Abstract
Epicardial adipose tissue (EAT) has been investigated in patients with both systolic and diastolic heart failure and suggested to play a pathogenic role, although results are not univocal. Experimental studies suggest a potential protective role of the brown fat features of EAT against the systolic heart failure. A function of EAT p53 and adiponectin has been also suggested in patients with heart failure. A neuromodulatory role of EAT in heart failure has been also considered. Clinically, diastolic heart failure is commonly associated with higher EAT. The role of EAT in the systolic heart failure is more controversial, as it could be influenced by concomitant confounders, such as coronary artery disease, diabetes and obesity. Some studies found that EAT volume was higher in patients with systolic heart failure, whereas other studies showed that either CT, MRI or ultrasound measured epicardial fat was actually lower in subjects with systolic heart failure. Epicardial fat may incur in fibrotic changes during chronic and advanced cardiac failure. Whether EAT plays a role in the long-term prognosis of heart failure requires future investigation.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Epicardial fat
- Epicardial adipose tissue
- Systolic heart failure
- Diastolic heart failure
- Ejection fraction
-
Epicardial adipose tissue plays a pathogenic role in heart failure. Thermogenic, paracrine, and neural factors seem to be involved.
-
Epicardial fat volume is higher in subjects with both systolic and diastolic heart failure.
-
Epicardial fat can be reduced in patients with severe heart failure due to fibrotic and apoptotic changes.
Introduction
Heart failure (HF) is a complex clinical syndrome that results from both structural and functional impairment of ventricular pump or filling. The causes leading to HF are multiple, and often co-occurring, such as coronary artery disease (CAD), atrial fibrillation (AF), preexisting cardiomyopathies, myocarditis, metabolic disorders, and acute organ failure. Heart failure can be classified into two types according to the left ventricular ejection fraction (reduced or preserved): systolic heart failure with reduced ejection fraction (HFrEF), and diastolic heart failure with preserved ejection fraction (HFpEF) [1].
Epicardial adipose tissue (EAT) has been investigated in patients with heart failure and suggested to play a pathogenic role. Given its peculiar location and intense metabolic activity, it is plausible to see the role of EAT in the development and progression of EAT. Also, abnormal or excessive EAT has shown to be correlated with both systolic and diastolic functions. Hence, it would be logical to attribute some action of EAT in affecting either the left ventricular pump or the filling phase. However, the results are quite scarce and controversial, also due to the occurrence of other conditions that can affect EAT. Hence, the role of EAT in independently causing or contributing to the development of HF is still uncertain.
Thermogenic Epicardial Fat and Heart Failure
EAT displays genetic and functional features similar to those of the brown fat [2]. Human EAT has the potential to serve as a thermogenic source for the myocardium and therefore protect it against hypothermia. A recent study found that patients with HFrEF expressed significantly lower thermogenic genes in EAT than those with HFpEF [3]. In fact, uncoupling protein 1 (UCP1), peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1α), and PR-domain-missing 16 (PRDM16) expression were significantly lower in EAT of patients with HRrEF than those with HFpEF. The correlation of EAT brown fat genes with HF was not independent. In fact, age, male gender, and different cardiovascular diseases were also associated with the levels of thermogenic genes expression. This study suggests that an adequate expression of thermogenic genes could serve a possible protective factor against congestive heart failure. Consequently, a loss of functional EAT brown-like features would contribute to the development of HFrEF. This hypothesis seems to be confirmed by another study in both mice and humans with obesity and heart failure [4]. The authors suggested a role of the epicardial fat heme oxygenase-1 (HO1) PGC-1α in modulating the left ventricle function. HO-1 PGC-1α and PRDM16 were inversely correlated with left ventricular ejection fraction. In summary, these studies suggest a potential protective role of the brown fat features of EAT against systolic heart failure. EAT thermogenic genes could serve as a therapeutic target in patients with HFrEF. Pharmacological manipulation would be certainly challenging as EAT brown fat-like properties tend to significantly decrease with aging [5]. However, these findings should be confirmed in larger and multicenter research trials.
Epicardial Fat Adipokines and Heart Failure
EAT is an active paracrine and endocrine organ. EAT secretome is large and includes both pro- and anti-inflammatory adipokines. Among these, EAT adiponectin has shown to provide potential cardioprotective effects. EAT adiponectin expression is downregulated in patients with coronary artery disease [6]. In a study with patients undergoing cardiac surgery, p53 and adiponectin mRNA expression was measured in frozen fat biopsies or explants culture from both EAT and subcutaneous fat (SAT). p53 expression in adipose tissue is involved in the development of insulin resistance and inflammation [7]. Higher p53 expression was linked to increased production of pro-inflammatory cytokines. p53 mRNA expression levels were higher in EAT of HF patients as compared to SAT. p53 was inversely correlated with adiponectin and regulated by sympathetic activation pathway in patients with HF. A neuromodulatory role of EAT in heart failure will be discussed in details in chap. 12.
Epicardial Fat in Diastolic and Systolic Heart Failure
The relationship between EAT and HF was evaluated by a number of studies, although few of these were focused to specifically address it. In these studies, populations were quite heterogeneous with patients with or without coronary artery disease (CAD) and other confounding factors such as diabetes and hypertension. The role of EAT in the development or progression of HF can be better interpreted if results are adjusted by these factors, and diastolic and systolic functions are factored in the relation. A recent meta-analysis included 26 studies accounting for more than 4000 patients [8]. This analysis was conducted on the basis of diagnosis of left ventricular diastolic and systolic dysfunction and their correlation with EAT. Left ventricular diastolic dysfunction (LVDD ) was defined echocardiographically by a ratio of early mitral valve flow velocity (E) to early diastolic lengthening velocity (e′; E/e′) during tissue Doppler imaging of ≥10; left ventricular systolic dysfunction (LVSD ) was defined with ejection fraction (LVEF) ≤50%. Patients with preserved systolic function (HFpEF ) are those with LVDD, whereas those with reduced systolic function (HFrEF) are presenting with LVSD. As previously discussed, EAT can be measured either with standard echocardiography or CT or CMR, as thickness or volume, respectively. The relationship between ultrasound measured EAT thickness and LVDD was reported in eight studies that included 775 cardiac patients with LVDD and 695 controls without LVDD [9,10,11,12,13,14,15]. Overall, EAT thickness was significantly higher in patients with LVDD as compared to those without it. The relationship between EAT volume and LVDD was investigated in six studies that included 433 cardiac patients with LVDD and 272 controls without LVDD [16,17,18,19,20]. Overall, EAT volume in cardiac patients with LVDD was increased compared to those without LVDD. One study measured EAT volume with cardiac magnetic resonance in patients with HFpEF defined by an LVEF >40% [21]. EAT volume was significantly higher in HFpEF patients compared to controls (107 mL/m2 vs. 77 mL/m2, P < 0.0001), despite similar body mass index. HF patients with atrial fibrillation and/or type 2 diabetes mellitus had higher EAT than HF patients without these comorbidities. We can conclude that EAT is higher in patients with LVDD as compared to patients without LVDD, irrespective of whether is measured as volume or thickness. Hence, diastolic heart failure is more commonly associated with increased EAT.
The role of EAT in systolic heart failure (HFrEF ) is less consistent, as findings are actually quite conflicting. The association between EAT and LV function was evaluated using either CMR or echocardiography. In some studies, EAT was found higher in subjects with LVSD [22,23,24,25]. One study showed that patients with HFrEF had significantly higher indexed EAT volume as measured with cardiac magnetic resonance imaging when compared with patients with HFpEF or the control group [22]. Another study reported an association between EAT and global longitudinal strain, a subclinical measure of myocardial function [23]. An independent correlation between echocardiographic EAT thickness and LVEF was also observed [24]. Epicardial fat thickness is associated with the severity of HF in patients with nonischemic dilated cardiomyopathy [25]. In fact, patients with HF had significantly lower epicardial fat thickness than those in the control group. Some of these results may be confounded by the co-occurrence of diabetes or CAD or obesity. Mechanical restriction from excessive EAT during the diastole may affect the ventricular filling and, consequently, reduce cardiac output in obese subjects [25].
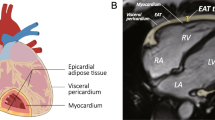
However, some other studies found opposite results. Doesch et al. reported a reduced EAT in patients with congestive, HFrEF [26,27,28]. When EAT was adjusted by LV end-diastolic mass was significantly reduced in patients with severe HF (EF < 35%) compared to healthy controls (Fig. 11.1). In the analysis performed by Doesch and colleagues, the reduction of EAT was irrespective of the underlying cause of the cardiomyopathy [26]. In fact, there were no differences in EAT between patients with or without CAD. Doesch attributes the reverse correlation between LVEF and indexed EAT to the left ventricular remodeling occurring in heart failure. Lower EAT in patients with poor systolic function was also confirmed in another study [24]. EAT volume, as measured with cardiac magnetic resonance, was also found to be significantly lower in obese patients with HFpEF, and no correlation between EAT and EF was observed [29].
EAT in healthy controls and patients with HFrEF . Volumetric measurement of EAT outlining the contours of EAT in end-diastolic images of short axis covering the left and right ventricles in a healthy control with normal EAT mass (Panel A) and in an HFrEF patient with reduced EAT mass (Panel B). CHF chronic heart failure, EAT Epicardial adipose tissue, and LV-EDM left ventricular end-diastolic mass. (From Doesch et al. [26], with permission)
EAT thickness, as measured according to the method first described by Iacobellis et al. [33, 30], was also found to be lower in subjects with HFrEF (EF < 50%) as compared to those without HF, independent of atrial fibrillation and HF [31]. Some mechanisms can be evoked to explain these findings. Epicardial fat reduction in HF subjects may reflect the overall fat mass reduction, commonly observed in these patients. It is also possible to hypothesize that epicardial fat pad may incur in fibrotic changes during chronic cardiac failure [32]. However, the exact interaction of EAT and HF is still unclear. Whether EAT plays a role in the long-term prognosis of HF requires future investigation.
References
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Colvin MM, et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Am Coll Cardiol. 2017;70:776–803.
Sacks HS, Fain JN, Holman B, Cheema P, Chary A, Parks F. Uncoupling protein-1 and related mRNAs in human epicardial and other adipose tissues: epicardial fat functioning as brown fat. J Clin Endocrinol Metab. 2009;94:3611–5.
Pérez-Belmonte LM, Moreno-Santos I, Gómez-Doblas JJ, et al. Expression of epicardial adipose tissue thermogenic genes in patients with reduced and preserved ejection fraction heart failure. Int J Med Sci. 2017;14:891–5.
Singh SP, McClung JA, Thompson E, Glick Y, Greenberg M, Acosta-Baez G, et al. Cardioprotective effect of heme oxygenase-1-PGC-1? Signaling in epicardial fat attenuates cardiovascular risk in humans as in obese mice. Obesity. 2019;27:1634–43.
Sacks HS, Fain JN. Human epicardial adipose tissue: a review. Am Heart J. 2007;153:907–17.
Iacobellis G, Pistilli D, Gucciardo M, Leonetti F, Miraldi F, Brancaccio G, et al. Adiponectin expression in human epicardial adipose tissue in vivo is lower in patients with CAD. Cytokine. 2005;29:251–5.
Krstic J, Reinisch I, Schupp M, Schulz TJ, Prokesch A. p53 functions in adipose tissue metabolism and homeostasis. Int J Mol Sci. 2018;19:2622.
Nerlekar N, Muthalaly RG, Wong N, Thakur U, Wong DTL, Brown AJ, Marwick TH. Association of volumetric epicardial adipose tissue quantification and cardiac structure and function. J Am Heart Assoc. 2018;7:e009975.
Lin HH, Lee JK, Yang CY, Lien YC, Huang JW, Wu CK. Accumulation of epicardial fat rather than visceral fat is an independent risk factor for left ventricular diastolic dysfunction in patients undergoing peritoneal dialysis. Cardiovasc Diabetol. 2013;12:127.
Topuz M, Dogan A. The effect of epicardial adipose tissue thickness on left ventricular diastolic functions in patients with normal coronary arteries. Kardiol Pol. 2017;75:196–203.
Turak O, Özcan F, Canpolat U, Işleyen A, Cebeci M, Öksüz F, et al. Increased echocardiographic epicardial fat thickness and high-sensitivity CRP level indicate diastolic dysfunction in patients with newly diagnosed essential hypertension. Blood Press Monit. 2013;18:259–64.
Lei P, Shi S, Li P, Liu Y, Cheng X, Li Y. Epicardial adipose thickness of hypertension patients correlated with left ventricular diastolic dysfunction (abstract GW25-e4319). J Am Coll Cardiol. 2014;64(16 Suppl C):C172.
Kaushik M, Alla VM, Saurav A, Smer A, Madan R, Khan S, et al. Epicardial adipose tissue thickness as assessed by echocardiography & left ventricular diastolic dysfunction (abstract 433). Cardiology. 2013;125(Suppl 2):447.
Manka M, Fenk S, Strack C, Kempinger S, Riegger G, Schmitz G, et al. Epicardial fat is related to left ventricular diastolic dysfunction in healthy obese and metabolic syndrome obese (abstract). Circulation. 2014;130:A19045.
Jung YH, Yoon SB, Kim WY, Park A. Epicardial fat as a contributing factor to diastolic dysfunction. Korean J Fam Pract. 2016;6:26–31.
Fontes-Carvalho R, Fontes-Oliveira M, Sampaio F, Mancio J, Bettencourt N, Teixeira M, et al. Influence of epicardial and visceral fat on left ventricular diastolic and systolic functions in patients after myocardial infarction. Am J Cardiol. 2014;114:1663–9.
Konishi M, Sugiyama S, Sugamura K, Nozaki T, Matsubara J, Akiyama E, et al. Accumulation of pericardial fat correlates with left ventricular diastolic dysfunction in patients with normal ejection fraction. J Cardiol. 2012;59:344–51.
Vural M, Talu A, Sahin D, Elalmis OU, Durmaz HA, Uyanik S, Dolek BA. Evaluation of the relationship between epicardial fat volume and left ventricular diastolic dysfunction. Jpn J Radiol. 2014;32:331–9.
Wu L, Chen W, Chen YJ, Wang KH, Qu KX. Correlation between volume of epicardial adipose tissue measured with MSCT and left ventricular diastolic function in coronary artery disease patients. Chin J Interv Imaging Ther. 2014;11:286–9.
Fuzhuang Z, Hong T, Yongmei W, Wenchuan W, Xiuying G, Xianxian Y, et al. Association of epicardial adipose tissue volume with coronary artery atherosclerosis and left ventricular diastolic function. J Cardiovasc Pulmon Dis. 2012;31:58–63.
van Woerden G, Gorter TM, Westenbrink BD, Willems TP, van Veldhuisen DJ, Rienstra M. Epicardial fat in heart failure patients with mid-range and preserved ejection fraction. Eur J Heart Fail. 2018;20:1559–66.
Wu CK, Tsai HY, Su MM, Wu YF, Hwang JJ, Lin JL, et al. Evolutional change in epicardial fat and its correlation with myocardial diffuse fibrosis in heart failure patients. J Clin Lipidol. 2017;11:1421–31.
Ng AC, Goo SY, Roche N, van der Geest RJ, Wang WY. Epicardial adipose tissue volume and left ventricular myocardial function using 3-dimensional speckle tracking echocardiography. Can J Cardiol. 2016;32:1485–92.
Khawaja T, Greer C, Chokshi A, Chavarria N, Thadani S, Jones M, et al. Epicardial fat volume in patients with left ventricular systolic dysfunction. Am J Cardiol. 2011;108:397–401.
Tabakci MM, Durmuş Hİ, Avci A, Toprak C, Demir S, Arslantaş U, et al. Relation of epicardial fat thickness to the severity of heart failure in patients with nonischemic dilated cardiomyopathy. Echocardiography. 2015;32:740–8.
Doesch C, Haghi D, Fluchter S, Suselbeck T, Schoenberg SO, Michaely H, et al. Epicardial adipose tissue in patients with heart failure. J Cardiovasc Magn Reson. 2010;12:40.
Doesch C, Haghi D, Suselbeck T, Schoenberg SO, Borggrefe M, Papavassiliu T. Impact of functional, morphological and clinical parameters on epicardial adipose tissue in patients with coronary artery disease. Circ J. 2012;76:2426–34.
Doesch C, Streitner F, Bellm S, Suselbeck T, Haghi D, Heggemann F, et al. Epicardial adipose tissue assessed by cardiac magnetic resonance imaging in patients with heart failure due to dilated cardiomyopathy. Obesity (Silver Spring). 2013;21:E253–61.
Haykowsky MJ, Nicklas BJ, Brubaker PH, Hundley WG, Brinkley TE, Upadhya B, et al. Regional adipose distribution and its relationship to exercise intolerance in older obese patients who have heart failure with preserved ejection fraction. JACC Heart Fail. 2018;6:640–9.
Iacobellis G, Willens HJ. Echocardiographic epicardial fat: a review of research and clinical applications. J Am Soc Echocardiogr. 2009;22:1311–9.
Iacobellis G, Assael F, Ribaudo MC, Zappaterreno A, Alessi G, Di Mario U, et al. Epicardial fat from echocardiography: a new method for visceral adipose tissue prediction. Obes Res. 2003;11:304–10.
Iacobellis G, Zaki MC, Garcia D, Willens HJ. Epicardial fat in atrial fibrillation and heart failure. Horm Metab Res. 2014;46:587–90.
Ruberg FL, Chen Z, Hua N, Bigornia S, Guo Z, Hallock K, et al. The relationship of ectopic lipid accumulation to cardiac and vascular function in obesity and metabolic syndrome. Obesity (Silver Spring). 2010;18:1116–21.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Iacobellis, G. (2020). Heart Failure and Epicardial Adipose Tissue. In: Iacobellis, G. (eds) Epicardial Adipose Tissue. Contemporary Cardiology. Humana, Cham. https://doi.org/10.1007/978-3-030-40570-0_11
Download citation
DOI: https://doi.org/10.1007/978-3-030-40570-0_11
Published:
Publisher Name: Humana, Cham
Print ISBN: 978-3-030-40569-4
Online ISBN: 978-3-030-40570-0
eBook Packages: MedicineMedicine (R0)