Abstract
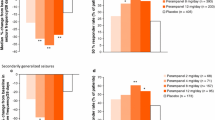
Rufinamide (RUF), originally granted orphan drug status for the adjunctive treatment of seizures in Lennox-Gastaut syndrome (LGS) in 2004, is an orally active anti-convulsant, indicated as adjunctive therapy in the treatment of seizures associated with Lennox-Gastaut syndrome in patients 1 year of age and older in the USA and Europe. It is structurally distinct from any other anti-convulsant drugs and suppresses neuronal hyperexcitability by prolonging the inactivation phase of voltage-gated sodium channels. It is extensively metabolized via enzymatic hydrolysis to a pharmacologically inactive derivative which is excreted in urine and feces. Overall response rate of RUF was observed in median 46.2% in controlled studies on patients with refractory epilepsies including LGS. Seizure freedom rates could be obtained in median 8.3%. Adverse events were observed in median 36.2%; no severe adverse events had been recorded in the studies. Most common AE’s were headache, dizziness, somnolence, vomiting, nausea, fatigue, and diplopia.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aldenkamp AP, Alpherts WCJ. The effect of the new antiepileptic drug rufinamide on cognitive functions. Epilepsia. 2006;47(7):1153–9. https://doi.org/10.1111/j.1528-1167.2006.00589.x.
Arzimanoglou A, Ferreira J, Satlin A, Olhaye O, Kumar D, Dhadda S, Bibbiani F. Evaluation of long-term safety, tolerability, and behavioral outcomes with adjunctive rufinamide in pediatric patients (≥1 to <4 years old) with Lennox-Gastaut syndrome: final results from randomized study 303. Eur J Paediatr Neurol. 2018; https://doi.org/10.1016/j.ejpn.2018.09.010.
Banzel Pediatric Safety Review. 2015. Online verfügbar unter https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/PediatricAdvisoryCommittee/UCM601631.pdf. Zuletzt geprüft am 9 Apr 2019.
Bauer B, Kluger G. Derzeitiger Stellenwert von Rufinamid in der Behandlung des Lennox-Gastaut-Syndroms. Z Epileptol. 2008;21(3):123–34. https://doi.org/10.1007/s10309-008-0314-0.
Borrelli EP, Lee EY, Descoteaux AM, Kogut SJ, Caffrey AR. Stevens-Johnson syndrome and toxic epidermal necrolysis with antiepileptic drugs: an analysis of the US Food and Drug Administration Adverse Event Reporting System. Epilepsia. 2018;59(12):2318–24. https://doi.org/10.1111/epi.14591.
Coppola G. Update on rufinamide in childhood epilepsy. Neuropsychiatr Dis Treat. 2011;7:399–407. https://doi.org/10.2147/NDT.S13910.
Coppola G, Grosso S, Franzoni E, Veggiotti P, Zamponi N, Parisi P, et al. Rufinamide in children and adults with Lennox-Gastaut syndrome: first Italian multicenter experience. Seizure. 2010;19(9):587–91. https://doi.org/10.1016/j.seizure.2010.09.008.
Grosso S, Coppola G, Dontin SD, Gobbi G, Pruna D, Accorsi P, et al. Efficacy and safety of rufinamide in children under four years of age with drug-resistant epilepsies. Eur J Paediatr Neurol. 2014;18(5):641–5. https://doi.org/10.1016/j.ejpn.2014.05.001.
Inovelon® Summary of Product Characteristics. 2019. Online verfügbar unter https://www.medicines.org.uk/emc/product/410. Zuletzt geprüft am 22 Jan 2019.
Jaraba S, Santamarina E, Miró J, Toledo M, Molins A, Burcet J, et al. Rufinamide in children and adults in routine clinical practice. Acta Neurol Scand. 2017;135(1):122–8. https://doi.org/10.1111/ane.12572.
Kessler SK, McCarthy A, Cnaan A, Dlugos DJ. Retention rates of rufinamide in pediatric epilepsy patients with and without Lennox-Gastaut syndrome. Epilepsy Res. 2015;112:18–26. https://doi.org/10.1016/j.eplepsyres.2015.02.003.
Kim JY, Lee CG, Yu HJ, Nam SH, Lee J, Lee M. The efficacy and tolerability of rufinamide in intractable pediatric epilepsy. J Epilepsy Res. 2012;2(2):33–7. https://doi.org/10.14581/jer.12009.
Kim SH, Lee JH, Ryu HW, Lim BC, Chae JH, Choi JE, et al. Short-term efficacy and tolerability of rufinamide adjunctive therapy in children with refractory generalised epilepsy. Epileptic Disord. 2013;15(1):49–54. https://doi.org/10.1684/epd.2013.0557.
Kluger G, Kurlemann G, Haberlandt E, Ernst J-P, Runge U, Schneider F, et al. Effectiveness and tolerability of rufinamide in children and adults with refractory epilepsy: first European experience. Epilepsy Behav. 2009;14(3):491–5. https://doi.org/10.1016/j.yebeh.2008.12.013.
Kluger G, Haberlandt E, Kurlemann G, Ernst J-P, Runge U, Schneider F, et al. First European long-term experience with the orphan drug rufinamide in childhood-onset refractory epilepsy. Epilepsy Behav. 2010;17(4):546–8. https://doi.org/10.1016/j.yebeh.2010.01.005.
May TW, Boor R, Rambeck B, Jürgens U, Korn-Merker E, Brandt C. Serum concentrations of rufinamide in children and adults with epilepsy: the influence of dose, age, and comedication. Ther Drug Monit. 2011;33(2):214–21. https://doi.org/10.1097/FTD.0b013e31820fa9ad.
McLean MJ, Schmutz M, Pozza M, Wamil A. The influence of rufinamide on sodium currents and action potential firing in rodent neurons. Epilepsia. 2005;46:296.. Abstract 3.062
Mourand I, Crespel A, Gelisse P. Dramatic weight loss with rufinamide. Epilepsia. 2013;54(1):e5–8. https://doi.org/10.1111/j.1528-1167.2012.03579.x.
Mueller A, Boor R, Coppola G, Striano P, Dahlin M, von Stuelpnagel C, et al. Low long-term efficacy and tolerability of add-on rufinamide in patients with Dravet syndrome. Epilepsy Behav. 2011;21(3):282–4. https://doi.org/10.1016/j.yebeh.2011.04.057.
National Library of Medicine (US). Drugs and Lactation Database (LactMed). Rufinamide. Bethesda: National Library of Medicine (US); 2006.
Panebianco M, Prabhakar H, Marson AG. Rufinamide add-on therapy for refractory epilepsy. Cochrane Database Syst Rev. 2018;4:CD011772. https://doi.org/10.1002/14651858.CD011772.pub2.
Patsalos PN, Bourgeois BFD. The epilepsy prescriber’s guide to antiepileptic drugs. Cambridge: Cambridge University Press; 2013.
Perucca E, Cloyd J, Critchley D, Fuseau E. Rufinamide: clinical pharmacokinetics and concentration–response relationships in patients with epilepsy. Epilepsia. 2008;49(7):1123–41. https://doi.org/10.1111/j.1528-1167.2008.01665.x.
Striano P, McMurray R, Santamarina E, Falip M. Rufinamide for the treatment of Lennox-Gastaut syndrome: evidence from clinical trials and clinical practice. Epileptic Disord. 2018;20(1):13–29. https://doi.org/10.1684/epd.2017.0950.
Thome-Souza S, Kadish NE, Ramgopal S, Sánchez Fernández I, Bergin AM, Bolton J, et al. Safety and retention rate of rufinamide in 300 patients: a single pediatric epilepsy center experience. Epilepsia. 2014;55(8):1235–44. https://doi.org/10.1111/epi.12689.
Vendrame M, Loddenkemper T, Gooty VD, Takeoka M, Rotenberg A, Bergin AM, et al. Experience with rufinamide in a pediatric population: a single center’s experience. Pediatr Neurol. 2010;43(3):155–8. https://doi.org/10.1016/j.pediatrneurol.2010.04.003.
Verrotti A, Loiacono G, Ballone E, Mattei PA, Chiarelli F, Curatolo P. Efficacy of rufinamide in drug-resistant epilepsy: a meta-analysis. Pediatr Neurol. 2011;44(5):347–9. https://doi.org/10.1016/j.pediatrneurol.2010.12.005.
Yıldız EP, Hızlı Z, Bektaş G, Ulak-Özkan M, Tatlı B, Aydınlı N, et al. Efficacy of rufinamide in childhood refractory epilepsy. Turk J Pediatr. 2018;60(3):238–43. https://doi.org/10.24953/turkjped.2018.03.002.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Hartlieb, T., Kluger, G. (2022). Anti-convulsant Agents: Rufinamide. In: Riederer, P., Laux, G., Nagatsu, T., Le, W., Riederer, C. (eds) NeuroPsychopharmacotherapy. Springer, Cham. https://doi.org/10.1007/978-3-030-62059-2_293
Download citation
DOI: https://doi.org/10.1007/978-3-030-62059-2_293
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-62058-5
Online ISBN: 978-3-030-62059-2
eBook Packages: MedicineReference Module Medicine