Abstract
The coronavirus disease 2019 (COVID-19) pandemic has resulted in worldwide research efforts to recognize people at greatest risk of developing critical illness and dying. Growing numbers of reports have connected obesity to more severe COVID-19 illness and death. Although the exact mechanism by which obesity may lead to severe COVID-19 outcomes has not yet been determined, the mechanisms appear to be multifactorial. These include mechanical changes of the airways and lung parenchyma, systemic and airway inflammation, and general metabolic dysfunction that adversely affect pulmonary function and/or response to treatment. As COVID-19 continues to spread worldwide, clinicians should carefully monitor and manage obese patients for prompt and targeted treatment.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
In late December 2019, a group of pneumonia patients with unknown origin was reported in Wuhan, China [1]. Since then, COVID-19, caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), has taken the world by storm and was officially announced as a pandemic by the World Health Organization (WHO) on March 11, 2020. SARS-CoV-2 is reported to be a member of the betacoronavirus family, associated with the severe acute respiratory syndrome (SARS) virus SARS-CoV [2]. Clinical signs of COVID-19 disease range from asymptomatic or mild infection to severe manifestations that are life-threatening. In China, those over 65 years old and/or with comorbidities were found to be at higher risk of a more severe course of SARS-CoV-2 infection. Among the comorbidities, the highest fatality rates were observed for individuals with cardiovascular disease (CVD) and diabetes mellitus, followed by chronic respiratory diseases, hypertension, cancer, immunosuppressive disorders, and organ failure [3, 4]. As both CVD and diabetes mellitus are linked with increased adipose tissue mass [5], a high body mass index (BMI) and other anthropometric indices associated with obesity might be key risk factors indicative of a more severe course of disease, including development of pneumonia. It has been established that both under- and overnutrition are related to a worse prognosis of viral infections, as occurred in the case of the devastating 1918 influenza pandemic [6]. In addition, recent evidence has shown that obesity and overweight are associated with more severe COVID-19 outcomes [7,8,9]. The parameters mediating this high risk are thought to be due to an impaired respiratory system in obese persons, mediated by elevated airway resistance, disrupted respiratory gas exchange, as well as low respiratory muscle strength and reduced lung volumes [10]. Furthermore, other studies have proposed that obesity is related to an impaired immune response which is a critical factor in COVID-19 disease [11, 12].
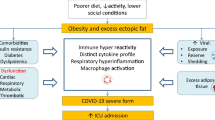
The obesity-related effects on the immune system play a key role in the pathogenesis and outcome of most viral infections such as COVID-19 disease, and obesity is also linked to an increased inflammation response in adipose tissue. In turn, the inflammatory response in adipose tissue can lead to metabolic dysfunction, potentially resulting in dyslipidemia, insulin resistance, diabetes mellitus, hypertension, and CVD [13]. In addition, anthropometric studies have shown that abdominal obesity can cause impaired mechanical ventilation at the base of the lungs, leading to decreased oxygenation of vital tissues [14]. The abnormal secretion of adipokines and cytokines such as tumor necrosis factor-alpha (TNF-α) and interferon (INF) is indicative of a chronic low-grade inflammation characteristic of abdominal-centered obesity, and this may further impair immune responses [15] and negatively impact lung physiology [16]. Figure 8.1 highlights some of the obesity-associated comorbidities related to COVID-19 disease severity.
The COVID-19 pandemic is now spreading all over the world, especially in Europe and the Americas, where obesity has a high prevalence [16]. Although this is only suggestive and does not necessarily imply causation, the links mentioned make a strong case for more thorough investigations into the potential associations between obesity and severity of COVID-19 disease. Obesity has already been identified as a risk factor for individuals experiencing a more severe course of infection during the 2009 influenza A H1N1 epidemic [15, 17, 18]. Taken together, these findings suggest that obesity is an independent risk factor for SARS-CoV-2 infection [19].
Since rapid diagnosis and early treatment appear to produce the best patient outcomes in many disease areas, recognition of risk factors for morbidity and mortality is important to protect the most vulnerable individuals in the society and to guide the most appropriate treatment response in a precision medicine manner. The aim of the present chapter was to investigate the hypothesis that having a higher BMI is a risk factor for COVID-19 infection and its progression to a more severe disease course, independent of other common risk factors. We also discuss potential key mechanisms by which obesity affects COVID-19 disease and may suggest potential therapeutic avenues.
2 Association Between COVID-19 and Obesity: Early Data
Much has been learned from influenza in patients with obesity . The Centers for Disease Control and Prevention (CDC) suggests that individuals with a BMI ≥ 40 kg/m2 have a higher risk of influenza complications [20]. During the H1N1 influenza pandemic, obesity was recognized as an independent risk factor for increasing disease severity [21]. A study also showed that individuals with obesity have reduced protection from influenza immunization [22]. Therefore, it has been deemed likely that obesity is an independent risk factor for COVID-19 severity.
A descriptive study between a small sample of 24 critically ill patients diagnosed with COVID-19 in the Seattle region was one of the first studies to report BMI data with 20 of the patients being either overweight or obese [23]. Although the numbers were too small for statistical analyses, 20 of the patients needed mechanical ventilation and 15 died. In a study in China, older age (≥65 years) and the presence of comorbidities were found to be related to a more severe course in patients infected with SARS-CoV-2 [3]. Across the recorded comorbidities, the highest fatality rates were reported for CVD (10.5%) and diabetes mellitus (7.3%), followed by chronic respiratory diseases (6.3%), hypertension (6.0%), and cancer (5.6%) [3].
Concern about the impacts of BMI was further demonstrated by preliminary data from Shenzhen, China [24], and New York City (NYC), USA [25]. In the Chinese study, it was found that obesity, particularly in men, significantly elevated the risk of developing severe COVID-19 [24]. In addition, the obese patients tended to have upper respiratory tract infection symptoms, including fever and cough, although no significant differences were observed in terms of disease duration or progression. In the New York study, having a BMI >40 kg/m2 was the next strongest independent predictor of hospitalization, after old age [25]. In addition, a small study at the University Hospital in Lille, France, reported that the need for mechanical ventilation in 124 COVID-19 patients was greater for those with a BMI ≥35 kg/m2, independent of other risk factors [26].
In California, USA, between April and August 2009, 1088 individuals were either hospitalized or died from H1N1 influenza infection [27]. Of these individuals, 58% were obese (BMI >30 kg/m2), and 67% had severe obesity (BMI >40 kg/m2). Approximately two-thirds of the patients with obesity had comorbidities, including chronic lung disease, asthma, cardiac problems, or diabetes.
In another investigation, Peng et al. conducted a retrospective analysis of 112 patients infected with SARS-CoV-2 who had been admitted to the Union Hospital in Wuhan, from January 20 to February 15, 2020 [28]. The BMI of the critical patients was higher than that of the general population, and 88% of the non-survivors had a BMI >25 kg/m2 compared to 18.9% of the survivors.
3 Association Between COVID-19 and Obesity: Recent Data
A number of more recent studies over the last 2 months have now been carried out investigating the effects of COVID-19 outcomes in terms of infection, disease severity, and risk of death. These are highlighted in the following sections.
3.1 Association of Obesity with a SARS-CoV-2-Positive Test
A study was carried out by the Oxford Royal College of General Practitioners (RCGP) Research, and Surveillance Center primary care network analyzed routinely collected data from patients tested for SARS-CoV-2 between Jan 28 and April 4, 2020 [29]. They used multivariable logistic regression models to identify risk factors associated with a positive test result. This showed that 20.9% of people with obesity tested positive for the disease compared with only 13.2% of normal-weight people [odds ratio (OR): 1.41, 95% confidence interval (CI): 1.04–1.91). Another study from the UK found that both BMI and waist circumference were positively associated with a positive test for COVID-19 [30]. This investigation also showed a dose-response-like relationship between BMI and a positive test for COVID-19, with odds ratios for overweight (BMI: 25–<30 kg/m2), obese (BMI: 30- < 35 kg/m2), and severely obese (BMI: ≥35 kg/m2) subjects of 1.31 (1.05–1.62), 1.55 (1.19–2.02), and 1.57 (1.14–2.17), respectively, compared to normal weight controls (BMI: 18.5–<25 kg/m2).
3.2 Association of Obesity with COVID-19 Disease Severity
A recent meta-analysis of three studies reported an increased need of invasive mechanical ventilation in COVID-19 patients with a BMI >35 kg/m2 with an OR of 7.36 (95% CI: 1.63–33.14, p = 0.021) [31]. A study in China which investigated the association between obesity and COVID-19 illness severity among patients with confirmed SARS-CoV-2 infection found that each unit increase in BMI was associated with a 12% increase in the risk of severe COVID-19 [32]. A study by Cummings et al. carried out in New York (NY), USA, found that of 257 individuals who were critically ill with COVID-19, 171 (67%) were males, 212 (82%) had at least one chronic illness, and 119 (46%) were obese [33]. Hur et al. analyzed data from 10 Chicago Illinois hospitals in the USA and found that among patients who required intubation, those who were older or more obese required longer intubation times [34].
3.3 Association of Obesity with Increased Risk of Death from COVID-19
A retrospective study of 13 young patients who died of COVID-19 and 40 matched survivors found that the deceased patients had higher BMIs (p = 0.010), increased C-reactive protein (CRP) inflammation biomarker (p = 0.014), increased troponin I (TPNI) cardiac biomarker (p = 0.005), and elevated D-dimer coagulation activity biomarker (p = 0.047) [35]. Klang et al. carried out a retrospective analysis of data from COVID-19 patients hospitalized in New York between March 1 and May 17, 2020, using multivariable logistic regression models and found that among the younger patients that died (<50 years old), having a BMI >40 kg/m2 was independently associated with mortality (OR: 5.1, 95% CI: 2.3–11.1) [36]. Another study in New York of 770 patients found that those who were obese were more likely to present with fever, cough, and shortness of breath, with a significantly higher rate of intensive care unit (ICU) admission or death (p = 0.002) [37].
A prospective study of 20,133 patients in the UK carried out by the International Severe Acute Respiratory and emerging infections consortium (ISARIC) World Health Organization (WHO) Clinical Characterization Protocol UK (CCP-UK) found that increasing age, male sex, and comorbidities, such as obesity and chronic cardiac, pulmonary, kidney, and liver diseases, were associated with higher mortality outcomes [38]. Bello-Chavolla and co-workers carried out a study in Mexico of 51,633 people with SARS-CoV-2 infection, which evaluated risk factors and proposed a lethality score for the disease [39]. In this study, 5332 of the individuals died, and it appeared that obesity increased risk for the need for ICU admission and intubation and was associated with 49.5% of the lethality. Another investigation assessed the obesity prevalence of the top 20 countries ranked according to total COVID-19-related deaths as of May 20, 2020 [40]. This showed that the USA had the highest obesity (36.2%) and overweight (31.7%) prevalence, as well as the highest number of total deaths. In addition, correlation analysis showed that the number of total deaths was significantly correlated with the obesity prevalence in each country (r = 0.464, p = 0.039).
4 Mechanisms
4.1 Inflammation
Obesity alters the innate and adaptive immune responses , which cause a state of chronic low-grade inflammation (Fig. 8.2) [41, 42]. This state is characterized by higher levels of pro-inflammatory cytokines such as TNF-α, macrophage chemoattractant protein I (MCP-1), and interleukin-6 (IL-6), which are mainly secreted from visceral and subcutaneous adipose tissue [43]. However, presentation of an antigen such as a virus results in decreased macrophage activation and blunted pro-inflammatory cytokine production, as well as exacerbation of viral symptoms [44]. This may explain the poorer vaccination response in obese individuals [45]. In addition, B- and T-cell responses are disrupted in obesity which elevates susceptibility to viral infections and a delay in their resolution. A study by Zhang et al. suggested that leptin resistance was a cofactor in the H1N1 influenza pandemic, as this hormone is an important regulator of B cell maturation, development, and performance [46]. In addition, obese patients may have impaired memory T-cell and antibody responses, which could also explain vaccine ineffectiveness [47].
A disturbed pro-inflammatory response is the likely cause of lung lesions observed in victims of influenza pandemics. In line with this, a study on influenza A virus infection in obese ob/ob mice showed elevated disease severity, increased secondary bacterial infections, and decreased vaccine efficacy [48]. A study by the same research group showed that serial passage of a human H1N1 influenza virus through diet-induced and genetic (ob/ob) models of obesity in mice leads to a more severe disease with elevated virulence and morbidity, which may br related to disruption of the INF response [49].
4.2 Impaired Insulin Signaling
Patients with obesity consume a higher than normal percentage of oxygen during respiratory work [50, 51]. Obesity is also linked with respiratory conditions, such as exertional dyspnea, obesity hypoventilation syndrome, chronic obstructive pulmonary disease (COPD), asthma, and aspiration pneumonia [52]. Obesity is a known risk factor for diabetes, metabolic syndrome, and CVD, which may also contribute to higher mortality in COVID-19 cases. Insulin resistance is a major feature of these conditions and can be caused by obesity [53]. Under normal conditions, binding of insulin to the insulin receptor in target tissues results in parallel signaling via the PI3K-Akt and Ras-MAPK networks in the regulation of metabolism and growth pathways (Fig. 8.3). Insulin-resistant states like obesity are characterized by specific impairments in the PI3K-Akt pathway, leading to compensatory hyperinsulinemia in order to maintain normal glycemia. This leads to excessive signaling via the growth pathway, contributing to increased inflammation, proliferation, and hypertrophy.
Insulin signaling in healthy and insulin-resistant states. Akt, protein kinase b; ERK, extracellular receptor kinase; IRS-1, insulin substrate receptor-1; MAPK, mitogen-activated protein kinase; MEK, mitogen-activated protein kinase/ extracellular receptor kinase; p38, p38 mitogen-activated protein kinase; PI3-K, phosphatidylinositol (PI)3-kinase; RAS, a small GTPase involved in signal transduction; RAF, proto-oncogene c-RAF
4.3 Other Mechanisms
Another factor might also lead to the elevated risk from COVID-19 for patients with obesity that was highlighted by a previous study which showed that adipose tissue can serve as a reservoir for human viruses [54]. More studies should be performed to determine if adipose tissue also serves as a focal point of SARS-CoV-2 infection and spreading to other organs.
5 Potential Treatment Avenues
5.1 Biomarkers
As described above, the presence of metabolic diseases such as obesity , hypertension, diabetes, and CVD is likely to contribute to a poorer prognosis in COVID-19 patients. Since these conditions are marked by insulin resistance and a latent chronic inflammatory state, it is possible that the application of approved antidiabetic drugs such as pioglitazone could lead to a better outcome for patients with such comorbidities [55]. As a preventative measure, obtaining a higher level of cardiorespiratory fitness by prior physical exercise may offer some innate immune-protection against SARS-CoV-2 infection by improving insulin signaling and attenuating the “cytokine storm syndrome “that can occur in high-risk individuals [56, 57]. The term cytokine storm describes an excessive and uncontrolled release of pro-inflammatory cytokines which can cause damage to the lungs and other tissues. High-risk patients could be identified by biomarker tests for insulin resistance, such as an oral glucose tolerance test or the measurement of the triglyceride and glucose index. Ren et al. showed that the latter index was closely associated with severity and morbidity in patients infected with SAR-CoV-2 [58]. Thus, the triglyceride-glucose index may be a useful marker for identification of those patients who are likely to experience a worse outcome of COVID-19 disease . These individuals could then be prioritized for specialized treatments. The successful use of some anti-inflammatory drugs in other hyperinflammation-related diseases like rheumatoid arthritis has generated much speculation about whether or not similar approaches could be useful in patients with COVID-19 disease and high inflammatory biomarker profiles [59, 60].
5.2 Physical Exercise and Dietary Changes
A large-scale population study of 387,109 men and women in the UK found that physical inactivity (relative risk = 1.32, 1.10–1.58), smoking (1.42, 1.12–1.79), and high BMI (2.05, 1.68–2.49) were related to cases of COVID-19 serious enough to warrant hospital admission [61]. Such problems may be compounded by obesity. For example, a study of 123 obese individuals under stay-at-home orders found that most reported increased anxiety and depression, increased stress eating, increased difficultly in achieving weight loss goals, and reduced exercise time and intensity [62]. This problem may have been exacerbated during the imposed lockdown in many countries due to negative effects on eating behaviors and dietary habits [63]. Physical inactivity is known to increase symptom severity and death outcomes in individuals with chronic diseases due to blunting of the immune response and macrophage activation, caused by the associated increased insulin resistance [64]. In contrast, exercise is known to reduce the risk of mortality from metabolic diseases through an increase in physiological reserve and enhanced immunological benefits.
Together, these findings argue for the development of specialized programs to encourage healthier lifestyles involving improved nutritional quality and increased physical activity to assist with disease management during and after the COVID-19 pandemic.
6 Conclusions and Future Perspectives
In conclusion, patients with obesity and, most importantly, those with severe obesity should take extra measures to avoid coming into contact with SARS-CoV-2-infected individuals during the current pandemic. Such individuals have a higher risk of more severe forms of COVID-19 disease due to impaired insulin signaling and chronic low grade inflammation. It is now accepted that researchers and clinicians should take these factors into account in order to offer the best possible therapeutic approach and to improve chances of a favorable outcome. This may include interventions such as the use of antidiabetic and anti-inflammatory drugs to potentially decrease the chances of the patient progressing to severe COVID-19 illness.
This current pandemic has highlighted that more should be done at the individual level to reduce the effects of obesity in our societies to minimize the effects of the current and future pandemics. As a preventative measure, policies should be adopted worldwide which encourage individuals to adopt a healthier lifestyle, involving improved nutrition and increased physical activity. This will also have the added benefit of decreasing the effects of other communicable and noncommunicable diseases on society and relieve the ever increasing burden on healthcare at a global level.
References
Zhu N, Zhang D, Wang W, Li X, Yang B, Song J et al (2020) A novel coronavirus from patients with Pneumonia in China, 2019. N Engl J Med 382(8):727–733
Epidemiology Working Group for NCIP Epidemic Response, Chinese Center for Disease Control and Prevention (2020) The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China. Zhonghua Liu Xing Bing XueZaZhi 41(2):145–151
Wu Z, McGoogan JM (2020) Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA 323:1239–1242
Wu Z, McGoogan JM (2020) Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA 323:1239–1242
Stefan N, Schick F, Häring HU (2017) Causes, characteristics, and consequences of metabolically unhealthy normal weight in humans. Cell Metab 26(2):292–300
Short KR, Kedziereska K, van de Sandt CE (2018) Back to the future: lessons learned from the 1918 influenza pandemic. Front Cell Infect Microbiol 8(8):343. https://doi.org/10.3389/fcimb.2018.00343
Dietz W, Santos-Burgoa C (2020) Obesity and its implications for COVID-19 mortality. Obesity (Silver Spring) 28(6):1005. https://doi.org/10.1002/oby.22818
Lighter J, Phillips M, Hochman S, Sterling S, Johnson D, Francois F et al (2020) Obesity in patients younger than 60 years is a risk factor for Covid-19 hospital admission. Clin Infect Dis:ciaa415. https://doi.org/10.1093/cid/ciaa415. Online ahead of print
Simonnet A, Chetboun M, Poissy J, Raverdy V, Noulette J, Duhamel A et al (2020) High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring). https://doi.org/10.1002/oby.22831. Online ahead of print
Murugan AT, Sharma G (2008) Obesity and respiratory diseases. Chron Respir Dis 5(4):233–242
Sheridan PA, Paich HA, Handy J, Karlsson EA, Hudgens MG, Sammon AB et al (2012) Obesity is associated with impaired immune response to influenza vaccination in humans. Int J Obes 36(8):1072–1077
Green WD, Beck MA (2017) Obesity impairs the adaptive immune response to influenza virus. Ann Am Thorac Soc 14(Suppl 5):S406–S409
Kassir R (2020) Risk of COVID-19 for patients with obesity. Obes Rev 21(6):e13034. https://doi.org/10.1111/obr.13034
Dixon AE, Peters U (2018) The effect of obesity on lung function. Expert Rev Respir Med 12(9):755–767
Huttunen R, Syrjänen J (2013) Obesity and the risk and outcome of infection. Int J Obes 37(3):333–340
Simonnet A, Chetboun M, Poissy J, Raverdy V, Noulette J, Duhamel A et al (2020) High prevalence of obesity in severe acute respiratory syndrome Coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring). https://doi.org/10.1002/oby.22831. Online ahead of print
Wang W, Chen H, Li Q, Qiu B, Wang J, Sun X et al (2011) Fasting plasma glucose is an independent predictor for severity of H1N1 pneumonia. BMC Infect Dis 11:104. https://doi.org/10.1186/1471-2334-11-104
Honce R, Schultz-Cherry S (2019) Impact of obesity on influenza A virus pathogenesis, immune response, and evolution. Front Immunol 10:1071. https://doi.org/10.3389/fimmu.2019.01071
Ryan DH, Ravussin E, Heymsfield S (2020) COVID 19 and the patient with obesity – the editors speak out. Obesity (Silver Spring) 28(5):847. https://doi.org/10.1002/oby.22808
Centers for Disease Control and Prevention. People at high risk for flu complications. https://www.cdc.gov/flu/highr isk/index.htm. Updated August 27, 2018. Accessed 19 Mar 2020
Louie JK, Acosta M, Winter K, Jean C, Gavali S, Schechter R et al (2009) Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA 302(17):1896–1902
Neidich SD, Green WD, Rebeles J, Karlsson EA, Schultz-Cherry S, Noah TL et al (2017) Increased risk of influenza among vaccinated adults who are obese. Int J Obes 41(9):1324–1330
Bhatraju PK, Ghassemieh BJ, Nichols M, Kim R, Jerome KR, Nalla AK et al (2020) Covid-19 in critically ill patients in the Seattle region- case series. N Engl J Med 382:2012–2022
Cai Q, Chen F, Wang T, Luo F, Liu X, Wu Q et al (2020) Obesity and COVID-19 severity in a designated hospital in Shenzhen, China. Diabetes Care:dc200576. https://doi.org/10.2337/dc20-0576. Online ahead of print
Petrilli CM, Jones SA, Yang J, Rajagopalan H, O'Donnell LF, Chernyak Y et al (2020) Factors associated with hospitalization and critical illness among 4,103 patients with COVID-19 disease in New York City. medRxiv. https://doi.org/10.1101/2020.04.08.20057794
Simonnet A, Chetboun M, Poissy J, Raverdy V, Noulette J, Duhamel A et al (2020) High prevalence of obesity in severe acute respiratory syndrome Coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring). https://doi.org/10.1002/oby.22831. Online ahead of print
Venkata C, Sampathkumar P, Afessa B (2010) Hospitalized patients with 2009 H1N1 influenza infection: the Mayo Clinic experience. Mayo Clin Proc 85(9):798–805
Peng YD, Meng K, Guan HQ, Leng L, Zhu RR, Wang BY et al (2020) Clinical characteristics and outcomes of 112 cardiovascular disease patients infected by 2019-nCoV. Zhonghuaxinxue Guan Bing Za Zhi 48(0):E004. https://doi.org/10.3760/cma.j.cn112148-20200220-00105
de Lusignan S, Dorward J, Correa A, Jones N, Akinyemi O, Amirthalingam G et al (2020) Risk factors for SARS-CoV-2 among patients in the Oxford Royal College of general practitioners research and surveillance Centre primary care network: a cross-sectional study. Lancet Infect Dis. https://doi.org/10.1016/S1473-3099(20)30371-6
Yates T, Razieh C, Zaccardi F, Davies MJ, Khunti K (2020) Obesity and risk of COVID-19: analysis of UK biobank. Prim Care Diabetes:S1751-9918(20)30196-0. https://doi.org/10.1016/j.pcd.2020.05.011. Online ahead of print
Tamara A, Tahapary DL (2020) Obesity as a predictor for a poor prognosis of COVID-19: a systematic review. Diabetes Metab Syndr 14(4):655–659
Gao F, Zheng KI, Wang XB, Sun QF, Pan KH, Wang TY et al (2020) Obesity is a risk factor for greater COVID-19 severity. Diabetes Care:dc200682. https://doi.org/10.2337/dc20-0682. Online ahead of print
Cummings MJ, Baldwin MR, Abrams D, Jacobson SD, Meyer BJ, Balough EM et al (2020) Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet 395(10239):1763–1770
Hur K, Price CPE, Gray EL, Gulati RK, Maksimoski M, Racette SD et al (2020) Factors associated with intubation and prolonged intubation in hospitalized patients with COVID-19. Otolaryngol Head Neck Surg. https://doi.org/10.1177/0194599820929640. Online ahead of print
Zhang F, Xiong Y, Wei Y, Hu Y, Wang F, Li G et al (2020) Obesity predisposes to the risk of higher mortality in young COVID-19 patients. J Med Virol. https://doi.org/10.1002/jmv.26039. Online ahead of print
Klang E, Kassim G, Soffer S, Freeman R, Levin MA, Reich DL et al (2020) Morbid obesity as an independent risk factor for COVID-19 mortality in hospitalized patients younger than 50. Obesity (Silver Spring). https://doi.org/10.1002/oby.22913. Online ahead of print
Hajifathalian K, Kumar S, Newberry C, Shah S, Fortune B, Krisko T et al (2020) Obesity is associated with worse outcomes in COVID-19: analysis of early data from new York City. Obesity (Silver Spring). https://doi.org/10.1002/oby.22923. Online ahead of print
Docherty AB, Harrison EM, Green CA, Hardwick HE, Pius R, ISARIC4C Investigators et al (2020) Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ 369:m1985. https://doi.org/10.1136/bmj.m1985
Bello-Chavolla OY, Bahena-López JP, Antonio-Villa NE, Vargas-Vázquez A, González-Díaz A, Márquez-Salinas A et al (2020) Predicting mortality due to SARS-CoV-2: A mechanistic score relating obesity and diabetes to COVID-19 outcomes in Mexico. J Clin Endocrinol Metab:dgaa346. https://doi.org/10.1210/clinem/dgaa346. Online ahead of print
Ekiz T, Pazarlı AC (2020) Relationship between COVID-19 and obesity. Diabetes Metab Syndr 14(5):761–763
Anderson CJ, Murphy KE, Fernandez ML (2016) Impact of obesity and metabolic syndrome on immunity. Adv Nutr 7:66–77
Ouchi N, Parker JL, Lugus JJ, Walsh K (2011) Adipokinesin inflammation and metabolic disease. Nat Rev Immunol 11(2):85–97
Richard C, Wadowski M, GorukS CL, Sharma AM, Field CJ (2017) Individuals with obesity and type 2 diabetes have additional immune dysfunction compared with obese individuals who are metabolically healthy. BMJ Open Diabetes Res Care 5(1):e000379. https://doi.org/10.1136/bmjdrc-2016-000379
Ahn SY, Sohn SH, Lee SY, Park HL, Park YW, Kim H et al (2015) The effect of lipopolysaccharide-induced obesity and its chronic inflammation on influenza virus-related pathology. Environ Toxicol Pharmacol 40(3):924–930
Karlsson EA, Hertz T, Johnson C, Mehle A, Krammer F, Schultz-Cherry S (2016) Obesity outweighs protection conferred by adjuvanted influenza vaccination. mBio 7(4):e01144-16. https://doi.org/10.1128/mBio.01144-16
Zhang AJ, To KK, Li C, Lau CC, Poon VK, Chan CC et al (2013) Leptin mediates the pathogenesisi of severe 2009 pandemic influenza A (H1N1) infection associated with cytokine dysregulation in mice with diet-induced obesity. J Infect Dis 207(8):1270–1280
Honce R, Schultz-Cherry S (2019) Impact of obesity on influenza, a virus pathogenesis, immune response, and evolution. Front Immunol 10:1071. https://doi.org/10.3389/fimmu.2019.01071
O’Brien KB, Vogel P, Duan S, Govorkova EA, Webby RJ, McCullers JA et al (2012) Impaired wound healing predisposes obese mice to severe influenza virus infection. J Infect Dis 205(2):252–261
Honce R, Karlsson EA, Wohlgemuth N, Estrada LD, Meliopoulos VA, Yao J et al (2020) Obesity related microenvironment promotes emergence of virulent influenza virus strains. mBio 11(2):e03341-19. https://doi.org/10.1128/mBio.03341-19
Baik I, Curhan GC, Rimm EB, Bendich A, Willett WC, Fawzi WW (2000) A prospective study of age and lifestyle factors in relation to community acquired pneumonia in US men and women. Arch Intern Med 160(20):3082–3088
Dixon AE, Peters U (2018) The effect of obesity on lung function. Expert Rev Respir Med 12(9):755–767
Zammit C, Liddicoat H, Moonsie I, Makker H (2010) Obesity and respiratory diseases. Int J Gen Med 3:335–343
de Luca C, Olefsky JM (2006) Stressed out about obesity and insulin resistance. Nat Med 12(1):41–42. discussion 42
Bourgeois C, Gorwood J, Barrail-Tran A, Lagathu C, Capeau J, Desjardins D et al (2019) Specific biological features of adipose tissue, and their impact on HIV persistence. Front Microbiol 10:2837. https://doi.org/10.3389/fmicb.2019.02837
Carboni E, Carta AR, Carboni E (2020) Can pioglitazone be potentially useful therapeutically in treating patients with COVID-19? Med Hypotheses 140:109776. https://doi.org/10.1016/j.mehy.2020.109776
Zbinden-Foncea H, Francaux M, Deldicque L, Hawley JA (2020) Does high cardiorespiratory fitness confer some protection against pro-inflammatory responses after infection by SARS-CoV-2? Obesity (Silver Spring). https://doi.org/10.1002/oby.22849. Online ahead of print
Kim J, Nam JH (2020) Insight into the relationship between obesity-induced low-level chronic inflammation and COVID-19 infection. Int J Obes (Lond) 22:1–2. https://doi.org/10.1038/s41366-020-0602-y. Online ahead of print
Ren H, Yang Y, Wang F, Yan Y, Shi X, Dong K et al (2020) Association of the insulin resistance marker TyG index with the severity and mortality of COVID-19. Version 2. Cardiovasc Diabetol 19(1):58. https://doi.org/10.1186/s12933-020-01035-2
Korakas E, Ikonomidis I, Kousathana F, Balampanis K, Kountouri A, Raptis A et al (2020) Obesity and COVID-19: immune and metabolic derangement as a possible link to adverse clinical outcomes. Am J Physiol Endocrinol Metab. https://doi.org/10.1152/ajpendo.00198.2020. Online ahead of print
Almerie MQ, Kerrigan DD (2020) The association between obesity and poor outcome after COVID-19 indicates a potential therapeutic role for montelukast. Med Hypotheses 143:109883. https://doi.org/10.1016/j.mehy.2020.109883
Hamer M, Kivimäki M, Gale CR, Batty GD (2020) Lifestyle risk factors, inflammatory mechanisms, and COVID-19 hospitalization: a community-based cohort study of 387,109 adults in UK. Brain Behav Immun:S0889-1591(20)30996-X. https://doi.org/10.1016/j.bbi.2020.05.059. Online ahead of print
Almandoz JP, Xie L, Schellinger JN, Mathew MS, Gazda C, Ofori A et al (2020) Impact of COVID-19 stay-at-home orders on weight-related behaviors among patients with obesity. Clin Obes Jun 9:e12386. https://doi.org/10.1111/cob.12386. Online ahead of print
Sidor A, Rzymski P (2020) Dietary choices and habits during COVID-19 lockdown: experience from Poland. Nutrients 12(6):E1657. https://doi.org/10.3390/nu12061657
Hudson GM, Sprow K (2020) Promoting physical activity during the COVID-19 pandemic: implications for obesity and chronic disease management. J Phys Act Health:1–3. https://doi.org/10.1123/jpah.2020-0318. Online ahead of print
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Editor(s) (if applicable) and The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Abiri, B., Guest, P.C., Vafa, M. (2021). Obesity and Risk of COVID-19 Infection and Severity: Available Evidence and Mechanisms. In: Guest, P.C. (eds) Clinical, Biological and Molecular Aspects of COVID-19. Advances in Experimental Medicine and Biology(), vol 1321. Springer, Cham. https://doi.org/10.1007/978-3-030-59261-5_8
Download citation
DOI: https://doi.org/10.1007/978-3-030-59261-5_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-59260-8
Online ISBN: 978-3-030-59261-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)