Abstract
Conjoined twins (CT) are a rare complication of monoamniotic twinning. The true incidence of this complication is difficult to estimate because most women with the diagnosis either choose not to continue the pregnancy or an intrauterine demise ensues. For continuing pregnancies, prognosis is mostly determined by the site fusion and the organs involved. Ultrasound, 3D ultrasound, and magnetic resonance imaging are critical for antenatal diagnosis, careful analysis of the area of fusion, and subsequent prognostication. Only a minority of twins that survive birth will go on to be candidates for surgical separation. Surgical separation should only be attempted at centers with expertise in these procedures, which are complex and frequently associated with residual morbidity. Because the ethical considerations of surgical separation are complex, a palliative care team is critical for understanding patient and her family’s goals and preferences.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Conjoined twins
- Monoamniotic twins
- Surgical separation
- Twin embryology
- Thoracopagus twins
- Omphalopagus twins
- Cephalopagus twins
- Ischiopagus twins
- Craniopagus twins
- Parapagus twins
- Rachipagus twins
- Pygopagus twins
Conjoined twins (CT) are a rare complication of monoamniotic twinning. The true incidence of this complication is difficult to estimate because most women with the diagnosis either choose not to continue the pregnancy or an intrauterine demise ensues. For continuing pregnancies, prognosis is mostly determined by the site fusion and the organs involved. Ultrasound, 3D ultrasound, and magnetic resonance imaging are critical for antenatal diagnosis, careful analysis of the area of fusion, and subsequent prognostication. Only a minority of twins that survive birth will go on to be candidates for surgical separation. Surgical separation should only be attempted at centers with expertise in these procedures, which are complex and frequently associated with residual morbidity. Because the ethical considerations of surgical separation are complex, a palliative care team is critical for understanding patient and her family’s goals and preferences.
FormalPara DefinitionsConjoined twins: A rare complication of monoamniotic twins in which there is fusion between anatomical structures of each twin. Specific organs along the site of fusion may be paired (i.e., one organ for each twin) or singular (i.e., one organ shared between twins).
Thoracopagus twins: The most common type of conjoined twins; they are fused anteriorly from the thorax to the umbilicus typically sharing cardiac structures.
Omphalopagus twins: A type of conjoined twins that are fused anteriorly typically from the sternum to the umbilicus. They are differentiated from thoracopagus twins in that they do not share a heart.
Cephalopagus twins: A type of conjoined twins that are fused primarily at the head and typically share cerebral structures. There are several possible orientations between the twins and fusion may extend rostrally to the level of the umbilicus.
Ischiopagus twins: A type of conjoined twins that are fused at the pelvic outlet. Fusion may extend caudally to the level of the diaphragm. Face-to-face and end-to-end orientations are possible.
Craniopagus twins: A type of conjoined twins that are fused at the head. Usually the cranium and meninges are involved; however, cerebral vasculature may also sometimes be involved. Fusion is not present below the head.
Pygopagus twins: A type of conjoined twins that are fused dorsally at the sacrum.
Parapagus twins: A type of conjoined twins that are fused anterolaterally with side-by-side orientation. The size of the fusion area and organs involved may vary dramatically between pairs of twins of this type.
Rachipagus twins: A type of conjoined twins that are fused posteriorly with the twins oriented facing away from one another. One of the twins is often parasitic.
FormalPara Learning Objectives-
A general understanding of the history, pathophysiology, embryology, and management of conjoined twins.
-
Awareness of the eight types of conjunction and how the site and complexity of fusion dictate the prognosis and availability of postnatal surgical separation.
-
An understanding of the roles of antenatal ultrasound, 3D ultrasound, and magnetic resonance imaging for delineation the organs involved in conjunction.
-
An overview of postnatal management of conjoined twins that survive birth. This includes non-operative management, emergent surgical separation, and interval elective separation.
-
An overview of the ethical issues involved in postnatal management.
1 Introduction
Conjoined twins (CT) are a rare complication of monochorionic twining historically viewed as a medical curiosity more than as a manageable condition. This has changed somewhat as successful surgical separation has become possible in selective cases with improved prenatal diagnosis and coordination of care. The true incidence of CT is unknown. Most estimates range between 1:50,000 and 1:250,000 live births; however, several recent reports suggest that the true incidence is more likely to be between 1:20,000 and 1:25,000 [1,2,3,4,5,6,7]. In perhaps the largest epidemiological study of CT, Mutchinick et al. calculated an incidence of 1.47:100,000 from a worldwide sample of 26.1 million births [8]. There is regional variation in rates of CT with greater numbers reported in China and Africa (>1:30,000) than in Europe and the United States (<1:50,000) [8]. Female CT are three times more common than male CT.
The first known historical artifact depicting CT is a 17-cm marble statue of parapagus twins, the so-called sisters of Catalhoyuk, dated 6000 BC; it is currently housed in the Anatolian Civilization Museum in Ankara, Turkey [9]. However, a few well-known historical examples of CT dominate the historical landscape. The “Biddenden Maids,” were a pair of parapagus sisters born in 1100 in the English agricultural village of Biddenden in the state of Kent. These are the first CT with an extensive historical literature describing their life and times. The twins lived for 34 years fused at the shoulders and hips prior to their demise within 6 hours of one another from an unknown illness [10]. They currently enjoy legendary status in Biddenden. The brothers Chang and Eng Bunker, born in Siam in 1911, are probably the best-known CT. The unfortunate story of their exploitation by PT Barnum at “Freak Shows” across Europe and the United states followed by coinage of the indecorous term “Siamese Twins” has contributed negatively to our cultural understanding and acceptance of CT [11, 12]. The medical circumstances underlying these stories are no less fascinating than the tales themselves.
2 Types of Conjoined Twins
The nomenclature of CT, as well as their prenatal and postnatal clinical management and outcomes, are dictated by their shared anatomy. The terminology describing conjunction has evolved, but eventually settled on eight types of twins proposed by Spencer et al., each ending with the suffix “pagus,” which means “fixed.” The types, in order of estimated incidence, are thoracopagus, omphalopagus, cephalopagus, ischiopagus, pygopagus, craniopagus, parapagus (diprosopus and dicephalus), and rachipagus [13]. Each of these types of CT is illustrated in ◘ Fig. 19.1.
Spencer classification of conjoined twins [13]
Thoracopagus twins (◘ Fig. 19.1a) are the most common subtype in most large series, accounting for 47–71% of the total [3, 14,15,16,17,18,19,20,21]. These twins usually lie face-to-face, fused from the anterior thorax to the umbilicus; they share a sternum, diaphragm, and upper abdominal wall and the majority share a pericardial sac, heart, and liver [22,23,24]. For twins with more extensive fusion inferiorly, the term thoraco-omphalopagus is sometimes used. Conjoined hearts, which are common in this group, are nearly always affected by additional major anomalies (◘ Fig. 19.2). In a recent series of 10 thoracopagus twins with conjoined hearts, every twin had additional cardiac anomalies including tricuspid atresia, double outlet right ventricle, transposition of the great arteries, hypoplastic left heart, complete atrioventricular canal defects, and totally anomalous pulmonary venous return among others [23]. This represents the primary source of mortality among these twins. Fusion of the extra-hepatic biliary system has also been described [25, 26].
Two sets of thoracopagus twins a 3D ultrasound image of the first pair showing anterior fusion from the sternum to the umbilicus. b 2D transverse ultrasound image showing a single shared heart with six chambers centrally located in the fusion between twins. c MRI showing the second pair fused more laterally than the first from the lower face to the pelvis. A liver and urinary bladder are shared; however, there are two hearts. d MRI showing the fused liver, which is outside of the abdomen
Omphalopagus twins (◘ Fig. 19.1b) make up about 20% of all CT. They are situated identically to thoracopagus twins (i.e., rostral fusion), but they have a less complex conjunction. Most of these twins share a liver (80%), and about a third share the terminal ileum and colon without rectal involvement [13, 27, 28]. There are always separate hearts (by definition), although the pericardium may be shared. Numerous case reports describe shared biliary anatomy and fusion extends from the xiphoid to the umbilicus [25]. Because they have the least complex conjunction among CT, they are more likely to survive postnatal surgical separation.
Cephalopagus twins (◘ Fig. 19.1c) are primarily joined at the head. Four varieties of facial presentation may occur including symmetric (two well-formed faces on opposite sides of the head), asymmetric (one well-formed face and one hypotrophic face on opposite sides), deradelphous (one midline face), and deradelphous diprosopous (one midline face with duplication of only some facial features) [29]. There is only one brain; however, some components may be duplicated (i.e., two cerebellums, ◘ Fig. 19.3). The fusion may extend rostrally as far as the umbilicus. When the thorax is involved, the twins tend to have separate hearts, lungs, and trachea [30, 31]. When fusion extends lower, the twins usually share the upper gastrointestinal tract with separate ileums, large bowels, and rectums [27, 32]. Cephalopagus twins are inseparable and frequently stillborn. Those that do survive delivery uniformly fail to survive the neonatal period.
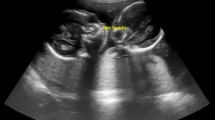
Cephalopagus twins fused from the head to the pelvis. a 3D ultrasound image showing anterior fusion of the cranium. There are four ears and one face. b MRI showing the twins fused to the umbilicus. There is fused liver and single heart with two spines. c Transverse 2D ultrasound image showing a single irregularly shaped cranium. There was a complex brain anomaly with two cerebral hemispheres, a single central ventricle, and two cerebellums. d Coronal ultrasound image showing a single liver with bilateral hyperextended spines
Ischiopagus twins (◘ Figs. 19.1d) are joined primarily at the pelvic outlet; however, fusion may present up to the level of the diaphragm and often includes the liver; the twins may be oriented face-to-face or end-to-end (◘ Fig. 19.4). This leads to tremendous variability in the angle between the twins’ spines and shared anatomy [28]. They may share portions of the cauda equina, thecal sac, and vertebral bodies [33]. Hemivertebrae and spinal lesions remote from the site of union are also common. There may be two, three, or four lower extremities (dipus, tripus, or tetrapus) and a wide variety of shared and unshared permutations of urinary, genital, and lower gastrointestinal tracts have been reported [33,34,35,36]. These include shared urethras and bladders (with varying numbers of ureteral orifices), ureters crossing between twins, cloacas, fused or shared genitals, and anal orifices with or without separate rectums. While the thoraces are separate, cardiac anomalies are present about 10% of the time [28]. These twins are frequently able to be separated when the anatomy if favorable [18].
Pygopagus twins (◘ Fig. 19.1e) are joined dorsally at the sacrum. They have similar urinary, lower gastrointestinal, and neurological malformations to ischiopagus twins without involvement of more superior structures. The twins usually share an anus with or without a common rectum and either one or two urinary bladders [28, 33]. The degree of spinal cord fusion typically determines whether separation is possible. Pygopagus and ischiopagus twins often have long-term urinary and bowel issues in cases of successful separation [33, 36].
Craniopagus twins (◘ Fig. 19.1f), like cephalopagus twins, are joined at the head but fusion typically involves only the skull and meninges while the face and brain are spared. However, cortical fusion and shared cerebral vasculature (typically venous) will be seen in upwards of 1/3 of cases. Numerous subclassification systems have been proposed based on the directions the twins are facing, the structures shared, and presence of dural venous communication [37]. While fusion is limited to the head, the majority of these twins have additional issues including cardiac, facial, and gastrointestinal anomalies [37]. Staged separation of these twins with a multidisciplinary team is possible with success often hinging on the degree of dural sinus communication [38,39,40].
Parapagus twins are fused anterolaterally with extremely variable organ involvement that may include the head, thorax, abdomen, or pelvis; they are positioned side by side [41,42,43,44]. The pelvis and abdomen are almost always involved with fusion of the liver and diaphragm; there may be two, three, or four upper limbs. Pelvic involvement may include anal atresia, colovesical fistula, and genitourinary abnormalities [45]. In parapagus disopropus CT, the fusion extends from the pelvis to the cranium, which is typically lethal (◘ Fig. 19.1h). The twins are deemed dithoracic if the pelvis and abdomen are fused with separation above the abdomen. However, when the twins are dicephalic (fused abdomen and thoraces), cardiac anomalies, and often a single heart, are common (◘ Figs. 19.1g and 19.5) [14, 46].
Rachipagus twins (◘ Fig. 19.1i) are fused posteriorly facing away from one another. The occiput and vertebral bodies are involved to varying degrees, and one of the twins is often parasitic [47,48,49]. These are the rarest of CT and our understanding of their fusion is limited to case reports [47, 48, 50].
3 Pathophysiology/Embryology
The pathophysiology of CT is unknown, and currently available theories do not explain all clinical observations. Nevertheless, the two prevailing theories of CT are fission of a single embryo versus fusion of two early embryos [51, 52]. The fission theory classically accounts for all major types of twins (dichorionic-diamniotic, monochorionic-diamniotic, monochorionic-monoamniotic, and conjoined) and argues that phenotype is determined by the embryological timing of the fission event [52].
According to the fusion theory, conjunction between twins lies along specific anatomic sites where embryological folding or closure occurs and ectoderm is at least transiently absent. Such sites include the diaphragm, oropharyngeal membrane, cloacal membrane, neural tube, and the periumbilical region [51, 53]. The events leading to union occur between 4 and 6 weeks after fertilization when the yolk sac is still prominent. Spencer proposed a “spherical theory” of union in which the two embryos’ relative positions on the surface of the yolk sac determine the final configuration [51]. Comparison of the two theories is beyond the scope of this text (reviewed by Spencer); however, some authors believe fusion to be more plausible in light of various clinical observations including cases of diamniotic CT [51, 53, 54]. Regardless of which theory is best describes clinical observations, the practical implication is that the process occurs early and CT are generally detectable at the first obstetric ultrasound.
4 Imaging for Detection of Conjoined Twins
CT are readily diagnosed in the first trimester of pregnancy by ultrasound as illustrated in a review of 54 cases diagnosed prior to 15 weeks. Only 18 pairs were suspected before 10 weeks with four of these being false positives [55]. However, diagnosis as early as 7 weeks has been reported [56]. The primary diagnosis in the case of false positives is monochorionic-monoamniotic pregnancy; the distinction between these diagnoses is often made in the late first trimester when fetal movement increases but the fetuses are not found to move independent of one another. The following findings, suspicious for a diagnosis of CT, have been suggested by several authors: two fetuses emerging from a single yolk sac, fetuses lying in the same constant relative position with body parts at the same level, an odd number limbs, shared organs, and single umbilical cords with more than three vessels [45, 57, 58]. Interestingly, increased nuchal translucencies are a common finding in thoracopagus twins thought to be caused by the single hearts’ failure to prevent lymphatic stasis in the posterior necks [59]. First trimester detection of CT is paramount to offering early termination, or in cases of triplet pregnancy, selective reduction of the conjoined pair [60,61,62].
In continuing pregnancies, prenatal imaging is critical for prognostication and planning of surgical separation. A comprehensive anatomy ultrasound at 20 weeks is the first step for estimating the extent of fusion and the organs involved. Fetal echocardiography should be performed in all CT as there is increased risk of heart anomalies even if the chest is not involved in the fusion; severity of cardiac lesions is often underestimated even with detailed prenatal imaging [57, 63]. Three-dimensional ultrasound may be useful in better defining the anatomy of fusion (◘ Figs. 19.2a and 19.3a); however, examples of this is clinically useful are limited to case reports [64,65,66,67,68]. Fetal MRI is increasingly used to provide an enhanced depiction of organ sharing and vascular anatomy. It has become an important tool for prenatal counseling and optimized planning of postnatal surgical management in continuing cases [15, 69,70,71] (◘ Figs. 19.2c, d and 19.3b).
5 Prenatal Issues and Management
Hundreds of documented cases of successfully separated CT and the surgical advancements leading to them have increased optimism for surgical management of CT; however, it is important to emphasize that these represent the minority of cases. In a large series of 36 pairs of CT, 30 (83.3%) were thought to have lethal anomalies [14]. Fourteen pregnancies were terminated after the diagnosis. Among the 16 twins born alive, none survived with seven expiring within 24 hours. In another series of 17 pairs of CT that survived to delivery, five were determined to be inoperable and all expired within 5 days. Seven pairs required emergency separation, of which four of 14 twins survived. The remaining 5 pairs underwent planned separation and experienced more favorable outcomes with eight twins attaining long-term survival [17]. Similarly, a series of 22 pairs of CT by Saguil et al. showed 64% overall mortality for twins born alive [16].
The stillbirth rates for CT are difficult to determine as frequently only live-born twins are reported. Furthermore, most series are from high-volume centers, which lack information regarding fetal demise prior to referral. The high incidence of stillbirth was better emphasized by Martinez-Frias et al., who found rates of stillborn CT to be 99.3 times that of live-born CT in a large database from Spain including 30 years of data [3]. True stillbirth estimates are confounded by the large number of women who terminate CT. In a review of 75 sets of CT detected in the first trimester, Chen et al. reported a termination rate of 80% [72]. This serves as a recapitulation of data from Pajkrt et al. who reported an 82% (41/50) termination rate among CT detected in the first trimester [55].
Termination of pregnancy should clearly be offered to any patient presenting with CT. There are limited data to inform the technical aspects of termination in these cases; however, routine dilation and curettage or evacuation are currently used for this process. After 20 weeks gestational age, transvaginal termination of CT may become technically difficult and hysterotomy may be required [55]. Hence, early detection is critical. While termination by vaginal delivery following induction has been described in the second trimester, this is not ideal [73].
For women who choose to continue the pregnancy, there are similarly scant data to guide antenatal monitoring. Given the high rates of stillbirth, serial ultrasound monitoring should be a cornerstone of any antenatal surveillance plan. As with all monoamniotic twins, delivery by planned Cesarean section likely carries less morbidity than vaginal delivery for these twins [74]. While successful vaginal delivery has been described in the literature, so too have associated birth traumas [75,76,77]. The optimal timing of delivery is unknown, but planned delivery at 35 weeks gestational age following administration of corticosteroids has been recommended [78].
6 Postnatal Management
The management of live-born CT is complex and specific to the site of fusion. Early referral to a tertiary care center with expertise in surgical separation is recommended for twins thought to be potential candidates. Separation requires a well-coordinated multidisciplinary approach with extensive planning and involvement of a variety of pediatric surgical subspecialties. There are three major categories of management for live-born CT:
-
1.
Non-operative management: These cases usually have complex neurological or cardiac fusion such that separation is likely to be lethal or severely disabling. Thoracopagus twins with heart fusion and/or associated complex heart anomalies and craniopagus, cephalopagus, or parapagus disopropus twins with complex cerebral fusion usually fall into this category [23, 79]. These twins are candidates for palliative care, and most survive less than a few days after birth.
-
2.
Emergent surgical separation: Several scenarios may arise where separation must be urgently undertaken. These include cases where one twin has died or has a lethal anomaly, cases with acute injury at the site of fusion, and cases where an omphalocele is present with necrotic tissue. Survival following emergent separation ranges from 8% to 50% [9, 14, 16, 17, 57, 80,81,82]. The ethical considerations in these cases may be complex. Generally, in cases where both twins may be salvageable, an equal chance should be given to each. In cases where only one twin is salvageable, it is reasonable to make decisions that increase likelihood of survival in one twin with appropriate palliative care provided to other twin [79].
-
3.
Interval elective separation: Carefully selected CT that survive the peripartum period have survival rates between 71% and 100% in experienced hands [9, 14, 16, 17, 57, 80,81,82]. Such outcomes are more likely for omphalopagus, ischiopagus, craniopagus (without cerebral involvement), and pygopagus twins, although there are examples of successful separation of thoracopagus twins [16]. Separation is usually performed between 6 and 24 months of age. Ethical considerations in this group are also very complex.
CT that survive planned surgical separation frequently have long-term medical complications and varying degrees of disability. Pygopagus and ischiopagus twins, for example, may require extensive post-separation neurologic surgery for a variety of issues including spinal column anomalies, tethered cords, syrinx, and hydrocephalus [33, 83]. To address downstream complications of urinary and lower gastrointestinal tract fusion, they may undergo ileostomy, colostomy, urinary diversion, and bladder augmentation among other procedures [33, 36, 83]. Complications following separation of fused hearts have led some groups towards palliation for thoracopagus twins with complex cardiac lesions [23]. Prediction of long-term sequelae following surgical separation of CT is a difficult task. Data are limited, and each pair of twins will have a unique set of post-operative challenges. This further highlights the importance of early referral to a specialized center with experienced multidisciplinary pediatric subspecialists.
6.1 Ethical Considerations
The ethics of managing CT is multifaceted and specific to each set of twins. A practical decision-oriented approach offered by Thomas et al. relies on quantifying the risk of morbidity and mortality of each twin prior to, versus after separation [84]. In cases where both twins are not expected to survive without separation, separation is obligatory as long as at least one survivor without major morbidity is expected. This may involve risk to, or sacrifice of, one of the pair for the other’s survival. If major morbidity is expected, separation may not be obligatory, but permissible depending on the values and desires of the patient and her family. If, on the other hand, the twins are expected to survive if they remain conjoined, separation is impermissible if one or both twins are expected to die following the procedure. If both twins are expected to survive, then morbidity and patient preferences become relevant to the permissibility of proceeding with separation. In all cases, involvement of a palliative care team is critical for understanding patient goals and preferences and aiding in communication between the patients (or their family) and the care team.
7 Conclusion
CT have captured the imagination of physicians as well as the general public throughout history. This is highlighted by renowned historic examples like the Bunker brothers and the Biddenden Maids. Behind these stories lie an extremely morbid, often lethal and always complex disease. The most common outcome of CT is termination of pregnancy followed by stillbirth and then neonatal demise. The embryology of the disease remains largely speculative, and the epidemiology is difficult to study. Treatment is available in a minority of well-selected cases, and post-separation complications are common.
From a diagnostic standpoint, CT are generally detectable during routine first trimester ultrasound. Prognostication may be improved by more advanced imaging techniques such as 3D ultrasound, fetal echocardiogram, and fetal MRI. Improvements in antenatal care may result from initiatives that promote early establishment of prenatal care and ultrasound. In the same vein, unhindered access to termination of pregnancy is of critical importance. For women who choose to continue their pregnancy, referral to an established and experienced tertiary care center is essential. Finally, advances in surgical techniques may allow more complicated separations to be achieved, but this will affect only a minority of cases.
7.1 Review Questions
-
1.
What are the two primary theories describing the embryologic pathophysiology of CT?
-
2.
What are the roles of ultrasound, 3D-ultrasound and fetal MRI in diagnosing and prognosticating cases of CT?
-
3.
What are the most common two outcomes for CT diagnosed in the first trimester?
-
4.
What are the three major categories of postnatal management for CT that survive delivery? What criteria should physicians use to determine the best management strategy?
-
5.
What are the primary ethical considerations in determining whether and when to surgically separate a pair of CT?
7.2 Multiple-Choice Questions
-
1.
What is the most likely outcome for the majority of conjoined twins of any type?
-
(a)
Termination of pregnancy or intrauterine fetal demise.
-
(b)
Live birth with demise shortly after birth.
-
(c)
Live birth with emergent separation surgery shortly after birth.
-
(d)
Live birth with interval elective separation surgery.
Answer: (a)
-
(a)
-
2.
Which of the following is most important to improve the likelihood of neonatal survival in women that choose to continue a pregnancy with a diagnosis of conjoined twins?
-
(a)
MRI and 3D ultrasound for improved prenatal delineation of the fusion site and organ involvement.
-
(b)
Early prenatal diagnosis and serial antenatal testing with non-stress tests, biophysical profiles, and Doppler interrogation of fetal vasculature.
-
(c)
Referral to a tertiary care center with expertise in surgical separation of conjoined twins.
-
(d)
A carefully considered plan for mode of delivery at an appropriate gestational age.
Answer: (c)
-
(a)
-
3.
Which of the following is true regarding the ethical considerations of surgical separation of conjoined twins.
-
(a)
Separation is obligatory, even if severe morbidity is expected after the procedure.
-
(b)
Involvement of a palliative team is a reasonable option, but not required, in the setting of planned surgical separation of twins.
-
(c)
Separation is obligatory in cases where both twins are expected to expire in the absence of intervention.
-
(d)
Separation is impermissible if it may involve severe morbidity or sacrifice of one of the twins.
Answer: (c)
-
(a)
References
Spitz L. Conjoined twins. Br J Surg. 1996;83(8):1028–30.
Edmonds LD, Layde PM. Conjoined twins in the United States, 1970–1977. Teratology. 1982;25(3):301–8.
Martinez-Frias ML, Bermejo E, Mendioroz J, et al. Epidemiological and clinical analysis of a consecutive series of conjoined twins in Spain. J Pediatr Surg. 2009;44(4):811–20.
Berezowski AT, Duarte G, Rodrigues R, et al. Conjoined twins: an experience of a tertiary hospital in Southeast Brazil. Rev Bras Ginecol Obstet. 2010;32(2):61–5.
Tang Y, Zhu J, Zhou GX, Dai L, Wang YP, Liang J. An epidemiological study on conjoined twins in China, from 1996 to 2004. Zhonghua Yu Fang Yi Xue Za Zhi. 2007;41(Suppl):146–9.
Liang J, Xu CI, Wang Y. Epidemiological survey of conjoined twins in China. Hua Xi Yi Ke Da Xue Xue Bao. 1999;30(1):56–8.
Savona-Ventura C, Grima S, Buttigieg GG. Conjoint twinning in the Maltese Islands. J Obstet Gynaecol. 2009;29(7):599–604.
Mutchinick OM, Luna-Munoz L, Amar E, et al. Conjoined twins: a worldwide collaborative epidemiological study of the International Clearinghouse for Birth Defects Surveillance and Research. Am J Med Genet C Semin Med Genet. 2011;157c(4):274–87.
Spitz L. Conjoined twins. Prenat Diagn. 2005;25(9):814–9.
Bondeson J. The Biddenden maids: a curious chapter in the history of conjoined twins. J R Soc Med. 1992;85(4):217–21.
Martin HE. Chang and Eng Bunker, “the original Siamese twins”: living, dying, and continuing under the spectator's gaze. J Am Cult (Malden). 2011;34(4):372–90.
Bogdan R. Freak show: presenting human oddities for amusement and profit. University of Chicago Press; 1990.
Spencer R. Anatomic description of conjoined twins: a plea for standardized terminology. J Pediatr Surg. 1996;31(7):941–4.
Brizot ML, Liao AW, Lopes LM, et al. Conjoined twins pregnancies: experience with 36 cases from a single center. Prenat Diagn. 2011;31(12):1120–5.
Mackenzie TC, Crombleholme TM, Johnson MP, et al. The natural history of prenatally diagnosed conjoined twins. J Pediatr Surg. 2002;37(3):303–9.
Saguil E, Almonte J, Baltazar W, et al. Conjoined twins in the Philippines: experience of a single institution. Pediatr Surg Int. 2009;25(9):775–80.
Spitz L, Kiely EM. Experience in the management of conjoined twins. Br J Surg. 2002;89(9):1188–92.
Tannuri AC, Batatinha JA, Velhote MC, Tannuri U. Conjoined twins: twenty years' experience at a reference center in Brazil. Clinics (Sao Paulo). 2013;68(3):371–7.
el-Gohary MA. Siamese twins in the United Arab Emirates. Pediatr Surg Int. 1998;13(2–3):154–7.
Castilla EE, Lopez-Camelo JS, Orioli IM, Sanchez O, Paz JE. The epidemiology of conjoined twins in Latin America. Acta Genet Med Gemellol. 1988;37(2):111–8.
Cywes S, Millar AJ, Rode H, Brown RA. Conjoined twins–the Cape Town experience. Pediatr Surg Int. 1997;12(4):234–48.
Hoyle RM. Surgical separation of conjoined twins. Surg Gynecol Obstet. 1990;170(6):549–62.
Thomas Collins R 2nd, Weinberg PM, Gruber PJ, St John Sutton MG. Conjoined hearts in thoracopagus twins. Pediatr Cardiol. 2012;33(2):252–7.
McMahon CJ, Spencer R. Congenital heart defects in conjoined twins: outcome after surgical separation of thoracopagus. Pediatr Cardiol. 2006;27(1):1–12.
Meyers RL, Matlak ME. Biliary tract anomalies in thoraco-omphalopagus conjoined twins. J Pediatr Surg. 2002;37(12):1716–9.
Saranrittichai S, Niramis R, Siripornadulsilp S, et al. Successful separation of thoracopagus conjoined twins with a single extra-hepatic biliary system. J Med Assoc Thail. 2007;90(5):994–1000.
McHugh K, Kiely EM, Spitz L. Imaging of conjoined twins. Pediatr Radiol. 2006;36(9):899–910. quiz 1002–1003
Winkler N, Kennedy A, Byrne J, Woodward P. The imaging spectrum of conjoined twins. Ultrasound Q. 2008;24(4):249–55.
Grassi R, Esposito V, Scaglione M, et al. Multi-detector row CT for depicting anatomic features of cephalothoracopagus varieties: revised approach. Radiographics. 2004;24(5):e21.
Kuroda K, Kamei Y, Kozuma S, et al. Cephalopagus conjoined twins. Ultrasound Obstet Gynecol. 2000;16(3):293.
Spencer R. Conjoined twins: theoretical embryologic basis. Teratology. 1992;45(6):591–602.
Mian A, Gabra NI, Sharma T, et al. Conjoined twins: from conception to separation, a review. Clin Anat. 2017;30(3):385–96.
Fieggen AG, Dunn RN, Pitcher RD, Millar AJ, Rode H, Peter JC. Ischiopagus and pygopagus conjoined twins: neurosurgical considerations. Childs Nerv Syst. 2004;20(8–9):640–51.
Shapiro E, Fair WR, Ternberg JL, Siegel MJ, Bell MJ, Manley CB. Ischiopagus tetrapus twins: urological aspects of separation and 10-year followup. J Urol. 1991;145(1):120–5.
Hoyle RM, Thomas CG Jr. Twenty-three-year follow-up of separated ischiopagus tetrapus conjoined twins. Ann Surg. 1989;210(5):673–9.
Lazarus J, Raad J, Rode H, Millar A. Long-term urological outcomes in six sets of conjoined twins. J Pediatr Urol. 2011;7(5):520–5.
Harvey DJ, Totonchi A, Gosain AK. Separation of craniopagus twins over the past 20 years: a systematic review of the variables that Lead to successful separation. Plast Reconstr Surg. 2016;138(1):190–200.
Dunaway D, Jeelani NU. Staged separation of craniopagus twins. Semin Pediatr Surg. 2015;24(5):241–8.
Staffenberg DA, Goodrich JT. Separation of craniopagus conjoined twins with a staged approach. J Craniofac Surg. 2012;23(7 Suppl 1):2004–10.
Pai KM, Naidu RC, Raja A, et al. Surgical nuances in the separation of craniopagus twins – our experience and a follow up of 15 years. Neurol India. 2018;66:426–33.
Watanabe K, Ono M, Shirahashi M, Ikeda T, Yakubo K. Dicephalus Parapagus conjoined twins diagnosed by first-trimester ultrasound. Case Rep Obstet Gynecol. 2016;2016:8565193.
Usang UE, Olasode BJ, Archibong AE, Udo JJ, Eduwem DA. Dicephalus parapagus conjoined twins discordant for anencephaly: a case report. J Med Case Rep. 2010;4:38.
Tansel T, Yazicioglu F. Cardiac and other malformations in parapagus twins. Arch Gynecol Obstet. 2004;269(3):211–3.
Melo A, Dinis R, Portugal A, Sousa AI, Cerveira I. Early prenatal diagnosis of parapagus conjoined twins. Clin Pract. 2018;8(2):1039.
Mathew RP, Francis S, Basti RS, et al. Conjoined twins – role of imaging and recent advances. J Ultrason. 2017;17(71):259–66.
Lopes LM, Brizot ML, Schultz R, et al. Twenty-five years of fetal echocardiography in conjoined twins: lessons learned. J Am Soc Echocardiogr. 2013;26(5):530–8.
Ratan SK, Rattan KN, Magu S, Rohilla S, Purwar P, Mathur SK. Thoracolumbar rachipagus parasite. Pediatr Surg Int. 2004;20(4):298–300.
Rattan KN, Singh J, Dalal P, Sonika P, Rattan A. Sacral Rachipagus parasite: a case report. J Neonatal Surg. 2016;5(2):16.
Chadha R, Lal P, Singh D, Sharma A, Choudhury SR. Lumbosacral parasitic rachipagus twin. J Pediatr Surg. 2006;41(1):e45–8.
Navaei AA, Habibi Z, Moradi E, Nejat F. Parasitic rachipagus twins; report of two cases. Childs Nerv Syst. 2015;31(6):1001–3.
Spencer R. Theoretical and analytical embryology of conjoined twins: part I: embryogenesis. Clin Anat. 2000;13(1):36–53.
Kaufman MH. The embryology of conjoined twins. Childs Nerv Syst. 2004;20(8–9):508–25.
Spencer R. Theoretical and analytical embryology of conjoined twins: part II: adjustments to union. Clin Anat. 2000;13(2):97–120.
Kapur RP, Jack RM, Siebert JR. Diamniotic placentation associated with omphalopagus conjoined twins: implications for a contemporary model of conjoined twinning. Am J Med Genet. 1994;52(2):188–95.
Pajkrt E, Jauniaux E. First-trimester diagnosis of conjoined twins. Prenat Diagn. 2005;25(9):820–6.
Hill LM. The sonographic detection of early first-trimester conjoined twins. Prenat Diagn. 1997;17(10):961–3.
Barth RA, Filly RA, Goldberg JD, Moore P, Silverman NH. Conjoined twins: prenatal diagnosis and assessment of associated malformations. Radiology. 1990;177(1):201–7.
Kingston CA, McHugh K, Kumaradevan J, Kiely EM, Spitz L. Imaging in the preoperative assessment of conjoined twins. Radiographics. 2001;21(5):1187–208.
Maymon R, Mendelovic S, Schachter M, Ron-El R, Weinraub Z, Herman A. Diagnosis of conjoined twins before 16 weeks' gestation: the 4-year experience of one medical center. Prenat Diagn. 2005;25(9):839–43.
Lam YH, Lee CP, Tang MH, Lau E. Thermocoagulation for selective reduction of conjoined twins at 12 weeks of gestation. Ultrasound Obstet Gynecol. 2000;16(3):267–70.
Goldberg Y, Ben-Shlomo I, Weiner E, Shalev E. First trimester diagnosis of conjoined twins in a triplet pregnancy after IVF and ICSI: case report. Hum Reprod. 2000;15(6):1413–5.
Sepulveda W, Munoz H, Alcalde JL. Conjoined twins in a triplet pregnancy: early prenatal diagnosis with three-dimensional ultrasound and review of the literature. Ultrasound Obstet Gynecol. 2003;22(2):199–204.
Andrews RE, Yates RW, Sullivan ID. The management of conjoined twins: cardiology assessment. Semin Pediatr Surg. 2015;24(5):217–20.
Yang PY, Wu CH, Yeh GP, Hsieh CT. Prenatal diagnosis of parapagus diprosopus dibrachius dipus twins with spina bifida in the first trimester using two- and three-dimensional ultrasound. Taiwan J Obstet Gynecol. 2015;54(6):780–3.
Ulker K, Akyer SP, Temur I, et al. First trimester diagnosis of parapagus diprosopus dibrachius dipus twins with cranirachischisis totalis by three-dimensional ultrasound. J Obstet Gynaecol Res. 2012;38(2):431–4.
Fang KH, Wu JL, Yeh GP, Chou PH, Hsu JC, Hsieh CT. Ischiopagus conjoined twins at 9 weeks of gestation: three-dimensional ultrasound and power Doppler findings. Ultrasound Obstet Gynecol. 2005;25(3):309–10.
Bonilla-Musoles F, Raga F, Bonilla F Jr, Blanes J, Osborne NG. Early diagnosis of conjoined twins using two-dimensional color Doppler and three-dimensional ultrasound. J Natl Med Assoc. 1998;90(9):552–6.
Biswas A, Chia D, Wong YC. Three-dimensional sonographic diagnosis of cephalothoracopagus janiceps twins at 13 weeks. Ultrasound Obstet Gynecol (England). 2001;18:289–90.
Watson SG, McHugh K. Conjoined twins: radiological experience. Semin Pediatr Surg. 2015;24(5):212–6.
Unal O, Arslan H, Adali E, Bora A, Yildizhan R, Avcu S. MRI of omphalopagus conjoined twins with a Dandy-Walker malformation: prenatal true FISP and HASTE sequences. Diagn Interv Radiol. 2010;16(1):66–9.
Teixeira Castro P, Werner H, Matos AP, Daltro P, Araujo Junior E. Symmetric and ventrally conjoined twins: prenatal evaluation by ultrasound and magnetic resonance imaging and postnatal outcomes. J Matern Fetal Neonatal Med. 2021;34(12):1955–62. https://doi.org/10.1080/14767058.2019.1651282. Epub 2019 Aug 12.
Chen CP, Hsu CY, Su JW, et al. Conjoined twins detected in the first trimester: a review. Taiwan J Obstet Gynecol. 2011;50(4):424–31.
Mitchell T, Cheng E, Jolley J, Delaney S. Successful induction of labor of late-second-trimester conjoined twins: an alternative to hysterotomy. Obstet Gynecol. 2014;123(2 Pt 2 Suppl 2):469–72.
Vaughn TC, Powell LC. The obstetrical management of conjoined twins. Obstet Gynecol. 1979;53(3 Suppl):67s–72s.
Harma M, Mil Z, Oksuzler C. Vaginal delivery of dicephalic parapagus conjoined twins: case report and literature review. Tohoku J Exp Med. 2005;205(2):179–85.
Tug N, Sahinoglu Z, Cam C, Eren S. Route of delivery of thoracoomphalopagus twins: analysis of three cases. Arch Gynecol Obstet. 2009;280(5):815–8.
Sinha M, Gupta R, Gupta P, Tiwari A. Assisted breech vaginal delivery of dicephalus dipus dibrachius conjoined twins: a case report. J Reprod Med. 2015;60(3–4):160–4.
O’Brien P, Nugent M, Khalil A. Prenatal diagnosis and obstetric management. Semin Pediatr Surg. 2015;24(5):203–6.
Spitz L. Ethics in the management of conjoined twins. Semin Pediatr Surg. 2015;24(5):263–4.
O’Neill JA Jr, Holcomb GW 3rd, Schnaufer L, et al. Surgical experience with thirteen conjoined twins. Ann Surg. 1988;208(3):299–312.
Rode H, Fieggen AG, Brown RA, et al. Four decades of conjoined twins at Red Cross Children’s Hospital–lessons learned. S Afr Med J. 2006;96(9 Pt 2):931–40.
Al Rabeeah A. Conjoined twins–past, present, and future. J Pediatr Surg. 2006;41(5):1000–4.
Votteler TP, Lipsky K. Long-term results of 10 conjoined twin separations. J Pediatr Surg. 2005;40(4):618–29.
Thomas A, Johnson K, Placencia FX. An ethically-justifiable, practical approach to decision-making surrounding conjoined-twin separation. Semin Perinatol. 2018;42(6):381–5.
Key Reading
Brizot ML, Liao AW, Lopes LM, et al. Conjoined twins pregnancies: experience with 36 cases from a single center. Prenat Diagn. 2011;31(12):1120–5.
Martinez-Frias ML, Bermejo E, Mendioroz J, et al. Epidemiological and clinical analysis of a consecutive series of conjoined twins in Spain. J Pediatr Surg. 2009;44(4):811–20.
Mathew RP, Francis S, Basti RS, et al. Conjoined twins – role of imaging and recent advances. J Ultrason. 2017;17(71):259–66.
Votteler TP, Lipsky K. Long-term results of 10 conjoined twin separations. J Pediatr Surg. 2005;40(4):618–29.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Brock, C., Johnson, A. (2021). Diagnosis and Management of Conjoined Twins. In: Khalil, A., Lewi, L., Lopriore, E. (eds) Twin and Higher-order Pregnancies. Springer, Cham. https://doi.org/10.1007/978-3-030-47652-6_19
Download citation
DOI: https://doi.org/10.1007/978-3-030-47652-6_19
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-47651-9
Online ISBN: 978-3-030-47652-6
eBook Packages: MedicineMedicine (R0)