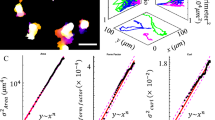
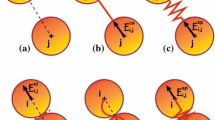
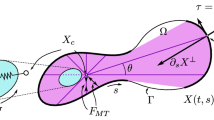
Abstract
Computational models have become an essential part of exploratory protocols in cell biology, as a complement to in vivo or in vitro experiments. These virtual models have the twofold advantage of enabling access to new types of data and validate complex theories. The design of mechanically functionalized biomaterials or scaffolds, to promote cell proliferation and invasion in the absence or in the complement of synthetic chemical coatings, can certainly benefit from these hybrid testing approaches. The underlying fundamental process of cell migration and in particular its dependence on the cell mechanical/geometrical environment remains crudely understood. Currently at least two theories explain the migration patterns observed by cells on curved topographies, involving either polymerization dynamics of actin or assembly dynamics of focal adhesions. We recently proposed a third mechanism relying on nucleus mechanosensitivity, which has been tested extensively experimentally and computationally. We now explore the hypothesis that nucleosensitivity could be a mechanism for cells to optimally find microenvironments suited for mitosis, providing mechanical stability and relaxation. By means of a computational mechanical model with intracellular structure detail, we investigate how the complex interplay between this new migration mechanism and the microenvironment topography can lead to more relaxed cells and organelles. To go further, we simulated in this study cell migration via a novel protocol in silico which generates dynamical ripple wave on a deformable substrate and changes topography over time. This kind of in silico protocols based on a new understanding of cell migration and nucleosensitivity could, therefore, inform the design of optimized scaffold topographies for cell invasion and proliferation.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Adey W (1983) Molecular aspects of cell membranes as substrates for interaction with electromagnetic fields. In: Synergetics of the Brain, Springer, Berlin, Heidelberg, pp 201–211
Almonacid M, Ahmed WW, Bussonnier M, Mailly P, Betz T, Voituriez R, Gov NS, Verlhac MH (2015) Active diffusion positions the nucleus in mouse oocytes. Nature cell biology 17(4):470–79
Barchiesi E, Spagnuolo M, Placidi L (2019) Mechanical metamaterials: a state of the art. Mathematics and Mechanics of Solids 24(1):212–234
Caballero D, Comelles J, Piel M, Voituriez R, Riveline D (2015) Ratchetaxis: long-range directed cell migration by local cues. Trends in cell biology 25(12):815–827
Chandorkar Y, Nava AC, Schweizerhof S, Van Dongen M, Haraszti T, Köhler J, Zhang H, Windoffer R, Mourran A, Möller M, et al (2019) Cellular responses to beating hydrogels to investigate mechanotransduction. Nature communications 10(1):1–13
Chen T, Callan-Jones A, Fedorov E, Ravasio A, Brugués A, Ong HT, Toyama Y, Low BC, Trepat X, Shemesh T, et al (2019) Large-scale curvature sensing by directional actin flow drives cellular migration mode switching. Nature physics 15(4):393–402
Clark P, Connolly P, Curtis A, Dow J, Wilkinson C (1991) Cell guidance by ultrafine topography in vitro. Journal of cell science 99(1):73–77
Czeisler C, Short A, Nelson T, Gygli P, Ortiz C, Catacutan FP, Stocker B, Cronin J, Lannutti J, Winter J, et al (2016) Surface topography during neural stem cell differentiation regulates cell migration and cell morphology. Journal of Comparative Neurology 524(17):3485–3502
Darling NJ, Sideris E, Hamada N, Carmichael ST, Segura T (2018) Injectable and spatially patterned microporous annealed particle (MAP) hydrogels for tissue repair applications. Advanced Science 5(11):1801,046
Del Vescovo D, Giorgio I (2014) Dynamic problems for metamaterials: review of existing models and ideas for further research. International Journal of Engineering Science 80:153–172
dell’Isola F, Seppecher P, Alibert JJ, et al (2019) Pantographic metamaterials: an example of mathematically driven design and of its technological challenges. Continuum Mechanics and Thermodynamics 31(4):851–884
Dillon R, Fauci L, Gaver III D (1995) A microscale model of bacterial swimming, chemotaxis and substrate transport. Journal of theoretical biology 177(4):325–340
Doyle AD, Wang FW, Matsumoto K, Yamada KM (2009) One-dimensional topography underlies three-dimensional fibrillar cell migration. Journal of cell biology 184(4):481–490
George D, Allena R, Remond Y (2018) A multiphysics stimulus for continuum mechanics bone remodeling. Mathematics and Mechanics of Complex Systems 6(4):307–319
George D, Allena R, Remond Y (2019) Integrating molecular and cellular kinetics into a coupled continuum mechanobiological stimulus for bone reconstruction. Continuum Mechanics and Thermodynamics 31(3):725–740
Giorgio I, Andreaus U, dell’Isola F, Lekszycki T (2017) Viscous second gradient porous materials for bones reconstructed with bio-resorbable grafts. Extreme Mechanics Letters 13:141–147
Griffin DR, Weaver WM, Scumpia PO, Di Carlo D, Segura T (2015) Accelerated wound healing by injectable microporous gel scaffolds assembled from annealed building blocks. Nature materials 14(7):737–744
Isenberg BC, DiMilla PA, Walker M, Kim S, Wong JY (2009) Vascular smooth muscle cell durotaxis depends on substrate stiffness gradient strength. Biophysical journal 97(5):1313–1322
Kloxin AM, Benton JA, Anseth KS (2010) In situ elasticity modulation with dynamic substrates to direct cell phenotype. Biomaterials 31(1):1–8
Lekszycki T, dell’Isola F (2012) A mixture model with evolving mass densities for describing synthesis and resorption phenomena in bones reconstructed with bio-resorbable materials. ZAMM-Zeitschrift für Angewandte Mathematik und Mechanik 92(6):426–444
Milan JL, Manifacier I, Beussman KM, Han SJ, Sniadecki NJ, About I, Chabrand P (2016) In silico CDM model sheds light on force transmission in cell from focal adhesions to nucleus. Journal of biomechanics 49(13):2625–2634
Mogilner A (2009) Mathematics of cell motility: have we got its number? Journal of mathematical biology 58(1-2):105
Nickaeen M, Berro J, Pollard TD, Slepchenko BM (2019) Actin assembly produces sufficient forces for endocytosis in yeast. Molecular biology of the cell 30(16):2014–2024
Pieuchot L, Marteau J, Guignandon A, Dos Santos T, Brigaud I, Chauvy PF, Cloatre T, Ponche A, Petithory T, Rougerie P, et al (2018) Curvotaxis directs cell migration through cell-scale curvature landscapes. Nature communications 9(1):1–13
Rens EG, Merks RM (2017) Cell contractility facilitates alignment of cells and tissues to static uniaxial stretch. Biophysical journal 112(4):755–766
Rodriguez ML, McGarry PJ, Sniadecki NJ (2013) Review on cell mechanics: experimental and modeling approaches. Applied Mechanics Reviews 65(6)
Schakenraad K, Ravazzano L, Sarkar N,Wondergem JA, Merks RM, Giomi L (2019) Topotaxis of active Brownian particles. arXiv preprint arXiv:190806078
Vassaux M (2018) mvassaux/adhsc: Initial release of the adhesion cell model (version v1.0.0). URL https://zenodo.org/record/1187087
Vassaux M, Milan J (2017) Stem cell mechanical behaviour modelling: substrate’s curvature influence during adhesion. Biomechanics and modeling in mechanobiology 16(4):1295–1308
Vassaux M, Pieuchot L, Anselme K, Bigerelle M, Milan JL (2019) A biophysical model for curvature-guided cell migration. Biophysical journal 117(6):1136–1144
Winkler B, Aranson IS, Ziebert F (2019) Confinement and substrate topography control cell migration in a 3D computational model. Communications Physics 2(1):1–11
Zigmond SH, Hirsch JG (1973) Leukocyte locomotion and chemotaxis: new methods for evaluation, and demonstration of a cell-derived chemotactic factor. The Journal of experimental medicine 137(2):387–410
Zink M, Szillat F, Allenstein U, Mayr SG (2013) Interaction of ferromagnetic shape memory alloys and RGD peptides for mechanical coupling to cells: from ab initio calculations to cell studies. Advanced Functional Materials 23(11):1383–1391
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Vassaux, M., Pieuchot, L., Anselme, K., Bigerelle, M., Milan, JL. (2020). Designing Optimal Scaffold Topographies to Promote Nucleus-Guided Mechanosensitive Cell Migration Using in Silico Models. In: Abali, B., Giorgio, I. (eds) Developments and Novel Approaches in Biomechanics and Metamaterials. Advanced Structured Materials, vol 132. Springer, Cham. https://doi.org/10.1007/978-3-030-50464-9_12
Download citation
DOI: https://doi.org/10.1007/978-3-030-50464-9_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-50463-2
Online ISBN: 978-3-030-50464-9
eBook Packages: Physics and AstronomyPhysics and Astronomy (R0)