Abstract
Cavernoma at pediatric age has been regarded as a relatively rare pathology, classified within the congenital vascular malformations of the central nervous system. Although majority of the cases are identified in adulthood due to expanding mass lesion or with the new onset of seizures following repeated hemorrhages. Advent of computed tomography (CT) and magnetic resonance imaging (MRI) their diagnosis both as an incidental finding and in childhood including infancy is increasing. Contrary to the other members of arteriovenous malformations of the brain, incidence, pathogenesis and the natural history of intracerebral cavernomas are not yet fully elucidated. Although they are originally regarded as congenital in origin with familial forms consisting almost one third of the diagnosed cases, documented cases of de novo formation after radiation therapy, stereotactic biopsy and association with developmental venous anomaly (DVA) bring controversy against the congenital hypothesis. Hemorrhage, as the main determinant of symptomatology in cavernomas is also diverse as it is almost always repetitive and intralesional resulting in expansion of the lesion rather than intraparenchymal disruption. Distinct pathology and pathogenesis of the cavernomas prevent sudden, life-threatening deterioration upon hemorrhage unlike true arteriovenous malformations. Nevertheless, seizures or progressive neurological deficits in a child have enough priority to treat a cavernoma to avoid neurocognitive decline even if incidentally detected. Surgical treatment is straightforward for symptomatic cases but a number of controversies exist related to the localization, multiplicity of the lesions especially in asymptomatic cavernomas. Regarding the brainstem, paraventricular or deep locations are more frequent in children, it is difficult to define a standard algorithm for surgical treatment of pediatric cavernomas.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
12.1 Introduction
In spite of intensive research, pathogenesis of cerebral cavernomas has not yet been elucidated. Cavernomas are originally believed to be primarily congenital in origin and loss–of-function mutations in at least three genes has been demonstrated in familial forms that are responsible for almost one third of the diagnosed cases. On the other hand, documented cases of de novo formation after radiation therapy, at stereotactic biopsy trajectory and association with developmental venous anomaly (DVA) brought controversy against the congenital hypothesis. Similar to the true arteriovenous malformations (AVM), where several theories have been proposed against congenital origin including inflammation, angiogenic and hormonal factors induced by hypoxia due to the presence of DVA. Nevertheless, cavernomas detected at pediatric age including fetal period suggest a congenital origin cannot be underestimated [1]. Diverse clinical course of the disease in both familial and isolated cases has been explained by the existence of powerful genetic and/or environmental disease modifiers [2]. Currently, there is no unifying hypothesis that would explain the occurrence of symptomatic cavernomas in a wide age spectrum beginning from fetal period. Ongoing debate on the origin and pathogenesis of cerebral cavernomas raises the question whether pediatric cavernomas should be regarded as a different disease entity or as a result of different germ-line mutations owing their diverse presentation compared to adults [3].
12.2 Epidemiology
Exact incidence of cavernomas remains unknown due to asymptomatic cases, revealed in autopsy. Prevalence estimations depend mostly on either autopsy or radiological series and the incidence has been reported to be 0.15–0.56 per 100,000 persons per year, with a prevalence of 0.17–0.9% while 70–95% of lesions remain life-time asymptomatic [93,4,5,6,7,9]. Previous estimates are largely based on adult series very few children have been included in those. Recent data argues they are not uncommon and its prevalence ranges from 0.37 to 0.53%, being second most common cause of spontaneous intracerebral hemorrhage in children, after AVMs [9,10,11,13].
12.3 Clinical Presentation
Seizure, hemorrhage or focal neurological deficits which are cardinal presentations of cerebral AVM are also valid for cavernomas. Cerebral cavernomas are typically presented with seizures and focal neurological deficits due to mass effect and hemorrhage [5, 13,14,15,17]. Hemorrhage is the most feared complication for a true AVM responsible for serious morbidity and mortality which all modes of treatment aim to avoid. Although hemorrhage has also been presented as a major complication in cavernomas, the magnitude and consequences of bleeding are incomparable to AVM’s. Owing to their morphology, hemorrhage is episodic, confined within the lesion; initially asymptomatic detected by radiology. While sudden bleeding from a high-flow AVM can result with acute symptoms secondary to direct parenchymal injury, cavernomas become symptomatic only after smaller, silent intralesional hemorrhages reach a critical mass to disturb surrounding parenchyma [14, 18]. Discrepancy between AVM and cavernoma bleeding is believed to cause confusion and misconception effecting especially natural history studies reporting a wide range of frequencies, partly due to differences in definition of hemorrhage [19, 20]. Several associations such as American Heart Association and Angioma Alliance have recommended standards to define cavernoma hemorrhage only with acute or subacute onset symptoms concordant with radiological location, surgical findings or pathological examination (Fig. 12.1). Presence of hemosiderin or enlarged diameter of the CM is not considered hemorrhage [19, 21]. Nevertheless, symptoms related to cavernoma hemorrhage following asymptomatic repeated episodes are closely related to size and location. Those who have reached enough size at the supratentorial area produce neurological deficits secondary to local mass effect usually preceded by sudden headache, impaired consciousness or seizure. Mixed series with adult predominance report hemorrhage in 8–37% cases as the initial presenting feature [22]. Among pediatric population, cavernomas represent 20–25% of spontaneous ICH in children. Hemorrhage was observed in 33–64% of the pediatric cases at the time of initial diagnosis [22,23,25]. Significant fatal hemorrhage is a rare phenomenon compared to AVM’s, with a reported annual risk of 0.25–6% in cavernomas [5, 16, 25,26,27,29]. Although cavernomas are identified by repeated microhemorrhages, extralesional/parenchymal, subarachnoid and intraventricular hemorrhages have also been described [30, 31]. Several clinical series report a higher propensity for overt hemorrhage in women and children [30, 31,32,34] (Fig. 12.2). Cavernomas situated in the vicinity of the ventricular system can cause blood oozing through the disrupted ependyma during episodes of hemorrhage. Shirvani and Hajimirzabeigi [35] have reviewed 136 intraventricular cavernoma cases in the literature where 23% of patients were in the pediatric and adolescent group and 19%, infants. They have concluded that lack of perilesional brain tissue inside the ventricles promotes the rapid growth and increases the probability of bleeding. These figures indicate that the ratio of intraventricular location is higher in the pediatric age among symptomatic cases (Fig. 12.3). It is not clear whether paraventricular location and relatively large size is an evidence indicating pediatric cavernoma to be a different disease entity compared to adults or simply early detection due to localization. Similar concern is valid for infratentorial localization, published large series report a higher incidence of infratentorial cavernomas in children over 30%, compared to adult series [25, 35,36,38]. Contrarily, reported high incidence has been attributed to the tendency to report such a less common location in the medical literature, rather than reflecting the actual incidence [37]. Nevertheless, brainstem cavernomas, both in adults and children, are presented with focal neurological signs almost always accompanied by cranial nerve involvement along with motor deficits, ataxia and even neuropathic pain [25, 36, 39] (Fig. 12.4). Neurological deficits are secondary to local mass effect rather than direct parenchymal injury although they invariably exhibit varying degrees of micro hemorrhage in the brainstem.
Criteria advised by Angioma Alliance Scientific Advisory Board for accurate definition of cavernoma hemorrhage with matching symptoms with radiological, pathological or surgical findings (Figure adapted from Salman RA-S et al. [21])
Cavernoma with extra-lesional, parenchymal hemorrhage presented with right sided hemiparesis following focal seizure in a 3 year old boy; T2 weighed axial (a, b), coronal (c) sections revealing a paraventricular lesion with mixed signal intensity surrounded by a rim of hypointensity consistent with cavernoma and hyperintense extralesional signal at the neighbouring cortex at the T1 weighed sagittal image (d), representing recent hemorrhage
MR study of a 23 month old boy due to sudden irritability and nuchal rigidity revealing subependymal lesion at the wall of the IV. Ventricle at medullar level, homogenous and hypointense in axial T2 (a) and coronal (c), and diffusion weighed (b) images; with adjacent DVA in T1 axial image with contrast (d), suggestive for a cavernoma apparent by oozing into the ventricle
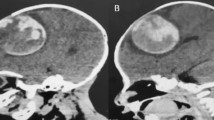
12 year old girl admitted with the complaint of facial weakness on the right, with MR request to rule-out a tumor revealing a non-homogenous lesion with hiper- and hypointense patchy appearance on axial T1 (a) and T2 (b) and sagittal (c) weighed images, at the pontomedullary junction at the level of facial nucleus, consistent with a cavernoma
An exclusive characteristic of cavernoma hemorrhage is that; even when they are too small to be symptomatic, they may contribute to seizure development. Progressive deposition of hemosiderin in the cerebral parenchyma surrounding the cavernous malformation is a potent epileptogenic agent with the iron content [40, 41].
Whether preceded by a recent hemorrhagic event or not, seizure is the most frequent single presenting symptom in most of the published series [23, 41,42,43,44,45,46,48]. Bilginer et al. [23], in their review of 36 operated pediatric cavernoma cases found that seizure was the most common single presenting symptom in 38.9%, added those with additional symptoms and signs, 61.1%of the patients had had at least one seizure on admission (Fig. 12.5).
Association of symptoms on admission in 36 pediatric cavernomas, demonstrating recent hemorrhage is imaging corresponds closely to the presentation except that only seizures where no recent hemorrhage was detected at the majority (Figure adapted from Bilginer et al. [23])
12.4 Natural History
“Understanding the natural history of a disease is an important prerequisite for designing studies that assess the impact of interventions, both chemotherapeutic and environmental…. may provide important indicators for drug targets and surrogate outcomes for clinical trials. However, collecting and visualizing data on natural history is challenging in part because disease processes are complex and evolve in different chronological periods for different subjects.” [49]. This concept is valid for central nervous system diseases with congenital background, including cavernomas. Cerebral AVM’s and pediatric aneurysms to some extent are vascular pathologies with congenital origin and may stay asymptomatic during life-time. Apart from those which become symptomatic by hemorrhage, intention to treat is to avoid hemorrhage, once detected. Consequence of hemorrhagic insult is well established as well as the life-time risks of bleeding with epidemiological natural history studies. Accumulated data on their natural history enables to make straightforward decisions for treatment in asymptomatic cases. Cavernomas, as a member of central nervous system vascular malformations have disadvantages in evaluating their natural history due to their diverse morphology and different flow dynamics. This can be appreciated from the diversity of the estimations for prevalence and hemorrhage rates reported so far.
Inconsistency among figures have several reasons; depending on the methodology of the study and composition of the target population. Above all, hemorrhage risk, as the main variable to be calculated has been appreciated differently in the studies. Unlike AVM’s, the hemorrhage referred is almost always intralesional recurrent oozing, not necessarily symptomatic. Symptoms usually depend on the size and location of the cavernomas rather than the parenchymal disruption due to hematoma. Adding cases with image based diagnosis to symptomatic cases may result with under- or overestimation of the bleeding rate. Moreover, existence of the familial vs. sporadic, congenital and de-novo, single or multiple, adult and pediatric forms all together have a potential selection bias. Like all other vascular malformations, estimate of the natural history of cavernomas is important to compare the untreated clinical course and outcome in terms of morbidity and mortality to risks and complications of potential treatment.
Pediatric cavernomas detected even in infants may be regarded on the congenital origin but estimates based on this assumption may result in an artificially low figures because annual rate of bleeds before surgery overall may be underestimated as it is impossible to quantify the length of time that a patient has had the lesion [19, 24]. On the other hand, calculations based on children who present with hemorrhage may result in artificially high hemorrhage risk estimates. In adult series, while the annual risk of hemorrhage in incidentally detected cases was found as 0.33%, while the risk rebleeding after the first hemorrhage increases up to 60% [7, 16, 28, 49,50,51,52,53,55]. Nevertheless, Al-Shahi Salman et al. [7], in their population based cohort study of 139 adult cases, concluded that the risk of a first-ever intracranial hemorrhage is low; functional impairment from hemorrhage is mild at initial presentation and although the risk of recurrence is higher than the risk of a first event, it seems to decline over time.
In limited number of recent publications in pediatric cavernoma series, hemorrhage rate was 0.2–1.6% in incidental cases and 7.4–18.2% after the first incident, which are apparently lower than the average values in adult series [14, 24]. On the other hand, brain stem location is brainstem was associated with a higher rate of symptomatic hemorrhage [38, 56, 57].
12.5 Treatment
For any cerebral vascular malformation whether symptomatic or has the potential of initiating morbidity or mortality, definitive treatment modality is surgical excision. This is also valid for cavernomas as a member of cerebral vascular malformations. From surgical point of view, cavernomas, adult or pediatric, more likely resemble cerebral benign tumors rather than vascular malformations. In fact, cavernomas do not demonstrate the adverse properties of intra-axial tumors and AVM’s that hamper resection. They are well circumscribed and can be differentiated from the surrounding brain parenchyma without any invasion as seen in most of the intra-axial tumors no high flow, arterial bleeding is expected within the lesion, unlike AVM’s. These properties offer a rather safe and easy excision of a cavernoma compared to most of the intra-axial lesions. On the other hand, as a presumably congenital anomaly, diverse natural history along with the variable size, multiplicity and eloquent location of cavernomas complicates the decision-making process. Pediatric patients with cavernomas have additional disadvantages such as having a higher probability of having hemorrhage with a longer life span, facing detrimental effects of neurological impairments and seizures to neurocognitive and psychosocial development. Moreover, although not universally confirmed, pediatric cavernomas are reported to be larger, more aggressive at eloquent locations [22, 58, 59]. Epidemiologic studies based on autopsy findings assume that as high as 95% of cavernomas may remain asymptomatic [60, 61]. This probability supports the decision of observation in incidentally detected cavernomas especially when they are single, located supratentorially at a deep or non-eloquent area. Although several studies have reported younger age as a risk factor for hemorrhage and worse natural history, exclusively pediatric case series stated comparable hemorrhage rates with adults indicating age at time of diagnosis did not correlate with hemorrhage risk [24, 38, 51, 62]. There is enough evidence for managing conservatively an asymptomatic incidental cavernoma in a child, both for presumably low risk of hemorrhage as well as minor consequences when become symptomatic, due to non-eloquent location. Once detected, there is no clear-cut policy on how frequently and for which criteria should an asymptomatic cavernoma be followed. Annual magnetic resonance imaging is a logical option although a life-time screening for a child seems to be irrational. There are conflicting conclusions on how to manage silent growth based on radiology alone; whether location or size is predictive for further hemorrhage and symptoms. While deep hemispheric and brainstem locations are regarded as a risk factor by some, most studies have failed to confirm this in adults [38, 62,63,64,65,66,67,69]. Size, as well as location in terms of predicting hemorrhage is not regarded as a major risk factor in most of the studies [28, 38, 42, 66, 67]. On the other hand, cavernomas associated with developmental venous anomaly (DVA) have a higher risk of hemorrhage [22, 47, 63, 69,70,71,72,74]. Nevertheless, incidentally detected pediatric cavernomas with eloquent cortex or brainstem location, especially when accompanied by a DVA require a close monitoring (Fig. 12.6).
Six year old boy, MR imaging after temporary conjugate gaze palsy following headache revealed a pontine heterogenous lesion consistent with cavernoma supplemented by hipointense rim and adjacent linear hypointensity (arrow), representing DVA (a). He was decided to be followed until a second insult 4 years later at an age of 10, due to a new lesion posterior to the regressed cavernoma (b)
Surgery is the single best treatment option for symptomatic as well as growing asymptomatic cavernomas. Anticipated long life span of children weighed against the natural history of cavernomas early aggressive treatment is advocated [74,75,76,78]. Mode of presentation related to the location of the lesion is the most important variable dictating the timing and extent of surgical intervention. In most locations, larger the size of the lesion provides easier access through the intervening parenchyma concerning the architecture of cavernomas have detectable demarcation and less chance to bleed compared to most tumors and AVM’s. On the contrary, small and deep seated cavernomas should not be recommended for surgery. Multiple lesions, especially in familial forms whether asymptomatic or with symptoms that cannot clearly be attributed to a single lesion, eloquent cortex and brainstem cavernomas, recently expanded lesions with perilesional reaction are difficult to decide to treat.
12.5.1 Supratentorial Cavernomas
Seizure by far is the most frequent symptom for newly diagnosed supratentorial cavernomas in adults except for those with extralesional bleeding. In children, neurological impairment due to mass effect is more frequent than adults, either alone or accompanied by seizures. Larger lesions tend to be more common as the age approaches to infancy. Lobar cavernomas with symptoms due to their size and remote to eloquent cortex can be removed without morbidity through the shortest trajectory from the non-eloquent cortex. Similarly, single lesions found incidentally with a considerable mass in children are advised to be resected as the probability of morbidity due to hemorrhage risk and further lesion growth during long life-span outweighs surgical morbidity [79]. Regardless their size, periventricular cavernomas incidentally with an exophytic component extending into the ventricle are candidates for surgery to avoid rupture into the ventricle [79,80,81,83]. Ventricular location provides safe approach without interrupting normal cortex and relatively direct access to the lesion through ventricular surface (Fig. 12.7).
Intraventricular cavernoma situated in the III. ventricle; operated through anterior interhemispheric transcallosal approach where encapsulated hematoma was observed at the foramen of Monroe (a), excised without any neural tissue interruption except for a half centimeters of corpus callosum (b). Yellow hemosiderin deposits over the ependyma (asterix), incision in corpus callosum (arrows)
Decision for surgery is complicated for lesions at the eloquent cortex and those presenting with seizures. Eloquent area cavernomas already symptomatic in children should be considered for early surgery as progressive morbidity is inevitable if left alone. Size and proximity to cortex may permit to reach the lesion through the already compromised cortex without adding further morbidity. As cavernomas have no intervening neural tissue or vasculature contributing to the surrounding, excision is less likely to increase the morbidity (Fig. 12.8). Contrarily, eliminating the mass effect is expected to counterbalance the injury of the limited cortical incision in amelioration of the symptoms. Frameless stereotaxy or intraoperative ultrasound is very helpful in determining the closest and less dangerous incision point and trajectory to reach the lesion. One exception is a deep and small lesion where reaching to the lesion requires considerable retraction. Observing for repeat hemorrhages until the size reaches to a resectable size or progressive neurological decline seems to be a more logical option.
Right insular cavernoma in a left handed 5 year old boy disclosed due a temporary, sudden speech arrest and right hemiparesis, surgery through Sylvian dissection with the aid of navigation (a, b) cavernoma easily identified within the cortex (c) and excision without interrupting the neighbouring cortex hemosiderin ring (d)
Cavernomas manifested with seizures may be problematic for the surgical decision in children. For a cavernoma apparent with a single seizure along with mass effect surgical treatment is straightforward. In those with drug resistant epilepsy or seizure semiology that does not correspond the location of the cavernoma surgical decision and the extent of resection is not easily justified (Fig. 12.9). With a single seizure in the pediatric age, which is the most common most common neurological symptom at the pediatric age, it may be challenging to prove cause and effect relationship. It might be more appropriate to observe the child if electrophysiology is uninformative and the cavernoma is rather silent in terms of size and location. Those with recurrent focal seizures and seizure semiology concordant with cavernoma location, excision provides the best chance for seizure freedom. When the pathophysiological background of seizure related to cavernoma is considered, excision of the hemosiderin deposition within normal parenchyma around the lesion is argued for a better seizure control [44, 84, 85]. Particularly for lesions that are located non-eloquently and in which the usually relatively small perilesional deposits can be resected safely [44].
One other focus discussion is the resection extent in temporal lobe epilepsy related cavernomas. As far as the surgery is aimed to eliminate the seizures rather than the mass effect of the cavernoma, question is whether lesionectomy is enough to achieve seizure control. For any given lesion at the mesial temporal lobe that has been found responsible for seizures, extended mesial resections including hippocampus has been proven to be superior to lesion excision [86]. Likewise, there is enough evidence that better postoperative seizure outcome is achieved with extended resections over lesionectomy and hemosiderin rim alone in temporal lobe epilepsy [44, 86,87,89]. In childhood temporal lobe epilepsy syndromes, extended cortical resections including amygdala and hippocampus is argued to be superior to lesionectomy for better seizure control even if the lesion including cavernomas is located in temporal neocortex [90] (Fig. 12.10). This requires a thorough preoperative evaluation and testing to identify the epileptogenic zone in specialized epilepsy centers.
Drug resistant temporal lobe epilepsy cases, 8-year-old boy with cavernoma at the right inferior temporal gyrus (a and b); 12-year-old girl, cavernoma detected on the left posterior parahippocampus (c and d). Temporal lobectomy and amigdalohippocampectomy where both cavernomas are situated within the limits of standard lobectomy borders were performed in both cases
12.5.2 Brainstem
Unlike their supratentorial counterparts, significant morbidity exists with the surgical treatment of brainstem cavernomas. On the other hand, Both subsequent amount of data is present in the literature that brainstem cavernomas have a higher rate of symptomatic hemorrhage than cavernous malformations at other locations (brainstem [38, 56, 57]). Children with incidentally detected brainstem cavernomas with lesions deep to the surface or prior hemorrhagic episode and acceptable neurologic deficits should be followed conservatively. Nevertheless, once bled and become symptomatic, progressive natural history of brainstem cavernomas ultimately require surgical resection. Pediatric cavernomas are found to be larger than the adult population and become symptomatic differently from their adult counterparts with their relatively reduced intracranial volume indicating a potential for a greater mass effect [37, 38]. Therefore, it is reasonable to accept that surgery may offer a high probability of altering the natural history of the disease by preventing future hemorrhages and avoiding neurologic decline, as demonstrated on published surgical series. Experience gained from brainstem surgery from low grade lesions of the brainstem in children promotes surgical intervention whose longer life expectancy increases the driving force to seek definitive treatment. As in the supratentorial locations, larger the cavernoma, less eloquent brainstem tissue is need to traversing to reach the lesion. In respect to the location of the cavernoma within the brainstem; mesencephalic, pontine or intramedullary, one of the standard posterior fossa approaches such as midline suboccipital, retrosigmoid, supracerebellar-infratentorial can be utilized. Lesions that are exophytic or apparent by a hemosiderin-stained area at the surface can be approached through a direct route (Fig. 12.11). Those imbedded within the neural tissue require careful preoperative planning to estimate the displaced tracts and find the safest entry point with minimal destruction. Displacement by the lesion may result with unexpected morbidity when previously defined safe entry zones are used. Again, once the lesion is reached, the morphology of the cavernoma allows safe excision with better appreciation of the normal tissue interface compared to tumors and without the risk of unexpected bleeding as in AVM’s. One important point to remember in both supra- and infratentorial cavernomas is to preserve the DVA, accompanying to subsequent number cavernomas. Reader is advised to refer to the related chapters in this book for further information about microsurgical techniques as there is no difference to mention separately for pediatric cavernomas.
Brain stem (medullary) cavernoma in a 11 year old boy with swallowing difficulty and conjugate gaze disturbance (a, b) operated through a midline posterior fossa telavolar approach; hematoma detected at the floor of the ventricle within hemosiderin staining (c), exposing the mass after enlarging the ruptured area of the hematoma (d), leaving the hemosiderin stained tissue, intact (e and f)
12.6 Radiosurgery for Pediatric Cavernomas
Stereotactic radiosurgery (SRS) has been used as an alternative treatment for symptomatic cavernomas in elequent areas although it is not clear whether cavernomas respond to high dose irradiation the same way as AVM’s [20, 90,91,93]. While studies report that hemorrhage rates is expected to decrease after 2–4 years, there appears to be a temporary increase in the hemorrhage rate up to 22.4% per patient per year with temporary morbidity rates varying from 10.5 to 59% and from 1.7 to 22.7%, permanently, in adult series (pri&pre). There is ongoing debate as to whether the effects of SRS in fact merely reflect the cavernomas’ natural history [20] As far as radiation exposure enhancing the genesis of new cavernoma formation in pediatric cases along with no clear information existing for SRS optimal dose to reduce hemorrhage and unknown late effects of focused radiation in children, it seems irrational to propose SRS for pediatric cavernomas.
References
Leung KY, Tang MH, Lam TP, Fan YW, Shek TW, Wong KY, Ngai CS. Prenatal diagnosis of a cavernous angioma associated with intracranial hemorrhage: report of one case and review of the literature. Ultrasound Obstet Gynecol. 2004;24(7):800–3.
Tang AT, Choi JP, Kotzin JJ, Yang Y, Hong CC, Hobson N, Girard R, Zeineddine HA, Lightle R, Moore T, Cao Y, Shenkar R, Chen M, Mericko P, Yang J, Li L, Tanes C, Kobuley D, Võsa U, Whitehead KJ, Li DY, Franke L, Hart B, Schwaninger M, Henao-Mejia J, Morrison L, Kim H, Awad IA, Zheng X, Kahn ML. Endothelial TLR4 and the microbiome drive cerebral cavernous malformations. Nature. 2017;545(7654):305–10.
Tirakotai W, Fremann S, Soerensen N, Roggendorf W, Siegel AM, Mennel HD, Zhu Y, Bertalanffy H, Sure U. Biological activity of paediatric cerebral cavernomas: an immunohistochemical study of 28 patients. Childs Nerv Syst. 2006;22(7):685–91.
Morris Z, Whiteley WN, Longstreth WT Jr, Weber F, Lee YC, Tsushima Y, Alphs H, Ladd SC, Warlow C, Wardlaw JM, Al-Shahi Salman R. Incidental findings on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ. 2009;339:b3016.
Kim DS, Park YG, Choi JU, Chung SS, Lee KC. An analysis of the natural history of cavernous malformations. Surg Neurol. 1997;48:9–17.
Moriarity J, Clatterbuck R, Rigamonti D. The natural history of cavernous malformations. Neurosurg Clin N Am. 1999;10:411–7.
Al-Shahi Salman R, Hall JM, Horne MA, Moultrie F, Josephson CB, Bhattacharya JJ, Counsell CE, Murray GD, Papanastassiou V, Ritchie V, Roberts RC, Sellar RJ, Warlow CP. Scottish audit of intracranial vascular malformations (SAIVMs) collaborators. Untreated clinical course of cerebral cavernous malformations: a prospective, population-based cohort study. Lancet Neurol. 2012;11:217–24.
Berry R, Alpers B, White J. The site, structure and frequency of intracranial aneurysms, angiomas and arteriovenous abnormalities. In: Millikan CH, editor. Research publications: Association for Research in nervous and mental disease. Baltimore: Williams and Wilkins; 1966. p. 4–72.
Otten P, Pizzolato G, Rilliet B, et al. 131 cases of cavernous angioma (cavernomas) of the CNS, discovered by retrospective analysis of 24,535 autopsies. Neurochirurgie. 1989;35:128–31.
Robinson JR, Awad IA, Neurosurg LJRJ. Natural history of the cavernous angioma. J Neurosurg. 1991;75(5):709–14.
Alexiou GA, Mpairamidis E, Sfakianos G, Prodromou N. Surgical management of brain cavernomas in children. Pediatr Neurosurg. 2009;45(5):375–8.
Rigamonti D, Hadley M, Drayer B, et al. Cerebral cavernous malformations. Incidence and familial occurrence. N Engl J Med. 1988;319:343–7.
Bergeson PS, Rekate HL, Tack ED. Cerebral cavernous angiomas in the newborn. Clin Pediatr (Phila). 1992;31(7):435–7.
Gross BA, Du R, Orbach DB, Scott RM, Smith ER. The natural history of cerebral cavernous malformations in children. J Neurosurg Pediatr. 2016;17(2):123–8.
Fortuna A, Ferrante L, Mastronardi L, Acqui M, d’Addetta R. Cerebral cavernous angioma in children. Childs Nerv Syst. 1989;5:201–7.
Aiba T, Tanaka R, Koike T, Kameyama S, Takeda N, Komata T. Natural history of intracranial cavernous malformations. J Neurosurg. 1995;83:56–9.
Frim DM, Scott RM. Management of cavernous malformations in the pediatric population. Neurosurg Clin N Am. 1999;10:513–8.
Zabramski J, Wascher T, Spetzler R, Johnson B, Golfinos J, Drayer BP, Brown B, Rigamonti D, Brown G. The natural history of familial cavernous malformations: results of an ongoing study. J Neurosurg. 1994;80:422–32.
Flemming KD, Brown RD Jr. Epidemiology and natural history of intracranial vascular malformations. In: Winn HR, editor. Youmans & Winn neurological surgery. 7th ed. Amsterdam: Elsevier; 2017. p. 3446–3463e7.
Akers A, Al-Shahi Salman RA, Awad I, et al. Synopsis of guidelines for the clinical management of cerebral cavernous malformations: consensus recommendations based on systematic literature review by the Angioma Alliance Scientific Advisory Board Clinical Experts Panel. Neurosurgery. 2017;80(5):665–80.
Salman RA-S, Berg M, Morrison L, et al. Hemorrhage from cavernous malformations of the brain: definition and reporting standards. Angioma Alliance Scientific Advisory Board. Stroke. 2008;39:3129–30.
Maraire J, Awad I. Intracranial cavernous malformations: lesion behavior and management strategies. Neurosurgery. 1995;37:591–605.
Bilginer B, Narin F, Hanalioglu S, Oguz KK, Soylemezoglu F, Akalan N. Cavernous malformations of the central nervous system (CNS) in children: clinico-radiological features and management outcomes of 36 cases. Childs Nerv Syst. 2014;30(8):1355–66.
Al-Holou WN, O’Lynnger TM, Pandey AS, Gemmete JJ, Thompson BG, Muraszko KM, Garton HJ, Maher CO. Natural history and imaging prevalence of cavernous malformations in children and young adults. J Neurosurg Pediatr. 2012;9(2):198–205.
Ghali MG, Srinivasan VM, Mohan AC, Jones JY, Kan PT, Lam S. Pediatric cerebral cavernous malformations: genetics, pathogenesis, and management. Surg Neurol Int. 2016;7(Suppl 44):S1127–34.
Raychaudhuri R, Batjer HH, Awad IA. Intracranial cavernous angioma: a practical review of clinical and biological aspects. Surg Neurol. 2005;63(4):319–28.
Pollock BE, Garces YI, Stafford SL, Foote RL, Schomberg PJ, Link MJ. Stereotactic radiosurgery for cavernous malformations. J Neurosurg. 2000;93:987–91.
Porter PJ, Willinsky RA, Harper W, Wallace MC. Cerebral cavernous malformations: natural history and prognosis after clinical deterioration with or without hemorrhage. J Neurosurg. 1997;87:190–7.
Del Curling O Jr, Kelly DL Jr, Elster AD, Craven TE. Ananalysis of the natural history of cavernous angiomas. J Neurosurg. 1991;75:702–8.
Barrow D, Krisht A. Cavernous malformations and hemorrhage. In: Awad I, Barrow D, editors. Cavernous malformations. Park Ridge: AANS; 1993. p. 65–80.
Ueda S, Saito A, Inomori S, et al. Cavernous angioma of the cauda equina producing subarachnoid hemorrhage. Case report. J Neurosurg. 1987;66:134–6.
Hubert P, Choux M, Houtteville J. Cerebral cavernomas in infants and children. Neurochirurgie. 1989;35:104–5.
Mazza C, Scienza R, Bernardin BD, et al. Cerebral cavernous malformations (cavernomas) in children. Neurochirurgie. 1989;35:106–8.
Scott R, Barnes P, Kupsky W, et al. Cavernous angiomas of the central nervous system in children. J Neurosurg. 1992;76:38–46.
Shirvani M, Hajimirzabeigi A. Intraventricular cavernous malformation: review of the literature and report of three cases with neuroendoscopic resection. J Neurol Surg A Cent Eur Neurosurg. 2017;78(3):269–80.
Li D, Hao SY, Tang J, Xiao XR, Jia GJ, Wu Z, Zhang LW, Zhang JT. Clinical course of untreated pediatric brainstem cavernous malformations: hemorrhage risk and functional recovery. J Neurosurg Pediatr. 2014;13(5):471–83.
Di Rocco C, Tamburrini G, Rollo M. Cerebral arteriovenous malformations in children. Acta Neurochir. 2000;142:145–56.
Abla AA, Lekovic GP, Garrett M, Wilson DA, Nakaji P, Bristol R, Spetzler RF. Cavernous malformations of the brainstem presenting in childhood: surgical experience in 40 patients. Neurosurgery. 2010;67(6):1589–98. discussion 1598–9.
Smit LM, Halbertsma FJ. Cerebral cavernous hemangiomas in childhood. Clinical presentation and therapeutic considerations. Childs Nerv Syst. 1997 Oct;13(10):522–5.
McCormick W. The pathology of vascular malformations. J Neurosurg. 1966;24:807–16.
Rosenow F, Alonso-Vanegas MA, Baumgartner C, et al. Cavernoma-related epilepsy: review and recommendations for management. Report of the Surgical Task Force of the ILAE Commission on therapeutic strategies. Epilepsia. 2013;54:2025–35.
Robinson J, Awad I, Little J. Natural history of the cavernous angioma. J Neurosurg. 1991;75:709–14.
Buckingham MJ, Crone KR, Ball WS, Berger TS. Management of cerebral cavernous angiomas in children presenting with seizures. Childs Nerv Syst. 1989;5(6):347–9.
Dammann P, Schaller C, Sure U. Should we resect peri-lesional hemosiderin deposits when performing lesionectomy in patients with cavernoma-related epilepsy (CRE)? Neurosurg Rev. 2017;40(1):39–43.
Del Curling O Jr, Kelly DL Jr, Elster AD, et al. An analysis of the natural history of cavernous angiomas. J Neurosurg. 1991;75:702–8.
Awad I, Jabbour P. Cerebral cavernous malformations and epilepsy. Neurosurg Focus. 2006;21(1):e7.
Gross BA, Lin N, Du R, et al. The natural history of intracranial cavernous malformations. Neurosurg Focus. 2011;30(6):E24.
Giombini S, Morello G. Cavernous angiomas of the brain. Account of fourteen personal cases and review of the literature. Acta Neurochir. 1978;40:61–82.
Jewell NP. Natural history of diseases: statistical designs and issues. Clin Pharmacol Ther. 2016;100(4):353–61.
Flemming KD. Predicting the clinical behaviour of cavernous malformations. Lancet Neurol. 2012;11:202–3.
Barker F, Amin-Hanjani S, Butler W, Lyons S, Ojemann RG, Chapman PH, et al. Temporal clustering of hemorrhages from untreated cavernous malformations of the central nervous system. Neurosurgery. 2001;49:15–25.
Mathiesen T, Edner G, Kihlstrom L. Deep and brainstem cavernomas: a consecutive 8-year series. J Neurosurg. 2003;99:31–7.
Wang W, Liu A, Zhang J, et al. Surgical management of brainstem cavernous malformations: report of 137 cases. Surg Neurol. 2003;59:444–4542.
Fritschi J, Reulen H, Spetzler R, et al. Cavernous malformations of the brain stem. A review of 139 cases. Acta Neurochir. 1994;130:35–46.
Kondziolka D, Monaco EA 3rd, Lunsford LD. Cavernous malformations and hemorrhage risk. Prog Neurol Surg. 2013;27:141–6.
Abla AA, Lekovic GP, Turner JD, de Oliveira JG, Porter R, Spetzler RF. Advances in the treatment and outcome of brainstem cavernous malformation surgery: a single-center case series of 300 surgically treated patients. Neurosurgery. 2011;68(2):403–14. discussion 414–5.
Garrett M, Spetzler RF. Surgical treatment of brainstem cavernous malformations. Surg Neurol. 2009;72(Suppl 2):S3–9. discussion S9–10.
Mottolese C, Hermier M, Stan H, Jouvet A, Saint-Pierre G, Froment JC, Bret P, Lapras C. Central nervous system cavernomas in the pediatric age group. Neurosurg Rev. 2001;24(2–3):55–71. discussion 72–3.
Van Lindert EJ, Tan TC, Grotenhuis JA, Wesseling P. Giant cavernous hemangiomas: report of three cases. Neurosurg Rev. 2007;30:83–92.
Jellinger K. Vascular malformations of the central nervous system: a morphological overview. Neurosurg Rev. 1986;9:177–216.
McCormick W. Pathology of vascular malformations of the brain. In: Wilson C, Stein B, editors. Intracranial arteriovenous malformations. Baltimore: Williams and Wilkins; 1984. p. 44–63.
Kupersmith MJ, Kalish H, Epstein F, Yu G, Berenstein A, Woo H, et al. Natural history of brainstem cavernous malformations. Neurosurgery. 2001;48:47–54.
Porter RW, Detwiler PW, Spetzler RF, Lawton MT, Baskin JJ, Derksen PT, et al. Cavernous malformations of the brainstem: experience with 100 patients. J Neurosurg. 1999;90:50–8.
Labauge P, Brunereau L, Lévy C, Laberge S, Houtteville JP. The natural history of familial cerebral cavernomas: a retrospective MRI study of 40 patients. Neuroradiology. 2000;42:327–32.
Cantu C, Murillo-Bonilla L, Arauz A, Higuera J, Padilla J, Barinagarrementeria F. Predictive factors for intracerebral hemorrhage in patients with cavernous angiomas. Neurol Res. 2005;27:314–8.
Kondziolka D, Lunsford LD, Kestle JR. The natural history of cerebral cavernous malformations. J Neurosurg. 1995;83:820–4.
Moriarity JL, Wetzel M, Clatterbuck RE, Javedan S, Sheppard JM, Hoenig-Rigamonti K, et al. The natural history of cavernous malformations: a prospective study of 68 patients. Neurosurgery. 1999;44:1166–73.
Sarwar M, McCormick WF. Intracerebral venous angioma. Case report and review. Arch Neurol. 1978;35:323–5.
McCormick WF. Intracerebral venous angioma. Case report and review. Arch Neurol. 1978;35:323–5.
Abdulrauf SI, Kaynar MY, Awad IA. A comparison of the clinical profile of cavernous malformations with and without associated venous malformations. Neurosurgery. 1999;44:41–7.
Cakirer S. De novo formation of a cavernous malformation of the brain in the presence of a developmental venous anomaly. Clin Radiol. 2003;58:251–6.
Campeau NG, Lane JI. De novo development of a lesion with the appearance of a cavernous malformation adjacent to an existing developmental venous anomaly. AJNR Am J Neuroradiol. 2005;26:156–9.
Detwiler PW, Porter RW, Zabramski JM, Spetzler RF. De novo formation of a central nervous system cavernous malformation: implications for predicting risk of hemorrhage. Case report and review of the literature. J Neurosurg. 1997;87:629–32.
Pozzati E, Acciarri N, Tognetti F, Marliani F, Giangaspero F. Growth, subsequent bleeding, and de novo appearance of cerebral cavernous angiomas. Neurosurgery. 1996;38:662–70.
Olivecrona H, Riives J. Arteriovenous aneurysms of the brain, their diagnosis and treatment. Arch Neurol Psychiatr. 1948;59:567–602.
Humphreys RP, Hoffman HJ, Drake JM, Rutka JT. Choices in the 1990s for the management of pediatric cerebral arteriovenous malformations. Pediatr Neurosurg. 1996;25:277–85.
LeBlanc R, Ethier R, Little JR. Computerized tomography findings in arteriovenous malformations of the brain. J Neurosurg. 1979;51:765–72.
Takahashi S, Sonobe M, Shirane R, Kubota Y, Kawakami H. Computer tomography of ruptured intracranial arteriovenous malformations in the acute stage. Acta Neurochir. 1982;66:87–94.
Ozgen B, Senocak E, Oguz KK, Soylemezoglu F, Akalan N. Radiological features of childhood giant cavernous malformations. Neuroradiology. 2011;53(4):283–9.
Nieto J, Hinojosa J, Muñoz MJ, Esparza J, Ricoy R. Intraventricular cavernoma in pediatric age. Childs Nerv Syst. 2003;19(1):60–2.
Katayama Y, Tsubokawa T, Maeda T, Yamamoto T. Surgical management of cavernous malformation of the third ventricle. J Neurosurg. 1994;80:64–72.
Reyns N, Assaker R, Louis E, Lejeune JP. Intraventricular cavernomas: three cases and review of the literature. Neurosurgery. 1999;44:648–53.
Tatagiba M, Schönmayr R, Samii M. Intraventricular cavernous angioma. A survey. Acta Neurochir. 1991;110:140–5.
Chusid JG, Kopeloff LM. Epileptogenic effects of pure metals implanted in motor cortex of monkeys. J Appl Physiol. 1962;17:697–700.
Kwon CS, Sheth SA, Walcott BP, Neal J, Eskandar EN, Ogilvy CS. Long-term seizure outcomes following resection of supratentorial cavernous malformations. Clin Neurol Neurosurg. 2013;115:2377–81.
Schramm J, Aliashkevich AF. Surgery for temporal mediobasal tumors: experience based on a series of 235 patients. Neurosurgery. 2008;62(6 Suppl 3):1272–82.
Shan YZ, Fan XT, Meng L, An Y, Xu JK, Zhao GG. Treatment and outcome of epileptogenic temporal cavernous malformations. Chin Med J. 2015;128:909–13.
Van Gompel JJ, Rubio J, Cascino GD, Worrell GA, Meyer FB. Electrocorticography-guided resection of temporal cavernoma: is electrocorticography warranted and does it alter the surgical approach? J Neurosurg. 2009;110:1179–85.
Yeon JY, Kim JS, Choi SJ, Seo DW, Hong SB, Hong SC. Supratentorial cavernous angiomas presenting with seizures: surgical outcomes in 60 consecutive patients. Seizure. 2009;18:14–20.
Ormond DR, Clusmann H, Sassen R, Hoppe C, Helmstaedter C, Schramm J, Grote A. Pediatric temporal lobe epilepsy surgery in Bonn and review of the literature. Neurosurgery. 2019;84(4):844–56.
Smith ER, Scott RM. Cavernous and venous malformations. In: Albright AL, Pollack IF, Adelson PD, editors. Principles and practice of pediatric neurosurgery. Third ed. New York: Thieme Medical Publishers; 2015. p. 858–65.
Gross BA, Batjer HH, Awad IA, Bendok BR, Du R. Brainstem cavernous malformations: 1390 surgical cases from the literature. World Neurosurg. 2013;80(1–2):89–93.
Sager O, Beyzadeoglu M, Dincoglan F, et al. Evaluation of linear accelerator (LINAC)-based stereotactic radiosurgery (SRS) for cerebral cavernous malformations: a 15-year single-center experience. Ann Saudi Med. 2014;34(1):54–8.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Akalan, N. (2020). Cavernomas in Children. In: Bradáč, O., Beneš, V. (eds) Cavernomas of the CNS. Springer, Cham. https://doi.org/10.1007/978-3-030-49406-3_12
Download citation
DOI: https://doi.org/10.1007/978-3-030-49406-3_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-49405-6
Online ISBN: 978-3-030-49406-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)