Abstract
Disturbance can open new habitats that follow different plant colonization patterns. The Churince hydrological system from the Cuatro Ciénegas Basin has been desiccating progressively since 2003 because of the disturbance of the underground hydrological flow. The terminal lagoon and the last part of the Churince river have had no surface water since 2009, and the Intermediate Lagoon is now dry. The drying up of the system generated three new habitats: sinkholes, formed by the dispersion of soils in the South bank of the Churince river, the bed of the dry river, and the lacustrine bed of the desiccated Churince lagoon. Sinkholes, which started forming around 2003 at a high rate, are of different shapes, sizes, and depths. New sinkholes can be formed while others can be closed by the accumulation of sand and plants; others have persisted during at least nine years since their formation. Our aim was to record early vegetation stages in these three newly created habitats.
We found continuous changes in water content, as well as in plant species richness and plant cover. While water flowed towards the plain, many sinkholes had water, but today they are dry. Sinkholes are colonized mainly by riparian species, e.g., Samolus ebracteatus var. coahuilensis and Flaveria chlorifolia, which are also establishing in the dry river and lacustrine beds. Species composition and frequency in the sinkholes change as water availability diminishes, reducing the frequency of the more hydrophilic species like Schoenus nigricans and S. ebracteatus var. coahuilensis and increasing that of less water demanding and more salt and gypsum tolerant plants, as Sporobolus airoides and F. chlorifolia. The same pattern is expected in the lake and river beds, where colonization started several years later than in the sinkholes. Vegetation patterns and species composition and cover were influenced by the availability of water, which diminished in time due to the desiccation of the hydrological system.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Disturbance is a relatively discrete event in time with disruptive effects on environmental physical and biotic features that can have structuring effects at different ecological scales affecting ecosystems, communities, and populations (Turner 1989). A common result of disturbance is the formation of new habitats because of the destruction and substitution of the original habitats, and the consequent effects on nearby places. Colonization processes usually follow this opening of new habitats (White and Jentsch 2001; White and Pickett 1985).
Desert springs and wetlands, as well as other isolated water bodies, are intensely used and overexploited all over the world. Several underground water systems have been recently reduced, or even have completely disappeared, thus deeply disturbing nearby and long-distance terrestrial ecosystems depending on them (Briggs 1996; Capon and Dowe 2007; Hendrickson et al. 2008; Unmack and Minckley 2008; Sivinski and Tonne 2011; Souza et al. 2018).
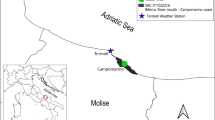
The disturbance of ground water systems in the Cuatro Ciénegas Basin (CCB) and in nearby valleys, like Ocampo and El Hundido, due to overexploitation of aquifers mainly for agriculture, is having huge ecological consequences (Evans 2005; Souza et al. 2006, 2018; Rodríguez et al. 2007; Wolaver et al. 2008; Pisanty et al. 2013), including the progressive desiccation of the Churince system (Fig. 16.1), which has shown alterations continuously since 2003 (Souza et al. 2006, 2018; Pisanty et al. 2013). The first parts to lose water in the Churince system were the furthest from the spring at poza Churince, located near the base of the San Marcos y Pinos sierra; the largest terminal lake of the basin (i.e., the Churince Lagoon) was the first to dry up in 2008, followed by the nearby river mouth (RM), the part of the river that drained in this lagoon, and by the Intermediate Lagoon, that dried recently, in 2018 (De Anda et al. 2018).
The first consequences of this major disturbance are the loss of surface water and, consequently, the change of the riparian habitats, followed by the opening of new disturbance-induced habitats represented by the dry beds of the Churince river (DRB) and Churince lagoon (DLB), the latter including the dry RM. The lagoon and the RM have been dry since 2009. As the lagoon dried, its bed first became marshy and then firmer, with less evidence of subsurface water. In the terminal part of the system, the river began to desiccate in 2009, and has been dry since 2012. However, during this study period there was still water in the last part before the Intermediate Lagoon, and today some water remains between the latter and the spring, implying a water gradient between the final and the initial part of the system. In 2015, seedlings and some mature plants of several species could be found in the part of the river where water was no longer flowing, along ca. 600 m from the RM. In the lake bed, one species (Flaveria chlorifolia) was the main colonizer. Additionally, a complex set of sinkholes (S) was formed starting in 2003 in the surroundings of the last part of the river and of the Intermediate Lagoon, due to the subsurface flow of the water that could not reach the RM (Pisanty et al. 2013).
In this chapter, using both community and population dynamics, we describe the plant colonization process of the different environments generated by the disturbance caused by the progressive drying of the Churince system, including sinkholes (S), the dry lake bed (DLB), and the dry river bed (DRB). These analyses allow us to understand the patterns of early successional stages in the three environments and the ecological behavior of the different plant species that colonize them, most of which characterized the original riparian habitat.
Methods
Study Site
Cuatro Ciénegas Basin is located at 27° 11′ 24″–26° 42′ 36” N and 102° 48′ 00″–101° 54′ 36” W, in the northern state of Coahuila, Mexico. It has an arid climate, with an annual average precipitation of 212 mm (CONANP 1999) which fluctuates considerably (Montiel-González et al. 2018). It is characterized by a high specific and ecosystem diversity (Minckley 1969; Souza and Eguiarte 2018) that includes aquatic and semiaquatic systems that belong to five different hydrological systems. Many pozas (spring fed pools) can be found in different parts of the basin. Pits and pozas, due to substrate characteristics and the flow of underground water, can be ephemeral or long lasting, so their exact number is impossible to determine, but Hendrickson et al. (2008) recognized that the basin contains more than 200 water bodies, including pozas, springs, and rivers, as well as two terminal lakes, and Meyer (1973) reported the formation of temporal, ephemeral pits. One of the systems, known as Churince System, is formed by the poza Churince, where the spring that feeds the system is located, the Churince River, an intermediate lagoon and the Churince Lagoon, the largest terminal (or desiccation) lake of the basin (Fig. 16.1).
Three different new environments were formed due to the desiccation process:
-
1.
Sinkholes (S) . A set of soil depressions formed since 2003 in ca. 600 m2, in a semi-parallel pattern along the South bank of the river in the terminal part of the system. These soil depressions are formed when water dissolves salt and gypsum particles, causing the loss of cohesion between them (Umesh et al. 2011). Sinkholes are relatively discrete environments, with either water or high humidity, where the extreme temperatures and strong winds of the desert are buffered (Fig. 16.2a). Originally, the flatland was recognized as a zone with no evident vegetation (Pinkava 1984), and only sparse, short stems of the halophyte Distichlis spicata (Poaceae) were found.
-
2.
Dry river bed (DRB) . The last part of the river remains without surface water since the end of 2009. Many seedlings of riparian species can be found since the end of 2015 (Fig. 16.2b).
-
3.
Dry lake bed (DLB) . Churince lagoon remains without surface water since 2009. The RM is included in this environment. Flaveria chlorifolia (Asteraceae), an herbaceous perennial plant associated with humid, saline and gypsic soils, started establishing in 2014, forming discrete patches (Fig. 16.2c).
Field Methods
-
1.
Sinkhole dynamics and colonization
A census of the sinkholes was made from January 2008 to September 2017. In a total of 51 census, all open sinkholes were considered, and new ones were identified and incorporated as such. When a sinkhole closed, it was also registered and kept under observation because some re-opened. Re-opened sinkholes were not considered as new ones, and they were counted as open.
In every census, the presence or absence of water was registered, and specific composition, richness, and individual plant cover were determined in every sinkhole. Specific cover was determined adding individual cover, approximated to the area of an ellipse.
-
2.
Colonization of the river bed
In June 2015, 13 plots were set every 50 m along the 600 m between the RM and the part of the river that still had surface water. Eight plots were 2 m × 10 m, and five, placed in the narrower part of the river, were 0.9 m × 10 m. Bimonthly, from June 2015 to January 2017, all plants in the plots were identified and plant cover was determined.
Frequency and plant cover of species colonizing the river bed from June 2015 to January 2017 were analyzed with an ANOVA. A Bray–Curtis similarity index was calculated for plant cover.
-
3.
Colonization of the lake bed
After identifying F. chlorifolia as almost the only species establishing in the DLB, the demography of this species was chosen as the tool to analyze the colonization of this environment, contrasting with the community approach used in the other two. Six plots of 5 m × 5 m were set where patches of the species were found. In each plot, all individuals were numbered and tagged. Height and reproductive stage of each individual were registered bimonthly from September 2015 to September 2017. Established plants were classified in four height categories (1 = 0.1–4 cm; 2 = 5–15 cm; 3 = 16–30 cm; 4 ≥ 30 cm), and additional categories were included for the seed stage and for adult plants with dry aerial parts. Height was chosen to build categories because it is more accurate than cover due to the growth form and branching pattern of this species.
A Lefkovitch average transition probability matrix was built for each zone, followed by projection matrices, using the standard iterative procedure (Caswell 2001). The finite growth rate (λ) was calculated once the stable stage distribution was obtained and elasticities (De Kroon et al. 1986; Caswell 2001) were calculated to evaluate the relative contribution of each process to the finite growth rate.
Results
1. Colonization of Sinkholes
A total of 247 sinkholes were censed between January 2008 and September 2017. In January 2008, when the census started, 126 sinkholes were identified. The maximum number of open sinkholes (157) was registered in June 2010, while the minimum (62) corresponds to August 2017 (Fig. 16.3). Sixty-two new sinkholes were formed between January 2008 and November 2011, while there was still water in the river, and another 61 opened since January 2012, when the latter was already dry. Contrastingly, only 22 sinkholes closed while the river had water and 162 did so when it was dry (Fig. 16.3).
Most sinkholes had plants throughout all the census period, but the presence of water was variable and, overall, the proportion of sinkholes with water decreased through time. In the first census, 50.79% of the sinkholes had water, but since 2012, when the river bed dried, only a few did. From May 2016 until September 2017 none had water (Fig. 16.3).
A total of 15 plant species colonized the sinkholes (Table 16.1). The average number of species in sinkholes with plants was 2.39, with a maximum of seven and a minimum of one species. Most of these species are riparian perennial herbs. Some sinkholes had no plants in some census. The most frequent species throughout the census period were Samolus ebracteatus var. coahuilensis, Flaveria chlorifolia, Distichlis spicata, Schoenus nigricans, and Sporobolus airoides (Table 16.1; Fig. 16.4). While water availability in the sinkholes was high, S. nigricans and S. ebracteatus var. coahuilensis were very frequent: however, as the river and the sinkholes lost their water, their frequency decreased (Fig. 16.4a, b). The frequency of F. chlorifolia (Fig. 16.4c) increased in 2015, and that of D. spicata remained relatively constant, although it periodically decreases in the cold season (Fig. 16.4d). S. airoides showed a low frequency during the first years, but since 2013 it increased until it was present in more than half of the sinkholes (Fig. 16.4e). Frequencies of the remaining species were much lower and are not shown in the figure.
Plant cover of the five most frequent species changed through time (Fig. 16.5). The loss of cover of S. ebracteatus var. coahuilensis coincides with the decrease of its frequency, while for S. nigricans frequency decreases more than cover, indicating the prevalence of large clumps in some sinkholes. The cover of S. airoides tends to increase with its frequency, while that of D. spicata remains low almost constantly, despite its high frequency.
2. Colonization of the River Bed
Ten different riparian species of plants colonized the DRB from June 2015 to June 2017 (Table 16.1). In this environment, three statistically significantly different groups based on species frequencies were identified with an ANOVA (F8,12 = 14.92, p < 0.0001): (1) F. chlorifolia, S. airoides, D. spicata, S. ebracteatus var. coahuilensis, and Sesuvium verrucosum (highest frequency); (2) S. verrucosum and S. nigricans (intermediate frequency); and (3) Sabatia tuberculata, Eustoma exaltatum, Prosopis glanudulosa, and Haploesthes robusta (lowest frequency).
Plant cover on the river bed changed in time (F8,12 = 14.92, p < 0.0001). The maximum cover values were registered on November 2015, and June and August 2016, while its minimum values were on January 2016 and 2017, showing a seasonal pattern. Lower plant cover values were registered in the coldest month (January) in both years. Specific plant cover varied between plots (Fig. 16.6a). The highest plant cover was in plots 7–13, and the lowest in plots 1–6. F. chlorifolia, S. airoides, and D. spicata were found in all plots (Fig. 16.6a); S. ebracteatus var. coahuilenis was not present in plots 3 and 13, and S. nigricans was found only from plot 6 to 12. All the other species were found in almost all plots but with a lower cover (Fig. 16.6b).
The Bray–Curtis similarity analysis shows three main clusters (Fig. 16.6b), one (S1) corresponding to the plots near the RM (1 to 6 plus 8), and two other (S2) closer to where water was still found, one including plots 7 and 9–11, and another formed by plots 12 and 13 (Fig. 16.6b).
3. Colonization of the Lake Bed
The DLB was the last disturbance-induced environment to be colonized. A total of 316 individuals of F. chlorifolia were monitored bimonthly. At the end of the study, only 20% of these individuals survived. Only two recruitments were observed.
During the coldest month (January) of 2016 and 2017, over 50% of the individuals entered the desiccated category but, after the cold period, meristems were activated and plants began to grow again, reaching the highest number of plants in the category of the tallest individuals (category 5) at the end of summer (Fig. 16.7).
The finite growth rate (λ = 0.77) indicates that the population is decreasing by 23%. The elasticity analysis showed that the two main processes that contribute the most to lambda are the stasis of the tallest individuals and of those that desiccated (0.13). The third most important process is the stasis in the category 3 (0.08) (Fig. 16.7).
Discussion
Disturbance in arid and semiarid ecosystems generates spatial heterogeneity, which plays an influential role in population and community dynamics, and explains, among other things, patchy plant distributions at both levels. Ecological succession , as a regional process, is seldom observed in these systems, because it is extremely slow; however, colonization of microenvironments or microhabitats, and differences in population or community behavior are common (Polis 1991; Mandujano et al. 1998; Vega and Montaña 2004). The same type of process is observed after disturbance both in the disturbed habitat and in its neighborhood.
Riparian habitats in deserts are more mesic than the surrounding areas (Briggs 1996). Species growing in different types of desert wetlands and springs face different selective pressures that include anoxic environments for the roots of plants growing on continuously or intermittent flooded sites, xeric conditions for aerial parts, and different types of saline or gypsic conditions (Briggs 1996; Ezcurra et al. 1998; Capon and Dowe 2007). The colonization potential of these species depends on their capacity to adapt to this complex set of selective forces. This capacity is evident in the riparian species , characteristic of the borders of undisturbed water bodies that colonize the environments created by disturbance as the S, DRB, and DLB analyzed here.
The three different habitats formed after the disturbance of the Churince system that we considered in this study have different characteristics and the periods in which colonization has been taking place vary among them; nonetheless, they are consistently and predominantly colonized by the same set of riparian species.
Sinkholes proved to be very dynamic since they can remain open, or close permanently or temporarily, and new ones can be formed. Formation of new sinkholes decelerated progressively as water from the river diminished together with the sublevel flow, for the dispersion of soils does not happen without the presence of water that dissolves salts and causes loss of particle cohesion (Umesh et al. 2011). Sinkholes closed more frequently after they stopped having water. Although the same five species remained as the most frequent through time, specific frequencies did change, together with cover. While the river had water, many sinkholes did also, and the most frequent species were the most hydrophilic, as S. ebracteatus var. coahuilensis and S. nigricans. As sinkholes became drier, species that are less water demanding, as S. airoides, and more tolerant to gypsum, as F. chlorifolia , became more frequent. Plant cover strongly depends on growth forms, but it reflects the success a species has in a given environment (Kent 2011). The loss through time of plant cover of some of the first colonizers, as S. ebracteatus and S. nigricans , reflects the change of conditions and of selective pressures as sinkholes become drier, as well as the limited responses some species have. Changes take place both seasonally and in a longer term. Thus, frequency reflects the colonizing ability of a species, while cover indicates its success after establishment. As underground and sublevel water continue to decrease, the more water demanding species, as S. ebracteatus var. coahuilensis and S. nigricans , are likely to continuously become less frequent and lose plant cover, and eventually disappear, while more resistant species as F. chlorifolia and S. airoides become dominant, at least temporarily. However, in a longer term, we can expect all riparian species will become locally extinct in the Churince system as desiccation continues. The concentration of salt and gypsum is probably changing due to the disturbance of the hydrological system, and some gypsophytes as Nerisyrenia incana and Nama spp. (see Ochoterena et al. 2020, this volume), formerly absent, can now be found on the flatland (pers. obs.).
The presence of water in the sinkholes depends on many factors, including sublevel availability of water and meteorological conditions, as seen in August 2010, when the number of sinkholes with water increased due to high precipitations caused by a hurricane. The peak of sinkholes with water in 2013 was caused because the main gate through which water is extracted from the basin through an artificial canal, that diverts it even beyond CCB, was closed for a short period, allowing the wetland and the river to recover water temporarily (Souza and Eguiarte 2018). As soon as the gate was opened again, water was lost and was never recovered.
The DRB was colonized much later than the sinkholes, because it only became an available habitat for terrestrial plants until the end of 2014. Despite the short period of colonization, the bed was quickly occupied by 10 riparian species, all found also in sinkholes with similar frequencies for that time period. Seeds allowing this colonization process were probably dispersed from recently formed fruits by plants on the sinkholes and from other cohorts represented in the seed bank. The DRB did not have the same conditions over space and time. The CCB has clear seasonal changes (De Anda et al. 2018) which explain the observed short term variations of plant cover on the DRB. Plant cover was higher in the rainy months than in the dry ones. The Bray–Curtis similarity analysis showed a spatially discontinuous establishment pattern along the DRB, determined by the distance between plots and the presence of nearby water. Two zones can be recognized: S1 was the zone of the DRB with drier soils, because it was the first to desiccate. In S2, since superficial water was intermittently present, water availability was higher, a fact confirmed by the presence of S. nigricans. The highest number of species was found in S2 apparently because the more tolerant plants, which can stand hydric stress produced by desiccation, were found in it as well as in S1. Plant cover of the more tolerant species was higher in S2 than in S1, confirming their hydrophilic character. Plant cover was relatively scarce except for plot 7, where it reached more than 100%, which could be explained by the higher humidity in this plot that enhanced the cover of F. chlorifolia .
Although the Churince lagoon was the first part to desiccate, it passed through a very swampy period before becoming suitable for plant establishment, which started later than in the sinkholes and on the DRB, probably due to its high gypsum content and low nutrient concentration (Escudero et al. 2014). Although scattered and scarce individuals of S. ebracteatus var coahuilensis and D. spicata could be found, F. chlorifolia was the main colonizer on the DLB , probably resulting of a massive, but punctual, germination and establishment event, associated to an unidentified environmental trigger and to its successful germination in natural conditions (Peralta-García et al. 2016). Establishment of F. chlorifolia was enhanced by its tolerance and relative affinity to gypsum soils (Powell 1978; Flores-Olvera et al. 2016). Despite its seasonal decline, its perennial character allows successful individuals to persist, an event relevant to its population dynamics, as observed in the elasticities values. The reactivation of meristems after the cold season is also crucial for the population, and it is the second major process of importance over the population growth rate. However, the high mortality rates in the DLB coincided with the death of most of the established shrubs in 2017 (pers. obs.), after a long and cold winter which, apparently, limited the survival of meristems.
Colonizers of the new habitats can show characteristics identified as favorable for the colonization of new microsites in deserts (Ezcurra et al. 2020, this volume), as high fecundity rates (Noy-Meir 1973, 1985), like in S. ebracteatus var. coahuilensis (Pisanty et al. 2013) and F. chlorifolia (Rodríguez-Sánchez 2018), and rapid germination responses, as those reported by Peralta-García et al. (2016) for them, E. exaltatum, and S. tuberculata, as well as for S. nigricans (Peralta-García et al. 2020, this volume). Clonality, present at least in S. ebracteatus var coahuilensis, S. nigricans, S. airoides, and D. spicata, has also been identified as a successful strategy for establishment in arid zones (Montaña et al. 1990; Mandujano 2007). All these characteristics play a relevant role in the way each species responds to disturbance.
Heterogeneity in arid and semiarid ecosystems plays an influential role in population and community dynamics, and explains, among other things, patchy distributions at both levels. Succession, as a regional process, is seldom observed in these systems, because it is extremely slow; however, colonization of microenvironments or microhabitats, and differences in population or vegetation dynamics are common (Polis 1991; Mandujano et al. 1998; Vega and Montaña 2004), although their effect at a larger scale are unknown.
The colonization of the three newly formed habitats reported in this chapter offers a unique possibility of observing succession after a disturbance on a semiarid zone in newly formed, different microenvironments that offer, at least temporarily, water availability, but are heterogeneous in space and in time. Changing conditions force the process to be dynamic in a relatively short time, and the fact that riparian species are the colonizers indicate the presence of some water that makes these habitats suitable for species that used to live close by.
The changing conditions , especially in sinkholes, are reflected by the change in frequency and cover of the main colonizing species. S. airoides can grow in alkaline and gypsic soils that offer water at least temporarily, and tolerate both drought and flooding once established. (USDA 2018), which explains the increase in frequency observed throughout this study. The increasing presence of P. glandulosa indicates there is still deep ground water available (Nilsen et al. 1983). Seeds of this species were spread by animals, mainly by horses that used to roam the study site grazing and looking for water.
The disappearance of the Gentianaceae Sabatia tuberculata from the borders of water bodies (pers. obs.) and its increasingly low frequency in the sinkholes is a cause of concern, because this is an endemic species (Williams 1982; Hendrickson et al. 2008; Vela Coiffier et al. 2015; Villarreal-Quintanilla et al. 2017) and it is not known whether it is endangered or not (SEMARNAT 2010; Pisanty and Rodríguez-Sánchez 2017).
The presence of riparian plants away from their natural habitats indicates the latter are being disturbed. When these species grow in areas where they could not establish before, they indicate the presence of water in areas that are usually dry, and their sole presence should be considered as an early warning of underground disturbance, even if this is not evident in the water bodies that seem undisturbed.
References
Briggs MK (1996) Riparian ecosystem recovery in arid lands. The University of Arizona Press, Tucson, AZ, USA
Capon S, Dowe JL (2007) Diversity and dynamics of riparian vegetation. In: Lovett S, Price P (eds) Principles of riparian lands management. Land & Water Australia, Canberra, Australia, pp 13–32
Caswell H (2001) Matrix population models. Construction, analysis, and interpretation. Sinauer Associates Inc, Sunderland, MA, USA
Comisión Nacional de Áreas Naturales Protegidas (CONANP) (1999) Programa de manejo del Área de Protección de Flora y Fauna Cuatrociénegas. Instituto Nacional de Ecología SEMARNAP, México, DF, Mexico
De Anda V, Zapata-Peñasco I, Eguiarte LE, Souza V (2018) Towards a comprehensive understanding of environmental perturbations in microbial mats from the Cuatro Ciénegas Basin by network inference. In: García-Oliva F, Elser J, Souza V (eds) Ecosystem ecology and geochemistry of Cuatro Ciénegas. How to survive in an extremely oligotrophic site. Springer International, Cham, Switzerland, pp 85–98
De Kroon H, Plaisier A, van Groenendael J, Caswell H (1986) Elasticity: the relative contribution of demographic parameters to population growth rate. Ecology 67(5):1427–1431
Escudero A, Palacio S, Maestre FT, Luzuriaga AL (2014) Plant life in gypsum: a review of its multiple facets. Biol Rev 90(1):1–18
Evans S (2005) Using chemical data to define flow systems in Cuatro Cienegas. MSc thesis, University of Texas in Austin, Austin, TX, USA
Ezcurra E, Felger RS, Russell AD, Equihua M (1998) Freshwater island in a desert sand sea: the hydrology, flora and phytogeography of the Grand Desierto oasis of Northwestern Mexico. Desert Plants 9:35–63
Ezcurra E, Martínez-Berdeja A, Villanueva-Almanza L (2020) The evolution of North American deserts and the uniqueness of Cuatro Ciénegas. In: Mandujano MC, Pisanty I, Eguiarte LE (eds) Plant diversity and ecology in the Chihuahuan Desert. Springer International, Cham, Switzerland
Flores-Olvera H, Czaja A, Estrada JL, Méndez UR (2016) Floristic diversity of halophytic plants of Mexico. In: Khan M, Boër B, Ȫzturk M, Clüsener M, Gul B, Breckle SW (eds) Sabkha ecosystems. Tasks for vegetation science, vol 48. Springer, Cham, Switzerland
Hendrickson DA, Marks JC, Moline AB, Dinger EC, Cohen AE (2008) Combining ecological research and conservation: a case study in Cuatro Ciénegas, Mexico. In: Stevens L, Meretsky VJ (eds) Aridland springs in North America: ecology and conservation. University of Arizona Press, Tucson, AZ, USA, pp 127–157
Kent M (2011) Vegetation description and data analysis. Willey-Blackwell, Chichester, UK
Mandujano MC, Montaña C, Méndez I, Golubov J (1998) The relative contributions of sexual reproduction and clonal propagation in Opuntia rastrera from two habitats in the Chihuahuan Desert. J Ecol 86:911–921
Mandujano MC (2007) La clonalidad y sus efectos en la biología de poblaciones. In: Eguiarte LE, Souza V, Aguirre X (eds) Ecología Molecular. Secretaría de Medio Ambiente y Recursos Naturales, Instituto Nacional de Ecología, Universidad Nacional Autónoma de México, Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México, DF, Mexico, pp. 215–250
Meyer ER (1973) Late-quaternary paleoecology of the Cuatro Ciénegas Basin, Coahuila, México. Ecology 54(5):982–995
Minckley WL (1969) Environment of the Bolsón of Cuatro Ciénegas, Coahuila, Mexico: with special reference to the aquatic biota. Science Series, vol 2. Texas Western Press, El Paso, TX, USA
Montaña C, Lopez-Portillo J, Mauchamp A (1990) The response of two woody species to the conditions created by a shifting ecotone in an arid ecosystem. The Journal of Ecology 78(3):789
Montiel-González C, Bautista F, Delgado C, García-Oliva F (2018) The climate of Cuatro Ciénegas Basin: drivers and temporal patterns. In: Souza V, Olmedo-Álvarez G, Eguiarte LE (eds). Cuatro Ciénegas Ecology, Natural History and Microbiology. Springer International, Cham, Switzerland, pp 35–44
Nilsen ET, Sharifi MR, Rundel PW, Jarrell WM, Virginia RA (1983) Diurnal and seasonal water relations of the desert phreatophyte Prosopis glandulosa (honey mesquite) in the Sonoran desert of California. Ecology 64:1381–1393
Noy-Meir I (1973) Desert ecosystems: Environment and producers. Annu Rev Ecol Syst 4(1):25–51
Noy-Meir I (1985) Desert ecosystem structure and function. In Evanari M, Noy Meir I, Goodall DW (eds) Ecosystems of the World. Elsevier, Amsterdam, Netherlands
Ochoterena H, Flores-Olvera H, Gómez-Hinostrosa C, Moore MJ (2020) Gypsum and plant species: a marvel of Cuatro Ciénegas and the Chihuahuan Desert. In: Mandujano MC, Pisanty I, Eguiarte LE (eds) Plant diversity and ecology in the Chihuahuan Desert. Springer International, Cham, Switzerland
Peralta-García C, Sánchez-Coronado ME, Orozco-Segovia A, Orozco-Segovia S, Pisanty-Baruch I (2016) Germination of four riparian species in a disturbed semi-arid ecosystem. S Afr J Bot 106:110–118
Peralta-García C, Pisanty I, Orozco-Segovia A, Sánchez-Coronado ME, Rodríguez-Sánchez M (2020) Germination of riparian species in natural and experimental conditions. In Mandujano MC, Pisanty I, Eguiarte LE (eds.) Plant diversity and Ecology in the Chihuahuan desert. Springer International, Cham, Switzerland
Pinkava DJ (1984) Vegetation and Flora of the bolson of Cuatro Cienegas region Coahuila, Mexico: IV. Summary, endemism and corrected catalogue. J Ariz Nev Acad Sci 19(1):23–47
Pisanty I, Pérez y Sosa M, Gálvez G (2013) Agriculture, water mismanagement and ecosystem transformations in the Cuatrociénegas Valley in the Chihuahuan Desert, Mexico. In: Schwabe K, Albaic J, Connor JD, Hassan RM, Meza L (eds) Drought in arid and semi-arid regions, a multi-disciplinary and cross-country perspective. Springer, Dordrecht, Netherlands, pp 199–216
Pisanty I, Rodríguez-Sánchez M (2017) Amenazas a la biodiversidad en Cuatro Ciénegas. In: La biodiversidad de Coahuila. Estudio de Estado. Vol. I CONABIO/Gobierno del Estado de Coahuila de Zaragoza, Saltillo, Coahuila, Mexico, pp 485–498
Polis GA (1991) The ecology of desert communities. The University of Arizona Press, Tucson, AZ, USA
Powell AM (1978) Systematic of Flaveria (Flaveriinae-Asteraceae). Ann Mo Bot Gard 65(2):590–636
Rodríguez JM, Souza V, Arriaga L (2007) Effect of the overexploitation of the aquifer of the Hundido valley and the impact on the ecological reserve of Cuatrociénegas, Coahuila. Ciencia FIC. Universidad Autónoma de Nuevo León 1:32-38
Rodríguez-Sánchez M (2018) Dinámica poblacional, germinación, establecimiento y crecimiento de Flaveria chorifolia (Asteraceae) en un sistema perturbado en el Valle de Cuatrociénegas, Coahuila, México. Master Thesis, Facultad de Ciencias, Universidad Nacional Autónoma de México, Mexico City, Mexico
Secretaría de Medio Ambiente y Recursos Naturales (SEMARNAT) (2010) Norma Oficial Mexicana NOM-059-SEMARNAT-2010. Published December 30, 2010, México, DF, Mexico
Sivinski RC, Tonne P (2011) Survey and assessment of aridland spring ciénegas in the southwest region. Section 6, Segment 25, Progress Report submitted to: NM Forestry Diversion Santa Fe and USDI-Fish and Wildlife Service, Region 2, Albuqerque, NM, USA
Souza V, Eguiarte LE (2018) In the beginning, there was fire: Cuatro Ciénegas Basin (CCB) and the long history of life on earth. In: Souza V, Olmedo-Álvarez G, Eguiarte LE (eds). Springer International, Cham, Switzerland, pp 21–34
Souza V, Espinosa L, Escalante A, Eguiarte LE, Farmer J, Forney L, Lloret L, Rodríguez J, Soberón X, Dirzo R, Elser J (2006) An endangered oasis of aquatic microbial biodiversity in the Chihuahuan desert. Proc Natl Acad Sci USA 103(17):6565–6570
Souza V, Eguiarte LE, Elser J, Travisano M, Olmedo-Álvarez G (2018) A microbial saga: how to study an unexpected hot spot of microbial biodiversity from scratch? In: Souza V, Olmedo-Álvarez G, Eguiarte LE (eds) Cuatro Ciénegas ecology, natural history and microbiology. Springer International, Cham, Switzerland, pp 1–18
Turner MG (1989) Landscape ecology: the effect of pattern on process. Annu Rev Ecol Syst 20:171–197
Umesh T, Dinesh S, Sivapullaiah P (2011) Characterization of dispersive soils. Mater Sci Appl 2(6):629–633
United States Department of Agriculture (USDA) (2018) Natural Resources Conservation Service. https://plants.usda.gov/core/profile?symbol=SPAI. Accessed 4 Jan 2019
Unmack PJ, Minckley WL (2008) The demise of desert springs. Aridland springs in North America: ecology and conservation. University of Arizona Press, Tucson, AZ, USA, pp 11–34
Vega E, Montaña C (2004) Spatio-temporal variation in the demography of bunch grass in a patchy semiarid environment. Plant Ecol 175(1):107–120
Vela Coiffier MP, Vela Coiffier EL, Lozano García DF (2015) Evaluación del conocimiento actual del APFF de Cuatrociénegas, Coahuila: FASE 1. Instituto Tecnológico y de Estudios Superiores de Monterrey. Centro de Calidad Ambiental. Informe final SNIB-CONABIO, proyecto No. JN002. México, D.F., Mexico
Villarreal-Quintanilla JA, Bartolomé JA, Estrada E, Ramírez H, Martínez SJ (2017) El elemento endémico de la flora vascular del Desierto Chihuahuense. Acta Bot Mex 118:65–96
White PS, Jentsch A (2001) The search for generality in studies of disturbance and ecosystem dynamics. In: Esser K, Lüttge U, Kadereit JW, Beyschlag W (eds) Progress in Botany, vol 62. Springer, Berlin, Germany, pp 399–450
White PS, Pickett STA (1985) Natural disturbance and patch dynamics: an introduction. In: Pickett STA, White PS (eds) The ecology of natural disturbance and patch dynamics. Academic Press, New York, NY, USA, pp 3–13
Williams JE (1982) A new species of Sabatia (Gentianaceae) from the Chihuahuan Desert. Southwest Nat 27(4):379–382
Wolaver B, Sharp JM Jr, Rodríguez JM, Ibarra Flores JC (2008) Delineation of regional arid karstic aquifers: an integrative data approach. Ground Water 46(3):396–413
Acknowledgements
This research was financed by programs UNAM/PAPIIT IN231811 and UNAM/PAPIIT IN217915, and supported by the Department of Ecology and Natural Resources (Facultad de Ciencias, UNAM) and the alliance WWF-Carlos Slim Foundation. MRS was supported by a scholarship from CONACyT. We thank Juan Carlos Ibarra, director of the Cuatro Ciénegas protected area, Valeria Souza, and Martín Carrillo for their constant support and commitment; Cristina Pérez y Sosa, and Erick Peña for their contributions, and Gabriel Gálvez and Jazmín Sánchez for their help. We thank the experts of the National Herbarium (UNAM), the Herbario de la Facultad de Ciencias (UNAM), and Patricia Dávila, Nelly Diego, Hilda Flores-Olvera, Manuel Villarreal-Quintanilla, and Manuel Rosas for the identification of species. We also acknowledge Israel Solano Zavaleta, Mariana Hernández-Apolinar, Pedro E. Mendoza-Hernández and Marco Romero Romero for their technical support, as well as the people of the Ejido Seis de Enero and Alma Zertuche. The advice of the late Salvador Contreras is fully appreciated. This chapter constitutes a partial fullfillment of the Doctorado en Ciencias (Biología) of the Universidad Nacional Autónoma de México (UNAM) of Irene Pisanty Baruch
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Pisanty, I., Rodríguez-Sánchez, M., Orozco, P.T., Granados-Hernández, L.A., Escobar, S., Mandujano, M.C. (2020). Disturbance and the Formation and Colonization of New Habitats in a Hydrological System in the Cuatro Ciénegas Basin. In: Mandujano, M., Pisanty, I., Eguiarte, L. (eds) Plant Diversity and Ecology in the Chihuahuan Desert. Cuatro Ciénegas Basin: An Endangered Hyperdiverse Oasis. Springer, Cham. https://doi.org/10.1007/978-3-030-44963-6_16
Download citation
DOI: https://doi.org/10.1007/978-3-030-44963-6_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-44962-9
Online ISBN: 978-3-030-44963-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)