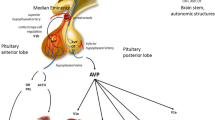
Abstract
Vasopressin (AVP) represents the key endocrine regulator of water balance. It is synthesized in the hypothalamic supraoptic and paraventricular nuclei and then transported to the neurohypophysis, where it is released in the bloodstream. Alterations in plasma osmolality represent the main input affecting AVP secretion, yet many other physiological and pathophysiological conditions can intervene in modulating its levels, as hypovolemia and hypotension. AVP primarily controls plasma osmolality and fluid volume by inducing synthesis and insertion of essential water transport proteins in the kidneys, thus reabsorbing water into blood circulation and reducing diuresis. Moreover, AVP exerts vascular and platelet control and intervenes in several metabolic pathways, including glycogenolysis and gluconeogenesis. Furthermore, AVP stimulates the release of adrenocorticotropic hormone (ACTH). AVP exerts its multiple effects by binding to its receptors, classified in different subtypes depending on tissue expression, function, and second messengers. Disorders in AVP synthesis or action can lead to clinical syndromes characterized by water and electrolyte imbalance.
Similar content being viewed by others
References
Agre P, Brown D, Nielsen S. Aquaporin water channels: unanswered questions and unresolved controversies. Curr Opin Cell Biol. 1995;7:472–83.
Ball SG. Vasopressin and disorders of water balance: the physiology and pathophysiology of vasopressin. Ann Clin Biochem. 2007;44:417–31.
Bankir L, Bichet DG, Morgenthaler NG. Vasopressin: physiology, assessment and osmosensation. J Intern Med. 2017;282:284–97.
Barat C, Simpson L, Breslow E. Properties of human vasopressin precursor constructs: inefficient monomer folding in the absence of copeptin as a potential contributor to diabetes insipidus. Biochemistry. 2004;43:8191–203.
Baylis PH, Thompson CJ. Osmoregulation of vasopressin secretion and thirst in health and disease. Clin Endocrinol. 1988;29:549–76.
Bichet DG, Arthus MF, Barjon JN, Lonergan M, Kortas C. Human platelet fraction arginine-vasopressin. Potential physiological role. J Clin Invest. 1987;79:881–7.
Boone M, Deen PMT. Physiology and pathophysiology of the vasopressin-regulated renal water reabsorption. Pflügers Arch – Eur J Physiol. 2008;456:1005–24.
Carter DA, Murphy D. Rapid changes in poly (A) tail length of vasopressin and oxytocin mRNAs form a common early component of neurohypophyseal peptide gene activation following physiological stimulation. Neuroendocrinology. 1991;53:1–6.
Christ-Crain M, Morgenthaler NG, Fenske W. Copeptin as a biomarker and a diagnostic tool in the evaluation of patients with polyuria-polydipsia and hyponatremia. Best Pract Res Clin Endocrinol Metab. 2016;30:235–47.
Côté M, Salzman KL, Sorour M, Couldwell WT. Normal dimensions of the posterior pituitary bright spot on magnetic resonance imaging. J Neurosurg. 2014;120:357–62.
Cunningham JT, et al. Cardiovascular regulation of supraoptic vasopressin neurons. Prog Brain Res. 2002;139:257–73.
Davison JM, Gilmore EA, Dürr J, Robertson GL, Lindheimer MD. Altered osmotic thresholds for vasopressin secretion and thirst in human pregnancy. Am J Phys. 1984;246:F105–9.
Dobsa L, Edozien KC. Copeptin and its potential role in diagnosis and prognosis of various diseases. Biochem Med. 2013;23:172–90.
du Vigneaud V, Gish DT, Katsoyannis PG. A synthetic preparation possessing biological properties associated with arginine vasopressin. J Am Chem Soc. 1954;76:4751–2.
Esposito P, Piotti G, Bianzina S, Malul Y, Dal Canton A. The syndrome of inappropriate antidiuresis: pathophysiology, clinical management and new therapeutic options. Nephron Clin Pract. 2011;119:c62–73.
Fenske W, et al. A copeptin-based approach in the diagnosis of diabetes insipidus. N Engl J Med. 2018;379:428–39.
Garrahy A, Moran C, Thompson CJ. Diagnosis and management of central diabetes insipidus in adults. Clin Endocrinol. 2019;90:23–30.
Hernando F, Schoots O, Lolait SJ, Burbach JPH. Immunohistochemical localization of the vasopressin V1b receptor in the rat brain and pituitary gland: anatomical support for its involvement in the central effects of vasopressin. Endocrinology. 2001;142:1659–68.
Holmes CL, Landry DW, Granton JT. Science review: vasopressin and the cardiovascular system part 2 – clinical physiology. Crit Care. 2004;8:15–23.
Ichaliotis SD, Lambrinopoulos TC. Serum oxytocinase in twin pregnancy. Obstet Gynecol. 1965;25:270–2.
Johnson AK, Thunhorst RL. The neuroendocrinology of thirst and salt appetite: visceral sensory signals and mechanisms of central integration. Front Neuroendocrinol. 1997;18:292–353.
Lauson HD. Metabolism of antidiuretic hormones. Am J Med. 1967;42:713–44.
Lindheimer MD, Barron WM, Davison JM. Osmoregulation of thirst and vasopressin release in pregnancy. Am J Phys. 1989;257:F159–69.
Lolait SJ, et al. Cloning and characterization of a vasopressin V2 receptor and possible link to nephrogenic diabetes insipidus. Nature. 1992;357:336–9.
McKenna K, Thompson C. Osmoregulation in clinical disorders of thirst appreciation. Clin Endocrinol. 1998;49:139–52.
Morgenthaler NG, Struck J, Alonso C, Bergmann A. Assay for the measurement of copeptin, a stable peptide derived from the precursor of vasopressin. Clin Chem. 2006;52:112–9.
Moses AM, Miller M. Osmotic threshold for vasopressin release as determined by saline infusion and by dehydration. Neuroendocrinology. 1971;7:219–26.
Muhsin SA, Mount DB. Diagnosis and treatment of hypernatremia. Best Pract Res Clin Endocrinol Metab. 2016;30:189–203.
Nielsen S, Knepper MA. Vasopressin activates collecting duct urea transporters and water channels by distinct physical processes. Am J Phys. 1993;265:F204–13.
Nielsen S, Smith BL, Christensen EI, Knepper MA, Agre P. CHIP28 water channels are localized in constitutively water-permeable segments of the nephron. J Cell Biol. 1993;120:371–83.
Nielsen S, et al. Aquaporins in the kidney: from molecules to medicine. Physiol Rev. 2002;82:205–44.
Novak J, et al. Relaxin is essential for renal vasodilation during pregnancy in conscious rats. J Clin Invest. 2001;107:1469–75.
Parodi AJ. Protein glucosylation and its role in protein folding. Annu Rev Biochem. 2000;69:69–93.
Robertson GL. Disorders of the neurohypophysis. In: Jameson JL, editor. Harrison’s endocrinology. USA: McGraw-Hill; 2013. p. 50–61.
Robinson AG, Verbalis JG. Posterior pituitary. In: Hetherington P, Ryan J, editors. Williams textbook of endocrinology. USA: Elsevier; 2011. p. 291–316.
Schally A. Hormones of the neurohypophysis. In: Lock W, Schally AV, editors. The hypothalamus and pituitary in health and disease. Springfield: Thomas; 1972. p. 154–71.
Spruce BA, Baylis PH, Burd J, Watson MJ. Variation in osmoregulation of arginine vasopressin during the human menstrual cycle. Clin Endocrinol. 1985;22:37–42.
Stout NR, Kenny RA, Baylis PH. A review of water balance in ageing in health and disease. Gerontology. 1999;45:61–6.
Szinnai G, et al. Changes in plasma Copeptin, the C-terminal portion of arginine vasopressin during water deprivation and excess in healthy subjects. J Clin Endocrinol Metab. 2007;92:3973–8.
Weitzman RE, Kleeman CR. The clinical physiology of water metabolism. Part I: The physiologic regulation of arginine vasopressin secretion and thirst. West J Med. 1979;131:373–400.
Zingg HH. Vasopressin and oxytocin receptors. Bailliere Clin Endocrinol Metab. 1996;10:75–96.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Nature Switzerland AG
About this entry
Cite this entry
Mantovani, G., Mangone, A., Sala, E. (2023). Physiology of Vasopressin Secretion. In: Caprio, M., Fernandes-Rosa, F.L. (eds) Hydro Saline Metabolism. Endocrinology. Springer, Cham. https://doi.org/10.1007/978-3-030-44628-4_2-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-44628-4_2-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-44628-4
Online ISBN: 978-3-030-44628-4
eBook Packages: Springer Reference MedicineReference Module Medicine