Abstract
The exchange of carbohydrates and mineral nutrients in the arbuscular mycorrhizal (AM) symbiosis must be controlled by both partners in order to sustain an evolutionarily stable mutualism. Plants downregulate their carbon (C) flow to the fungus when nutrient levels are sufficient, while the mechanism controlling fungal nutrient transfer is unknown. Here, we show that the fungus accumulates nutrients when connected to a host that is of less benefit to the fungus, indicating a potential of the fungus to control the transfer of nutrients. We used a monoxenic in vitro model of root organ cultures associated with Glomus intraradices, in which we manipulated the C availability to the plant. We found that G. intraradices accumulated up to seven times more nutrients in its spores, and up to nine times more in its hyphae, when the C pool available to the associated roots was halved. The strongest effect was found for phosphorus (P), considered to be the most important nutrient in the AM symbiosis. Other elements such as potassium and chorine were also accumulated, but to a lesser extent, while no accumulation of iron or manganese was found. Our results suggest a functional linkage between C and P exchange.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
3.1 Introduction
Paddy soil is a type of anthropogenic wetlands on earth. Arbuscular mycorrhizal fungi (AMF) are usually found in paddy soil, their efficient role in such environments has usually been probed or overlook. They may originate from any type of soil in pedological terms but are highly modified by anthropogenic activities. Anaerobic condition alternations due to paddy organization regulator microbial community structure and role and thus short-range biogeochemical processes (Kögel-Knabner et al. 2010; Suzuki et al. 2015). It is presented that short- and long-term effects of waterlogging (Fig. 3.1). All of these factors influenced nutrient bioavailability and microbial activity.
Rice (Oryza sativa L.) is one of the important crops grown expansively in numerous countries. Paddy used to cultivate rice is the most prevalent method of farming in some sites, where it utilizes a small fraction of the total land yet feeds the common of the country population (Anonymous 2019).
The most usual symbiotic association of plants with microbes is Arbuscular mycorrhiza (AM). Arbuscular mycorrhizal fungi (AMF) are typically found in wetlands, their efficient role in such surroundings has been probed (Khan 2004; Bao et al. 2019). Some studies showed a number benefits of AMF application. In the following diagram, we summarized these (Fig. 3.2).
In addition to these benefits to plants, AMF may progress soil structure, decrease drought and salinity stress, and affect the variety of plant communities (Van der Heijden et al. 1998; Rillig and Mummey 2006; Smith et al. 2010). The benefits of AMF are serious in increasing agricultural production and efficiency in a low-input method.
The most important fungi are AMF because they organized impel symbioses that supply phosphorus and nitrogen to 70–90% of the plant, as well as almost all crops including cereals, vegetables, and fruit trees; therefore, they had efficient application in sustainable agriculture. It helps plants to uptake nutrients such as phosphorus, sulfur, nitrogen, and micronutrients from the soil (Yadav et al. 2019). Some researchers regarded that the expansion of the AMF symbiosis had a vital role in the primary colonization of land by plants. Formation of unique structures, arbuscules, and vesicles by fungi of the phylum Glomeromycota are characteristics symbiotic relationship. Also, AMF were existed in the rhizosphere of the first land plants and more than 80% of vascular plants, as well as most crops (Barbour et al. 1980; Brundrett 2002, 2009; Smith and Read 2010; Taylor et al. 2017; Brundrett and Tedersoo 2018; Bao et al. 2019).
However, the occurrence of AMF in aquatic ecosystem has been known for lasting, and evidence present that AMF is widespread these places (Sondergaard and Laegaard 1977; Nielsen et al. 2004; Wilde et al. 2009; Wang et al. 2010; Wang et al. 2011; Wang et al. 2014; Dolinar and Gaberščik 2010). Recently, the existence and accumulation of AMF has been specially attended in wetland environments (Wilde et al. 2009; Wang et al. 2010, 2011, 2014; Lumini et al. 2011).
AMF-facilitated improvement in inorganic uptake may lead to increased growth and development of plants and may consult survived to abiotic and biotic stress (Liu et al. 2007; Smith and Read 2008; Gianinazzi et al. 2010). Along with all of their benefits, there are some problems with their application, conceptually (Fig. 3.3).
Arbuscular mycorrhizal fungi (AMF) are necessary for proper control of advantageous symbiosis in an agricultural production ecosystem. The present chapter describes the biodiversity and functional role of Arbuscular Mycorrhizal Fungal from various sources and its applications in paddy fields as a maker of bio-active compounds in agriculture as plant growth prompter and harmless and bio-control factors. In the end, it will be discussed that the current study methods, concerns and appropriate future for its application in agricultural paddy soils.
3.2 Biodiversity of AM Fungi and Plant in Paddy Soils
Some of the researches have been accompanied by plant and AMF biodiversity in paddy soils (Kosaka et al. 2006, 2006; Lakshmipathy et al. 2012). The following are some studies being detailed in the following:
Kosaka et al. (2006) reported a paddy plant was arranged in order of irrigation regimes from a shorter to longer hydroperiod by DCA. A total of 184 wild herbaceous species and 17 cultivated species were recorded by Kosaka et al. (2006) in the wetland of their case study. The factors associated with high species diversity were: (1) the species unique to various paddy types; (2) the remnant species from original vegetation; and (3) the effect of agricultural practices. DWARF 14 LIKE is gene among the 26 genes deleted in Habiba the one responsible for the loss of symbiosis. Some consequences discovered an unanticipated plant recognition strategy for AMF and a previously unknown signaling association among symbiosis and plant improvement (Gutjahr et al. 2015b). Rice seedlings grown under natural sunlight exhibited various MGR to F. mosseae, from −4.4% to 118.9%, depending on cultivar. ARC5955, an indica rice, showed the highest MGR, whereas Nipponbare, popular japonica rice, showed an MGR of only 18.7%. Suzuki et al. (2015) focused on these two cultivars. However, un-inoculated roots observed no fungal structure because MGR and root length colonization were not powerfully associated in their research and individual differences in root colonization were large Smith and Read (2008) did not further examine the colonization. Other studies showed different results. In contrast to F. mosseae, ARC5955 and Nipponbare showed little reaction to Rhizophagus irregularis (formerly Glomus intraradices). Plant growth responses to AM fungi be contingent on the combination of plant and fungus (Smith and Read 2008).
All of the AM fungal species are positioned in four orders, i.e. Archaeosporales , Diversisporales , Glomerales , and Paraglomerales which comprise 13 families and 19 genera that belong to class Glomeromycetes of the phylum Glomeromycota (Sieverding and Oehl 2006; Oehl et al. 2008; Palenzuela et al. 2008). The plant species is in the specified ecosystem determined its inhabitants and diversity, so these fungi are obligate symbionts (Harinikumar and Bagyaraj 1988). According to Wang et al. (2015), the 639 AMF sequences from roots and soils were clustered into 101 OTUs based on 97–100% sequence resemblance by the mothur program and finally based on phylogenetic analysis, it assigned to 16 phylotypes. Wang et al. (2015) showed that AMF collected at the seedling, tillering, heading, and ripening stages in the roots of wetland rice (Oryza sativa L.) in four paddy wetlands that had been under a high-input and severely irrigated rice cultivation for more than 15 years. It was found that AMF colonization was mainly formed in the heading and ripening stages. The AMF community was observed in the root and rhizosphere area of paddy soils (LT and HN) (Fig. 3.4).
Distribution pattern of AMF phylotypes in the roots collected at the tillering, heading, and ripening stages of rice in two paddy soils. The numbers in the parentheses indicate the number of constructed SSU-ITS-LSU libraries at the corresponding growth stage (Wang et al. 2015)
Two AMF phylotypes (Rhi1 and Par6) were found to be the dominant AMF groups in roots throughout the tillering, heading, and ripening stages in the LT paddy wetland (Fig. 3.4), it representing that rice development caused well adapted in the environment of rice roots. Watanarojanaporn et al. (2013) showed that it is also obligatory to study whether any variations in species composition may be expressively affected by nutrient availability and plant growth since the AMF community changed by cultivation system and the application of compost types. Two major AMF in the genera Glomus and Acaulospora were distinguished from rice roots with or without compost application in this study.
Lakshmipathy et al. (2012) investigated that the effect of land-use intensity on the community (abundance and diversity) of arbuscular mycorrhizal fungi (AMF) was explored at various land-use types. In pre-monsoon season, the spore density and number of infective propagules were higher compared to the post-monsoon season in other land-use types, this is on condition that the land flora is different with the season and many species of herbs are documented. The dominant herbaceous weeds are in that area including Panicum repens, Grangea maderaspatana, Centella asiatica, Blumea barbata. The AMF community composition and diversity may be influenced by plant growth and community composition (Lin et al. 2015). In general, the AMF community is directly related to the cultivated plant species and this relationship is a two-way interaction (AMF-plant species) in paddy soil. Also, it changes according to the AMF community, the cultivation conditions such as organic matter application, the variability of soil moisture content and land-use history, and so on.
3.3 Colonization of Rice by Arbuscular Mycorrhizal Fungi
Inside the root and in the soil is the AMF community. The AMF community exists inside root and soil. The hyphae and other fungal structures, such as arbuscules and vesicles are present intraradical mycelium; the extraradical mycelium forms spores in soil for nutrient element uptake (Tommerup and Sivasithamparam 1990). The relationship between arbuscular mycorrhizal fungi (AMF) and bacteria within the rhizosphere section can motivate plant growth by a symbiotic. These communications are vital for increasing soil fertility, productivity, and sustainability, in addition to food security (Okonji et al. 2008). Legumes have benefited from biologically fixed nitrogen that association or symbiosis with root-nodulating bacteria, however, most non-leguminous plants, being the Gramineae (cereals), do not have a natural association (Hurek and Reinhold-Hurek 2003). So, the combination of these plants with AMF application can be important in terms of field product quality .
Vascular plants, including most crops, are widely established that the symbiotic arbuscular mycorrhizal fungi (AMF; phylum Glomeromycota) play an important function in suitable production systems and that rice freely organizes a symbiotic association (Lumini et al. 2011).
The arbuscules separated in the peri-arbuscular membrane significantly improved contact surface. In order to energizing of nutrient transport and the peri-arbuscular membrane, the symbiotic interface is acidified (Fig. 3.5) (Alexander et al. 1989; Guttenberger 2000; Krajinski et al. 2014; Wang et al. 2014) in paddy soil.
Distinguishing of AM fungal arbuscules . (a) Arbuscules are highly branched hyphal structures that nearly fill the cortex cells of the host. Green staining of fungal structures with wheat germ agglutinin (WGA) coupled to fluorescein isothiocyanate (FITC), red staining of the cell wall with propidium iodide (from Kretzschmar et al. 2012). (b) Transmission electron micrograph of a colonized host cell with an arbuscular branch (fungus), surrounded by the peri-arbuscular membrane. (c) A colonized root stained with Neutral Red which accumulates in acidic compartments, in this case, the space between the peri-arbuscular membrane and the fungal cell wall [compare with (b)]. Size bars, 20 μm in (a, c); 1 μm in (b) (Chen et al. 2018)
A vastly branched hyphal an arbuscule grows in the cortical cells and helps for nutrient exchange between the host plant (which make available photosynthates) and the AMF (which provides mainly phosphate along with other nutrients). To survive in phosphorus-deficient soil , external hyphae growing from the mycorrhizal roots permit host plants to assimilate phosphate from outside the root region (Nakagawa and Imaizumi-Anraku 2015) (Fig. 3.6).
Schematic view of mycorrhization and rice genes directly involved at each stage. SLs derived from host roots induce hyphal branching of AM fungi. Myc factors secreted from AM fungi induce symbiotic Ca2+ spiking. Hyphopodia are organized on the epidermis of host roots at the tips of branched hyphae. Immediately beneath the hyphopodia, the pre-penetration apparatus (PPA) develops to guide hyphal penetration. In the inner cortex, arbuscules develop and act as a nutrient exchanger between the host plants and AM fungi (Nakagawa and Imaizumi-Anraku 2015)
Bernaola et al. (2018) indicated that root colonization proportion alternating from around 2% to 62% in all the rice samples given from various locations over many years were colonized by AMF. These organs tend to decomposition rapidly, a few arbuscules were observed in some studies. Hyphae were the most common structures for delivery of mineral nutrients in the symbiosis interaction (Parniske 2008); vesicles were set up in more quantity. N1, N2, and N3 represent for N chemical fertilization at 100%, 75%, and 50% of the optimum rate, respectively; P1, P2, and P3 represent for P chemical fertilization at 100%, 75%, and 50% of the optimum rate, respectively; M, mycorrhizal fungi. Accordingly to Hoseinzade et al. (2016), the least rate of colonization (%) was observed by N1P1M (N1, N2, and N3 represent for N chemical fertilization at 100, 75, and 50% of the ideal rate, respectively); the highest rate of colonization was resulted by N2P3M (47), N3P2M (46), and N3P3M (48). Some results indicating the significant effects of bio-active fertilizers including H. seropedicae and G. mosseae, on the rice yield, the time of appropriate application is presented in the following table during the rice plant growth stages (Hoseinzadeh et al. 2016)
Hattori et al. (2013) showed that in the flooding treatment , most root nodules were organized on the adventitious roots, especially. Among two treatments including flooding and non-flooding, there was no significant difference in the number of nodules on the adventitious roots and the total number of nodules, because the values had a significant difference between plants, completely in the flooding. AM colonization and nodule formation on the two different types of roots were detected in the flooding treatment , that is, the root system was divided into (primary and lateral roots as) basal roots, and adventitious roots appeared below the lower place of the hypocotyl. The hypocotyls were dispersed on the soil surface in the shallow soil layer, in the flooding treatment. Hence, the roots are not damaged by poorer oxygen situations in anaerobic conditions. In fact, the roots had a higher AM colonization ratio and many root nodules were formed in wetland soil. Enlarged diameter and partial cracking of the surface tissues were found in the flooding treatment , they were characteristic morphological modifications in water logging hypocotyls on the soil surface (Fig. 3.7).
Aerenchymatous tissues in soybean seedlings after flooding. (a) Irrigation, (b) flooding (Hattori et al. 2013)
According to Lynch (2007), adventitious roots have more plenty of aerenchyma than other root types. The ratio of the aerenchyma/stele area in the roots was more than 80% in the flooding treatment. The adventitious roots have high rates of AM infection and nodule in persuaded in the flooding treatment because the microorganisms are aerobic (Ray and Inouye 2006; Maekawa et al. 2011). As follows, two approaches were founded for AMF application in paddy soil. Suzuki et al. (2015) show that rice under well-drained conditions (aerobic) is positively quick to respond to AM fungi and that colonization and growth response were different widely.
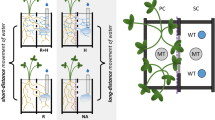
Gutjahr et al. (2015a) showed that the jasmonate-deficient rice (Oryza sativa) mutant hebiba cannot establish symbiosis with either of two AMF rhizophagus irregularis and Gigaspora rosea—as reflected by the absence of hyphopodia, intraradical colonization and induction of colonization marker genes (Fig. 3.8a–c). Growing hebiba alongside colonized wild-type plants caused the lack of fungal interaction persisted upon increased inoculum strength imposed (Fig. 3.8d). This suggested that during pre-symbiotic signaling, the mutant is compromised at a very early stage of the interaction.
AM phenotype of hebiba . (a and b) Roots of hebiba and wild type (WT) stained with trypan blue to visualize AM fungal structures 6 weeks post-inoculation (wpi) with R. irregularis (a) and G. rosea (b). Labels refer to A, arbuscule; AC, arbuscular coil; AX, auxiliary cell; EH, extraradical hypha; HP, hyphopodium; V, vesicle; scale bar, 100 mm. (c) Expression of early (AM1 and AM3, left) and late (AM14 and PT11, right) AM marker genes in hebiba and WT 6 wpiFig. 3.8 (continued) with R. irregularis as assessed by quantitative PCR. Expression values were normalized to those of the housekeeping gen Cyclophilin2 (LOC_Os02g02890). Means and SEs of four biological replicates are shown. Expression was significantly less in hebiba than WT (P = 0.02 for each gene; Kruskal–Wallis test with Benjamini–Hochberg adjustment for multiple testing). (d) Percentage of root length colonization (RLC) by R. irregularis of two central “tester” surrounded by six “donor” plants at 7 wpi. Means and standard errors of five biological replicates are shown. ext hyphae, extraradical hyphae; int hyphae, intraradical hyphae. For each of the six fungal structures in the figure, separate Kruskal–Wallis tests were performed, using the Benjamini–Hochberg adjustment for the post hoc tests. The P values were as follows: P (total) ≤ 0.01, P (ext. hyphae) = 0.43, P (hyphopodia) ≤ 0.05, P (int. hyphae, arbuscules, vesicles) ≤ 0.001.The letters above each bar show growth conditions that were not significantly different in the post hoc pairwise comparisons (Gutjahr et al. 2015b)
3.4 Arbuscular Mycorrhiza Community Under Different Irrigation Regimes in Paddy Soils
Irrigation regimes are determined by the rate, number, and time of water applications to agricultural products. It depends on the biologic characteristics of plants, climatic, soil, and hydrologic conditions. Water is a vital section of soil fertility. Other views, some crops are planted in upland fields that have been converted from rice paddies in the several countries of Asia. In such fields, waterlogging conditions extensively damage this crop during the rainy season. The lack of oxygen in the root system caused these damages including prevented the growth and nutrient uptake and often reduced seed yields (Hattori et al. 2013; Linkemer et al. 1998; Miao et al. 2012).
Wetlands are productive bio-systems that collect large amounts of organic matter in the soil, so functioning as significant C sinks (Mitra et al. 2005). In these places, waterlogging leads to soils dominated by anaerobic processes. The presence and practical role of AMF in wetlands have largely been overlooked due to the aerobic nature of AMF (Khan and Zaidi 2007; Vallino et al. 2014). Also, most studies being about Arbuscular mycorrhiza have been carried out on upland rice (not flooding irrigation) and the number of studies is relatively little in lowland (Yimyam et al. 2008; Rajeshkannan et al. 2009) but anthro-saturation occurs as soil is deliberately flooded and ponded for agronomic aims. Most of this occurs are in the crop growing of rice (paddy soils). Paddy soils are consequent from different kinds of parent materials under waterlogging cultivation over various parts. They are exposed through alternate processes of oxidation and reduction. They are usually anaerobic and possess a low redox potential during the growing period of rice. Vesicles, hyphae, and arbuscules (Characteristic AMF structures) were observed in all roots inoculated with R. irregularis, and there were no AMF colonization in the non-mycorrhizal (NM) controls (Bao et al. 2019).
The explored paddy fields were under waterlogged conditions most of the time, and as AMF is aerobic, the occurrence of AMF in paddy wetlands could be correlated to the expansion of aerenchyma in the rice plant. At the heading and ripening stages, the aerenchyma provides oxygen in rice roots; but the aerenchyma tissue has not been well developed at the early developmental stages (Wang et al. 2013) and the AMF may be oxygen incomplete. Although results of some research showing that AMF are usually present in about types of wetlands , such as mangrove (Wang et al. 2011, 2013, 2014), salt marsh (Wilde et al. 2009), the AMF community in wetland habitats poorly known. The relatively high number of AMF phylotypes in the surveyed conventional paddy wetlands from this study indicates that the AMF diversity in an aquatic ecosystem is not unavoidably low (Wang et al. 2011). This can be explored by aerenchyma in rice roots (Fig. 3.9) and the high ecological adaptability of some AMF species. Up to now, the role of AMF in wetland ecosystems was poorly understood (Wang et al. 2011; Zhang et al. 2014), but several researches have been found that the AMF colonization of aquatic plants produces a wide range of advantages to AMF-plant partners (Wang et al. 2011; Zhang et al. 2014; Stevens et al. 2011). It offered that AMF is an important component in the traditional rice cultivation systems in paddy soil. Considering the high fertilizer inputs and the frequent disruption that happens in the paddy fields, the observed high AMF diversity indicated that some AMF groups can be adaptive to a high-input and severely managed ecological unit.
Differences in lysigenous aerenchyma formation and patterns of radial O2 loss (ROL) in rice roots under drained soil conditions and waterlogged soil conditions (Nishiuchi et al. 2012)
The AM colonization ratio in flooding was lower than in the irrigation regime. Some results showing different rates of AM colonization and root nodule formation between the two different types of roots increase the appreciative of responses of soybeans grown in drying-rewetting (lowland and upland) fields (Hattori et al. 2013). Generally, rice is grown under thin flooded or wet paddy conditions, but it is also cultured where floodwater with meters deep; therefore, rice appears to have a high water requirement. The system of rice intensification (SRI) is another system for rice cultivation, made in Madagascar in the early 1980s to cultivate rice, and these managements have now been approved in over 20 countries. Five main differences in SRI compared to conventional practices (constant waterlogging) including (1) transplanting younger seedlings, (2) transplanting seedlings singly, (3) widespread spacing application, (4) interchanging soil flooding with draining during the vegetative growth phase, and (5) applying organic amendment (as compost) rather than mineral fertilizer (Watanarojanaporn et al. 2013).
Other studies showed that AMF is usually present in high-input and intensification irrigated rice cultivation management and that AMF colonization and community are dependent on the growing stage of the rice plant (Wang et al. 2015), proposing that development stage plays an important role in their relations. Solaiman and Hirata (1995) showed that the role of AMF varies in nutrient uptake ratio in plant organs, plant growth stages, and application intensity. AMF considerably reduced shoot N concentration and did not affect shoot P concentration at the development stage. N and P concentrations in unhulled grain were significantly increased due to AMF inoculation only under flooded conditions. These results recommend that AMF may accelerate N and P transmission from shoots and soils to rice grains even under flooded conditions and along with the trend to rise the harvest index under the flooded conditions, AMF increased the harvest index due to a decrease in shoot dry matter production while under non-flooded conditions, AMF decreased it due to a decrease in the dry matter of unhulled grains itself. There was a significant relation between AMF inoculation and irrigation regimes on AMF colonization (Solaiman and Hirata 1995).
Previous studies have reported a significant effect of hydrology management on the AMF community structure in waterlogging conditions (Wang et al. 2015, 2016). In the all typical AMF structures, the root colonization was generally lesser following increased flooding intensities at the rice and showed an increasing tendency at the second growth stage compared to the first (Bao et al. 2019). The percentage of AMF colonization by the three types [Glomus versiforme (GV), Glomus mosseae (GM), and Glomus diaphanum (GD)] on the rice cultivars ranged from 30% to 70%. Mycorrhizal colonization of upland rice cultivars had a large effect on plant development by increasing the shoot and root biomass (Zhang et al. 2005).
AMF community structure was affected by different irrigation systems, alternating flooding, and draining of water management in paddy fields. Rice plants grown under SRI (the system of rice intensification) had a more various AMF community than those grown under conventional rice cultivation systems (Watanarojanaporn et al. 2013). The AMF colonization ratio was 12.5% in the primary and lateral roots and 14.5% in the adventitious roots in the irrigation treatment. The ratios in the irrigation treatment were significantly more than those in flooding treatment. The AM colonization ratio in the adventitious roots (7.5%) was significantly high in the primary and lateral roots, while it was not necessarily low in the flooding treatment (0.8%). Under upland conditions, rice plants readily form mycorrhizal associations, but under water logging conditions, infection is rare due to anaerobic conditions (Ilag et al. 1987). Nevertheless, AMF has necessitated aerobes in nature but can stay alive under submerged conditions, and this is sustained by the fact that AMF as Glomus etunicatum showed properly high colonization in rice roots and best survival under water logging conditions (Vallino et al. 2009).
3.5 Arbuscular Mycorrhiza Under Water: Carbon–Phosphorus Exchange Between Rice and Arbuscular Mycorrhizal Fungi
It has before been recommended that mycorrhiza should be combined into global models of C and mineral cycling for the plan of ecosystem reactions and responses to climate change. Global carbon and phosphorus cycling are significantly contributed by AM symbiosis (Averill et al. 2014; Terrer et al. 2016; Treseder 2016). AMF organizes mutualistic associations with the roots of most plants, including many crops. These mutualistic associations have shown the potential to increase crop efficiency, thereby playing a key role in the functioning and sustainability of agricultural systems (Gianinazzi et al. 2010). The most important role of these symbiotic associations consist of the transfer of nutrients such as organic carbon in the form of sugars and lipids (Jiang et al. 2017; Luginbuehl et al. 2017), to the fungi by the plants, and the transfer of phosphorus (P) and nitrogen (N) to the plants by the fungi (Smith and Read 2008).
Bao et al. (2019) observed that alternative water logging led to no obvious decline in AMF colonization intensity and, although sharp decreases in C–P exchange under flooding treatments at growth stages. These consequences describe the significant dynamic force of flooding in AM symbiosis in wetlands (Vallino et al. 2014), and showed that it can be observed nutrient influence from both AMF and plants change in response to flooding before a significant decrease in mycorrhizal colonization. The flooding-induced reduction in oxygen availability could describe the reduced role of the AM symbiosis, particularly in the extra-radical mycelium, which had lesser availability to the transported oxygen through root aerenchyma, despite established aerenchyma in rice.
It was observed that the improved P uptake due to AMF colonization under non-flooding conditions by Vallino et al. (2014) and Bao et al. (2019) but not during flooding and, the intraradical: extraradical ratio of 13C allocation increased with increased flooding intensity. Flooding conditions caused a decrease in C-P exchange may also be related to the change in morphology of the rice root (Vallino et al. 2014). Hydrologic cycle is being intensified the global climate change, and the occurrence of crop flooding can be probable to increase in the future (Milly et al. 2002). The response of AM symbiotic functionality to flooding observed by Bao et al. (2019) study. The plant C distribution to AMF declined at increased flooding intensity and was significantly greater at the growth stage when the rice plants had a higher nutrient necessity.
The relationship between the arbuscular and the root cell of the plant is demonstrated as a model in Fig. 3.13. The sucrose is released root cortical cells into the interfacial apoplast, acid invertase hydrolyzed it into hexoses that can be taken up by the fungus (Fig. 3.13, [1]). Once taken up by the fungus, the C is integrated into lipids and transported throughout the fungal mycelium. The glyoxylate cycle metabolized the lipids and characterize the major C source for supportive the spread of the extra-radical mycelium in the soil. The hyphal tips catch P via high-affinity mineral P (Pi) transporters (Fig. 3.13). Mainly in the form of polyphosphate, P is collected and transported along the hyphae to the roots of the host plant (polyP), phosphoanhydride-bound Pi polymer residues. Alkaline phosphatases depolymerized PolyP in the intraradical mycelium, and Pi is abandoned into the between apoplast (Fig. 3.13, [3]). Plants catch the Pi by mycorrhiza-specific Pi transporters in the periarbuscular membrane (Fig. 3.13, [4]); (Smith et al. 1994; Harrison and van Buuren 1995; Bago et al. 2000; Douds et al. 2000; Ezawa et al. 2002; Bucking and Shachar-Hill 2005; Schaarschmidt et al. 2006; Schüβler et al. 2001; Rausch et al. 2001; Javot et al. 2007; Xu et al. 2007; Hijikata et al. 2010). This process is illustrated in Fig. 3.10, completely.
The nutrient exchange between plants and AMF is a theoretical model. The flows are identified by numerals and adjustment mechanisms as letters in square brackets. Mycorrhiza-specific Pi transporters give Pi to plants in the peri-arbuscular membrane (a) Under high plant P levels C release to the fungus declines, and plant peri-arbuscular P transporters are down-regulated premature arbuscule senescence is induced under low P delivery (b) Fungal nutrients can motivate plant C statement (c) Hyphal P adsorb is P induced, as the P transporter gene is regulated by the environmental P concentration but C flow to the fungus does not influence P uptake from the medium place (d) researchers hypothesized that P transfer to plants can be controlled by the fungus that provides low-C benefit (Hammer et al. 2011)
Hammer et al. (2011) showed that G. intraradices gathered more than six times more nutrients in its spores, and up to more eight times more in its hyphae, when the C sink available decreased by 50%. Potassium and chorine were also collected, while no accumulation of iron or manganese was established. They recommended a useful relation between C and P exchange. The highest effect they establish for phosphorus (P) considered to be the most vital nutrient in the AM symbiosis .
3.6 The Role of Arbuscular Mycorrhizal Fungi in Improving Yield, Nutrient Uptake Management and Stress Tolerance in Paddy Soils
AM fungi are present in the majority of natural ecosystems and they provide a range of important environmental services, in particular by improving plant nutrition, tension resistance and tolerance, physic and chemical soil characteristics (Kour et al. 2019). Basic research of the past decade has exposed the existence of a devoted recognition and signaling pathway that is required for AM. Additionally, recent evidence delivered a new vision into the exchange of nutritional benefits between the symbiotic partners. The great potential for the application of AM has given an increase to a prosperous industry for AM-associated products for agriculture, horticulture, and landscaping work. Here, we debate new developments in these fields, and we highlight prospect potential and limits toward the use of AM fungi for plant production (Chen et al. 2018).
The basis for AM symbiosis is this exchange of resources, and it is responsible for considerable nutrient transfer globally and is critical for carbon sequestration in earthly ecosystems. Plants transfer averaged (∼5 billion tonnes of C per year: 20% of their photosynthetic products) to fungal symbionts (Bago et al. 2000) and receive up more than 80% of the mineral nutrients from AMF (Smith and Smith 2011). The improvement of the plant’s ability in tolerating toxic elements, the AMF has been shown to increment the nutrient station of their host plant (Aggangan et al. 1998). Low-C availability to the plant did not cause to reduce the hyphal length colonization, but the number of arbuscules was lower, indicating that P transfer was reduced, which established by isotope probing that mycorrhizal 33P transfer was intensely reduced to root that was C-ravenous in a communal mycorrhizal network (Lekberg et al. 2010; Hammer et al. 2011).
Although phosphorus (P) shortage strongly restricts rice production in some soils (Nishigaki et al., 2019), P uptake and plant growth are increased by colonization of plant roots with AMF (Bernaola et al., 2018; Hajiboland et al., 2009). Although AMF colonization did not increase rice growth, a significant amount of P was transported to plants through the mycorrhizal way under wetland conditions. This is understood by the gene expression profiles and rice P levels (Bao et al. 2019). Two pathways are present for nutrient absorption in AM (arbuscular mycorrhizal) roots, which include: the root epidermis and root hairs (the direct pathway (DP)); and root cortical cells are the mycorrhizal pathway (MP) via AM fungal hyphae (Smith and Smith 2011). Optimistic effects escalate principally from the increasing P uptake via the MP, improving P efficiency (Smith and Read 2008; Smith and Smith 2011).
Mycorrhiza had supporting effects against the toxicity of Cu, Zn, Pb, and Cd in the contaminated soil conditions. In the two cultivars, colonization by AMF reduced the translocation of heavy metals from root to shoot. The mixture of Cu, Zn, Pb, and Cd and immobilization of the heavy metals in roots decrease the potential toxicity to shoots. When un-inoculated condition, the two rice cultivars presented significant differences in uptake of Cu, Zn, Pb, and Cd. AMF inoculation had the most protective effects under the combined soil contamination (Zhang et al. 2005). Mycorrhiza had supporting effects against the toxicity of Cu, Zn, Pb, and Cd in the contaminated soil conditions. In the two cultivars, colonization by AMF reduced the translocation of heavy metals from root to shoot. The mixture of Cu, Zn, Pb, and Cd and immobilization of the heavy metals in roots decrease the potential toxicity to shoots. When un-inoculated condition, the two rice cultivars presented significant differences in uptake of Cu, Zn, Pb, and Cd. AMF inoculation had the most protective effects under the combined soil contamination (Zhang et al. 2005).
Significant effects of biological fertilizers including H. seropedicae and G. mosseae, on the rice production as well as on soil factors, were gained. However, biological fertilizers almost related to the soil and plants were importantly affected by the bacteria and the fungi (Hoseinzade et al. 2016). The K and Mn concentration appeared somewhat higher with colonization. On the other, the levels of Fe, Cu, Zn, B, and Al reduced with colonization in some India rice cultivars. Co contents in AM roots increased significantly in both cultivars of rice (Nipponbare and ARC5955). Colonization of F. mosseae imitated the above growth motivation, and also improved the P concentration. Response of rice to F. mosseae was stated by the increase of P concentration in colonization of F. mosseae and increase beneficial elements concentration and decrease deleterious element concentration (Suzuki et al. 2015). P accumulated in definite regions within the spores, together with K the elemental maps obtained with PIXE (Fig. 3.11). In vacuole-like structure, P accumulation was seemed spot-wise. It revealed that the concentrations of K and Ca were the highest near the spore wall in the full-C treatment, analysis was done on a cross-section of two separate spores from mycelium (Hammer et al. 2011). Organic amendments including paddy straw, maize straw, and Pongamia leaf improved the mycorrhizal activity in the paddy soils. Addition of Pongamia leaf enhanced AM fungal infection to the maximum, and then followed by maize straw in three amendments addition (Harinikumar and Bagyaraj 1988).
PIXE elemental distributions of Ca, K, P, and S in hyphae and spores from two areas of the mycelium [(a) full-C; (b) half-C] of Glomus intraradices. The color scale shows the gradient from low to high elemental concentrations (Hammer et al. 2011)
3.7 Effect of Arbuscular Mycorrhiza Community on Rice Cultivation Systems (Growing Steps)
Rice (Oryza sativa L.) is the main food crop cultivated in wetland environments and one of the financially most important plant production worldwide (Wang et al. 2014). It is a semi-aquatic plant species that are adapted and staple food to a partially flooded environment. It is for more than half of the world’s population (FAO 2004). In non-legume plants, rice displays one of the perfect model plants used for understanding the molecular, biochemical, and physiological origin of AM symbiosis (Paszkowski et al. 2002; Yang et al. 2012; Fiorilli et al. 2015). Rice production be accomplished in wetlands characterized by noticeable differences in soil flooding more than 85% of global (FAO 2004). AMF colonization of rice roots under wetland conditions has even been the subject of debate because little is known about the nutrient transfer between AMF and rice roots in flooded fields, and the (Vallino et al. 2009, 2014; Lumini et al. 2011; Watanarojanaporn et al. 2013). In recent last 50 years, some areas of Asia, rice production has more than tripled, mostly due to increased grain yield. The today concern is that serious challenges regarding water and soil pollution has been induced the overuse of nitrogen and phosphorus fertilizers as well as pesticides in rice production. “Green” rice production system is calling for a serious need to develop without a yield decrease that can replace the traditional intensive cultivation (Wang and Bjorn 2014; Wang et al. 2014).
Rice rhizosphere had a key role in the uptake of water and nutrients from paddy soil. Some researchers showed that there was a positive relationship between the presence of arbuscular mycorrhizal fungi (AMF), the number of spores in separate root systems, and the development of rice (Sanni 1976). Mycorrhizal inoculums application could be one approach to increase rice production and decrease fertilizer use (Wang et al. 2015). All arbuscular mycorrhizal fungi (AMF or AM fungi) are obligate symbionts and are clustered in the phylum Glomeromycota (Schüβler et al. 2001). Wang et al. (2015) showed that a total of 639 AMF sequences were obtained, and these were finally apportioned to 16 phylotypes founded on a phylogenetic analysis, including 12 phylotypes from Glomeraceae, one phylotype from Claroideoglomeraceae, two phylotypes from Paraglomeraceae. There was an obvious difference between the communities gained from root and soil, but the AMF phylotype compositions in the soils were alike between the two measured places. The comparatively high number of AMF phylotypes at the surveyed sites suggests that the conditions are suitable for some species of AMF and that they may have an important role in traditional rice cultivation systems. The species enriched of root-colonizing AMF improved along with the rice growth, and next researches should study the developmental stages of this crop in the survey of AMF role in paddy soils (Wang et al. 2015).
According to Fig. 3.12, regarding management patterns of P distribution, +m rice displayed higher panicle P: shoot P proportions, but lower leaf P: shoot P ratios than −m treated rice across all fertilizer concentrations (Fig. 3.12). Stem P: shoot P ratios of −m rice at low fertilizer levels were significantly higher than those of +m rice. These correlations display the role that treatment-mediated alterations in biomass portion play in responsible patterns of nutrient element portion within rice shoots (Zhang et al. 2014).
Effect of fertilization and inoculation on plant biomass making and portion to shoots and roots. F0, F20, F40, F60, F80, and F100, delivered with 0, 20, 40, 60, 80, and 100% of the local norm of nutrient input (Zhang et al. 2014)
Two kinds of spores were identified as Glomussppat (Fig. 3.13a, b) the nursery during inoculation: Internal hyphae (Fig. 3.13c–e) and external hyphae (Fig. 3.13f). They showed widespread colonization at the different time.
AMF sporulation (a, b) and colonization (c) at the nursery stage and colonized roots at 30 days (d); 52 days (e); external hyphae at 130 days (f) after transplanting under field conditions; Bars = 50 μm (Solaiman and Hirata 1995)
Arbuscular mycorrhizal fungi (AMF) form symbiotic associations with the roots they live in, and potentially change resistance against pests. Rice-AMF associations were the greatest in Arkansas followed by Mississippi and Texas (Fig. 3.14) (Bernaola et al. 2018).
Examples of arbuscular mycorrhizal fungal structures used as indicators of rice root colonization collected from Mississippi (a), Arkansas (b), Texas (c), and Louisiana (d, Crowley) rice fields (Bernaola et al. 2018)
AMF colonization intensity is various at the different growth stages of rice. AMF constructions were lacking or very infrequent at the seedling and tillering periods. On the other hand, typical vesicular and arbuscular structures of AMF were generally observed at the heading and ripening stages. AMF colonization intensities (including hyphal, vesicular, and arbuscular colonization rates) at the heading stage were significantly lower than at the ripening stage. AMF colonization intensities (including hyphal, vesicular, and arbuscular colonization rates) at the heading stage were significantly lower than at the ripening stage. AMF colonization intensities at HN, BY, and LT were lower than at TX those, while at both heading and ripening stages, the colonization intensities among the last three sites were similar (Wang et al. 2015).
Another possible explanation for the higher P levels in the more slowly growing low-C mycelium might be that younger hyphae could contain higher element concentrations than older Hyphae (Hammer et al. 2011). In general, soil moisture, the inorganic nutrient available in the soil, pH, species of AMF, the host plant species, and several agricultural management practices have effects on the AMF community (abundance and activity). A long with these, soil biological communities could influence the establishment of the AMF symbiosis and its impact on plant agricultural production (Fig. 3.15) (Lekberg and Koide 2005; Bernaola et al. 2018).
3.8 AMF Research
Laboratory or greenhouse experiments were involved in the majority of research on AMF associations, in which plants are cultivated in sterilized soil, with specific AMF species. They disregard native AMF species that could change plant responses or compete with the AMF inoculant (Munkvold et al. 2004). Some of the measurement methods are taken the following;
3.8.1 Quantification of AMF Colonization
Mycorrhizal colonization by AMF structures was determined by considering five slides with ten segments per slide from each sample and calculating the amount of colonization using the extended connections method of McGonigle et al. (1990) with negligible changes.
The seven to ten samples root colonization percentage averaged at each site and calculated by the following formula: Root colonization percentage (%) = No. of segments colonized with AMF/No. of segments observed × 100 (Bernola et al. 2018)
3.8.2 PIXE Analysis
A multi-elemental analytical technique is particle-induced X-ray emission founded on the detection of characteristic X-rays produced by MeV ions (protons or heavier ions). When protons pass through the sample, STIM is based on the detection of energy loss. Several spores, hyphae from a particular sample and detailed elemental maps of at least P, S, K, Ca, and Fe were analyzed (Hammer et al. 2011).
3.9 Conclusion and Future of AMF in Paddy Soils
Rice is one of the economically most important plants worldwide cultivated in wetland environments, so finding several AMF species that are capable of surviving in the flooding conditions makes a great help in symbiotic relationships. Our review showed that natural AMF communities that are more appropriate than others. Improving the activity of native arbuscular mycorrhizal fungi (AMF) has benefits in wetland agriculture.
It is a shortage severely limits in rice production worldwide, while colonization of plant roots with AMF often enhances nutrient element uptake and water logging plant growth. Little is known about the nutrient exchange between AMF and rice roots in flooded fields, under wetland conditions has even been the subject of debate. Colonization intensity between AMF and rice at different growth stages. The role of AMF is more noticeable in the early stages of growth terms of increased yield, nutrient concentration, and reduced heavy metals in plant tissue of cultivation in paddy fields. The AM colonization ratio in flooding was lower than in irrigation. Therefore, by adjusting the irrigation regimes and the application of organic matter amendments along with AMF application, it can be achieved maximum crop yields in paddy soils. This review will help appeal research to colonization of AMF in paddy soils of the worldwide that can impact future rice production by facilitating agricultural application of the AMF symbiosis.
By comparing the results and discussion of Van der Heijden et al. (2015), a better future can be considered for AMF application and increased production in paddy soil by following:
The various biochemical and genetic endeavors describe supporting by enormous efforts to structure the genomes of various fungal symbionts in paddy soils. Determination of the role of mycorrhizal fungi in waterlogging plant rhizosphere, paddy soil structure, and responses to environmental changes, and global C and nutrient cycles. Investigation of symbiotic gene networks in the molecular dialogue between wetland plants and fungus is a key challenge to understand plant-fungal cohabitation and the elements responsible for the organization of AMF symbiosis. The main challenge is to make known the fluxes of energy, metabolites, signaling molecules, and nutrients through mycorrhizal networks in paddy fields. Understand coevolutionary processes between wetland plants and mycorrhizal fungi. Now, we need to improve biogeochemical models that help us to forecast when, and under what conditions, application of mycorrhizal technology is profitable.
References
Aggangan NS, Dell B, Malajczuk N (1998) Effects of chromium and nickel on growth of the ectomycorrhizal fungus Pisolithus and formation of ectomycorrhizas on Eucalyptus urophylla S.T. Blake. Geoderma 84:15–27
Alexander IJ (1989) Mycorrhizas in tropical forests. In: J. Proctor (Ed.), Mineral nutrients in tropical forest and savannah ecosystems. Blackwell, Oxford. Pp. 169–188
Anonymous (2019). https://www.britannica.com/topic/paddy
Averill C, Turner BL, Finzi AC (2014) Mycorrhiza-mediated competition between plants and decomposers drives soil carbon storage. Nature 505:543–545
Bago B, Pfeffer PE, Shachar-Hill Y (2000) Carbon metabolism and transport in arbuscular mycorrhizas. Plant Physiol 124:949–958
Bao X, Wanga Y, Olsson PA (2019) Arbuscular mycorrhiza under water—carbon–phosphorus exchange between rice and arbuscular mycorrhizal fungi under different flooding regimes. Soil Biol Biochem 129:169–177
Barbour MG, Burk JH, Pitts WD (1980) Terrestrial plant ecology. Benjamin/Cummings Publishing Company, Menlo Park
Bernaola L, Cange G, Way MO, Gore J, Gore J, Stout MJ (2018) Natural colonization of rice by arbuscular mycorrhizal fungi in different production areas. Rice Sci 25:169–174
Bravin MN, Travassac F, Floch ML, Hinsinger P, Granier JM (2008) Oxygen input controls the spatial and temporal dynamics of arsenic at the surface of a flooded paddy soil and in the rhizosphere of lowland rice (Oryza sativa L.): amicrocosmstudy. Plant Soil 312:207–218
Brundrett MC (2002) Coevolution of roots and mycorrhizas of land plants. New Phytol 154:275–304
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77
Brundrett MC, Tedersoo L (2018) Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol 220:1108–1115
Bucking H, Shachar-Hill Y (2005) Phosphate uptake, transport and transfer by the arbuscular mycorrhizal fungus Glomus intraradices is stimulated by increased carbohydrate availability. New Phytol 165:899–912
Chen M, Arato M, Borghi L, Nouri E, Reinhardt D (2018) Beneficial services of arbuscular mycorrhizal fungi – from ecology to application. Front Plant Sci 9:1–14
Dolinar N, Gaberščik A (2010) Mycorrhizal colonization and growth of Phragmites australis in anintermittent wetland. Aquat Bot 93:93–98
Douds DD, Pfeffer PE, Shachar-Hill Y (2000) Application of in vitro methods to study carbon uptake and transport by AM fungi. Plant Soil 226:255–261
Ezawa T, Smith SE, Smith FA (2002) P metabolism and transport in AM fungi. Plant Soil 244:221–230
FAO (2004) International year of rice. http://www.fao.org/rice2004/en/rice4.htm
Fiorilli V, Vallino M, Biselli C, Faccio A, Bagnaresi P, Bonfante P (2015) Host and non-host roots in rice: cellular and molecular approaches reveal differential responses to arbuscular mycorrhizal fungi. Front Plant Sci 6:1–17
Gianinazzi S, Gollotte A, Binet MN, Van Tuinen D, Redecker D, Wipf D (2010) Agroecology: the key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 20:519–530
Greipsson S (1995) Effect of iron plaque on roots of rice on growth of plants in excess zinc and accumulation of phosphorus in plants in excess copper or nickel. J Plant Nutr 18:1659–1665
Gutjahr C, Gobbato E, Choi J, Riemann M, Johnston MG, Summers W, Carbonnel S, Mansfield C, Yang SY, Nadal M (2015a) Rice perception of symbiotic arbuscular mycorrhizal fungi requires the karrikin receptor complex. Science 350:1521–1524
Gutjahr C et al (2015b) Rice perception of symbiotic arbuscular mycorrhizal fungi requires the karrikin receptor complex. Science 350:1521–1524
Guttenberger M (2000) Arbuscules of vesicular-arbuscular mycorrhizal fungi inhabit an acidic compartment within plant roots. Planta 211:299–304
Hajiboland R, Aliasgharzad N, Barzeghar R (2009) Phosphorus mobilization and uptake in mycorrhizal rice (Oryza sativa L.) plants under flooded and non-flooded conditions. Acta Agric Slov 93(2):153
Hammer EC, Pallon J, Wallander H, Olsson PA (2011) Tit for tat? A mycorrhizal fungus accumulates phosphorus under low plant carbon availability. FEMS Microbiol Ecol 76:236–244
Harinikumar KM, Bagyaraj DJ (1988) Effect of crop rotation on native vesicular arbuscular mycorrhizal propagules in soil. Plant Soil 110:77–80
Harrison MJ, Van Buuren ML (1995) A phosphate transporter from the mycorrhizal fungus Gomus versiforme. Nature 378:626–629
Hattori R, Matsumura A, Yamawaki K, Tarui A, Daimon H (2013) Effects of flooding on arbuscular mycorrhizal colonization and root-nodule formation in different roots of soybeans. Agri Sci 4:673–677
Hijikata N, Murase M, Tani C, Ohtomo R, Osaki M, Ezawa T (2010) Polyphosphate has a central role in the rapid and massive accumulation of phosphorus in extraradical mycelium of an arbuscular mycorrhizal fungus. New Phytol 186:285–289
Hoseinzade H, Ardakani MR, Shahdi A, Asadi Rahmani H, Noormohammadi G, Miransari M (2016) Rice (Oryza sativa L.) nutrient management using mycorrhizal fungi and endophytic Herbaspirillum seropedicae. J Integr Agric 15:1385–1394
Hurek T, Reinhold-Hurek B (2003) Azoarcus sp. strain BH72 as a model for nitrogen-fixing grass endophytes. J Biotechnol 106:169–178
Ilag LL, Rosales AM, Elazegui FA, Mew TW (1987) Changes in the population of infective endomycorrhizal fungi in a rice-based cropping system. Plant Soil 103:67–73
Javot H, Penmetsa RV, Terzaghi N, Cook DR, Harrison MJ (2007) A Medicago truncatula phosphate transporter indispensable for the arbuscular mycorrhizal symbiosis. Proc Natl Acad Sci USA 104:1720–1725
Jiang YN, Wang WX, Xie QJ, Liu N, Liu LX, Wang DP, Zhang XW, Yang C, Chen XY, Tang DZ, Wang ET (2017) Plants transfer lipids to sustain colonization by mutualistic mycorrhizal and parasitic fungi. Science 356:1172–1175
Khan A (2004) Mycotrophy and its significance in wetland ecology and wetland management. In: Wong MH (ed) Wetlands ecosystems in Asia: function and management. Elsevier, Amsterdam
Khan MS, Zaidi A (2007) Synergistic effects of the inoculation with plant growth-promoting rhizobacteria and an arbuscular mycorrhizal fungus on the performance of wheat. Turk J Agri For 31:355–362
Kögel-Knabner I, Amelung W, Cao Zh S, Frenzel FP, Jahn R, Kalbitz K, Kölbl A, Schloter M (2010) Biogeochemistry of paddy soils. Geoderma 157:1–14
Kosaka Y, Takeda S, Sithirajvongsa S, Xaydala K (2006) Plant diversity in paddy fields in relation to agricultural. Econ Bot 60:49–61
Kour D, Rana KL, Yadav N, Yadav AN, Singh J, Rastegari AA et al (2019) Agriculturally and industrially important fungi: current developments and potential biotechnological applications. In: Yadav AN, Singh S, Mishra S, Gupta A (eds) Recent advancement in white biotechnology through fungi, Perspective for value-added products and environments, vol 2. Springer, Cham, pp 1–64. https://doi.org/10.1007/978-3-030-14846-1_1
Krajinski F, Courty PE, Sieh D, Franken P, Zhang HQ, Bucher M et al (2014) The HC-ATPase HA1 of Medicago truncatula is essential for phosphate transport and plant growth during arbuscular mycorrhizal symbiosis. Plant Cell 26:1808–1817
Kretzschmar T, Kohlen W, Sasse J, Borghi L, Schlegel M, Bachelier JB, Reinhardt D, Bours R, Bouwmeester HJ, Martinoia E (2012) A petunia ABC protein controls strigolactone-dependent symbiotic signalling and branching. Nature 483(7389):341–344
Lakshmipathy R, Balakrishna AN, Bagyaraj DJ (2012) Abundance and diversity of AM fungi across a gradient of land use intensity and their seasonal variations in Niligiri biosphere of the Western Ghats, India. J Agr Sci Tech 14:903–918
Lekberg Y, Koide RT (2005) Is plant performance limited by abundance of arbuscular mycorrhizal fungi? A meta-analysis of studies published between 1988 and 2003. New Phytol 168:189–204
Lekberg Y, Hammer EC, Olsson PA (2010) Plants as resource islands and storage units – adopting the mycocentric view of arbuscular mycorrhizal networks. FEMS Microbiol Ecol 74:336–345
Lin G, McCormack ML, Guo D (2015) Arbuscular mycorrhizal fungal effects on plant competition and community structure. J Ecol 103(5):1224–1232
Linkemer G, Board JE, Musgrave ME (1998) Waterlogging effects on growth and yield components in late–planted soybean. Crop Sci 38:1579–1584
Liu WJ, Zhu YG, Smith FA (2005) Effects of iron and manganese plaques on arsenic uptake by rice seedings (Oryza sativa L.) grown in solution culture supplied with arsenate and arsenite. Plant Soil 277:127–138
Liu J, Maldonado-Mendoza I, Lopez-Meyer M, Cheung F, Town CD, Harrison MJ (2007) Arbuscular mycorrhizal symbiosis is accompanied by local and systemic alterations in gene expression and an increase in disease resistance in the shoots. Plant J 50:529–544
Luginbuehl LH, Menard GN, Kurup S, Van Erp H, Radhakrishnan GV, Breakspear A, Oldroyd GE, Eastmond PJ (2017) Fatty acids in arbuscular mycorrhizal fungi are synthesized by the host plant. Science 6:763–775
Lui H, Zhang J, Christie P, Zhang F (2008) Influence of iron plaque on uptake and accumulation of Cd by rice (Oryza sativa L.) seedling grown in soil. Sci Total Environ 394:361–368
Lumini E, Vallino M, Alguacil MM, Romani M, Bianciotto V (2011) Different farming and water regimes in Italian rice fields affect arbuscular mycorrhizal fungal soil communities. Ecol Appl 21:1696–1707
Lynch JP (2007) Roots of the second green revolution. Aust J Bot 55:493–512
Maekawa T, Shimamura S, Shimada S (2011) Effects of short-term waterlogging on soybean nodule nitrogen fixation at different soil reductions and temperatures. Plant Proc Sci 14:49–358
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Mejstrik V (1965) Study on the development of endotrophic mycorrhiza in the association of Cladietum marisci. In: Macura J, Vancura V (eds) Plant microbe relationships. Czechoslovak Academy of Sciences, Prague, pp 283–290
Miao S, Shi H, Jian J, Judong L, Xiaobing L, Guanghua W (2012) Effects of short-term drought and flooding on soybean nodulation and yield at key nodulation stage under pot culture. J Food Agric Environ 10:819–824
Milly PCD, Wetherald RT, Dunne K, Delworth TL (2002) Increasing risk of great floods in a changing climate. Nature 415:514–517
Mitra S, Wassmann R, Vlek PL (2005) An appraisal of global wetland area and itsorganic carbon stock. Curr Sci 88:25–35
Munkvold L, Kjøller R, Vestberg M, Rosendahl S, Jakobsen I (2004) High functional diversity within species of arbuscular mycorrhizal fungi. New Phytol 164:357–364
Nakagawa T, Imaizumi-Anraku H (2015) Rice arbuscular mycorrhiza as a tool to study the molecular mechanisms of fungal symbiosis and a potential target to increase productivity. Rice 8:1–9
Nielsen KB, Kjoller R, Schweiger PF, Andersen FO, Rosendahl S (2004) Colonization and molecular diversity of arbuscular mycorrhizal fungi in the aquatic plants Littorella uniflora and Lobelia dortmanna in southern Sweden. Mycol Res 6:616–625
Nishigaki T, Tsujimoto Y, Rinasoa S, Rakotoson T, Andriamananjara A, Razafimbelo T (2019) Phosphorus uptake of rice plants is affected by phosphorus forms and physicochemical properties of tropical weathered soils. Plant Soil, 435(1–2):27–38
Nishiuchi S, Yamauchi T, Takahashi H, Kotula L, Nakazono M (2012) Mechanisms for coping with submergence and waterlogging in rice. Rice 5:1–14
Oehl F, Souza FA, Sieverding E (2008) Revision of Scutellospora and description of five new genera and three new families in the arbuscular mycorrhiza-forming Glomeromycetes. Mycotaxon 106:311–360
Okonji RE, ALadesanmi OT, Kuku A, Agboola FK (2008) Isolation and some properties of pattially purified Rhodanese from the hepatopancreas of giant freshwater prawn (Macrobrachium rosenbergii De Man). Ife J Sci 10:255–262
Palenzuela J, Ferrol N, Boller T, Azcón-Aguilar C, Oehl F (2008) Otospora bareai, a new fungal species in the Glomeromycetes from a dolomitic shrub-land in the National Park of Sierra de Baza (Granada, Spain). Mycologia 100:282–291
Parniske M (2008) Arbuscular mycorrhiza: the mother of plant root endosymbioses. Nat Rev Microbiol 6:763–775
Paszkowski U, Kroken S, Roux C, Briggs SP (2002) Rice phosphate transporters include an evolutionarily divergent gene specifically activated in arbuscular mycorrhizal symbiosis. Proc Natl Acad Sci USA 99(20):13324–13329
Rajeshkannan V, Sumathi Ch S, Manian S (2009) China National Rice Research Institute. Arbuscular mycorrhizal fungi colonization in upland rice as influenced by agrochemical application. Rice Sci 16:307–313
Rausch C, Daram P, Brunner S et al (2001) A phosphate transporter expressed in arbuscule-containing cells in potato. Nature 414:462–466
Ray AM, Inouye RS (2006) Effects of water-level fluctuations on the arbuscular mycorrhizal colonization of Typha latifolia L. Aquat Bot 84:210–216
Rillig MC, Mummey DL (2006) Mycorrhizas and soil structure. New Phytol 171:41–53
Sanni SO (1976) Vesicular-arbuscular mycorrhiza in some Nigerian soils: the effect of Gigaspora gigantea on the growth of rice. New Phytol 77:673–674
Schaarschmidt S, Roitsch T, Hause B (2006) Arbuscular mycorrhiza induces gene expression of the apoplastic invertase LIN6 in tomato (Lycopersicon esculentum) roots. J Exp Bot 57:4015–4023
Schüβler A, Schwarzott D, Walker C (2001) A new fungal phylum, the Glomeromycota: phylogeny and evolution. Mycol Res 105:1413–1421
Sieverding E, Oehl F (2006) Revision of Entrophospora and description of Kuklospora and Intraspora, two new genera in the arbuscular mycorrhizal Glomeromycetes. J Appl Bot Food Qual 80:69–81
Simpson EH (1949) Measurement of diversity. Nature 163:688
Smith SE, Read DJ (2008) Mycorrhizal Symbiosis. (3rdedn) Academic Press, San Diego, CA.
Smith SE, Read DJ (2010) Mycorrhizal symbiosis, 3rd edn. London, Academic
Smith SE, Smith FA (2011) Roles of arbuscular mycorrhizas in plant nutrition and growth: new paradigms from cellular to ecosystem scales. Annu Rev Plant Biol 62:227–250
Smith SE, Gianinazzi-Pearson V, Koide R, JWG C (1994) Nutrient transport in mycorrhizas – structure, physiology and consequences for efficiency of the symbiosis. Plant Soil 159:103–113
Smith SE, Facelli E, Pope S, Smith FA (2010) Plant performance in stressful environments: interpreting new and established knowledge of the roles of arbuscular mycorrhizas. Plant Soil 326:3–20
Solaiman MZ, Hirata H (1995) Effects of indigenous arbuscular mycorrhizal fungi in paddy fields on rice growth and N, P, K nutrition under different water regimes. Soil Sci Plant Nutr 41:505–514
Sondergaard M, Laegaard S (1977) Vesicular–arbuscular mycorrhiza in some aquatic vascular plants. Nature 268:232–233
Stevens KJ, Wall CB, Janssen JA (2011) Effects of arbuscular mycorrhizal fungi on seedling growth and development of two wetland plants, Bidens frondosa L, and Eclipta prostrata (L) L, grown under three levels of water availability. Mycorrhiza 21:279–288
Suzuki S, Kobae Y, Sisaphaithong T, Tomioka R, Takenaka C et al (2015) Differential growth responses of rice cultivars to an arbuscular mycorrhizal fungus, Funneliformis mosseae. J Hortic 2:142
Taylor JD, Helgason T, Öpik M (2017) Molecular community ecology of arbuscular mycorrhizal fungi. In: Dighton J, White JF (eds) The fungal community: its organization and role in the ecosystem, 4th edn. CRC Press, Boca Raton, pp 1–26
Terrer C, Vicca S, Hungate BA, Phillips RP, Prentice IC (2016) Mycorrhizal association as a primary control of the CO2 fertilization effect. Science 353:72–74
Tommerup IC, Sivasithamparam K (1990) Zygospores and asexual spores of Gigaspora decipiens: an arbuscular mycorrhizal fungus. Mycol Res 94:897–900
Treseder KK (2016) Model behavior of arbuscular mycorrhizal fungi: predicting soil carbon dynamics under climate change. Botany 94:417–423
Trolldenier G (1988) Visualisation of oxidising power of rice roots and of possible participation of bacteria in iron deposition. Z Pflanzenernaehr Bodenkd 151:117–121
Vallino M, Greppi D, Novero M, Bonfante P, Lupotto E (2009) Rice root colonisation by mycorrhizal and endophytic fungi in aerobic soil. Annu Appl Biol 154:195–204
Vallino M, Fiorilli V, Bonfante P (2014) Rice flooding negatively impacts root branching and arbuscular mycorrhizal colonization, but not fungal viability. Plant Cell Environ 37:557–572
Van der Heijden MGA, Klironomos JN, Ursic M, Moutoglis P, Streitwolf-Engel R, Boller T, Wiemken A, Sanders IR (1998) Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396:69–72
Van der Heijden MG, Martin FM, Selosse MA, Sanders IR (2015) Mycorrhizal ecology and evolution: the past, the present, and the future. New Phytol 205(4):1406–1423
Wang YT, Björn LO (2014) Heavy metal pollution in Guangdong province, China, and the strategies to manage the situation. Front Environ Sci 2:1–12
Wang YT, Qiu Q, Yang ZY, Hu ZJ, Tam NFY, Xin GR (2010) Arbuscular mycorrhizal fungi in two mangroves in South China. Plant Soil 331:181–191
Wang YT, Huang YL, Qiu Q, Xin GR, Yang ZY, Shi SH (2011) Flooding greatly affects the diversity of arbuscular mycorrhizal fungi (AMF) communities in the roots of wetland plants. PLoS One 6:e24512
Wang X, Yao H, Wong MH, Ye Z (2013) Dynamic changes in radial oxygen loss and iron plaque formation and their effects on Cd and As accumulation in rice (Oryza sativa L.). Environ Geochem Health 35:779–788
Wang YT, Li T, Li YW, Qiu Q, Li SS, Xin GR (2014) Distribution of arbuscular mycorrhizal fungi in four semi-mangrove plant communities. Ann Microbiol 65:603–610
Wang Y, Li T, Li Y, Bjorn LO, Rosendahl S, Olsson PA, Li S, Fud X (2015) Community dynamics of arbuscular mycorrhizal fungi in high-input and intensively irrigated rice cultivation systems. Appl Environ Microbiol 81:2958–2965
Wang Y, Li Y, Bao X, Björn LO, Sh L, Olsson PA (2016) Response differences of arbuscular mycorrhizal fungi communities in the roots of an aquatic and a semiaquatic species to various flooding regimes. Plant Soil 403:361–373
Watanarojanaporn N, Boonkerd N, Tittabutr P, Longtonglang A, Peter J, Young W, Teaumroong N (2013) Effect of rice cultivation systems on indigenous arbuscular mycorrhizal fungal community structure. Microb Environ 28:316–324
Wilde P, Manal A, Stodden M, Sieverding E, Hildebrandt U (2009) Biodiversity of arbuscular mycorrhizal fungi in roots and soils of two salt marshes. Environ Microbiol 11:1548–1546
Xu GH, Chague V, Melamed-Bessudo C et al (2007) Functional characterization of LePT4: a phosphate transporter in tomato with mycorrhiza-enhanced expression. J Exp Bot 58:2491–2501
Yadav AN, Singh S, Mishra S, Gupta A (2019) Recent advancement in white biotechnology through fungi. vol 3: Perspective for sustainable environments, Springer, Cham
Yang SY, Gronlund M, Jakobsen I, Grotemeyer MS, Rentsch D, Miyao A, Hirochika H, Kumar CS, Sundaresan V, Salamin N (2012) Nonredundant regulation of rice arbuscular mycorrhizal symbiosis by two members of the Phosphate Transporter1 gene family. The Plant Cell 24:4236–4251
Yimyam NS, Youpensuk J, Wongmo A, Kongpan B, Rerkasem K, Rerkasem B (2008) Arbuscular mycorrhizal fungi - An underground resource for sustainable upland agriculture. Biodivers Agric 9:61–63
Zhang XH, Zhu YG, Chen BD, Lin AJ, Smith SE, Smith FA (2005) Arbuscular mycorrhizal fungi contribute to resistance of upland rice to combined metal contamination of soil. J Plant Nutr 28:2065–2077
Zhang Q, Sun QX, Koide RT, Peng ZH, Zhou JX, Gu XG, Gao WD, Yu M (2014) Arbuscular mycorrhizal fungal mediation of plant-plant interactions in a marshland plant community. Scientific World J 2014:923610
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Novair, S.B., Hosseini, H.M.S., Etesami, H., Razavipour, T., Pirmoradian, N. (2020). The Role of Arbuscular Mycorrhizal Fungal Community in Paddy Soil. In: Yadav, A., Mishra, S., Kour, D., Yadav, N., Kumar, A. (eds) Agriculturally Important Fungi for Sustainable Agriculture. Fungal Biology. Springer, Cham. https://doi.org/10.1007/978-3-030-45971-0_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-45971-0_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-45970-3
Online ISBN: 978-3-030-45971-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)