibular cortical width in children. JDR Clin Trans Res. 2019;4(1):58–67.
Calciolari E, Donos N, Park JC, Petrie A, Mardas N. Panoramic measures for oral bone mass in detecting osteoporosis: a systematic review and meta-analysis. J Dent Res. 2015;94(3 Suppl):17S–27S.
Rossi RC, Rossi NJ, Filho EI, Rossi NJ, Chilvarquer I, Fujita RR, Pignatari SS. Differences in volume and area of the upper airways in children with OSA compared to a healthy group. J Dent Sleep Med. 2016;3(3):81–7.
Alsufyani NA, Noga ML, Witmans M, Cheng I, El-Hakim H, Major PW. Using cone beam CT to assess the upper airway after surgery in children with sleep disordered breathing symptoms and maxillary-mandibular disproportions: a clinical pilot. J Otolaryngol Head Neck Surg. 2017;46(1):31.
Horwood L, Brouillette RT, McGregor CD, Manoukian JJ, Constantin E. Testing for pediatric obstructive sleep apnea when health care resources are rationed. JAMA Otolaryngol Head Neck Surg. 2014;140(7):616–23.
Wootton DM, Luo H, Persak SC, Sin S, McDonough JM, Isasi CR, et al. Computational fluid dynamics endpoints to characterize obstructive sleep apnea syndrome in children. J Appl Physiol (1985). 2014;116:104e12.
Hu C, Han D, Zhou B, Zhang L, Li Y, Zang H, Li L. Investigation of resectability degree for adenoidal surgery in OSA children with the method of computational fluid dynamics. Acta Otolaryngol. 2017;137(1):82–5.
Mihaescu M, Murugappan S, Gutmark E, Donnelly LF, Kalra M. Computational modeling of upper airway before and after adenotonsillectomy for obstructive sleep apnea. Laryngoscope. 2008;118:360e2.
Luo H, Sin S, McDonough JM, Isasi CR, Arens R, Wootton DM. Computational fluid dynamics endpoints for assessment of adenotonsillectomy outcome in obese children with obstructive sleep apnea syndrome. J Biomech. 2014;47(10):2498–503.
Nguyen TD, Su E, Lazarow FB, Wheatley FC, Chin-Loy A, Wang A, Protsenko D, Ahuja GS, Chen Z, Wong BJ-F. Constructing 3D models of the pediatric upper airway from long range optical coherence tomography images. In: Choi B, Kollias N, Zeng H, Kang HW, Wong BJ, Ilgner JF, Tearney GJ, Gregory K, Marcu L, Mandelis A, Morris MD, editors. Photonic therapeutics and diagnostics X, vol. 8926. Bellingham: Society of Photo- Optical Instrumentation Engineers (SPIE); 2014. p. 8926L-8921–12.
Ridgway JM, Ahuja G, Guo S, et al. Imaging of the pediatric airway using optical coherence tomography. Laryngoscope. 2007;117(12):2206–12.
Jing JC, Chou L, Su E, Wong BJ, Chen Z. Anatomically correct visualization of the human upper airway using a high-speed long range optical coherence tomography system with an integrated positioning sensor. Sci Rep. 2016;6:39443.
Lazarow FB, Ahuja GS, Chin Loy A, Su E, Nguyen TD, Sharma GK, Wang A, Jing J, Chen Z, Wong BJ. Intraoperative long range optical coherence tomography as a novel method of imaging the pediatric upper airway before and after adenotonsillectomy. Int J Pediatr Otorhinolaryngol. 2015;79(1):63–70.
Kimbell JS, Basu S, Garcia GJM, Frank-Ito DO, Lazarow F, Su E, Protsenko D, Chen Z, Rhee JS, Wong BJ. Upper airway reconstruction using long-range optical coherence tomography: effects of airway curvature on airflow resistance. Lasers Surg Med. 2019;51(2):150–60.
Friedman NR, Parikh SR, Ishman SL, Ruiz AG, El-Hakim H, Ulualp SO, Wootten CT, Koltai PJ, Chan DK. The current state of pediatric drug-induced sleep endoscopy. Laryngoscope. 2017;127(1):266–72.
Tejan J, Medina M, Ulualp SO. Comparative assessment of drug-induced sleep endoscopy scoring systems in pediatric sleep apnea. Laryngoscope. 2019;129(9):2195–8.
Chan DK, Liming BJ, Horn DK, Parikh SR. A new scoring system for upper airway pediatric sleep endoscopy. JAMA Otolaryngol Head Neck Surg. 2014;140:595–602.
Lam DJ, Weaver EM, Macarthur CJ, et al. Assessment of pediatric obstructive sleep apnea using a drug-induced sleep endoscopy rating scale. Laryngoscope. 2016;126:1492–8.
Chen J, He S. Drug-induced sleep endoscopy-directed adenotonsillectomy in pediatric obstructive sleep apnea with small tonsils. PLoS One. 2019;14(2):e0212317.
Park JS, Chan DK, Parikh SR, Meyer AK, Rosbe KW. Surgical outcomes and sleep endoscopy for children with sleep-disordered breathing and hypotonia. Int J Pediatr Otorhinolaryngol. 2016;90:99–106.
Galluzzi F, Pignataro L, Gaini RM, Garavello W. Drug induced sleep endoscopy in the decision-making process of children with obstructive sleep apnea. Sleep Med. 2015;16(3):331–5.
Gazzaz MJ, Isaac A, Anderson S, Alsufyani N, Alrajhi Y, El-Hakim H. Does drug-induced sleep endoscopy change the surgical decision in surgically naive non-syndromic children with snoring/ sleep disordered breathing from the standard adenotonsillectomy? A retrospective cohort study. J Otolaryngol Head Neck Surg. 2017;46(1):12.
Boudewyns A, Verhulst S, Maris M, Saldien V, Van de Heyning P. Drug-induced sedation endoscopy in pediatric obstructive sleep apnea syndrome. Sleep Med. 2014;15(12):1526–31.
Akkina SR, Ma CC, Kirkham EM, Horn DL, Chen ML, Parikh SR. Does drug induced sleep endoscopy-directed surgery improve polysomnography measures in children with Down Syndrome and obstructive sleep apnea? Acta Otolaryngol. 2018;138(11):1009–13.
Miller C, Kirkham E, Ma CC, Filipek N, Horn DL, Johnson K, Chen ML, Parikh SR. Polysomnography outcomes in children with small tonsils undergoing drug-induced sleep endoscopy-directed surgery. Laryngoscope. 2019;129(12):2771–4.
Maris M, Verhulst S, Saldien V, Van de Heyning P, Wojciechowski M, Boudewyns A. Drug-induced sedation endoscopy in surgically naive children with Down syndrome and obstructive sleep apnea. Sleep Med. 2016;24:63–70.
Esteller E, Mulas D, Haspert R, Matiñó E, López R, Girabent-Farrés M. Drug-induced sleep-endoscopy in children's sleep related breathing disorders. Acta Otorrinolaringol Esp. 2016;67(4):212–9.
Barrera JE. Virtual surgical planning improves surgical outcome measures in obstructive sleep apnea surgery. Laryngoscope. 2014;124(5):1259–66.
Mylavarapu G, Subramaniam D, Jonnagiri R, Gutmark EJ, Fleck RJ, Amin RS, Mahmoud M, Ishman SL, Shott SR. Computational modeling of airway obstruction in sleep apnea in down syndrome: a feasibility study. Otolaryngol Head Neck Surg. 2016;155(1):184–7.
Mylavarapu G, Mihaescu M, Fuchs L, Papatziamos G, Gutmark E. Planning human upper airway surgery using computational fluid dynamics. J Biomech. 2013;46(12):1979–86.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Slaats, M.A.L.J., Verhulst, S.L. (2021). Upper Airway Imaging in Pediatric Obstructive Sleep Apnea. In: Gozal, D., Kheirandish-Gozal, L. (eds) Pediatric Sleep Medicine. Springer, Cham. https://doi.org/10.1007/978-3-030-65574-7_16
Download citation
DOI: https://doi.org/10.1007/978-3-030-65574-7_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-65573-0
Online ISBN: 978-3-030-65574-7
eBook Packages: MedicineMedicine (R0)