Abstract
The median nerve originates from the lateral (C5–7) and medial cords (C8, T1) of the brachial plexus. The median nerve enters the arm lateral to the brachial artery, crosses anterior to the artery near the insertion of the coracobrachialis, and descends medial to the artery until reaching the cubital fossa. In the fossa, the nerve situates in between the bicipital aponeurosis and brachialis. The median nerve enters the forearm between the two heads of the pronator teres, crosses lateral to the ulnar artery, and passes posterior to a tendinous bridge between the humeroulnar and radial heads of the flexor digitorum superficialis before descending the forearm along the flexor digitorum superficialis and anterior to the flexor digitorum profundus. It emerges from behind the lateral edge of the flexor digitorum superficialis 5 cm proximal to the flexor retinaculum, and lies superficially between the flexor digitorum superficialis and flexor carpi radialis tendons. The median nerve enters the hand deep to the flexor retinaculum and innervates the thenar muscles and the two radial lumbricals via its motor branches, and innervates the palmar aspect of the first, second, and third fingers and the radial half of the fourth finger via its sensory cutaneous branches.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Hand variations
- Hand anomalies
- Martin-Gruber anastomoses
- Marinacci communications
- Gantzer’s muscle
- Accessory muscles
- Anomalous muscles
- Accessory ossicles
Nerves
Median Nerve
Normal Course
The median nerve originates from the lateral (C5–7) and medial cords (C8, T1) of the brachial plexus (Fig. 17.1). The median nerve enters the arm lateral to the brachial artery, crosses anterior to the artery near the insertion of the coracobrachialis, and descends medial to the artery until reaching the cubital fossa. In the fossa, the nerve situates in between the bicipital aponeurosis and brachialis. The median nerve enters the forearm between the two heads of the pronator teres, crosses lateral to the ulnar artery, and passes posterior to a tendinous bridge between the humeroulnar and radial heads of the flexor digitorum superficialis before descending the forearm along the flexor digitorum superficialis and anterior to the flexor digitorum profundus. It emerges from behind the lateral edge of the flexor digitorum superficialis 5 cm proximal to the flexor retinaculum, and lies superficially between the flexor digitorum superficialis and flexor carpi radialis tendons. The median nerve enters the hand deep to the flexor retinaculum and innervates the thenar muscles and the two radial lumbricals via its motor branches, and innervates the palmar aspect of the first, second, and third fingers and the radial half of the fourth finger via its sensory cutaneous branches [1].
Variations and Pathology
Carpal tunnel syndrome (CTS) , a common pathology affecting the median nerve, is often treated by surgical decompression. Awareness of the variations of the median nerve near the carpal tunnel is important when treating CTS. One particular branch to be cognizant of during carpal tunnel surgery is the thenar branch or recurrent motor branch. This branch normally divides from the median nerve radially at the distal margin of the transverse carpal ligament. However, the thenar branch has also been found to pierce through the transverse carpal ligament or leave the median nerve within the carpal tunnel under the transverse carpal ligament (Fig. 17.2) [2]. The thenar branch has even been found to divide from the median nerve ulnarly. Lanz [2] organized these variations of the thenar branch into one group and additionally described three other groups of median nerve variations at the wrist including accessory branches at the distal carpal tunnel, a high division of the median nerve (sometimes in association with a median artery), and accessory branches proximal to the carpal tunnel [2,3,4,5,6,7,8,9,10].
The thenar branch normally leaves the median nerve distal to the transverse carpal ligament. However, the thenar branch can pierce through the transverse carpal ligament (transligamentous) or leave the median nerve under the transverse carpal ligament (subligamentous) [2]
Variations of the median nerve may also exist more proximally. A rare variation of the median nerve supplying the anterior compartment of the arm in the absence of a musculocutaneous nerve bilaterally has been reported [11,12,13]. Variation of a distal union of the lateral and medial cords and piercing of the coracobrachialis by the proximal lateral cord instead of the musculocutaneous nerve has also been reported. Due to the possible use of a coracobrachialis flap in postmastectomy reconstruction, it is important to be aware of this variation [14].
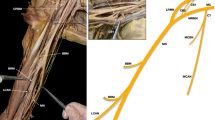
Multiple communicating branches may pass from the median nerve (and sometimes the anterior interosseous nerve) to the ulnar nerve called Martin-Gruber anastomoses (Fig. 17.3). Martin-Gruber anastomoses are present in about 17% of the global population. These branches arise from the median nerve proximally and pass medially between the flexor digitorum superficialis and the flexor digitorum profundus, deep to the ulnar artery, and join the ulnar nerve. This results in median nerve innervation of a variable number of intrinsic muscles of the hand, and presumably explains cases of functional “ulnar intrinsics” muscles despite ulnar nerve transection, and functional “median intrinsics” muscles despite median nerve transection [15]. Rarely, the reverse may occur, where communicating branches from the ulnar nerve pass to the median nerve, usually in the distal forearm near the carpal tunnel. This is termed a Marinacci communication (Fig. 17.3). Hand surgeons should be aware of these communications between the median and ulnar nerve, which can mask upper extremity nerve lesions [1, 14, 15].
Martin-Gruber anastomoses and Marinacci communications . Martin-Gruber anastomoses are communicating branches that pass from the median nerve or anterior interosseous nerve to the ulnar nerve. Marinacci communications are the reverse, where communicating branches from the ulnar nerve pass to the median nerve
Palmar Cutaneous Branch of the Median Nerve
Normal Course
The palmar cutaneous nerve (PCN) branches off the radial aspect of the median nerve at an average of 4–5 cm proximal to the wrist flexion crease (Fig. 17.4) [16, 17]. It runs in a small tunnel of sheath ulnar to the flexor carpi radialis and deep to the antebrachial fascia between the flexor carpi radialis (FCR) and palmaris longus (PL). It then pierces and emerges from the antebrachial fascia, where it runs superficial to the flexor retinaculum and divides into medial and lateral branches. The lateral branches supply the thenar skin and connect with the lateral cutaneous nerve of the forearm. Medial branches supply the central palmar skin and connect with the palmar cutaneous branch of the ulnar nerve.
Variations and Pathology
A few variations of the PCN should be noted because surgeons frequently operate near the PCN, and injury to the PCN can result in complex regional pain syndrome at a rate of 3–10% [16, 18]. Takeoff of the PCN from the ulnar side of the median nerve is seen in about 15% of patients, and in these cases, the PCN tends to position closer to (mean distance of 0.3 cm) or crossover to the ulnar side of the palmaris longus (PL) . Therefore, care should be taken to draw longitudinal incisions at 1 cm ulnar to the PL during its harvest to avoid damaging the PCN [19]. In other cases, the PCN is noted to either insert into or pass through PL tendon 1–1.5 cm proximal to its insertion into the aponeurosis. Transecting the PL at a more proximal position (at least 2 cm from the aponeurotic insertion) is recommended to avoid PCN damage [17, 20].
The PCN may sometimes crossover or enter the FCR sheath and course along its radial aspect. It is important to recognize this variant in cases of volar plating for distal radius fracture, because the PCN variant will be at risk for injury during the FCR radial dissection [16].
Taleisnik et al. demonstrated that the PCN can have a trans-retinaculum course, which is at risk for injury in the longitudinal division of the ligament during carpal tunnel releases. He recommended a curved longitudinal incision on the ulnar side of the axis of the ring finger [20]. However, Hobbs et al. suggested that Taleisnik’s incision may jeopardize the palmar branch of the ulnar nerve and recommended, instead, a skin incision 1 cm ulnar to the axis of the third metacarpal, in the axis of the ring finger [21]. They also noted that in 8% of hands a communication exists between the superficial radial nerve and the PCN that should be considered in surgeries requiring radial palmar wrist incisions (e.g., volar wrist ganglion cyst) [22].
Anterior Interosseous Nerve (AIN)
Normal Course
The anterior interosseous nerve (AIN) arises from the median nerve posteriorly between the two heads of pronator teres about 5 cm distal to the medial epicondyle. This motor branch of the median nerve supplies the anterior compartment of the forearm, classically innervating the flexor pollicis longus (FPL), the pronator quadratus (PQ), and the radial half of the flexor digitorum profundus (FDP) (Fig. 17.5).
Variation and Pathology
Anterior interosseous nerve syndrome (AINS) or Kiloh-Nevin syndrome is a rare entrapment syndrome of the AIN that affects the motor function of the FPL, the PQ, and the radial half of FDP (i.e., second and third digit). The compression is usually caused by the fibrous bands from the deep heads of the pronator teres to the brachialis fascia, but compression from other fibrous bands can also occur. The AIN and ulnar nerve can variably innervate the radial and ulnar FDPs. In one study of 50 hands, 50% of hands had AIN innervation to all FDPs, with the ulnar nerve providing dual innervation to the FDP of the third-fifth digits. The second most common finding (34%) was dual innervation to the third and fourth FDPs [23]. Clinically, these variations manifest as variable symptoms in AINS: the second digit is consistently affected, whereas the third digit and the PQ are occasionally spared depending on the innervation pattern [24, 25]. The presence of Martin-Gruber anastomoses (see above) in the setting of AINS can also lead to palsy of the intrinsic muscles of the hand (normally innervated by the ulnar nerve).
Ulnar Nerve
Normal Course
The ulnar nerve carries fibers from the C8 and T1 nerve roots as it originates from the medial cord of the brachial plexus (occasionally also from the C7 nerve root of the lateral cord) (Fig. 17.6). The ulnar nerve descends along the posteromedial aspect of the humerus. It lies posterior and posteromedial to the intermuscular septum and brachial artery, respectively, and anterior to the medial head of the triceps brachii muscle. As the ulnar nerve descends in the upper arm, the arcade of Struthers, a band of fascia attached to the intermuscular septum, covers the ulnar nerve about 8 cm proximal to the medial epicondyle of the humerus. The ulnar nerve descends posterior to the medial epicondyle of the humerus and medial to the olecranon of the ulna before entering the cubital tunnel. The cubital tunnel is defined by the medial epicondyle, the olecranon, and a fascial layer extending from the flexor carpi ulnaris muscle and the arcuate ligament of Osborne.
After exiting the cubital tunnel, the ulnar nerve enters the anterior compartment of the forearm between the ulnar and humeral heads and under the aponeurosis of the flexor carpi ulnaris muscle. The ulnar nerve then courses along the ulna with the ulnar artery, deep to the flexor carpi ulnaris muscle. It gives off three branches in the forearm: the muscular branch (near the elbow), the dorsal branch (approximately 5 cm proximal to the wrist), and the palmar branch (the continuation of the ulnar nerve) [1]. The muscular branch innervates the flexor carpi ulnaris and the ulnar half of the flexor digitorum profundus muscles. The dorsal branch passes backward beneath the flexor carpi ulnaris, perforates the deep fascia, and runs along the back of the wrist and hand as two dorsal digital branches to provide sensory innervation to the fifth finger and the ulnar side of the ring finger. The palmar branch/ulnar nerve enters the hand by passing superficial to the flexor retinaculum and deep to the superficial palmar carpal ligament via Guyon’s canal before dividing it into the superficial and deep branches, which provide sensory and motor innervation to the hand.
Variations and Pathology
A few variations occur near the elbow that are important for the surgeon to be aware of, particularly when managing ulnar nerve pathology such as in cubital tunnel syndrome. The ulnar nerve can pass in front of the medial epicondyle of the humerus instead of passing posterior to it. The ulnar nerve may also send a variable number of sensory branches to the elbow joint capsule at various distances from the elbow [26, 27]. Several studies found that the motor branch from the ulnar nerve providing innervation to the flexor carpi ulnaris originated anywhere from 40 mm proximal to 73 mm distal to the medial epicondyle [28, 29]. Although not normally a branch of the ulnar nerve, the medial antebrachial cutaneous nerve also can send a variable number of superficial branches to the elbow region [30]. One report described that the medial antebrachial cutaneous nerve originated from the ulnar nerve in the region of the elbow [31]. These branches are all susceptible to iatrogenic injury when surgically managing compression of the ulnar nerve not only in the cubital tunnel, but also in any pathology involving the elbow region.
The dorsal branch of the ulnar nerve follows a fairly consistent course on the dorsum of the wrist and hand by crossing a line drawn between the ulnar styloid and the fourth web space, on average 2.4 cm distal to the ulnar styloid (Fig. 17.7) [32]. However, aberrant branches that cross the wrist immediately distal to the ulnar styloid or ulnar head to innervate the radial side of the hand have been reported. Standard portals for wrist arthroscopy involve placement of the 6-radial portal within the proximal fifth of a line drawn between the ulnar styloid and fourth web space to avoid damage to the dorsal branch of the ulnar nerve. However, extreme care must still be taken as variations described above have been shown to cross this standard area of portal placement [32].
Dorsal branch of the ulnar nerve . The dorsal branch of the ulnar nerve normally travels along the dorsum of the hand crossing radially over a line drawn between the ulnar styloid and the fourth web space 2.4 cm distal to the ulnar styloid [32]
Ulnar tunnel syndrome , or Guyon’s canal syndrome , occurs due to compression of the ulnar nerve within Guyon’s canal. The location of the compression within Guyon’s canal is categorized by three zones: zone I, the bifurcation; zone II, along the deep branch; and zone III, along the superficial branch (Fig. 17.8). Compressions at zone I, zone II, and zone III typically produce motor and sensory deficits, exclusively motor deficits, and exclusively sensory deficits, respectively. However, division patterns of the ulnar nerve within Guyon’s canal are variable, with several different patterns described. Lindsey and Watumull described a trifurcated nerve pattern resulting in a motor branch and two sensory branches within Guyon’s canal [33]. Murata et al. categorized these variations into five types (Fig. 17.9) [34]. Interestingly, several instances of communications between the distal branches were found among the different types. Varying symptoms are described depending on the level of compression.
Types of ulnar nerve branching within Guyon’s canal. In type I, the ulnar nerve bifurcates into the deep branch and the superficial trunk (normal). In type II, the ulnar nerve trifurcates into the deep branch, the common digital nerve of the ring finger, and the proper digital nerve. In type III, the ulnar nerve bifurcates into radial and ulnar trunks. In type IV, an additional superficial branch divides from the ulnar nerve proximally. In type V, the dorsal branch of the ulnar nerve communicates with the proper digital nerve [34]
Although not a variation of the ulnar nerve itself, the contents of Guyon’s canal may differ, which are thought to contribute to compression of the ulnar nerve. An accessory abductor digiti minimi or accessory flexor digiti minimi traveling in Guyon’s canal and a fibrous arch between the hook of hamate and the pisiform have both been described as common occurrences and possible contributors to ulnar nerve entrapment [35].
Sensory innervation to the hand varies significantly. The medial proper palmar digital nerve of the fifth finger has been described as originating from the dorsal branch of the ulnar nerve and traveling outside of Guyon’s canal before reaching the palmar medial fifth finger [36,37,38,39,40]. The ulnar nerve can provide sensory innervation to more than the typical 1.5 digits of the hand both palmarly and dorsally, and innervation of the dorsal first phalanx has even been reported [36, 41, 42]. In these specific situations, compression of the ulnar nerve can result in sensory symptoms different from those in the ring and little finger.
First illustrated by Berrettini in 1741, communications exist between the common digital nerves of the ulnar and median nerves. These are called Berrettini anastomoses (Fig. 17.10a) [43]. The incidence of Berrettini anastomoses is thought to be over 80%, which would technically make them a normal structure rather than an anatomic variant. Damage to these anastomoses resulting in sensory deficits has been described during surgeries such as carpal tunnel release [44]. Therefore, care must be taken to avoid damaging these neural communications.
Riche and Cannieu first described a neural connection between the deep branches of the ulnar nerve and the recurrent branch of the median nerve at the thenar eminence, which was later termed the Riche-Cannieu anastomosis (RCA) [45]. This anastomosis has been reported in up to 83.3% of cases (Fig. 17.10b) [45, 46]. The ulnar nerve can innervate any number of the thenar muscles and intrinsic hand muscles normally innervated by the median nerve through this neural connection, and should be kept in mind in the presence of atypical muscle deficits in carpal tunnel syndrome or ulnar nerve compression at Guyon’s canal or more proximally [47,48,49,50,51].
Radial Nerve
Normal Course
The radial nerve originates from the posterior cord of the brachial plexus and carries fibers from the C5–T1 nerve roots (Fig. 17.11). It splits from the axillary nerve just proximal to the quadrangular space and travels posterior to the axillary and brachial arteries and anterior to the long head of the triceps brachii muscle through the triangular interval. The radial nerve then proceeds posterolaterally in conjunction with the brachial artery deep to the long head and lateral head of the triceps brachii and superficial to the medial head of the triceps brachii along the spiral groove (or radial sulcus). Innervation of the triceps brachii occurs along this course. Emerging from the spiral groove on the lateral side of the humerus, the nerve then pierces the lateral intermuscular septum, entering the anterior compartment of the upper arm approximately 10 cm proximal to the lateral epicondyle of the humerus. It then courses between the brachialis and brachioradialis muscles, sending branches to the brachialis muscle and the brachioradialis muscle. The radial nerve passes anterior to the lateral epicondyle as it enters into the forearm, branching into superficial (sensory) and deep branches (posterior interosseous nerve).
Variations and Pathology
Although the musculocutaneous nerve primarily innervates the brachialis [52], the radial nerve sends between one and three accessory branches to the brachialis muscle in up to 86% of cases [53, 54]. The radial nerve can also send one, two, and three branches to the brachioradialis muscle 46.5%, 40%, and 14% of the time, respectively, according to a study by Latev and Dalley [55]. Care should be taken to avoid cutting these nerves during tendon-transfer procedures using the brachioradialis. Atypical signs and symptoms can occur in injuries to the proximal radial nerve in variations where it communicates with the ulnar nerve and when it provides motor innervation to the extensor carpi radialis brevis, which is normally provided by the deep branch of the radial nerve further distally. This is known to occur in up to 20% of cases [56].
Superficial Branch of the Radial Nerve
Normal Course
The superficial branch of the radial nerve, or the radial sensory nerve (RSN) , descends in the forearm underneath the brachioradialis (Fig. 17.12). In the middle-to-distal forearm, it passes between the tendons of the brachioradialis and the extensor carpi radialis longus by passing through the fascia that binds the two tendons to enter into the subcutaneous tissue of the posterior forearm. From there it divides into lateral and medial branches that go on to give sensory innervation to the dorsal aspect of the thumb and index finger and the dorsoradial aspect of the middle finger.
Variations and Pathology
Variations of the RSN have been found as it travels from underneath the brachioradialis to reach the subcutaneous tissue. Abrams et al. discovered cases where the RSN pierced the brachioradialis tendon itself as it traveled superficially, and this variation is thought to be present in 3–10% of individuals [57]. Other variations include one in which the RSN pierces a fused brachioradialis and extensor carpi radialis longus tendon [58], and another in which the RSN emerges from the fascia between a split brachioradialis tendon [59]. These variations may all predispose to compression neuropathy, otherwise known as Wartenberg’s syndrome, and may be encountered during surgical correction of this pathology.
Compression neuropathy of the radial sensory nerve produces paresthesia, pain, or numbness along with its typical distribution. Variations in the RSN can also produce more atypical signs and symptoms that can confuse the clinical picture. The RSN commonly gives varying degrees of sensory innervation to the ulnar dorsum of the hand, which can lead to a mistaken diagnosis of ulnar nerve pathology for actual RSN or radial nerve pathology [60,61,62,63,64,65]. The RSN can provide motor innervation to the extensor carpi radialis brevis in up to 32% of individuals, which is normally provided by the posterior interosseous nerve. The RSN also occasionally provides motor innervation to the brachioradialis, which is normally innervated by the radial nerve more proximally [53].
Appleton first described a case where the RSN was completely absent and the lateral antebrachial cutaneous nerve (LACN) and ulnar nerve provided sensory innervation to the typical RSN territory. Other subsequent studies have found similar findings [66,67,68]. While this variation is rare, communication between the LACN and the RSN is more common, with one study finding the overlap between these two nerves in as many as 75% of individuals [69]. These variations can lead to errors in the diagnosis of injuries to the RSN.
Deep Branch of the Radial Nerve
Normal Course
The deep branch of the radial nerve passes under the arcade of Fröhse, a fibrous arch formed from the superior part of the superficial layer of the supinator, and proceeds between the two heads of the supinator. It winds around the lateral head of the radius between the two planes of the supinator to reach the posterior forearm, after which it is known as the posterior interosseous nerve (PIN) of the forearm (Fig. 17.13). It passes over the abductor pollicis longus muscle origin and then descends along the posterior interosseous membrane, providing motor innervation to the muscles of the posterior compartment of the forearm. The PIN enters the wrist on the radial side of the fourth extensor compartment, providing sensory fibers to the dorsal wrist capsule.
Variations and Pathology
The typical order of innervation to the muscles of the forearm (proximal to distal) by the deep branch of the radial nerve/posterior interosseous nerve is as follows: extensor carpi radialis brevis (ECRB), supinator, extensor digitorum (ED), extensor carpi ulnaris (ECU), extensor digiti minimi (EDM), abductor pollicis longus (APL), and extensor indicis (EI). This order of innervation can vary. Innervation by multiple branches (e.g., supinator) and variable innervation by different branches (radial, superficial sensory, or posterior interosseous nerve innervation to the ECRB) have also been reported. Awareness of the normal and variable innervation patterns is helpful when treating pathologies like radial tunnel syndrome and posterior interosseous nerve compression syndrome [70].
The course of the PIN itself can vary. Though normally it passes between the two planes of the supinator muscle, it has been reported to pass superficial or deep to the supinator muscle instead. The terminal branch of the PIN has also been occasionally found on the ulnar side of the fourth extensor compartment, which surgeons should be aware of when harvesting the PIN as a nerve graft [71].
Muscles and Tendons
Anterior Compartment
Superficial Muscles
The superficial muscles of the anterior compartment of the forearm include the flexor carpi ulnaris, flexor carpi radialis, palmaris longus, and the pronator teres (Fig. 17.14).
Flexor Carpi Ulnaris
The flexor carpi ulnaris (FCU) has two heads, with the humeral head originating proximally from the medial epicondyle of the humerus and the ulnar head from the medial border of the olecranon and posterior border of ulna, and inserting onto the base of fifth metacarpal bone, the hook of hamate, and pisiform. The FCU functions to flex and adduct the wrist joint.
Flexor Carpi Radialis
flexor carpi radialis (FCR) originates proximally from the common flexor tendon attached to the medial epicondyle of the humerus, and inserts onto the base of second and third metacarpals, with a small slip to the trapezial tuberosity. It functions to flex and abduct at the wrist joint.
Variations
FCR is often harvested for the reconstruction of forearm and hand defects. Although rare, FCR agenesis has been reported in the literature and should be kept in mind when planning to harvest this muscle for reconstruction [72, 73].
Palmaris Longus
The palmaris longus (PL) originates from the common flexor tendon on the medial epicondyle of the humerus and inserts onto the palmar aponeurosis. The PL functions as a weak flexor of the wrist and elbow.
Variations
The PL is commonly used as a tendon graft donor because its function is redundant and its absence has no functional or aesthetic consequence. It is important to note that the PL is one of the most variable muscles in the human body. The PL is absent in 11.8% of the United States population, but absent in only 2.9% of Asian and 4.5% of African American populations. Additionally, the absence of the PL is more likely in females and in the left upper extremity [74]. In the absence of the PL, the FCU can take over by sending fibers to and strengthening the palmar aponeurosis. A reverse PL is a rare anatomic anomaly. It is essentially the opposite of a normal PL: the tendon originates proximally and the muscle belly inserts onto the palmar aponeurosis distally [75, 76]. This reversed muscle belly can also divide into multiple muscle slips, inserting onto the flexor retinaculum, the flexor carpi radialis, and the palmar carpal ligament [77]. A reversed PL can hypertrophy due to overuse, causing effort-related distal median nerve compression, or less commonly, Guyon’s syndrome [78, 79]. Both duplication and bifurcation of the PL either at the level of the muscle belly or at the tendon has also been documented. Finally, both the origin and the insertion of the PL can vary. The PL can arise from the antebrachial fascia, bicipital aponeurosis, FCR, FCU, or FDS. The PL can insert onto the antebrachial fascia, fascia of the thenar eminence, carpal bones, expansion of the FCU insertion over the wrist, FDS, FDP, or FCR [76, 80]. Surgeons need to be aware of these variations of the PL when planning to harvest the PL as a tendon graft (Fig. 17.15).
Variations of the palmaris longus . Palmaris longus variations can include (a) a palmaris longus duplication; (b) a palmaris longus with a distal muscle belly (reverse palmaris longus); (c) a palmaris longus with a central muscle belly; (d) a bifid palmaris longus; and (e) a palmaris longus with a divided tendon [76]
Pronator Teres
The pronator teres (PT) consists of two heads: the humeral head and the ulnar head. The humeral head arises from the common flexor tendon attached to the medial epicondyle of the humerus, and the ulnar head arises from the coronoid process of the ulna. The PT inserts distally onto the lateral surface of the middle-third radius.
Variations
The origin of the PT can extend proximally to Struthers’ ligament, a tendinous arch between the medial epicondyle and the supracondylar process of the humerus. This variation along with the presence of Struthers’ ligament can entrap the median nerve, causing pronator teres syndrome. Symptoms include pain, weakness, and paresthesia. Ischemic pain and embolization of the hand and fingers may result if the brachial artery becomes entrapped between the elbow joint and Struthers’ ligament [81].
Intermediate Muscle
The intermediate muscle of the anterior compartment of the forearm includes the flexor digitorum superficialis (FDS). The FDS is sometimes considered part of the superficial layer of the anterior compartment of the forearm.
Flexor Digitorum Superficialis
The flexor digitorum superficialis (FDS) consists of two heads: the humeral head and the radial head. The humeral head arises from the common flexor tendon attached to the medial epicondyle of the humerus and the coronoid process of the ulna, and the radial head arises from the diaphysis of the radius. The four FDS tendons travel through the carpal tunnel and insert onto the volar base of the middle phalanges of the second to fifth digits. The FDS functions to flex the digits at the proximal interphalangeal joint (Fig. 17.16).
Variations
FDS insertions are variable in the literature. Bernardes and Cassell both reported cases where the PL tendon replaced the FDS tendon insertion at the base of the middle phalanx of the fourth digit [82]. The FDS instead gave digastric medial head tendons to the second and fifth digits, and a lateral head tendon to the third digit [80]. The FDS tendon to the fifth digit is found to be absent in 18.5% of the patients [83]. The FDS tendons have also been reported to form muscle bellies distal to the flexor retinaculum, causing symptomatic “pseudotumors” in the palm. These were treated through debulking or transection [84,85,86,87]. Another case reported an FDS tendon penetrating the median nerve 4 cm proximal to the wrist crease, which caused numbness and tingling in the median nerve distribution [88]. Many different variations of the FDS have been described in the literature. Elliot et al. categorized these anomalies into five types: (1) FDS tendon to FDS tendon attachment, (2) FDS tendon to flexor retinaculum attachment, (3) FDS tendon with a digastric muscle, (4) FDS muscle belly extension distally, and (5) FDS anomalies in the forearm [87] (Fig. 17.17).
Variations of the flexor digitorum superficialis (FDS) . FDS variations can be categorized into five types. Type I: An anomalous muscle can arise on the FDS tendon and reinsert back onto the FDS tendon more distally. Type II: An anomalous muscle can arise from the flexor retinaculum and insert onto the FDS tendon more distally. Type III: A digastric muscle can be found in the FDS tendon in the palm. Type IV: The FDS muscle belly can extend distally as far as into the carpal tunnel. Type V: Other anomalies of the FDS can occur proximally in the forearm [87]
Deep Muscles
The deep muscles of the anterior compartment of the forearm include the flexor digitorum profundus, flexor pollicis longus, and the pronator quadratus (Fig. 17.18).
Flexor Digitorum Profundus
The flexor digitorum profundus (FDP) originates proximally on the upper anterior surface of the ulna and interosseous membrane. The FDP divides into four tendons, which travel through the carpal tunnel and insert onto the volar surface of the distal second through fifth phalanges. It functions to flex the distal interphalangeal, proximal interphalangeal, and wrist joints.
Variations
Rarely, the FDP tendons to the fourth and fifth fingers share a common tendon that bifurcates at the mid-palmar level. This common FDP tendon has a configuration that shifts more loads to the fifth finger, which is therefore at risk for spontaneous rupture when flexing against increased resistance [89, 90]. Another variation is the presence of an accessory head of the FDP, called Gantzer’s muscle, which most commonly arises from the deep portion of the flexor digitorum superficialis muscle and courses distally to insert onto the FDP (14–25%). Gantzer’s muscle can also differentially insert onto the flexor pollicis longus. Because this accessory head lies between the median nerve anteriorly and AIN posteriorly, it has been reported to cause AINS or restricted movement of the FDP [91, 92] (Fig. 17.19).
Flexor Pollicis Longus
The flexor pollicis longus (FPL) originates proximally on the anterior surface of the radius and interosseous membrane, travels through the carpal tunnel, and inserts onto the volar surface of the first distal phalanx. It functions to flex the first interphalangeal joint.
Variations
An accessory head of the FPL (also called Gantzer’s muscle ) most commonly arises from the deep portion of the flexor superficialis muscle and courses distally to insert onto the FPL in 14–25% of the global population (Fig. 17.20). Like Gantzer’s muscle inserting onto the FDP, this accessory head can also cause AINS because of its relative position [91, 92].
Pronator Quadratus
The pronator quadratus (PQ) originates proximally on the anterior surface of the distal quarter of the ulna and inserts onto the anterior surface of the distal quarter of the radius. It functions to pronate the forearm.
Anomalous Muscles
Flexor Carpi Radialis Brevis
Flexor carpi radialis brevis (FCRB) is a rare anomalous muscle of the flexor compartment, occurring in approximately 2.6–7.5% of the global population. It originates from the lower third of the radius between the origin of the flexor pollicis longus (FPL) and insertion of the PQ, runs inside the FCR sheath parallel to the FCR, and inserts variably onto the second through fourth metacarpals or radial side carpal bones [93, 94] (Fig. 17.20).
Palmaris Profundus
The palmaris profundus is a duplicated PL reported to coexist with or replace the PL. It has an incidence of 0.8% in 1600 extremities studied by Reimann et al. [76]. Whereas an accessory PL shares an origin with the PL, the palmaris profundus has a different origin than the PL, and it inserts into a palmar aponeurosis or its miniature duplicate (a duplicate palmar aponeurosis that passes dorsal to the normal palmar aponeurosis) (Fig. 17.21). The palmaris profundus has the potential to cause carpal tunnel syndrome by compressing on the median nerve that is not released with conventional surgery, but requires detachment of the duplicated tendon [6, 95, 96].
Palmaris profundus . The palmaris profundus is a duplicated palmaris longus that inserts onto the palmar aponeurosis. The palmaris profundus can originate from the proximal radius, from the FDS, or from the ulna [96]
Posterior Compartment
Superficial Muscles
The superficial muscles of the posterior compartment of the forearm include the anconeus, brachioradialis, extensor carpi radialis longus, extensor carpi radialis brevis, extensor digitorum communis, extensor digiti minimi, and the extensor carpi ulnaris (Fig. 17.22).
Anconeus
The anconeus originates proximally from the lateral epicondyle of the humerus and inserts onto the posterior and lateral olecranon of the ulna. The anconeus functions to assist the triceps brachii in extending the elbow and is considered by some to be a continuation of the triceps brachii.
Brachioradialis
The brachioradialis originates proximally on the proximal aspect of the lateral supracondylar ridge of the humerus and lateral intermuscular septum. It inserts on the distal radius, just proximal to the styloid process. Although it is part of the posterior compartment, the brachioradialis actually functions to flex the forearm at the elbow.
Extensor Carpi Radialis Longus
The extensor carpi radialis longus (ECRL) originates proximally from the distal aspect of the lateral supracondylar ridge of the humerus and lateral intermuscular septum. It inserts on the dorsal base of the second metacarpal. The ECRL functions to extend and abduct the wrist.
Extensor Carpi Radialis Brevis
The extensor carpi radialis brevis (ECRB) originates proximally from the lateral epicondyle of the humerus. It inserts on the dorsal base of the third metacarpal. The ECRB primarily functions to extend and abduct the wrist.
Extensor Digitorum Communis
The extensor digitorum communis (EDC) originates proximally from the lateral epicondyle of the humerus. It separates into four tendons in the distal forearm where they travel into the hand to pass over each MCP joint. Each tendon separates into three bands after the MCP joint. The central band inserts on the dorsal base of the middle phalanx and the two lateral bands continue distally to rejoin and insert on the dorsal base of the distal phalanx. The EDC primarily functions to extend the wrist, MCP, and PIP joints.
Variations
Numerous variations of the EDC to each of the second through fifth fingers have been described in the literature. The index finger most commonly receives one EDC tendon but has been rarely described receiving two tendons [97]. Both the long finger and ring finger commonly receive one, two, or three tendons from the EDC. Each finger has even been described as receiving four EDC tendons [97, 98]. The EDC tendon contribution to the small finger is usually as a single tendon or absent [97]. The single tendon contribution to the small finger has been described as coming from one common tendon that bifurcates to insert on the ring and little fingers, one independent tendon that inserts onto the little finger, and an intertendinous connection that extends to the small finger from the EDC to the ring finger (Fig. 17.23) [99].
Extensor digitorum communis (EDC) variations to the small finger. (a) A single EDC to the small finger; (b) a single EDC bifurcates and inserts onto the ring and the small finger; (c) an intertendinous connection extends to the small finger from the EDC to the ring finger [99]
Extensor Digiti Minimi
The extensor digiti minimi (EDM) originates proximally from the lateral epicondyle of the humerus. It inserts on the dorsal base of fifth proximal phalanx joined with the EDC tendon to the small finger when present. The EDM functions to extend the MCP and PIP joints of the small finger.
Variations
The EDM commonly has multiple tendon slips that insert variably onto the ring and little finger. In a meta-analysis of the reported number of EDM tendons by Yammine, 11.5% had one EDM tendon, 77.6% had two EDM tendons, 7% had three EDM tendons, and 0.6% had four EDM tendons [100]. The variability of the EDM and its insertions has been suggested as a contributing factor to the development of tenosynovitis around the EDM. It is important to note that the EDM tendon is frequently used for tendon transfer procedures, particularly to correct abduction deformities of the little finger. EDM tendon transfer in the absence of an EDC tendon to the small finger may result in the loss of small finger extension [101].
Extensor Carpi Ulnaris
The extensor carpi ulnaris (ECU) originates proximally from the lateral epicondyle of the humerus. It inserts on the dorsal base of the fifth metacarpal. The ECU functions to extend and adduct the wrist.
Variations
An accessory tendinous slip has been described arising from the ECU in up to 34% of upper limbs studied [102]. This accessory tendinous slip can insert onto the base, midsection, or head of the fifth metacarpal [102]. Tenosynovitis of this accessory slip has been described as the cause of wrist pain and dysfunction [102, 103].
Deep Muscles
The deep muscles of the posterior compartment of the forearm include the supinator, abductor pollicis longus, extensor pollicis longus, extensor pollicis brevis, and extensor indicis proprius (Fig. 17.24).
Supinator
The supinator consists of two muscle planes. The superficial plane originates from the lateral epicondyle and the radial collateral and annular ligaments. The deep plane originates from the supinator crest and fossa of the ulna. The supinator inserts on the lateral, anterior, and posterior surfaces of the proximal third of the radius. As its name suggests, the supinator functions to supinate the arm.
Abductor Pollicis Longus
The abductor pollicis longus (APL) originates from the interosseous membrane and adjacent posterior surfaces of the radius and ulna in the mid forearm. It inserts onto the radial, dorsal base of the first metacarpal. The APL functions to abduct the thumb at the CMC joint. Along with the extensor pollicis brevis (EPB), it forms the radial border of the anatomical snuffbox.
Variations
The APL has multiple accessory tendons in the majority of cases, with or without accessory muscle bellies, and has even been described with seven accessory tendons [104, 105]. In a cadaver study of the APL by Kuthanan and Chareonwat, 11% of all APLs had one tendon, 63% had two tendons, and 26% had three tendons [106]. These accessory tendons of the APL are thought to contribute to the development of De Quervain syndrome. Intersection syndrome is caused by the confining nature of the area where the APL and EPB intersect over the ECRL and ECRB in the forearm. The presence of accessory tendons can add additional risk to the development of this syndrome [107,108,109].
While the classic insertion of the APL tendon is onto the base of the first metacarpal, the APL tendon more commonly inserts onto the trapezium, thenar muscle, and thenar muscle fascia. These variations have been reported as the cause of trapeziometacarpal joint laxity and subluxation [107, 110].
Extensor Pollicis Longus
The extensor pollicis longus (EPL) originates from the posterior surface of the ulna and interosseous membrane in the mid forearm and inserts onto the dorsal base of the distal phalanx of the thumb. The EPL functions to extend the thumb at the MCP and IP joints. It forms the ulnar border of the anatomical snuffbox.
Variations
Duplication of the EPL, with or without a separate muscle belly, can rarely occur. Both the EPL and its duplicate have been described coursing through the third dorsal compartment, in other dorsal compartments (e.g., first, fourth), or separately through different dorsal compartments [111,112,113,114,115]. When an additional tendon is present, both tendons can rejoin in the hand to have a single insertion onto the distal phalanx or have separate and anomalous insertions. The anomalous course of the EPL and a duplicate EPL can contribute to the development of tenosynovitis and wrist pain [115, 116].
Extensor Pollicis Brevis
The extensor pollicis brevis (EPB) originates from the posterior surface of the radius and interosseous membrane in the mid forearm. It inserts onto the dorsal base of the proximal phalanx of the thumb. The EPB functions to extend and abduct the thumb at the MCP joint. Along with the APL, it forms the radial border of the anatomical snuffbox.
Variations
The EPB is absent in up to 7% of cadaver dissections in a UK study [117]. When present, it can be fused to a variable extent to the APL [117]. In addition to its typical insertion onto the proximal phalanx, the EPB has been commonly described inserting onto the distal phalanx, both the distal and proximal phalanx, and uniting with the EPL as a single insertion [118]. The EPB is thought to play a more minor role in thumb function, and is therefore used as a donor in a number of different hand procedures [118]. Knowledge of these EPB variations may help in surgical planning as well as in the identification of the EPB during surgery.
Extensor Indicis Proprius
The extensor indicis proprius (EIP) originates from the posterior surface of the ulna and interosseous membrane in the distal forearm, medial and distal to the origin of the EPL. It runs to the ulnar side to the EDC-index tendon at the metacarpal head, and inserts on to the extensor hood of the index finger. The EIP functions to extend the index finger, allowing it to move independently of the other digits.
Variations
The EIP can lie palmar or radial to the EDC-index tendon over the metacarpal head [97, 119]. The EIP tendon has important uses as a graft and can be confused for EDC-index tendon in cases of tendon transfers and transplants. The EIP can also have two or even three tendon slips, which insert variably onto the thumb, index, or long fingers (Fig. 17.25) [97, 99, 119].
Extensor indicis proprius (EIP) variations . (a) EIP lies ulnar to the EDC; (b) EDC flanked by two EIP tendons; (c) two EIP tendons lie ulnar to the EDC; (d) EIP lies ulnar to two EDC tendons; (e) EIP lies radial to the EDC; (f) EIP lies volar to the EDC [119]
Anomalous Muscles
Extensor Pollicis et Indicis
This anomalous muscle has been described arising from the ulna between a normal EIP and EPL and splitting into two slips before inserting into the extensor hood of both the thumb and index finger or to either digit alone (Fig. 17.26) [120,121,122]. Its incidence has been variably reported in less than 1% and up to 5% [123,124,125].
Extensor pollicis et indicis . The extensor pollicis et indicis arises between the extensor indicis proprius and extensor pollicis longus and splits to insert onto both the thumb and index finger or to either digit alone [120]
Extensor Digitorum Brevis Manus
The extensor digitorum brevis manus (EDBM) is an anomalous muscle that appears in about 4% of the global population [126]. It can originate from the dorsal wrist joint capsule, the distal end of the radius, the dorsal metacarpal surface, or from the dorsal radiocarpal ligament [126]. The EDBM travels within the fourth dorsal wrist compartment and usually inserts onto the extensor hood of the index or middle finger, but has also been described inserting onto the ring finger, little finger, or multiple fingers (i.e., multiple tendons) [127]. The EDBM shares a close relationship with the EIP when found together, and is thought to compensate for the EIP in its absence (Fig. 17.27) [128]. Surgeons should be aware of such cases particularly when the EDBM is the only independent index finger extensor. The EDBM has been implicated as a cause of chronic dorsal wrist pain and can be easily mistaken for a ganglion, synovial condition, or benign tumor [129].
Extensor digitorum brevis manus (EDBM) . The EDBM can be classified into three types. Type I: The EDBM can insert onto the dorsal hood of the index finger in the absence of an extensor indicis proprius (EIP). Type II: Both the EIP and EDBM can insert onto the index finger. Type III: The EDBM can insert onto the long finger while the EIP inserts onto the index finger. (Adapted from Ogura et al. [128])
Extensor Medii Proprius
The extensor medii proprius (EMP) is another anomalous muscle with a reported incidence of up to 10%. The EMP originates in the distal ulna just distal and ulnar to the EIP (Fig. 17.28). It has been described inserting onto the dorsal hood of the long finger ulnar to the EDC tendon or onto the intertendinous fascia proximal to the third metacarpophalangeal joint. The EMP lies palmar to the EDC and can be missed, particularly when it inserts proximally to the MCP joint [130].
Carpal Bones
Normal Anatomy
The wrist, or carpus, is a complex association of bones that includes the distal radius and ulna, the eight carpal bones, and the proximal metacarpals (Fig. 17.29). The eight carpal bones are grouped into two rows. The proximal row articulates with the distal radius and ulna and consists of the scaphoid, lunate, triquetrum, and pisiform. The distal row articulates with the bases of the metacarpals and consists of the trapezium, trapezoid, capitate, and hamate. The concave, palmar side of the carpus forms the carpal tunnel.
Variations
Carpal Bipartition and Fusion/Coalition
Carpal bipartitions , or divided carpals , are very rare anatomic variants. The bipartite scaphoid is the best known of the carpal bipartitions and has a prevalence of 0.13% in a study of 743 radiographs by O’Rahilly [131]. An absence of trauma and radiographic signs (e.g., absence of degenerative changes, smooth and regular edges, symmetry) can help to distinguish these bipartitions from carpal fractures.
Carpal fusions have a prevalence of roughly 0.1% in the general US population and are more commonly found in females and African Americans [131, 132]. While almost every possible combination has been described, the most common are a fusion between the lunate and triquetrum (lunotriquetral), which represents almost 90% of all carpal fusions [133]. Some patients with a lunotriquetral fusion may have a widened scapholunate joint space on radiographs. However, this is a normal variant without decreased joint stability [134].
Accessory Ossicles
Accessory ossicles, or secondary ossification centers , are normal variants of which over 20 have been described [131, 135]. They are largely congenital in origin and their prevalence is thought to range anywhere from 0.4% to 1.6% [131, 136]. Of these accessory ossicles, the more common ones include the lunula, the os styloideum, the triangulare, the epilunate, the trapezium secondarium, and the os hamuli (Fig. 17.30) [135]. It is important not to mistake these accessory ossicles for fractures or loose bodies.
The os styloideum (carpal boss) is located over the dorsum of the base of the second or third metacarpal and is sometimes fused with the metacarpal base over which it lies. The os styloideum is important because it can undergo osteoarthritic changes or affect an overlying ganglion, limiting wrist motion and causing wrist pain [137, 138].
Lunate Variation
The lunate can be categorized anatomically into two types. The type I lunate has a single facet that articulates with the midcarpal joint. The type II lunate has an additional medial facet that articulates with the hamate (Fig. 17.31). The prevalence of the type II lunate ranges from 65.5% to 73% [139]. Viegas, in a cadaveric dissection of 165 wrists, found that about 44% of type II lunates demonstrated significant cartilage erosion at the proximal pole of the hamate, while only 0–2% of type I lunates exhibited such pathology [140]. Since this erosion is not easily identifiable on radiographs, type II lunates may be a cause of unidentified ulnar-sided wrist pain.
References
Standring S. Gray’s anatomy: the anatomical basis of clinical practice. 40th ed. Spain: Churchill-Livingstone; 2008.
Lanz U. Anatomical variations of the median nerve in the carpal tunnel. J Hand Surg Am. 1977;2(1):44–53.
Mitchell R, Chesney A, Seal S, McKnight L, Thoma A. Anatomical variations of the carpal tunnel structures. Can J Plast Surg. 2009;17(3):e3–7.
Amadio PC. Bifid median nerve with a double compartment within the transverse carpal canal. J Hand Surg Am. 1987;12(3):366–8.
Fernandez-Garcia S, Pi-Folguera J, Estallo-Matino F. Bifid median nerve compression due to a musculotendinous anomaly of FDS to the middle finger. J Hand Surg Br. 1994;19(5):616–7.
Jones DP. Bilateral palmaris profundus in association with bifid median nerve as a cause of failed carpal tunnel release. J Hand Surg Am. 2006;31(5):741–3.
Schultz RJ, Endler PM, Huddleston HD. Anomalous median nerve and an anomalous muscle belly of the first lumbrical associated with carpal-tunnel syndrome. J Bone Joint Surg Am. 1973;55(8):1744–6.
Szabo RM, Pettey J. Bilateral median nerve bifurcation with an accessory compartment within the carpal tunnel. J Hand Surg Br. 1994;19(1):22–3.
Takami H, Takahashi S, Ando M. Bipartite median nerve with a double compartment within the transverse carpal canal. Arch Orthop Trauma Surg. 2001;121(4):230–1.
Beris AE, Lykissas MG, Kontogeorgakos VA, Vekris MD, Korompilias AV. Anatomic variations of the median nerve in carpal tunnel release. Clin Anat. 2008;21(6):514–8.
Bhanu PS, Sankar KD. Bilateral absence of musculocutaneous nerve with unusual branching pattern of lateral cord and median nerve of brachial plexus. Anat Cell Biol. 2012;45(3):207–10.
Sarkar A, Saha A. Bilateral absence of musculocutaneous nerve: a case report. J Clin Diagn Res. 2014;8(9):AD06–7.
Nakatani T, Tanaka S, Mizukami S. Absence of the musculocutaneous nerve with innervation of coracobrachialis, biceps brachii, brachialis and the lateral border of the forearm by branches from the lateral cord of the brachial plexus. J Anat. 1997;191(Pt 3):459–60.
Butz JJ, Shiwlochan DG, Brown KC, Prasad AM, Murlimanju BV, Viswanath S. Bilateral variations of brachial plexus involving the median nerve and lateral cord: an anatomical case study with clinical implications. Australas Med J. 2014;7(5):227–31.
Leibovic SJ, Hastings H 2nd. Martin-Gruber revisited. J Hand Surg Am. 1992;17(1):47–53.
Jones C, Beredjiklian P, Matzon JL, Kim N, Lutsky K. Incidence of an anomalous course of the palmar cutaneous branch of the median nerve during volar plate fixation of distal radius fractures. J Hand Surg Am. 2016;41(8):841–4.
Dowdy PA, Richards RS, McFarlane RM. The palmar cutaneous branch of the median nerve and the palmaris longus tendon: a cadaveric study. J Hand Surg Am. 1994;19(2):199–202.
McCann PA, Clarke D, Amirfeyz R, Bhatia R. The cadaveric anatomy of the distal radius: implications for the use of volar plates. Ann R Coll Surg Engl. 2012;94(2):116–20.
Cheung JW, Shyu JF, Teng CC, Chen TH, Su CH, Shyr YM, et al. The anatomical variations of the palmar cutaneous branch of the median nerve in Chinese adults. J Chin Med Assoc. 2004;67(1):27–31.
Taleisnik J. The palmar cutaneous branch of the median nerve and the approach to the carpal tunnel. An anatomical study. J Bone Joint Surg Am. 1973;55(6):1212–7.
Hobbs RA, Magnussen PA, Tonkin MA. Palmar cutaneous branch of the median nerve. J Hand Surg Am. 1990;15(1):38–43.
Engber WD, Gmeiner JG. Palmar cutaneous branch of the ulnar nerve. J Hand Surg Am. 1980;5(1):26–9.
Oh CS, Won HS, Lee KS, Chung IH, Kim SM. Anatomic variation of the innervation of the flexor digitorum profundus muscle and its clinical implications. Muscle Nerve. 2009;39(4):498–502.
Hill NA, Howard FM, Huffer BR. The incomplete anterior interosseous nerve syndrome. J Hand Surg Am. 1985;10(1):4–16.
Wertsch JJ. AAEM case report #25: anterior interosseous nerve syndrome. Muscle Nerve. 1992;15(9):977–83.
Mazurek MT, Shin AY. Upper extremity peripheral nerve anatomy: current concepts and applications. Clin Orthop Relat Res. 2001;383:7–20.
Khoo D, Carmichael SW, Spinner RJ. Ulnar nerve anatomy and compression. Orthop Clin North Am. 1996;27(2):317–38.
Sunderland S, Hughes ES. Metrical and non-metrical features of the muscular branches of the ulnar nerve. J Comp Neurol. 1946;85:113–25.
Gonzalez MH, Lotfi P, Bendre A, Mandelbroyt Y, Lieska N. The ulnar nerve at the elbow and its local branching: an anatomic study. J Hand Surg Br. 2001;26(2):142–4.
Lowe JB 3rd, Maggi SP, Mackinnon SE. The position of crossing branches of the medial antebrachial cutaneous nerve during cubital tunnel surgery in humans. Plast Reconstr Surg. 2004;114(3):692–6.
Chow JC, Papachristos AA, Ojeda A. An aberrant anatomic variation along the course of the ulnar nerve above the elbow with coexistent cubital tunnel syndrome. Clin Anat. 2006;19(7):661–4.
Tindall A, Patel M, Frost A, Parkin I, Shetty A, Compson J. The anatomy of the dorsal cutaneous branch of the ulnar nerve – a safe zone for positioning of the 6R portal in wrist arthroscopy. J Hand Surg Br. 2006;31(2):203–5.
Lindsey JT, Watumull D. Anatomic study of the ulnar nerve and related vascular anatomy at Guyon’s canal: a practical classification system. J Hand Surg Am. 1996;21(4):626–33.
Murata K, Tamai M, Gupta A. Anatomic study of variations of hypothenar muscles and arborization patterns of the ulnar nerve in the hand. J Hand Surg Am. 2004;29(3):500–9.
Bozkurt MC, Tağil SM, Ozçakar L, Ersoy M, Tekdemr I. Anatomical variations as potential risk factors for ulnar tunnel syndrome: a cadaveric study. Clin Anat. 2005;18(4):274–80.
Kaplan EB. Variation of the ulnar nerve at the wrist. Bull Hosp Joint Dis. 1963;24:85–8.
Bozkurt MC, Cezayirli E, Tagil SM. An unusual termination of the ulnar nerve in the palm. Ann Anat. 2002;184(3):271–3.
Windisch G. Unusual vascularization and nerve supply of the fifth finger. Ann Anat. 2006;188(2):171–5.
McCarthy RE, Nalebuff EA. Anomalous volar branch of the dorsal cutaneous ulnar nerve: a case report. J Hand Surg Am. 1980;5(1):19–20.
Konig PS, Hage JJ, Bloem JJ, Prose LP. Variations of the ulnar nerve and ulnar artery in Guyon’s canal: a cadaveric study. J Hand Surg Am. 1994;19(4):617–22.
Bozkurt MC, Tagil SM, Ersoy M, Tekdemir I. Muscle variations and abnormal branching and course of the ulnar nerve in the forearm and hand. Clin Anat. 2004;17(1):64–6.
Martin CH, Seiler JG 3rd, Lesesne JS. The cutaneous innervation of the palm: an anatomic study of the ulnar and median nerves. J Hand Surg Am. 1996;21(4):634–8.
Dogan NU, Uysal II, Seker M. The communications between the ulnar and median nerves in upper limb. Neuroanatomy. 2009;8(1):15–9.
May JW Jr, Rosen H. Division of the sensory ramus communicans between the ulnar and median nerves: a complication following carpal tunnel release. A case report. J Bone Joint Surg Am. 1981;63(5):836–8.
Kimura I, Ayyar DR, Lippmann SM. Electrophysiological verification of the ulnar to median nerve communications in the hand and forearm. Tohoku J Exp Med. 1983;141(3):269–74.
Harness D, Sekeles E. The double anastomotic innervation of thenar muscles. J Anat. 1971;109(Pt 3):461–6.
Kim BJ, Date ES, Lee SH, Lau EW, Park MK. Unilateral all ulnar hand including sensory without forearm communication. Am J Phys Med Rehabil. 2004;83(7):569–73.
Ganes T. Complete ulnar innervation of the thenar muscles combined with normal sensory fibres in a subject with no peripheral nerve lesion. Electromyogr Clin Neurophysiol. 1992;32(10–11):559–63.
Dumitru D, Walsh NE, Weber CF. Electrophysiologic study of the Riche-Cannieu anomaly. Electromyogr Clin Neurophysiol. 1988;28(1):27–31.
Gutmann L. AAEM minimonograph #2: important anomalous innervations of the extremities. Muscle Nerve. 1993;16(4):339–47.
Refaeian M, King JC, Dumitru D, Cuetter AC. Carpal tunnel syndrome and the Riche-Cannieu anastomosis: electrophysiologic findings. Electromyogr Clin Neurophysiol. 2001;41(6):377–82.
Moore KL, Dalley AF, Agur AM. Clinically oriented anatomy. 7th ed. Baltimore: Lippincott Williams & Wilkins; 2014.
Mackinnon SE, Novak CB. Compression neuropathies. In: Wolfe SW, Pederson WC, Hotchkiss RN, Kozin SH, Cohen MS, editors. Green’s operative hand surgery. Philadelphia: Elsevier Churchill Livingstone; 2011. p. 977–1014.
Mahakkanukrauh P, Somsarp V. Dual innervation of the brachialis muscle. Clin Anat. 2002;15(3):206–9.
Latev MD, Dalley AF 2nd. Nerve supply of the brachioradialis muscle: surgically relevant variations of the extramuscular branches of the radial nerve. Clin Anat. 2005;18(7):488–92.
Khullar M, Kalsey G, Laxmi V, Khullar S. Variations in the nerve supply to the extensor carpi radialis brevis. J Clin Diagn Res. 2012;6(1):13–6.
Abrams RA, Brown RA, Botte MJ. The superficial branch of the radial nerve: an anatomic study with surgical implications. J Hand Surg Am. 1992;17(6):1037–41.
Linell EA. The distribution of nerves in the upper limb, with reference to variabilities and their clinical significance. J Anat. 1921;55(Pt 2–3):79–112.
Ehrlich W, Dellon AL, Mackinnon SE. Classical article: cheiralgia paresthetica (entrapment of the radial nerve). A translation in condensed form of Robert Wartenberg’s original article published in 1932. J Hand Surg Am. 1986;11(2):196–9.
Auerbach DM, Collins ED, Kunkle KL, Monsanto EH. The radial sensory nerve. An anatomic study. Clin Orthop Relat Res. 1994;308:241–9.
Bas H, Kleinert JM. Anatomic variations in sensory innervation of the hand and digits. J Hand Surg Am. 1999;24(6):1171–84.
Peterson AR, Giuliani MJ, McHugh M, Shipe CC. Variations in dorsomedial hand innervation. Electrodiagnostic implications. Arch Neurol. 1992;49(8):870–3.
McCluskey LF. Anomalous superficial radial sensory innervation of the ulnar dorsum of the hand: a cause of “paradoxical” preservation of ulnar sensory function. Muscle Nerve. 1996;19(7):923–5.
Kuruvilla A, Laaksonen S, Falck B. Anomalous superficial radial nerve: a patient with probable autosomal dominant inheritance of the anomaly. Muscle Nerve. 2002;26(5):716–9.
Leis AA, Wells KJ. Radial nerve cutaneous innervation to the ulnar dorsum of the hand. Clin Neurophysiol. 2008;119(3):662–6.
Appleton AB. A case of abnormal distribution of the n. Musculo-cutaneus, with complete absence of the ramus cutaneus n. radialis. J Anat Physiol. 1911;46(Pt 1):89–94.
Huanmanop T, Agthong S, Luengchawapong K, Sasiwongpakdee T, Burapasomboon P, Chentanez V. Anatomic characteristics and surgical implications of the superficial radial nerve. J Med Assoc Thail. 2007;90(7):1423–9.
Stopford JS. The variation in distribution of the cutaneous nerves of the hand and digits. J Anat. 1918;53(Pt 1):14–25.
Mackinnon SE, Dellon AL. The overlap pattern of the lateral antebrachial cutaneous nerve and the superficial branch of the radial nerve. J Hand Surg Am. 1985;10(4):522–6.
Branovacki G, Hanson M, Cash R, Gonzalez M. The innervation pattern of the radial nerve at the elbow and in the forearm. J Hand Surg Br. 1998;23(2):167–9.
Missankov AA, Sehgal AK, Mennen U. Variations of the posterior interosseous nerve. J Hand Surg Br. 2000;25(3):281–2.
Innocenti M, Tani M, Carulli C, Ghezzi S, Raspanti A, Menichini G. Radial forearm flap plus flexor carpi radialis tendon in Achilles tendon reconstruction: surgical technique, functional results, and gait analysis. Microsurgery. 2015;35(8):608–14.
Rumball KM, Tonkin MA. Absence of flexor carpi radialis. J Hand Surg Br. 1996;21(6):778.
Ioannis D, Anastasios K, Konstantinos N, Lazaros K, Georgios N. Palmaris longus muscle’s prevalence in different nations and interesting anatomical variations: review of the literature. J Clin Med Res. 2015;7(11):825–30.
Murabit A, Gnarra M, Mohamed A. Reversed palmaris longus muscle: anatomical variant – case report and literature review. Can J Plast Surg. 2013;21(1):55–6.
Reimann AF, Daseler EH, Anson BJ, Beaton LE. The palmaris longus muscle and tendon. A study of 1600 extremities. Anat Rec. 1944;89(4):495–505.
Acikel C, Ulkur E, Karagoz H, Celikoz B. Effort-related compression of median and ulnar nerves as a result of reversed three-headed and hypertrophied palmaris longus muscle with extension of Guyon’s canal. Scand J Plast Reconstr Surg Hand Surg. 2007;41(1):45–7.
Bencteux P, Simonet J, el Ayoubi L, Renard M, Attignon I, Dacher JN, et al. Symptomatic palmaris longus muscle variation with MRI and surgical correlation: report of a single case. Surg Radiol Anat. 2001;23(4):273–5.
Depuydt KH, Schuurman AH, Kon M. Reversed palmaris longus muscle causing effort-related median nerve compression. J Hand Surg Br. 1998;23(1):117–9.
Bernardes A, Melo C, Pinheiro S. A combined variation of palmaris longus and flexor digitorum superficialis: case report and review of literature. Morphologie. 2016;100(331):245–9.
Jelev L, Georgiev GP. Unusual high-origin of the pronator teres muscle from a Struthers’ ligament coexisting with a variation of the musculocutaneous nerve. Romanian J Morphol Embryol. 2009;50(3):497–9.
Cassell MD, Bergman RA. Palmaris longus muscle substituting for the ring finger slip of flexor digitorum superficialis. Anat Anz. 1990;171(3):201–4.
Guler F, Kose O, Turan A, Baz AB, Akalin S. The prevalence of functional absence of flexor digitorum superficialis to the little finger: a study in a Turkish population. J Plast Surg Hand Surg. 2013;47(3):224–7.
Coenen L, Biltjes I. Pseudotumor of the palm due to an anomalous flexor digitorum superficialis muscle belly. J Hand Surg Am. 1991;16(6):1046–51.
Sanger JR, Krasniak CL, Matloub HS, Yousif NJ, Kneeland JB. Diagnosis of an anomalous superficialis muscle in the palm by magnetic resonance imaging. J Hand Surg Am. 1991;16(1):98–101.
Still JM Jr, Kleinert HE. Anomalous muscles and nerve entrapment in the wrist and hand. Plast Reconstr Surg. 1973;52(4):394–400.
Elliot D, Khandwala AR, Kulkarni M. Anomalies of the flexor digitorum superficialis muscle. J Hand Surg Br. 1999;24(5):570–4.
Skie M, Ciocanel D. Anomaly of flexor digitorum superficialis penetrating through the median nerve: case report. J Hand Surg Am. 2010;25(1):27–9.
Davis C, Armstrong J. Spontaneous flexor tendon rupture in the palm: the role of a variation of tendon anatomy. J Hand Surg Am. 2003;28(1):149–52.
Masaki F, Isao T, Aya Y, Ryuuji I, Yohjiroh M. Spontaneous flexor tendon rupture of the flexor digitorum profundus secondary to an anatomic variant. J Hand Surg Am. 2007;32(8):1195–9.
Jones M, Abrahams PH, Sañudo JR, Campillo M. Incidence and morphology of accessory heads of flexor pollicis longus and flexor digitorum profundus (Gantzer’s muscles). J Anat. 1997;191(Pt 3):451–5.
Pai MM, Nayak SR, Krishnamurthy A, Vadgaonkar R, Prabhu LV, Ranade AV, et al. The accessory heads of flexor pollicis longus and flexor digitorum profundus: incidence and morphology. Clin Anat. 2008;21(3):252–8.
Lee YM, Song SW, Sur YJ, Ahn CY. Flexor carpi radialis brevis: an unusual anomalous muscle of the wrist. Clin Orthop Surg. 2014;6(3):361–4.
Mantovani G, Lino W Jr, Fukushima WY, Cho AB, Aita MA. Anomalous presentation of flexor carpi radialis brevis: a report of six cases. J Hand Surg Eur Vol. 2010;35(3):234–5.
Mathew AJ, Sukumaran TT, Joseph S. Versatile but temperamental: a morphological study of palmaris longus in the cadaver. J Clin Diagn Res. 2015;9(2):AC01–3.
Pirola E, Hébert-Blouin MN, Amador N, Amrami KK, Spinner RJ. Palmaris profundus: one name, several subtypes, and a shared potential for nerve compression. Clin Anat. 2009;22(6):643–8.
von Schroeder HP, Botte MJ. Anatomy of the extensor tendons of the fingers: variations and multiplicity. J Hand Surg Am. 1995;20(1):27–34.
Zilber S, Oberlin C. Anatomical variations of the extensor tendons to the fingers over the dorsum of the hand: a study of 50 hands and a review of the literature. Plast Reconstr Surg. 2004;113(1):214–21.
el-Badawi MG, Butt MM, al-Zuhair AG, Fadel RA. Extensor tendons of the fingers: arrangement and variations–II. Clin Anat. 1995;8(6):391–8.
Yammine K. The prevalence of the extensor digiti minimi tendon of the hand and its variants in humans: a systematic review and meta-analysis. Anat Sci Int. 2015;90(1):40–6.
Hirai Y, Yoshida K, Yamanaka K, Inoue A, Yamaki K, Yoshizuka M. An anatomic study of the extensor tendons of the human hand. J Hand Surg Am. 2001;26(6):1009–15.
Nakashima T. An accessory extensor digiti minimi arising from extensor carpi ulnaris. J Anat. 1993;182(Pt 1):109–12.
Barfred T, Adamsen S. Duplication of the extensor carpi ulnaris tendon. J Hand Surg Am. 1986;11(3):423–5.
Baba MA. The accessory tendon of the abductor pollicis longus muscle. Anat Rec. 1954;119(4):541–7.
Shiraishi N, Matsumura G. Anatomical variations of the extensor pollicis brevis tendon and abductor pollicis longus tendon–relation to tenosynovectomy. Okajimas Folia Anat Jpn. 2005;82(1):25–9.
Kulthanan T, Chareonwat B. Variations in abductor pollicis longus and extensor pollicis brevis tendons in the Quervain syndrome: a surgical and anatomical study. Scand J Plast Reconstr Surg Hand Surg. 2007;41(1):36–8.
Fabrizio PA, Clemente FR. A variation in the organization of abductor pollicis longus. Clin Anat. 1996;9(6):371–5.
Stein AH Jr. Variations of the tendons of insertion of the abductor pollicis longus and the extensor pollicis brevis. Anat Rec. 1951;110(1):49–55.
Allison DM. Pathologic anatomy of the forearm: intersection syndrome. J Hand Surg Am. 1986;11(6):913–4.
Martinez R, Omer GE Jr. Bilateral subluxation of the base of the thumb secondary to an unusual abductor pollicis longus insertion: a case report. J Hand Surg. 1985;10(3):396–9.
Bryce TH. Myology. In: Quain’s elements of anatomy, vol. 4, part II. London: Longmans, Green, and Co; 1923. p. 153–5.
Chiu DT. Supernumerary extensor tendon to the thumb: a report on a rare anatomic variation. Plast Reconstr Surg. 1981;68(6):937–9.
Cohen BE, Haber JL. Supernumerary extensor tendon to the thumb: a case report. Ann Plast Surg. 1996;36(1):105–7.
De Greef I, De Smet L. Accessory extensor pollicis longus: a case report. Eur J Plast Surg. 2006;28(8):532–3.
Rubin G, Wolovelsky A, Rinott M, Rozen N. Anomalous course of the extensor pollicis longus: clinical relevance. Ann Plast Surg. 2011;67(5):489–92.
Beatty JD, Remedios D, McCullough CJ. An accessory extensor tendon of the thumb as a cause of dorsal wrist pain. J Hand Surg Br. 2000;25(1):110–1.
Dawson S, Barton N. Anatomical variations of the extensor pollicis brevis. J Hand Surg Br. 1986;11(3):378–81.
Kulshreshtha R, Patel S, Arya AP, Hall S, Compson JP. Variations of the extensor pollicis brevis tendon and its insertion: a study of 44 cadaveric hands. J Hand Surg Eur Vol. 2007;32(5):550–3.
Gonzalez MH, Weinzweig N, Kay T, Grindel S. Anatomy of the extensor tendons to the index finger. J Hand Surg Am. 1996;21(6):988–91.
Culver JE Jr. Extensor pollicis and indicis communis tendon: a rare anatomic variation revisited. J Hand Surg Am. 1980;5(6):548–9.
Kaplan EB, Nathan P. Accessory extensor pollicis longus. Bull Hosp Joint Dis. 1969;30(2):203–7.
Macalister A. Additional observations on muscular anomalies in human anatomy (third series) with a catalogue of the principal muscular variations hitherto published. Trans R Ir Acad Sci. 1875;25:1–34.
Cauldwell EW, Anson BJ, Wright RR. The extensor indicis proprius muscle-a study of 263 consecutive specimens. Q Bull Northwest Univ Med Sch. 1943;17(4):267–79.
Wood J. On some variations in human myology. Proc R Soc Lond. 1863;13:299–303.
Wagenseil F. Untersuchungen über die muskulatur der chinesen. Z Morphol Anthropol. 1936;36(H. 1):39–150.
Yammine K. The prevalence of extensor digitorum brevis manus and its variants in humans: a systematic review and meta-analysis. Surg Radiol Anat. 2015;37(1):3–9.
Rodriguez-Niedenführ M, Vázquez T, Golanó P, Parkin I, Sañudo JR. Extensor digitorum brevis manus: anatomical, radiological and clinical relevance. A review. Clin Anat. 2002;15(4):286–92.
Ogura T, Inoue H, Tanabe G. Anatomic and clinical studies of the extensor digitorum brevis manus. J Hand Surg Am. 1987;12(1):100–7.
Nakano M, Watanabe Y, Masutani M. A case of extensor digitorum brevis manus. Dermatol Online J. 2003;9(5):21.
Klena JC, Riehl JT, Beck JD. Anomalous extensor tendons to the long finger: a cadaveric study of incidence. J Hand Surg Am. 2012;37(5):938–41.
O’Rahilly R. A survey of carpal and tarsal anomalies. J Bone Joint Surg Am. 1953;35–A(3):626–42.
Pfirrmann CW, Zanetti M. Variants, pitfalls and asymptomatic findings in wrist and hand imaging. Eur J Radiol. 2005;56(3):286–95.
Delaney TJ, Eswar S. Carpal coalitions. J Hand Surg Am. 1992;17(1):28–31.
Metz VM, Schimmerl SM, Gilula LA, Viegas SF, Saffar P. Wide scapholunate joint space in lunotriquetral coalition: a normal variant? Radiology. 1993;188(2):557–9.
Timins ME. Osseous anatomic variants of the wrist: findings on MR imaging. AJR Am J Roentgenol. 1999;173(2):339–44.
Bogart FB. Variations of the bones of the wrist. Am J Roentgenol. 1932;28:638–46.
Conway WF, Destouet JM, Gilula LA, Bellinghausen HW, Weeks PM. The carpal boss: an overview of radiographic evaluation. Radiology. 1985;156(1):29–31.
Resnick D. Bone and joint imaging. Philadelphia: Saunders; 1989. p. 1073.
Viegas SF. Advances in the skeletal anatomy of the wrist. Hand Clin. 2001;17(1):1–11.
Viegas SF, Wagner K, Patterson R, Peterson P. Medial (hamate) facet of the lunate. J Hand Surg Am. 1990;15(4):564–71.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kim, S., Xu, X., Clune, J.E., Narayan, D. (2021). Upper Extremity Variations. In: Narayan, D., Kapadia, S.E., Kodumudi, G., Vadivelu, N. (eds) Surgical and Perioperative Management of Patients with Anatomic Anomalies. Springer, Cham. https://doi.org/10.1007/978-3-030-55660-0_17
Download citation
DOI: https://doi.org/10.1007/978-3-030-55660-0_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-55658-7
Online ISBN: 978-3-030-55660-0
eBook Packages: MedicineMedicine (R0)