Abstract
Neotropical social wasps have a set of chimeric characteristics that make them unique and difficult to fit into current theories on the evolution of social behavior (Noll and Wenzel, Biol J Linn Soc 93: 509–22, 2008). Among them, the presence of more than one functional queen (polygyny), absence of strong morphological differentiation, and flexibility between casts result in relaxation of ovarian control and breeding opportunity for workers (West-Eberhard, Science 200: 441–3, 1978; Noll, Sociobiology 60: 347–54, 2013). Adding to these particularities, the Polistinae wasps, especially those belonging to the Epiponini, reached a great radiation and evolutionary success in the Neotropical region due to the unique mode of social organization: colony foundation by a swarm (Jeanne, The swarm-founding Polistinae. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca, pp 191–231, 1991), where several or many reproductive females (queens) tolerate each other laying eggs. This chapter deals in general with aspects of the complex caste delimitation, the division of labor in the colony, the recruitment and foraging, and the lack of colony in social wasps, with emphasis on swarm founders.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Castes
The origin of the morphological variation that establishes the social role (caste) is one of the definitive elements of the most sophisticated insect societies (Noll and Wenzel 2008). The study of castes differences in Epiponini species in Brazil began with the works of Richards (1978). Among Brazilian authors, studies of Shima et al. (1994, 1996a, b, 1998, 2000) were the pioneers. Understanding the mechanisms that lead to this differentiation is therefore of great importance for understanding the evolution of eusociality (Jeanne and Suryanarayanan 2011). The degree of distinction between breeding and worker caste varieties is often used as part of the definition of the degree of sociality (Noll et al. 2004), with highly social species presenting a complex division of labor, caste morphological differentiation, and a large number of individuals (Bourke 1999).
In Vespidae, this degree of differentiation ranges from no difference in the subfamily Stenogastrinae to large differences in size and morphology in Vespinae (Jeanne and Suryanarayanan 2011). In species presenting distinct morphological castes, the developmental difference between the future queens and workers should begin in the larval stage (Jeanne and Suryanarayanan 2011). In the independent-founding species of Polistinae, although the castes are morphologically identical, they present physiological differences that also should have origin in the larval stage (O’Donnell 1998a). This fact is supported by evidence that differences in mRNA expression and levels of hexameric storage proteins are already evident in the fifth and last larval instar in Polistes metricus Say (Jeanne and Suryanarayanan 2011; Hunt et al. 2007; Toth et al. 2007). In the case of Epiponini species, although described as highly social (Noll et al. 2004), the distinction between caste is not very evident and often difficult to identify (Richards and Richards 1951; Richards 1978, cited as Polybiini, junior synonym of Epiponini, see Carpenter 1993, 1997). According to Jeanne (2003), the complexity in this tribe is more related to the number of behaviors exhibited by the workers than to the presence of morphological differences.
There is no evidence of a genetic basis for caste differentiation in wasps (Jeanne and Suryanarayanan 2011). Differences in size among castes appeared independently in several taxa of this tribe, and caste evolved in different ways in the various lineages of Epiponini (Noll et al. 2004). A phylogenetic interpretation showed several distinct syndromes , representing a more complex scenario than previously thought (Noll et al. 2004; Noll and Wenzel 2008), challenging the defined standards. The four syndromes are (1) absence of caste differentiation (no differences in size or shape associated with reproduction); (2) physiological castes (absence of morphometric differences between castes, but females can be separated by degree of ovarian development); (3) larger queens, but with the same shape; and (4) queens who are different in size and shape from the workers (Noll et al. 2004; Chavarría 2013; Fig. 6.1 and Table 6.1).
The four syndromes related to the determination of castes in Polistinae. In (a, b) castes do not present morphological differences in size or shape. However, in (b) females can be separated by the degree of development of the ovaries (physiological caste). In (c) queens are larger in size, but there is no difference in the shape, and in (d) queens differ in size and shape from workers. The crowns identify the reproductive caste (queens)
As expected for highly social insects, pre-imaginal determination, given by nutritional differences during larval development (Hunt et al. 1996; Sakagami et al. 1996) or changes in the longitudinal axis of the body due to different programs in developmental parameters in the pre-adult stage (Jeanne et al. 1995), has been reported for the genera Agelaia (Fig. 6.2), Apoica, Chartergus, Polybia, Protopolybia, and Pseudopolybia (Noll et al. 2004). On the other hand, breeding females in Chartergellus, Metapolybia, Parachartergus, and Synoeca are morphologically similar to non-breeding and present no physiological discontinuities. In these cases, castes are flexible, and the determination is imaginal, given by the disputes between adult females instead by larval manipulation (West-Eberhard 1981; Mateus et al. 2004; Noll and Wenzel 2008; Chavarría 2013). Theoretically, in species with imaginal determination, all females when emerging could develop the ovaries and reach the status of queen, a condition that could lead to conflicts among the individuals of the colony, since the workers could also lay eggs (Hart and Ratnieks 2005).
In fact, in many colonies, workers with functional ovaries (named intermediates by Richards and Richards 1951) have been identified. The role of intermediate is debatable, being considered as producers of trophic eggs, males (Richards 1971), or even young queens (Forsyth 1978; West-Eberhard 1978; Gastreich et al. 1993). The level of ovarian development is inversely proportional to the number of queens, that is, when a few females are present, the intermediates present more developed ovaries (Fig. 6.3) and vice versa (Richards 1971; West-Eberhard 1978). Nevertheless, intermediates are absent in many other taxa (revised in Noll et al. 2004). Anyway, the presence of this type of female is part of a complex scenario related to the evolution of castes (Noll 2013).
The origin of queens that are highly tolerant of each other brings some consequences. The first is that many females could aspire for a chance of reproduction, which would lead to the totipotency of the caste (Strassmann et al. 2002). As a consequence, it would be expected to find a generalized ovarian development in members of polygynic societies, and, in fact, in several Epiponini species, workers who lay eggs are found (Noll et al. 2004). Several basal genera of Epiponini fit into the proposed scenario for polygynic groups (West-Eberhard 1978, 1981), that is, caste flexibility due to the absence of morphological differences between them and ovarian development in all females. This condition is found mainly in Angiopolybia, Pseudopolybia, Parachartergus, Chartergellus, and Leipomelles (Noll et al. 2004). Thus, the distribution of this characteristic (presence of workers laying eggs) actually constitutes a plesiomorphic rather than an apomorphic state (Noll 2013).
Due to the absence of morphologically distinct castes, the social way of life in Epiponini has already been considered as little complex. However, Jeanne (2003) suggested the presumption of self-organization rather than the presence of distinct morphologies as a characteristic that would define complex societies. In this case, the presence of self-organization would remove the need for evolution in two distinct forms in Epiponini (Noll 2013). Indeed, as suggested by West-Eberhard (2003), the origin of morphologically distinct castes is much more a condition for colony stability and defense than indicative of social complexity. From this perspective, the complex Epiponini societies may have undergone a phase of totipotent and monomorphic females for several morphologically distinct queen systems in different lineages. Such a theory proved to be perfectly congruent when optimized in a cladogram of Epiponini genera (Noll et al. 2004).
Reproductive control in social wasps can be done in two ways: through physical aggression (primitively eusocial wasps) or through chemical control. Within the colonies of primitively eusocial wasps (Polistes, Mischocyttarus, Belonogaster, Parapolybia, and Ropalidia), there is a hierarchy of dominance, where the female with greater ovarian development is solely in charge of egg laying (Fletcher and Ross 1985). Because any female can lay eggs, the queen status is maintained within the colony through physical aggression and oophagy. In Polistes , dominance interactions are more frequent than in Mischocyttarus (Spradbery 1991), and, in Ropalidia there is little or no direct interaction, with this genus presenting the least dominance repertoire among Polistinae wasps (Spradbery 1991; Kardile and Gadagkar 2002; Bhadra et al. 2010). After establishment of dominance, a reduction in the levels of aggression is observed (West-Eberhard 1986; Hughes et al. 1987), suggesting that the reproductive dominance can also be maintained by chemical signals (West-Eberhard 1986; Dapporto et al. 2007; Bhadra et al. 2010), possibly by mixtures of hydrocarbons secreted by the Van der Vecht gland (Dapporto et al. 2007). The factors of endocrine control and oogenesis are not yet clear, however. Dominance behavior alone seems to be insufficient to maintain reproductive control (Röseler 1991). In the absence of the original queen, one of the female co-founders of the nest or the female presenting the highest ovarian development may assume a role in reproduction (Spradbery 1991). The queen monopolizes the production of eggs but can also forage occasionally and starts the construction of new cells (Chavarría 2013).
For Epiponini, physical aggression was observed only during the phase of establishment and selection of queens (Mateus 2005); however, physical aggression on the part of the queen against the workers, to maintain their status within the colony, is not common. For this reason, it is believed that reproductive control is also made through chemical signaling or hormonal control (Naumann 1970; Forsyth 1978; Fletcher and Ross 1985; West-Eberhard 1989a; Spradbery 1991), although no study has proven this phenomenon. In addition to chemical signals, queens use ritualized postures to communicate their status within the colony (West-Eberhard 1978; Spradbery 1991). The queens, in Epiponini, do little more than lay eggs and perform no other task in the colony (Herman et al. 2000).
If the queens in Epiponini do not exercise a strong reproductive control, the workers do. The workers test and remove queens from the colony (West-Eberhard 1978, 1981; Herman et al. 2000; Platt et al. 2004) and favor the production of queens in colonies where there is a low number of queens and the production of males in colonies with high number of queens (Queller et al. 1993). Moreover, workers favor production of males by queens because they are more genetically related to the “sons” of the queens than to the “sons” of other workers (Hastings et al. 1998), which seems good to maintain the “workers’ interest” (Strassmann et al. 1997, 1998). This type of control is probably done in species with imaginal determination, although other control mechanisms may be related (Chavarría 2013). Studies in colonies of Protopolybia sedula (de Saussure) (Naumann 1970, still as P. pumila in his works), Metapolybia aztecoides Richards (West-Eberhard 1973, 1978), Parachartergus colobopterus (Lichtenstein) (Herman et al. 2000), Asteloeca ujhelyii (Ducke) (Nascimento et al. 2004), Parachartergus fraternus (Gribodo) (Mateus 2005), and Metapolybia docilis Richards (Chavarría 2009) demonstrated that worker behavior may be an important component in the mechanism of delimitation of functions in the colony.
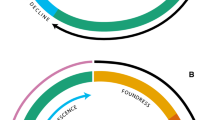
A peculiar feature that occurs in Polistinae is the presence of polygyny (presence of several queens coexisting in the same nest). This characteristic appeared three times within the subfamily – in the genera Polybioides and Ropalidia, and in the tribe Epiponini (Carpenter 1991) – and is associated with the colony’s foundation mode by swarming (the colony is constructed by a large number of workers accompanied by more than one queen) and complex social organization (Carpenter 1991; Jeanne 1991; Fig. 6.4). Epiponini species may exhibit permanent polygyny or variation in queen numbers (decrease) during the colony cycle, resulting in monogyny (West-Eberhard 1978; Richards 1978; Jeanne 1991). The presence of polygyny in the most basal clades indicates that the presence of multiple functional queens was already present in the common ancestor of the tribe (Carpenter 1991; Noll 2013).
Evolution of polygyny in Epiponini. (a) In a plesiomorphic society with short-term monogyny, the dominant female is intolerant of the reproduction of other females. The queen performs policing, while the workers are tolerant to the reproduction of other females. Occasionally, the workers reproduce. (b) In Epiponini no intolerant queens are present; instead, reproduction is performed by several tolerant females. The role of policing is adopted by workers, who suppress each other, as well as select among breeders. (Modified from Noll 2013)
It is not difficult to imagine that the origin of polygyny was molded by the same factors as swarm foundation and nest architecture and that ant predation was an important factor (Jeanne 1979). There is a consensus that eusociality of wasps originated in the tropics (Evans and West-Eberhard 1970), where ants are the most important predators of the colonies (Richards and Richards 1951). From this point of view, it is appropriate to state that the most peculiar characteristics of the Epiponini lifestyle are the result of the improvement of defense against ants (Richards 1971, 1978; Jeanne 1975, 1991; Starr 1990; Simões et al. 1996). Thus, the rapid abandonment of the nest through the migratory behavior (swarming) would be an important strategy, assured by polygyny, that led to the selection of queens morphologically undifferentiated (less body mass, easier to fly) and numerically more abundant (greater opportunity for reestablishment of the colony in a new place) (Mateus 2005). Considering that the loss of a single queen could be fatal, the presence of several queens could be advantageous for the colony in terms of survival (Noll 2013).
The presence of several queens laying eggs could represent a problem regarding “conflict of interest” occurring in the colonies of social insects (Noll 2013). Although degrees of relatedness may be low in colonies of the independent-founding wasps Polistes (Pickett et al. 2006), an additional problem arises in Epiponini: the division of reproduction among several queens would result in an even lower degree of kinship among the females of one colony (Hastings et al. 1998), which would not reinforce the sterility of the workers. However, kinship analyses for Epiponini colonies have indicated that the degree of relationship between females is relatively high (Queller et al. 1988, 1993; Strassmann et al. 1992; Gastreich et al. 1993; Hughes et al. 1993; Hastings et al. 1998). These results are in line with what West-Eberhard (1978, 1981) observed in colonies of Metapolybia aztecoides . After the swarm, at the beginning of the cycle, several queens and workers are present. Throughout the development of the colony, some of the queens disappear or begin to display worker functions, and, later, the number of queens is reduced until there remains a few or only one. Queen production and swarms occur predominantly after the establishment of monogyny or oligogyny, restricting the production of future queens to only a few individuals. This pattern is observed in several other species, such as Polybia occidentalis (Olivier) (West-Eberhard 1978; Queller et al. 1993), Parachartergus colobopterus (Strassmann et al. 1991), Polybia emaciata Lucas (Strassmann et al. 1992), Protopolybia exigua (de Saussure) (Gastreich et al. 1993), Brachygastra mellifica (Say) (Hastings et al. 1998), and Agelaia multipicta (Haliday) (West-Eberhard 1990). The term cyclic oligogyny was coined to define the decrease in the number of queens and the production of new ones during the period when there are few of them in the colony. That way, the relationship between the queen and daughters is increased, as predicted by the kinship selection theory (Hamilton 1964a, b, 1972). The high kinship is also maintained by the fact that queens in Epiponini are inseminated only once (Goodnight et al. 1996). The possibility that the wasps of this tribe are inflexible in terms of loss of these elements of sociality should not be ruled out either (Noll 2013).
6.2 Communication
6.2.1 Alarm Pheromone
Swarm-founding wasps and their populous colonies are engaged in sophisticated communication systems to coordinate the activities of all members of the colony (Jeanne 1991). The defense behavior in Polybia occidentalis involves two steps. First, in response to a sudden movement or mechanical disturbance to the nest, a large number of individuals are quickly recruited to the outer surface of the envelop. Second, a number of these individuals immediately fly and attack the intruder or predator. Alarm recruitment has been demonstrated experimentally in Polybia occidentalis (Jeanne 1981, 1991). The workers’ venom contains a pheromone that immediately recruits a large number of adults, which move to the surface of the envelope. Outside the nest, the attack occurs immediately against dark-colored objects that move close to the nest. The chemical alarm communication certainly occurs in most of the swarming wasps, but it is still necessary to investigate whether it occurs in colonies with low numbers of individuals (Jeanne 1981, 1991). The alarm and defense behavior of the nest in Synoeca surinama (Linnaeus) is coordinated by visual factors and by odors, which are recognized by the individuals of the same colony. In the presence of intruders, these factors together incite and produce the wasp attack (Castellón 1981).
6.2.2 Chemical Communication During Migration
Communication during migration depends on the chemical signals left by the scout workers on different substrates indicating a direction to follow (Naumann 1975; Jeanne 1975; Forsyth 1978; West-Eberhard 1982; Mateus 2005; Mateus 2011). For migration to occur, three major challenges need to be overcome. First, the scouts select a location for the construction of the new nest. Second, these workers create a “trail” of chemical signals, left on prominent objects (such as leaves) along the way to the new site. Finally, the rest of the colony follows the chemical trail to reach the site of the new nest (Sonnentag and Jeanne 2009; Mateus 2011).
The chemical communication during swarming in Epiponini seems to be related to a gland present in the fifth gastral sternite. Reports of the presence of this gland were made by Richards (1971) and later studied by Jeanne and Post (1982) and Jeanne et al. (1983). The “dragging behavior”, which consists of rubbing the gaster on a substrate to make the chemical pathway that is followed by the rest of the colony during migration, was first described by Naumann (1975) and observed in species of the genera Agelaia, Angiopolybia, Leipomeles, and Polybia. Jeanne (1975, 1981) experimentally confirmed the findings of Naumann (1975) observing the wasps followed an artificial trail made with substances removed from the gland of the fifth gastral sternite. However, this seems not to be the only strategy used. Studies with Apoica pallens (Fabricius) (Howard et al. 2002) showed a different strategy of chemical communication during the swarm. This species does not make a chemical trail on a substrate and does not previously select the location of the new nest. After leaving the old nest, the population forms clusters in leaves of plants, and the recruitment is done when the wasps elevated the abdomen and exposes the base of the fifth and sixth sternites. This behavior suggests that the wasps are emitting a pheromone in the air (“calling display”) to group the population and lead it to a site to build the new nest (Howard et al. 2002).
In Parachartergus fraternus , although the presence of scouts doing “dragging behavior” during migration has been observed by some authors (Jeanne et al. 1983; Smith et al. 2002), no evidence of external glands was found in the gastral sternites (Jeanne et al. 1983). In this species, the communication of the end point of the chemical pathway was established by dozens of scouts, creating an area of easy recognition due to the strong odor of the substance applied to the substrate. In the moments that preceded the migration, it was observed that the number of scouts sprinkling venom on the substrate of the new nest site is high. Simultaneously, in the old nest, the scouts who returned there make lateral movements with the abdomen (“dragging behavior”) in the substrate and oral contacts to signal and stimulate the population to migrate. The strategy of marking the place of the new nest by sprinkling venom possibly prevents the action of possible predators until the colony is established (Mateus 2005, 2011). Venom spraying is well known in the Parachartergus (Jeanne et al. 1995; West-Eberhard 1982), which has specialized muscles in the venom reservoir (Maschwitz and Kloft 1971). Chemical communication during migration in the Epiponini has been described as very efficient, since the loss of individuals during the process has been reported as low. The main causes of loss are related to predation or to the fact that individuals are newly emerged and not fully capable (Bouwma et al. 2003; Mateus 2011).
There are two distinct types of migration in Epiponini, “diffuse swarms” and “clumped swarms”. For both types, use of chemical communication as a signal of the path to the new nest was observed for all species studied (West-Eberhard 1982). In diffuse swarms, observed in Agelaia areata (Say) (Jeanne 1975), Synoeca surinama (West-Eberhard 1982), and Parachartergus fraternus (Mateus 2011), the wasps spread over an area of more than 100 m in length by 20–50 m in width, in search of the chemical track. In the clumped swarm, several clusters are formed on a migration route, and, as the population advances along the migration path using the chemical trail, the clusters disintegrate behind the advance (West-Eberhard 1982). This type of migration was observed in Parachartegus apicalis (Fabricius), Polybia ignobilis (Haliday), Polybia raui Bequaert, Polybia occidentalis (Forsyth 1981), and Polybia sericea (Olivier) (Jeanne 1981).
6.2.3 Recruitment for Food Forage
Wasps can be considered as the great flying hunters and gatherers of the world of social insects. As far as we know, all social wasps are omnivorous, feeding on both animal proteins and sugar-rich carbohydrate sources. The sources of foraged proteins are predominantly living arthropods, which provide nutrients for immature growth. Carbohydrate sources serve primarily as an energy source for adults (Jeanne and Taylor 2009). Unlike other social Hymenoptera, bees and ants, in wasps there is no sophisticated communication system to inform nest mates of location of the source or when and where to forage for food. Thus, the foragers of social wasps seem to act individually (Jeanne and Taylor 2009).
Some hypotheses have been suggested by Jeanne et al. (1995). One is that wasps do not have sufficient genetic variability to give rise to structures or behavior to inform nest mates about the location of resources, which seems unlikely. Swarm-founding wasps exhibited sufficient genetic variability for the evolution of trail pheromones, which guide swarms to new nests (Jeanne 1981, 1991), a mechanism that should probably be able to function in the recruitment of nest mates to food sources. The lack of a signal-based recruitment system may be due also to a social constraint. A critical colony size may be necessary for the benefits of recruitment to outweigh the costs (Beckers et al. 1989), and it can be argued that the number of foragers in most social wasp species is too low to effectively defend food sources against competitors such as some species of ants and stingless bees. A third possibility is that ecological factors may make recruitment an unproductive strategy in social wasps. For recruitment to be worth it, the resources need to be clustered in space and persist for considerable periods of time. However, according to Jeanne and Taylor (2009), it may be premature to generalize that all social wasps do not recruit for food sources, since only a few species have been studied carefully and there are many other species to be studied.
6.2.4 Division of Labor
The division of labor is a central theme in the study of social insects (Garcia and Noll 2013). The ecological success of these groups can be credited to their ability to coordinate their colonies for feeding, breeding, cleaning, and nesting (Beshers and Fewell 2001; Wilson 1979; Wilson and Hölldobler 2005). In ants and termites, the division of labor among castes is regulated by ontogenetic and physiological mechanisms (Miura 2005). In Epiponini, as in bees, the division of labor is associated with age polyethismFootnote 1 and specialization of tasks (Chavarría and Noll 2013; Garcia and Noll 2013).
Workers of independent foundation species begin to forage at the age of several days and combine foraging activities with activities within the nest throughout their lives. Foragers bring food (nectar and prey) and material (vegetable matter and water) to build the nest, and division of labor among them may occur by preference for a particular type of activity (polyethism) and in the way of processing the load (division of tasks). The forager can perform all the necessary handling alone or divide its load of material totally or partially with other members of the colony. The frequency of load partitioning increases with colony growth (Post et al. 1988; Rusina 2006), and the food division is often more frequent than the construction material division (Rusina et al. 2011). In Polistes fuscatus (Fabricius), for example, three functional groups are present: (1) workers who prefer to hunt than to build and tend to pass protein to other individuals, (2) workers who deliver construction material and do not pass prey to other workers, and (3) non-forage workers who engage exclusively in activities within the nest (Post et al. 1988). A similar pattern was found for P. dominula (Christ), P. nimpha (Christ), and P. gallicus (Linnaeus). In the latter two species, however, the constructors were involved in establishing and maintaining the dominance structure of the workers (Rusina 1999, 2006). Dominant workers in Mischocyttarus mastigophorus Richards collect building material and obtain protein food from prey forage more frequently than subordinate individuals (O’Donnell 1998b).
In Epiponini, the high level of social organization resulted in the presence of age-related polyethism and in the specialization of foragers. Handling of the delivered material (building material, water, prey, and nectar) is divided into two sub-tasks: collection and use. Foragers of the two functional groups bring food (prey and nectar) and construction material (wood pulp and water) to the nest and pass to other workers. A forager can deliver a specific type or both types of material throughout its active period. If a forager changes to a different type of load, the change usually occurs within the same group (food or nest material) (Rusina et al. 2011). Construction activity in these wasps is not only subdivided into delivery of pulp and construction but also involves the transportation of water by specialized workers. In addition, pulp delivered by foragers is generally too large to be used directly in the construction, so they have to pass at least part of the material to the workers in the nest who then distribute the material among themselves (Jeanne 1986; O’Donnell and Jeanne 1990; Karsai and Wenzel 1998). According to Jeanne (2003), the main difference between the independent and swarm-founding species is in the way of dealing with the material collected and in the fact that the foragers transfer the material to another worker (and continue to forage), allowing the development of the age polyethism and a more complex organization of the colony. In general, young workers perform tasks within the nest, such as cleaning and caring for the offspring. Meanwhile, the middle-aged workers are concerned with the construction and repair of the nest. Older workers, in turn, act as foragers, carrying material for construction and water and food for the colony, in addition to defending the nest (O’Donnell 2001). Studies with Polybia, Protopolybia, and Agelaia found that young and middle-aged workers perform tasks in the nest such as construction, care of the offspring, nest maintenance, and defense, while older workers forage (Simões 1977; Forsyth 1978; Jeanne et al. 1988, 1992). Chavarría and Noll (2013) observed a similar number of young and old female workers performing tasks such as cell inspection, construction, and foraging in Metapolybia miltoni Andena and Carpenter colonies. However, younger workers tend to forage less than the older ones (Chavarría and Noll 2013).
Specialization , that is, the presence of workers who perform a single type of task, is rare in most social wasps (Robinson 1992; Sendova-Franks and Franks 1999; O’Donnell 1998c; Karsai and Wenzel 2000; Beshers and Fewell 2001; Johnson 2003). Karsai and Wenzel (2000) did not find specialization in colonies of Metapolybia aztecoides and M. mesoamerica Smethurst and Carpenter. The same was observed by Chavarría and Noll (2013) for Metapolybia miltoni. Mateus (2005) also found flexibility in performing tasks in Parachartergus fraternus, where a worker who acted as a scout during the pre-swarm was subsequently observed laying eggs and, when dissected, the presence of ovaries developed with mature oocytes was found. Other scouts of the same nest performed construction tasks after nest establishment (Mateus 2005). In Polybia occidentalis and Metapolybia sp., there was greater stability in the preference for collection of a type of material when colonies were large (Jeanne 1986; Karsai and Wenzel 1998, 2000). Data obtained for Polybia aequatorialis (Zavattari) indicate that workers with specialization for the same activity have a close genetic relationship (O’Donnell 1996, 1998c). When there is little variation of age in the population, the workers should be allocated to tasks according to the needs of the colony, not being very relevant to the age of the individuals (Chavarría and Noll 2013). On the other hand, in colonies with individuals of different ages, polyethism seems to be more important in the delimitation of tasks, as demonstrated in the works of Jeanne et al. (1988, 1992). In short, the workers’ choice of task depends on the circumstances of the colony, and the workers can be allocated to this or that task according to need (Karsai and Wenzel 2000).
Workers in any insect society retain some degree of behavioral flexibility, which is decisive for the survival of the colony in changing situations (Robinson 1992). Likewise, caste flexibility is an important factor for colony survival in the swarm-founding wasps, allowing these insects to respond efficiently to changes that may occur (Chavarría and Noll 2013). As evidenced by Noll and Wenzel (2008), cast dimorphism evolved at least eight times in Epiponini, and social organization derives directly from an ancestor with incipient caste dimorphism in most taxa. Given this fact, general patterns are insufficient to explain the different strategies adopted by the species of this tribe (Beshers and Fewell 2001; Chavarría and Noll 2013).
6.2.5 Colony Defense
“I suspect that social wasps pay much more attention to ants in their environment than we think”. The phrase by Jeanne (1991) reinforces the idea that in the tropics, the ants represent the most important predators of the colonies of social wasps (Richards and Richards 1951). Predation by these insects may have strongly influenced the evolution of these wasps (Jeanne 1979). The rapid abandonment of the nest and swarm, the complex architecture of nests, and the presence of breeds without clear morphological differences suggest evolutionary adaptations against ant predation as well as optimization of work (Jeanne 1975; Wenzel 1998). While independent-founding species (Polistes and Mischocyttarus) rely heavily on secreted chemical repellents to keep ants away from their offspring, the swarming wasps depend primarily on the envelope surrounding the combs, coupled with the presence of workers guarding the nest entrance (Jeanne 1975). In addition, in the face of an ant attack, abandoning the nest by the rapid migration of the adult population is certainly an important and unique strategy in Epiponini (Mateus 2005).
In addition to envelope protection, workers actively guard the nest (Jeanne 1975, 1991). When the ants get too close, the wasps can resort to a series of behaviors to drive away the invaders. Protopolybia exigua and Polybia occidentalis, for example, exhibit “wing buzzing” and “burst” behaviors, where female workers swing their bodies up and backward by buzzing their wings rapidly and producing a snap (Chadab 1979). In P. exigua, this behavior is shown when visual or olfactory stimuli, as well as the odor of formic acid, are detected by the wasps and act as a warning signal to the companions who can quickly evacuate the nest if the ants reach this one (Chadab 1979). When an ant reaches a P. occidentalis nest, it can be stung by a worker who immediately uses the jaws to trap and carry the ant away from the nest (Jeanne 1991). As described by Chadab (1979), buzzing and burst behaviors cause small bursts of air directed at the ants whose antennae curve at each flight wrist. These pulses paralyze the ants momentarily, and the display of these behaviors persistently by the wasps results in the retreat of the ants (Jeanne 1991). Workers of Clypearia sulcata (de Saussure), Metapolybia aztecoides, and Polybia occidentalis use the jaws to scrape the substrate of the nest where the ants passed in order to remove the pheromone trail left by them (Chadab 1979; West-Eberhard 1989b; Jeanne 1991). Thus, the chances of ants reaching the nest in the future are reduced (Jeanne 1991).
Although most species bet on the active defense of the nest against invasive ants, a different strategy has evolved in some species. Nectarinella xavantinensis Mateus and Noll, N. championi (Dover), and Leipomelles dorsata (Fabricius) protect access to the nest with the placement of “sticky pillars” in areas near the entrance of the nest, apparently produced from an oral secretion (Jeanne 1991; Wenzel 1991; Mateus and Noll 1997; Carpenter and Marques 2001). Wasps of these species are quite small, and defending the nest actively against many ant species would be impossible (Jeanne 1991). Envelope protection, active defense, and building sticky traps can be very effective against small ants, but they are not enough against a group of ants, in particular the army ants. The majority of social wasps seem to have no effective defense against army ants, and the tactic against their arrival is the immediate abandonment of the nest by the entire adult population so that they do not end up as prey of the invaders, which will occur with the abandoned larvae and pupae (Jeanne 1991). In some species of Polistinae, however, ways of escaping of predation by these ants have evolved. One of them is to build very high nests in the canopy, as do Agelaia areata, Epipona tatua (Cuvier), and Chartergus metanotalis Richards (Jeanne 1991; Carpenter and Marques 2001). Other species with more resistant envelopes like Synoeca septentrionalis Richards, S. chalibea de Saussure, Polybia emaciata, and P. jurinei de Saussure prevent the entry of army ants using the body itself to block the entrance of the nest.
Curiously, despite the antagonistic relations between the ants and the social wasps, some species developed interspecific associations. Polybia rejecta (Fabricius), a very aggressive social wasp, often builds its nest in association with colonies of the ant Azteca (Richards 1978; Somavilla et al. 2012). This association may be one of the few defenses that tropical wasps have against invasions by ants of the army ants (Richards and Richards 1951). Similarly, wasps exhibit aggressive behavior when the ant colony is disturbed, protecting the ants primarily against predatory mammals such as anteaters (Virgínio et al. 2015). Synoeca chalybea can also construct nests in association with ants of the genus Azteca (Souza et al. 2013).
In addition to barring the ants, the presence of the envelope also seems to be an effective defense against parasites. When these are found, they are far less numerous when compared to the large infestations that may occur in non-enveloped nests of independent-founding species. Forsyth (1978) analyzed 141 colonies of Metapolybia azteca and Polybia occidentalis finding parasites in only 7% of them and less than 1% of offspring infestation. On the other hand, Nelson (1968) observed that about 60% of the colonies of Polistes metricus analyzed by him were parasitized by the moth Chalcoela iphitalis, with some nests reaching 90–95% infestation of the offspring.
The most famous defense mechanism of social wasps is undoubtedly the painful sting. In spite of the bad reputation, the sting apparatus in its origin has as primary function the capture of prey (Macalintal and Starr 1996). However, they assumed an important defense role against bigger enemies, especially in social lineages (Macalintal and Starr 1996). The behavior of stinging would have evolved due to the predation pressure exerted by vertebrates attracted by the increase in colony size, particularly in the tropics, in open and seasonally dry forests (Starr 1985; Carpenter and Marques 2001). This form of defense is undoubtedly quite effective in discouraging potential predators, although, it is not uncommon for wasp colonies to be attacked by vertebrates such as birds, bats, and primates (Jeanne 1975, 1991). Nevertheless, different mechanisms were adopted by other species in an attempt to minimize the risk of being attacked by these animals. Species with small colonies construct nests camouflaged for the purpose of hindering the visualization by vertebrates and parasites. Leipomeles dorsata, for example, constructs its nest at the bottom of a broad leaf and covers the envelope with green details in order to mimic plant venation (Williams 1928; Richards 1978; Jeanne 1991). Other species build large and sturdy nests, inaccessible to most vertebrates.
In Apis mellifera Linnaeus, defense and foraging activities are carried out by different groups of workers (Breed et al. 1990), that is, the workers who act in the defense of the colony are specialized in this task. In Epiponini, Jeanne et al. (1992) analyzing colonies of Polybia occidentalis did not find a subgroup of specialized workers in this task, concluding that the wasps that defended the nest also performed other activities inside the nest and of foraging, for example (Fig. 6.5a). In fact, there is a positive correlation since forager workers are more likely to act in defense than non-forager ones (Jeanne et al. 1992). Still for P. occidentalis, the likelihood of a wasp displaying defense response increases significantly among workers aged between 8 and 12 days, stabilizing after the 13th day (Jeanne et al. 1992; Fig. 6.5b). During a mechanical stimulus, young wasps also leave the nest in response to the attack signal, but only the older workers effectively attack the intruder. This fact makes it clear that the behaviors of “leaving the nest” and “attacking” represent different responses (Jeanne et al. 1992) and that the participation of a large number of individuals in the exit response suggests that this behavior functions as an aposematic sign to the predator (Jeanne et al. 1992).
Defense behavior according to age. (a) Frequency of task performance according to the age of the worker. Number of tasks related to nest maintenance, foraging trips, and defensive responses. (b) Frequency of task performance as a function of relative age (age is relative to the transition of each individual from the work in the nest to the foraging). (Modified from Jeanne et al. 1992)
Thanks to its defensive potential, the sting apparatus is considered a key factor that favored the origin of social behavior several times within Aculeata. Added to this is the fact that only females act as workers in the societies of wasps, bees, and ants (Silveira and Silveira 1994). An evolutionary analysis of the sting apparatus in Vespidae showed that some variations in the characteristics of this structure are related to the highly social way of life (Silva et al. 2014). In addition to the defense function, the use of the sting apparatus in a phylogenetic reconstruction supports the unique origin of eusociality , which would have occurred in the ancestor of Vespinae + Polistinae (Silva et al. 2014).
6.3 General Conclusion
The peculiar features presented by Neotropical social wasps, specially the swarm-founding Epiponini, resulted in a great diversity of species. Among the representatives of this tribe, all stages of caste differentiation are present, from castes morphologically very distinct to forms in which there are no external differences. Physiological differentiation also follows this rule, giving rise, in some species, to females with ovary developed but not fertilized, so-called intermediates. Because of polygyny, many females are able to lay eggs, tolerating each other, and the egg layers are tested and occasionally removed by sterile workers. Chemical communication guides the swarms and defense of the nest, and the tasks performed inside and outside the nest are divided according to the age of the wasps.
Notes
- 1.
The phenomenon whereby an animal shows different forms of behavior at different ages. In social insects, certain members of the colony may perform different tasks as they get older. In Epiponini genera Polybia, Protopolybia, and Agelaia, for example, young and middle-aged workers perform nest tasks (building, brood care, nest maintenance, defense), while old workers forage.
References
Baio MV, Noll FB, Zucchi R, Simões D (1998) Non-allometric caste differences in Agelaia vicina (Hymenoptera, Vespidae, Epiponini). Sociobiology 32:465–476
Baio MV, Noll FB, Zucchi R (2003a) Morphological caste differences, variation according to colony cycle, and non-sterility of workers in Brachygastra augusti (Hymenoptera, Vespidae, Epiponini), a Neotropical swarm-founding wasp. J N Y Entomol Soc 111:243–253
Baio MV, Noll FB, Zucchi R (2003b) Shape differences rather than size differences between castes in the Neotropical swarm-founding wasp Metapolybia docilis (Hymenoptera: Vespidae, Epiponini). BMC Evol Biol 3:1–9
Beckers R, Goss S, Deneubourg JL, Pasteels JM (1989) Colony size, communication and ant foraging strategy. Psyche 96:239–256
Beshers SN, Fewell JH (2001) Models of division of labor in social insects. Annu Rev Entomol 46:413–440
Bhadra A, Mitra A, Deshpande S, Chandrasekhar K, Naik D, Hefetz A, Gadagkar R (2010) Regulation of reproduction in the primitively eusocial wasp Ropalidia marginata: on the trail of the queen pheromone. J Chem Ecol 36:424–431
Bourke A (1999) Colony size, social complexity and reproductive conflict in social insects. J Evol Biol 12:245–257
Bouwma AM, Bouwma PE, Nordheim EV, Jeanne RL (2003) Adult mortality rates in young colonies of a swarm-founding social wasp (Polybia occidentalis). J Zool 260:11–16
Breed MD, Robinson GE, Page RE Jr (1990) Division of labor during honey bee colony defense. Behav Ecol Sociobiol 27:395–401
Carpenter JM (1991) Phylogenetic relationships and the origin of social behavior. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca, pp 7–32
Carpenter JM (1993) Biogeographic patterns in the Vespidae (Hymenoptera): two views of Africa and South America. In: Goldblatt P (ed) Biological relationships between Africa and South America. Yale University Press, New Haven, pp 139–155
Carpenter JM (1997) A note on the names of paper wasp tribes (Insecta: Hymenoptera: Vespidae). Ibaraki Univ Nat Hist Bull 1:15–16
Carpenter JM, Marques OM (2001) Contribuição ao Estudo de Vespídeos do Brasil (Insecta, Hymenoptera, Vespoidea, Vespidae). [CD - ROM]. Universidade Federal da Bahia, série Publicações Digitais, 2, Cruz das Almas
Carpenter JM, Ross KG (1984) Colony composition in four species of Polistinae from Suriname, with a description of the larva of Brachygastra scutellaris (Hymenoptera, Vespidae). Psyche 91:237–250
Castellón EG (1981) Alarma e defesa no ninho de Synoeca surinama (L) (Hymenoptera: Vespidae). Acta Amazônica 11:377–382
Chadab R (1979) Army-ant predation on social wasp. PhD Dissertation, University of Connecticut, Storrs
Chavarría L (2009) Comportamento, atividade e interações sociais entre raonhas e operárias de Metapolybia docilis (Vespidae: Polistinae Epiponini). Dissertação de mestrado em Ciências (Entomologia) - Universidade de São Paulo, Ribeirão Preto, São Paulo, 132 f
Chavarría L (2013) Sobre a produção e seleção de rainhas em diferentes fases do ciclo colonial em Epiponini (Vespidae: Polistinae). Tese de doutorado em Ciências (Entomologia) - Universidade de São Paulo, Ribeirão Preto, São Paulo, 139 f
Chavarría L, Noll FB (2013) Age polyethism in the swarm-founding wasp Metapolybia miltoni (Andena and Carpenter) (Hymenoptera: Vespidae; Polistinae, Epiponini). Sociobiology 60:214–216
Dapporto L, Santini A, Dani FR, Turillazzi S (2007) Workers of a Polistes paper wasp detect the presence of their queen by chemical cues. Chem Senses 32:795–802
Evans HE, West-Eberhard MJ (1970) The wasps. University of Michigan Press, Michigan, 265p
Fletcher DJC, Ross KG (1985) Regulation of reproduction in eusocial Hymenoptera. Annu Rev Entomol 30:319–343
Forsyth AB (1978) Studies on the behavioral ecology of polygynous social wasps. Dissertation, Harvard University
Forsyth AB (1981) Swarming activity of polybiine social wasps (Hymenoptera: Vespidae: Polybiini). Biotropica 13:93–99
Garcia ZJ, Noll FB (2013) Age and morphological changes in the Epiponini wasp Polybia paulista Von Ihering (Hymenoptera: Vespidae). Neotrop Entomol 42:293–299
Gastreich KR, Strassmann JE, Queller DC (1993) Determinants of high genetic relatedness in the swarm-founding wasp, Protopolybia exigua. Ethol Ecol Evol 5:529–539
Goodnight KF, Strassmann JE, Klinger CJ, Queller DC (1996) Single mating and its implications for kinship structure in a multiple-queen wasp, Parachartergus colobopterus. Ethol Ecol Evol 8:91–198
Hamilton WD (1964a) The genetical evolution of social behaviour I. J Theor Biol 7:1–16
Hamilton WD (1964b) The genetical evolution of social behaviour I. J Theor Biol 7:17–52
Hamilton WD (1972) Altruism and related phenomena, mainly in the social insects. Annu Rev Ecol Syst 3:193–232
Hart AG, Ratnieks FLW (2005) Crossing the taxonomic divide: conflict and its resolution in societies of reproductively totipotent individuals. J Evol Biol 18:383–395
Hastings MD, Queller DC, Eischen F, Strassmann JE (1998) Kin selection, relatedness and worker control of reproduction in a large-colony epiponine wasp, Brachygastra mellifica. Behav Ecol 9:573–581
Herman RA, Queller DC, Strassmann JE (2000) The role of queens in colonies of the swarm-founding wasp Parachartergus colobopterus. Annu Behav 59:841–848
Howard KJ, Smith AR, O’Donnell S, Jeanne RL (2002) Novel method of swarm emigration by the epiponine wasp, Apoica pallens (Hymenoptera Vespidae). Ethol Ecol Evol 14:365–371
Hughes CR, Beck MO, Strassmann JE (1987) Queen succession in the social wasp Polistes annularis. Ethology 76:124–132
Hughes CR, Queller DC, Negrón-Stomayor JA, Strassmann JE, Solis C, Gastreich KR (1993) The maintenance of high genetic relatedness in multi-queen colonies of social wasps. In: Keller L (ed) Queen number and sociality in insects. Oxford University Press, New York, pp 153–170
Hunt JH, Schmidt DK, Mulkey SS, Williams MA (1996) Caste dimorphism in Epipona guerini (Hymenoptera: Vespidae): further evidence for larval determination. J Kansas Entomol Soc 69:362–369
Hunt JH, O’Donnell S, Chernoff N, Brownie C (2001) Observations on two neotropical swarm-founding wasps, Agelaia yepocapa and A. panamaensis (Hymenoptera: Vespidae). Ann Entomol Soc Am 94:555–562
Hunt JH, Kensinger BJ, Kossuth JA, Henshaw MT, Norberg K, Wolschin F et al (2007) A diapause pathway underlies the gyne phenotype in Polistes wasps, revealing an evolutionary route to caste-containing insect societies. Proc Natl Acad Sci U S A 104:14020–14025
Jeanne RL (1975) The adaptiveness of social wasp nest architecture. Q Rev Biol 50:267–287
Jeanne RL (1979) A latitudinal gradient in rates of ant predation. Ecology 60:1211–1224
Jeanne RL (1981) Chemical communication during swarm emigration in the social wasp Polybia sericea (Olivier). Anim Behav 29:102–113
Jeanne RL (1986) The organization of work in Polybia occidentalis: costs and benefits of specialization in a social wasp. Behav Ecol Sociobiol 19:333–341
Jeanne RL (1991) The swarm-founding Polistinae. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca, pp 191–231
Jeanne RL (1996) Non-allometric queen-worker dimorphism in Pseudopolybia difficilis. J Kansas Entomol Soc 69:370–374
Jeanne RL (2003) Social complexity in the Hymenoptera, with special attention to the wasps. In: Kikuchi T, Azuma N, Higashi S (eds) Genes, behaviors and evolution of social insects, proceedings of the 14th congress of the IUSSI. Hokkaido University Press, Sapporo, pp 81–130
Jeanne RL, Fagen R (1974) Polymorphism in Stelopolybia areata (Hymenoptera, Vespidae). Psyche 81:155–166
Jeanne RL, Post DC (1982) Richards´ gland and associated cuticular modifications in social wasps of the genus Polybia (Hymenoptera: Vespidae: Polistinae: Polybiini). Insects Soc 29:280–294
Jeanne RL, Suryanarayanan S (2011) A new model for caste development in social wasps. Commun Integr Biol 4:373–377
Jeanne RL, Taylor BJ (2009) Individual and social foraging in social wasps. In: Jarau S, Hrncir M (eds) Food exploitation by social insects: ecological, behavioral, and theoretical approaches. CRC Press, Boca Raton, pp 53–73
Jeanne RL, Downing HA, Post DC (1983) Morphology and function of sternal glands in polistine wasps (Hymenoptera: Vespidae). Zoomorphology 103:149–164
Jeanne RL, Downing HA, Post DC (1988) Age polyethism and individual variation in Polybia ocidentalis, and advance eusocial wasp. In: Jeanne RL (ed) Interindividual behavioral variability in social insects. Westview Press, Boulder, pp 323–357
Jeanne RL, William NM, Yandell BS (1992) Age polyethism and defense in tropical social wasps (Hymenoptera: Vespidae). J Insect Behav 5:211–227
Jeanne RL, Graf CA, Yandell BS (1995) Non-size-based morphological castes in a social insect. Naturwissenschaften 82:296–298
Johnson BR (2003) Organization of work in the honeybee: a compromise between division of labor and behavioral flexibility. Proc R Soc Lond 270:147–152
Kardile SP, Gadagkar R (2002) Docile sitters and active fighters in paper wasps: a tale of two queens. Naturwissenschaften 89:176–179
Karsai I, Wenzel JW (1998) Productivity, individual-level and colony-level flexibility, and organization of work as consequences of colony size. Proc Natl Acad Sci U S A 95:8665–8669
Karsai I, Wenzel JW (2000) Organization and regulation of nest construction behavior in Metapolybia wasp. J Insect Behav 13:111–140
Macalintal EA, Starr CK (1996) Comparative morphology of the stinger in the social wasp genus Ropalidia (Hymenoptera: Vespidae). Mem Entomol Soc Wash 17:108–115
Maschwitz UWJ, Kloft W (1971) Morphology and function of the venom apparatus of insects-bees, wasps, ants, and caterpillars. In: Bücherl EE (ed) Venomous animals and their venoms. Academic Press, New York and London, pp 1–60
Mateus S (2005) Análise dos comportamentos envolvidos na organização social e no processo de enxameio de Parachartergus fraternus (Hymenoptera, Polistinae, Epiponini). Tese de doutorado em Ciências (Entomologia) – Universidade de São Paulo, Ribeirão Preto, São Paulo, Brasil. 160 f
Mateus S (2011) Observations on forced colony emigration in Parachartergus fraternus (Hymenoptera: Vespidae: Epiponini): New Nest Site Marked with Sprayed Venom. Psyche, Volume 2011, Article ID 157149, 8 pages
Mateus S, Noll FB (1997) Nectarinella xavantinensis, a new Neotropical social wasp (Hymenoptera: Vespidae; Polistinae). J N Y Entomol Soc 105:45–49
Mateus S, Noll FB, Zucchi R (1997) Morphological caste differences in Neotropical swarm-founding Polistinae wasps: Parachartergus smithii (Hymenoptera, Vespidae). J N Y Entomol Soc 105:129–139
Mateus S, Noll FB, Zucchi R (1999) Caste differences and related bionomic aspects of Chartegellus communis, a Neotropical swarm-founding Polistinae wasps (Hymenoptera: Vespidae: Polistinae: Epiponini). J N Y Entomol Soc 107:390–405
Mateus S, Noll FB, Zucchi R (2004) Caste flexibility and variation according to the Colony cycle in the swarm-founding wasps, Parachartergus fraternus (Gribodo) (Hymenoptera; Vespidae: Epipoini). J Kansas Entomol Soc 77:470–483
Miura T (2005) Developmental regulation of caste-specific characters in social-insect polyphenism. Evol Dev 7:122–129
Nascimento FS, Tannure-Nascimento IC, Zucchi R (2004) Behavioral mediators of cyclical oligogyny in the Amazonian swarm-fouding wasp Asteloeca ujhelyii (Vespidae, Polistinae, Epiponini). Insect Soc 51:17–23
Naumann MG (1970) The nesting behavior of Protopolybia pumila in Panama (Hymenoptera: Vespidae). PhD Dissertation, University of Kansas, Lawrence
Naumann MG (1975) Swarming behavior: evidence for communication in social wasps. Science 189:642–644
Nelson JM (1968) Parasites and symbionts of nests of Polistes wasps. Annu Entomol Soc Am 61:1528–1539
Noda SCM, Shima SN, Noll FB (2003) Morphological and physiological caste differences in Synoeca cyanea (Hymenoptera, Vespidae, Epiponini) according to the ontogenetic development of the colonies. Sociobiology 41:547–570
Noll FB (2013) “Marimbondos”: a review on the neotropical swarm-founding polistines. Sociobiology 60:347–354
Noll FB, Wenzel JW (2008) Caste in the swarming wasps: “Queenless” societies in highly social insects. Biol J Linn Soc 93:509–522
Noll FB, Zucchi R (2000) Increasing caste differences related to life cycle progression in some neotropical swarm-founding polygynic polistine wasps (Hymenoptera, Vespidae, Epiponini). Ethol Ecol Evol 12:43–65
Noll FB, Zucchi R (2002) Castes and the influence of the colony cycle in swarm-founding polistine wasp (Hymenoptera, Vespidae, Epiponini). Insect Soc 48:01–13
Noll FB, Mateus S, Zucchi R (1996) Morphological caste differences in neotropical swarm-founding Polistinae wasps V: Protopolybia exigua exigua (Hymenoptera, Vespidae). J N Y Entomol Soc 104:62–69
Noll FB, Simões D, Zucchi R (1997a) Morphological caste differences in the Neotropical swarm-founding Polistinae wasps: Agelaia n. multipicta and A. p. pallipes (Hymenoptera, Vespidae). Ethol Ecol Evol 9:361–372
Noll FB, Zucchi R, Mateus S (1997b) Morphological caste differences in the Neotropical swarm-founding and polygynous Polistinae wasp Polybia scutellaris. Stud Neotrop Fauna Environ 32:76–80
Noll FB, Yamane S, Zucchi R (2000) Morphological caste differences in the Neotropical swarm-founding polistine wasps. IX. Polybia (Myrapetra) occidentalis (Hymenoptera, Vespidae). Entomol Sci 3:491–497
Noll FB, Wenzel JW, Zucchi R (2004) Evolution of caste in Neotropical swarm-founding wasps (Hymenoptera: Vespidae; Epiponini). Am Mus Novit 3467:1–24
O’Donnell S (1996) RAPD markers suggest genotypic effects on forager specialization in a eusocial wasp. Behav Ecol Sociobiol 38:83–88
O’Donnell S (1998a) Reproductive caste determination in eusocial wasps (Hymenoptera: Vespidae). Annu Rev Entomol 43:323–346
O’Donnell S (1998b) Dominance and polyethism in the eusocial wasp Mischocyttarus mastigophorus (Hymenoptera: Vespidae). Behav Ecol Sociobiol 43:327–331
O’Donnell S (1998c) Genetic effects on task performance, but not on age polyethism, in a swarm-founding eusocial wasp. Anim Behav 55:417–426
O’Donnell S (2001) Worker biting interactions and task performance in a swarm-founding eusocial wasp (Polybia occidentalis, Hymenoptera: Vespidae). Behav Ecol 12:353–359
O’Donnell S, Jeanne RL (1990) Forager specialization and the control of nest repair in Polybia occidentalis Olivier. Behav Ecol Sociobiol 27:359–364
Pickett M, Carpenter JM, Wheeler WC (2006) Systematics of Polistes (Hymenoptera: Vespidae), with a phylogenetic consideration of Hamilton’s haplodiploidy hypothesis. Ann Zool Fenn 43:390–406
Platt TG, Queller DC, Strassmann JE (2004) Aggression and worker control of caste fate in a multiple-queen wasp Parachartergus colobopterus. Anim Behav 67:1–10
Post DC, Jeanne RL, Erickson HE (1988) Variation in behavior among workers of the primitively social wasp Polistes fuscatus variatus. In: Jeanne RL (ed) Interindividual behavior variability in social insects. Boulder, Westview, pp 283–321
Queller DC, Hughes CR, Strassmann JE (1988) Genetic relatedness in colonies of tropical wasps with multiple queen. Science 242:1155–1157
Queller DC, Negrón-Stomayor HCR, Strassmann JE (1993) Queen number and genetic relatedness in a Neotropical wasp, Polybia occidentalis. Behav Ecol 4:7–13
Richards OW (1971) The biology of the social wasps (Hymenoptera: Vespidae). Biol Rev 46:483–528
Richards OW (1978) The social wasps of the Americas excluding the Vespinae. British Museum (Natural History), London, 580p
Richards OW, Richards MJ (1951) Observations on the social wasps of South America (Hymenoptera, Vespidae). Trans R Entomol Soc Lond 102:1–170
Robinson GE (1992) Regulation of division of labor in insect societies. Annu Rev Entomol 37:637–665
Röseler PF (1991) Reproductive competition during colony establishment. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University, Ithaca, pp 309–335
Rusina LY (1999) Task partitioning in Polistine wasps (Hymenoptera, Vespidae). Vestnik Zool 33:105–108
Rusina LY (2006) Polistine wasps in natural and anthropogenic landscapes of the lower Dnieper area. Kherson Gos. Univ. [in Russian]
Rusina LY, Firman LA, Yu RY, Starr CK (2011) Pulp partitioning and worker specialization in Polistine wasps (Hymenoptera, Vespidae, Polistinae). Entomol Rev 91:820–827
Sakagami SF, Zucchi R, Yamane S, Noll FB, Camargo JMF (1996) Morphological caste differences in Agelaia vicina, the neotropical swarm-founding polistine wasp with the largest colony size among social wasps (Hymenoptera: Vespidae). Sociobiology 28:207–223
Sendova-Franks AB, Franks NR (1999) Self- assembly, self- organization and division of labour. Phil Trans R Soc Lond 1388:1395–1405
Shima SN, Yamane S, Zucchi R (1994) Morphological caste differences in some Neotropical swarm-founding Polistinae wasps I. Apoica flavissima (Hymenoptera, Vespidae). Jpn J Entomol 62:811–822
Shima SN, Yamane S, Zucchi R (1996a) Morphological caste differences in some Neotropical swarm-founding Polistinae wasps. II. Polybia dimidiata (Hymenoptera; Vespidae). Jpn J Entomol 64:131–144
Shima SN, Yamane S, Zucchi R (1996b) Morphological caste differences in some Neotropical swarm-founding Polistinae wasps. III. Protonectarina sylveirae (Hymenoptera; Vespidae). Bull Fac Educ Ibaraki Univ 45:57–67
Shima SN, Noll FB, Zucchi R, Yamane S (1998) Morphological caste differences in the Neotropical swarm-founding Polistinae wasps IV: Pseudopolybia vespiceps, with preliminary considerations on the role of intermediate females in the social organization of the Epiponini (Hymenoptera; Vespidae). J Hymenopt Res 7:280–295
Shima SN, Noll FB, Zucchi R (2000) Morphological caste differences in the Neotropical swarm-founding Polistinae wasp, Brachygastra lecheguana (Hymenoptera: Vespidae, Polistinae, Epiponini). Sociobiology 36:41–52
Silva M, Noll FB, Carpenter JM (2014) The usefulness of the sting apparatus in phylogenetic reconstructions in vespids, with emphasis on the Epiponini: more support for the single origin of eusociality in the Vespidae. Neotrop Entomol 43:134–142
Silveira O, Silveira A (1994) Comparative morphology of skeletal parts of the sting apparatus in Neotropical Polistinae social wasps (Hymenoptera, Vespidae, Polistinae). Sociobiology 25:295–327
Simões D (1977) Etologia e diferenciação de casta em algumas vespas sociais (Hymenoptera, Vespidae). Tese de Doutorado em Ciências – Universidade de São Paulo, Ribeirão Preto, São Paulo, 169 f
Simões D, Noll FB, Zucchi R (1996) Duration of protopolybia exigua nest and related aspects as influence by phorid fly infestation (Vespidae, Polistinae, Epiponini). Sociobiology 28:212–229
Smith AR, O’Donnell S, Jeanne RL (2002) Evolution of swarm communication in eusocial wasps (Hymenoptera: Vespidae). J Insect Behav 15:751–764
Somavilla A, Oliveira ML, Silveira OT (2012) Guia de identificação dos ninhos de vespas sociais (Hymenoptera, Vespidae, Polistinae) na Reserva Ducke, Manaus, Amazonas, Brasil. Rev Bras Entomol 56:405–414
Sonnentag PJ, Jeanne RL (2009) Initiation of absconding swarm emigration in the social wasp Polybia occidentalis. J Insect Sci 9, article 11
Souza MM, Pires EP, Prezoto F (2013) Nidification of Polybia rejecta (Hymenoptera: Vespidae) associated to Azteca chartifex (Hymenoptera: Formicidae) in a fragment of Atlantic Forest, in the state of Minas Gerais, southeastern Brazil. Biota Neotrop 13:390–392
Spradbery JP (1991) Evolution of queen number and queen control. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca, pp 336–388
Starr CK (1985) Enabling mechanisms in the origin of sociality in the Hymenoptera - the sting’s the thing. Ann Entomol Soc Am 78:836–840
Starr CK (1990) Holding the fort: colony defense in some primitively social wasps. In: Evans DL, Schmidt JO (eds) Insect defenses, adaptive mechanisms and strategies of prey and predators. State University of New York Press, New York, pp 421–463
Strassmann JE, Queller DC, Solis CR, Hughes CR (1991) Relatedness and queen number in the neotropical wasp, Parachartergus colobopterus. Ann Behav 42:461–470
Strassmann JE, Gastreich KR, Queller DC, Hughes CR (1992) Demographic and genetic evidence for cyclical changes in queen number in a neotropical wasp, Polybia emaciata. Am Nat 140:363–372
Strassmann JE, Solis CR, Hughes CR, Goodnight KF, Queller DC (1997) Colony life history and demography of a swarm founding social wasp. Behav Ecol Sociobiol 40:71–77
Strassmann JE, Goodnight KF, Klinger CJ, Queller DC (1998) The genetic structure of swarms and the timing of their production in the queen cycles of neotropical wasps. Mol Ecol 7:709–718
Strassmann JE, Sullender BW, Queller DC (2002) Caste totipotency and conflict in a large-colony social insect. Proc R Soc L Ser B 269:263–270
Tanaka GM Jr, Soleman RA, Noll FB (2010) Morphological and physiological variation between queens and workers of Protonectarina sylveirae (de Saussure) (Hymenoptera, Vespidae, Epiponini). Rev Brasil Entomol 54:104–109
Toth AL, Varala K, Newman TC, Miguez FE, Hutchison SK, Willoughby DA et al (2007) Wasp gene expression supports an evolutionary link between maternal behavior and eusociality. Science 318:441–444
Virgínio NF, Maciel TT, Barbosa BC (2015) Nidificação de Polybia rejecta (Fabricius) (Hymenoptera: Vespidae) associada à Azteca chartifex Forel (Hymenoptera: Formicidae) em ecótono de Bioma Caatinga/Mata Atlântica, no Estado do Rio Grande do Norte. EntomoBrasilis 8:242–245
Wenzel JW (1991) Evolution of nest architecture. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University, Ithaca, pp 7–29
Wenzel JW (1998) A generic key to the nests of hornets, yellowjackets, and paper wasps worldwide (Vespidae: Vespinae, Polistinae). Am Mus Novit 3224:1–39
West-Eberhard MJ (1973) Monogyny in “polygynous” social wasps. In: Proceedings of the 7th international congress of International Union Study for the Social Insects, London, pp 396–403
West-Eberhard MJ (1978) Temporary queens in Metapolybia wasps: non-reproductive helpers without altruism? Science 200:441–443
West-Eberhard MJ (1981) Intragroup selection and the evolution of insect societies. In: Alexander RD, Tinkle DW (eds) Natural selection and social behavior. Chiron Press, New York, pp 3–17
West-Eberhard MJ (1982) The nature and evolution of swarming in tropical social wasps (Vespidae, Polistinae, Polybiini). In: Jaisson P (ed) Social insects in the tropics, vol 1. Universit’e de Paris-Nord, Paris, pp 97–128
West-Eberhard MJ (1986) Dominance behavior in Polistes canadensis (L.), a tropical social wasp. Monit Zool Ital 20:263–281
West-Eberhard MJ (1989a) The establishment of reproductive dominance in social wasp colonies. In: Proceedings of the 8th International Congress of the International Union for the Study of Social Insects, Wageningen, pp 223–227
West-Eberhard MJ (1989b) Scent-trail diversion, a novel defense against ants by tropical social wasps. Biotropica 21:280–281
West-Eberhard MJ (1990) The genetic and social structure of polygynous social wasp colonies (vespidae: Polistinae). In: Veeresh GK, Mallik D, Viraktamath CS (eds) Social insects and the environment. Oxford and IBH, New Delhi, pp 254–255
West-Eberhard MJ (2003) Developmental plasticity and evolution. Oxford University Press, New York, 816p
Williams FX (1928) Studies in tropical wasps – their hosts and associates (with descriptions of new species). Bull Exp Station Hawaii Sugar Planters’ Assoc (Entomol) 19:1–179
Wilson EO (1979) The evolution of caste systems in social insects. Proc Am Philos Soc 123:204–210
Wilson EO, Hölldobler E (2005) Eusociality: origin and consequences. PNAS 102:13367–13371
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
da Silva, M., Mateus, S., Noll, F.B. (2021). Castes and Polymorphisms in Neotropical Social Wasps. In: Prezoto, F., Nascimento, F.S., Barbosa, B.C., Somavilla, A. (eds) Neotropical Social Wasps. Springer, Cham. https://doi.org/10.1007/978-3-030-53510-0_6
Download citation
DOI: https://doi.org/10.1007/978-3-030-53510-0_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-53509-4
Online ISBN: 978-3-030-53510-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)