Abstract
The bird dawn chorus has fascinated humans since ancient times, but still today numerous questions remain unclear. This chapter will explore this puzzling phenomenon, a communal display that likely involves the highest level of sound complexity found among animal signals. Covering from the first descriptive studies to recent multidisciplinary approaches, we review the physiological, behavioural and environmental factors affecting dawn chorus. In addition, we provide a critical assessment of the supporting evidence for the functional hypotheses proposed so far to disentangle its proximal and ultimate causes. We find that, despite the latest empirical and theoretical studies, there is still a good degree of confusion, and that four out of the nine hypotheses proposed so far in the literature have not been empirically tested. We show that most of these hypotheses are not incompatible with each other, and that their explanatory value changes depending on the species and the season. We argue that, at any rate, a single explanation may not be a reasonable expectation. The best-supported hypotheses for early singing provide three complementary lines of explanation: (1) singing at dawn has a relatively low energetic cost, most likely because it does not interfere with feeding; (2) is optimal to manipulate female mating or settle territory boundaries; and (3) may promote a handicap mechanism that prevents dishonest signalling. Thus, it follows that a combination of hypotheses based on both an optimality standpoint and costliness assumptions is needed to understand the phenomenon. We provide a series of specific suggestions for further research to refine our knowledge of this intriguing aspect of animal behaviour.
Thou hearest the Nightingale begin the Song of Spring:
The Lark, sitting upon his earthy bed, just as the morn
Appears, listens silent, then, springing from the waving corn-field, loud
He leads the Choir of Day—trill! trill! trill! trill!
William Blake (Milton: a Poem, 1810)
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
3.1 Introduction
The bird dawn chorus has fascinated humans and aroused their interest since ancient times. The use of roosters as clocks is likely as old as chicken domestication itself, which recent studies trace back to 3000 BT (Peters et al. 2016). The classical Greek and Latin literature, as well as the Bible or later the writings of the Romantics, is rife with references to bird song being used as an announcement of sunrise (Armstrong 1963; Doggett 1974). Romeo famously leaves Juliet’s chamber in a hurry when he realises that it was a lark, and not a nightingale, the bird that woke him up (Shakespeare 1597). Today the bird dawn chorus keeps intriguing us, and numerous questions about this puzzling behavioural phenomenon are still unclear.
It is impossible to envisage a review on this question without building on previous efforts, such as those of Kacelnik and Krebs (1983), Mace (1987b) and more recently Staicer et al. (1996), which have been seminal in shaping modern research on the dawn chorus. Staicer et al. (1996) summarised 12 different hypotheses on the dawn chorus, classified in three groups (intrinsic, social and environmental), and examined them in relation to empirical support. Now, more than 20 years since this key review, we wish devote this chapter to revisiting the status of the different hypotheses in the light of the studies published in the last decades. As the approach and terminology in Staicer et al. (1996) has been widely applied, here we proceed using a similar classification of hypotheses (Table 3.1).
Starting by briefly describing the general features of the bird dawn chorus, this chapter outlines a series of issues that appear when we try to disentangle its proximal and ultimate causes. On this overall basis, we move on to discuss, one by one, the main factors affecting this phenomenon and the explanations that have been proposed to explain why birds sing at this particular time. To do so, we use the following structure: (1) we report the patterns, variations and known facts about the bird dawn chorus in relation to physiological, behavioural or environmental factors; (2) we present functional hypotheses that are derived from these observations; and (3) we critically assess the supporting evidence for such hypotheses and its primary predictions, pointing out remaining questions and proposing further studies to test them.
An important omission of our review is female song. A series of studies have emphasised that female song is more widespread than traditionally assumed (Langmore 1998), actually being the evolutionarily ancestral state in birds (Odom et al. 2014). However, research on female song is still underrepresented in the literature (Riebel et al. 2005) and we have found almost no reference to female song in the dawn chorus literature. We believe that very interesting insights may be gained by comparing male and female patterns of singing with respect to dawn in those species that show female song. But in the remainder of the chapter we will deal with male song only.
3.2 The Bird Dawn Chorus
The dawn chorus is a daily period of high singing activity shown by most passerine and some non-passerine species in the breeding season. This burst of singing typically starts for the earliest species around nautical twilight (sun at 12° under the horizon), and is followed in a rather orderly fashion by other species until sometime around sunrise (Thomas et al. 2002). Although the time of the first song is a species-specific trait (Berg et al. 2006), during this period many species end up overlapping their songs, leading to a more or less cacophonic chorus (Fig. 3.1) that characterises the soundscapes of different avian communities (Farina et al. 2015). As a result, probably no other daily period involves larger levels of sound complexity among animal group displays (Burt and Vehrencamp 2005). Depending on latitude, season and species composition, choruses can be more or less intense and prolonged, but they occur in most breeding bird habitats (Staicer et al. 1996).
Spectrograms of a temperate and a tropical avian community: (a) Forêt domaniale de la Commanderie (Île-de-France, France: 48° 17′ N, 2° 40′ E, 85 m.a.s.l; April 2014) and (b) Ducke Reserve (Manaus, Brazil: 2° 57′ S, 59° 55′ W, 92 m.a.s.l; October 2009). The following avian species can be detected: (a) Turdus merula, Turdus philomelos, Erithacus rubecula, Periparus ater and Regulus regulus; (b) Attila spadiceus, Momotus momota, Ramphotrigon ruficauda, Dendrocolaptes certhia and Dendrexetastes rufigula (in addition to at least one anuran: Dendropsophus reticulatus). From unpublished recordings by Diego Llusia (France) and Thiago Bicudo (Manaus). Figure created in Python v.3.7.4. Listen to sound clips at: http://sono.behavecol.es/en/landscapes
The first studies on the dawn chorus in the twentieth century were rather descriptive, and dealt primarily with the order in which species sang (the bird-clock), and with the effects of weather and moonlight on singing time (Allard 1930; Allen 1913; Wright 1912). Further studies started to carefully measure the specific light level that triggered the first song of each species, reaching more conclusive evidence for a species-specific light singing level (Leopold and Eynon 1961; Scheer 1952). However, we have to wait until the 1980s to find the first considerations of the dawn chorus from the perspective of behavioural ecology. In that sense, the pioneering study by Alex Kacelnik and John Krebs (1983) is noteworthy, combining physiological, ecological and behavioural perspectives. Since then, dawn song has been examined from a multitude of angles, including physiology (Shimmura and Yoshimura 2013), sexual selection (Dolan et al. 2007), reproduction (Kempenaers et al. 2010), energy allocation (Hutchinson et al. 1993), noise pollution (Gil et al. 2015) and soundscapes (Farina et al. 2015). Thus, a comprehensive review of this topic needs to cover a very wide range of subjects.
From the point of view of its features, the dawn chorus has also been studied using a relatively broad range of measures. Undoubtedly, dawn chorus timing, and particularly the time at which each species or individual starts to sing within the chorus (i.e. the time of the first song), has been one of the most frequently employed parameters (Allard 1930; Gil et al. 2015; Leopold and Eynon 1961; Poesel et al. 2006; Scheer 1952). However, other researchers have focused on additional aspects, such as dawn-related variations in song repertoire (Allard 1930; Allen 1913; Demko et al. 2013; Kroodsma et al. 1989; Nelson and Croner 1991; Staicer et al. 1996), song rate (Liu and Kroodsma 2007; Pärt 1991) or song characteristics (Halfwerk et al. 2011; Lambrechts and Dhondt 1986).
In addition to the dawn chorus, it is important to note that a second peak of singing at the end of the day, the dusk chorus, is also common (Cuthill and Macdonald 1990; Møller 1988; Morse 1989), although less intense and dramatic (for a noteworthy exception see McCabe 1951). This double-peaked circadian pattern is typical of many activity patterns across diverse vertebrate taxa (Rusak 2013). Although we only consider birds in this review, circadian cycles of signalling have also been described in other animal groups, including crickets, crabs, fish, amphibians and mammals (Au et al. 2000; D’Spain and Batchelor 2006; Green 1990; Greenfield 2015; Ord 2008). These similarities suggest two alternatives: (1) that there is widespread selection for acoustic communication to exploit optimal times; (2) that selection focus on synchronization, allowing comparisons between signallers by ritualizing the displays at the same time (West-Eberhard 1979; Zahavi 1980).
It is important to emphasise that the dawn chorus is a phenomenon that comprises two levels: individuals within species, and species within communities. For most functional studies, the dawn chorus implies the song of different individuals of the same species at dawn. In that sense, the multispecies chorus would be the additive consequence of selective pressures acting within species. Although some studies have analysed interactions between species that may reduce song-overlapping time (Planqué and Slabbekoorn 2008), no study that we know of proposes a multispecies function of dawn chorusing in general. Nevertheless, some studies tentatively suggest that some species within the chorus may converge in timing as a way of competing among them or of extracting public information (Phelps et al. 2006; Tobias et al. 2014).
Differences among species in the time of the first song at dawn are strong and well known (Fig. 3.2), and were the main object of early descriptive studies (Allard 1930; Allen 1913). Later on, Scheer (1952) transformed specific times into light intensity levels for a few species, putting forward the concept of a species-specific light intensity for first morning song (Singhelligkeit). Further studies recognised that these values varied with seasonal and meteorological factors, although repeatability in the order of species singing was still high (Leopold and Eynon 1961).
Mean time of the first song for a temperate (blue) and a tropical (orange) avian community (same areas as in Fig. 3.1). Grey scales represents dawn period, from nautical twilight to sunrise, lasting for 70 min in Île-de-France and 45 min in Manaus. Unpublished data by Diego Llusia, Thierry Aubin and Jérôme Sueur (France) and Diego Gil and Marconi Campos-Cerqueira (Brazil). Figure created in R v.3.5.2
So, what is the cause of these interspecific differences? Armstrong was the first author to recognise that eye size could explain dawn song timing, claiming that one could get a good idea of what time a bird starts to sing by looking at its eyes (Armstrong 1963). The first test of this hypothesis was conducted with European species, showing that species with larger eyes start to sing earlier than species with smaller eyes (Thomas et al. 2002). They also found that body size was an additional predictor, with larger species starting to sing later than smaller species, although this effect was less important than that of eye size. Further studies in other avian communities have confirmed this relationship between eye size and dawn song time (Berg et al. 2006; Chen et al. 2015; Gil et al. 2015). A study conducted in an Ecuadorian rainforest showed that, in addition to eye size and body size, dawn song timing was also explained by feeding stratum: birds in the well-lit canopy sang earlier than birds foraging in the dark understory (Berg et al. 2006). An important consideration in these studies is the need to control for phylogeny, since dawn timing is a phylogenetically conserved trait, and a bias may appear because of the particular sampling of species of a given study (for instance suboscines sing earlier than oscines: Berg et al. 2006).
Larger eye size implies larger pupils and corneas and an increased number of photoreceptors, leading to improved visual acuity, a larger visual field width, and the possibility of perceiving objects at lower light intensities (Martin 1993; Thomas et al. 2002). Larger eye size has evolved in connection to nocturnal habits, capture of mobile prey, use of dark habitats and increased flight distances (Garamszegi et al. 2002; Martínez-Ortega et al. 2014; Møller and Erritzøe 2010).
3.3 Song Function Versus Dawn Chorus Function: Why at This Time Precisely?
Before dealing with the question of why singing early, we need to answer the question of why singing at all. Bird song is mostly produced by males in temperate regions, and is generally said to have two main general functions: female attraction and territory or resource defence (Catchpole and Slater 2008; Kroodsma and Byers 1991). Innumerable correlative and observational studies suggest a role for these functions (Catchpole 1982), and there are also a series of clever manipulations that provide experimental support. Thus, for female attraction, there is strong evidence in several species that nest boxes equipped with song playback attract more females than silent boxes (Eriksson and Wallin 1986; Mountjoy and Lemon 1991). With regard to territory defence, some studies show that playback-defended territories take longer to be occupied than silent areas (Falls 1988; Krebs et al. 1978). Finally, a classical study in which males were temporarily muted showed that song is necessary to keep intruders away and to attract females in the seaside sparrow (Ammospiza maritima) (McDonald 1989). In addition, there exists some less commonly studied functions of song, such as deterring predators (Cresswell 1994) and self-stimulation in the case of laying females (Cheng 1992). Moreover, song produced by females also deserves more attention and much work is needed to clarify its particular function (Riebel et al. 2005).
Although the role of song in female attraction and resource defence can be generalised to many songbirds, the two functions do not have the same importance in different species. In addition, there are large species-specific differences in the relative importance of the two functions at different stages of the breeding season (Catchpole 1982). A multitude of factors could influence this balance: differences in brooding rate (multiple vs. single-brooded species), migratory behaviour (sedentary vs. migratory species), risk of extra-pair paternity, intrusion rate, etc. It is indeed very difficult to know to which audience or audiences precisely a bird is singing at a given time. The same signal can be directed to a variety of intended receivers with different results: to attract potential mates; to its already acquired mate, who may modify her investment in response to the song (Gil et al. 2004); to its neighbours, who may use information on his competitive abilities to update the territorial status quo (Sexton et al. 2007); or to floaters, who may use song information to decide future settlement patterns (Amrhein and Erne 2006; Amrhein et al. 2004a). Deciding which is the most relevant receiver at a given time is a difficulty that permeates most research on the dawn chorus, since adaptive hypotheses about this trait are, by definition, intimately related to its function.
We must stress here that many studies attempt to explain the function of the dawn chorus without addressing the key question: why singing at this precise time? In other words, it is not enough to know that the dawn song is directed to the female or to male neighbours; we must also explain why more singing occurs at this time than later or earlier in the day. In this chapter, we will consider in turn all hypotheses that have been proposed to explain dawn song in birds, even some for which the evidence is still scant or negative (see Table 3.1 for a list of hypotheses).
3.4 Factors Affecting the Dawn Chorus and Related Functional Hypotheses
The dawn chorus has been the subject of a large number of disciplines, from physiology of circadian rhythms to ecology of predation, energetics, visual perception and extra-pair paternity. We have classified the information gathered by these different areas in the following sections, with the aim of providing a wide view of the levels of variation shown by this phenomenon. To allow the reader to follow this information more easily, we have associated each factor with the hypotheses that most closely derive from that subject area.
3.4.1 Hormones and Other Internal Drivers: Proximal Causation of the Dawn Chorus
Most of the temporal organization of the behaviour and physiology of animals is affected by circadian rhythms, i.e. daily cycles of endogenous origin (Gwinner and Brandstatter 2001). These rhythms are selectively responsive to specific environmental factors, mainly to light and ambient temperature. In the case of birds, a complex system that includes the retina, the pineal gland and the hypothalamus is responsible for circadian organization (Cassone and Westneat 2012; Gwinner and Brandstatter 2001). The key messenger within this system is melatonin, a hormone that controls circadian pacemaking of behaviour and physiology. Secreted by the pineal gland, melatonin has a primary role in regulating sleep and daily rhythms (Bentley 2001; Haldar and Singh 2001; Rodríguez et al. 2001). Studies in several species have established that daily cycles of singing are regulated by a central clock system, and that neither light nor sound stimuli are always necessary to trigger the response (Shimmura and Yoshimura 2013; Wang et al. 2012). In zebra finches (Taeniopygia guttata), pinealectomy stops circadian rhythms of singing in constant environmental conditions, but these can be restored by melatonin implants, showing a role of this hormone in regulating circadian singing cycles (Wang et al. 2012).
Data show that circadian period length is highly heritable in great tits (Parus major), suggesting that it might be an object of selection in response to ecological pressures (Helm and Visser 2010). External factors, such as light, sound and singing by conspecifics (see sections below) can interact with melatonin secretion and strongly modify singing patterns. For instance, in the case of roosters (Gallus gallus domesticus), both light levels and listening to other roosters increase the probability and rate of crowing (Shimmura and Yoshimura 2013). It is not known whether modifications in timing are brought about by a change in the responsiveness of the organism to internal hormone secretions, or by a modification of hormone levels (for a similar consideration in the case of testosterone see Hau 2007). Studies showing that urban life leads to reduction of melatonin levels in Eurasian blackbirds (Turdus merula) suggest that the second mechanism is a likely possibility (Dominoni et al. 2013a, b).
In addition to the effect of melatonin, testosterone is also seasonally related to the timing and performance of singing behaviour. For instance, studies in several species show a seasonal match between the dawn chorus and a yearly peak of testosterone levels (Foerster et al. 2002; Van Duyse et al. 2003). Besides, male great tits whose androgen receptors had been experimentally blocked were less likely to show a dawn song than control birds (Van Duyse et al. 2005), and a correlative study with Eurasian blue tits (Cyanistes caeruleus) found a positive relationship between male testosterone levels and dawn singing output before breeding (Foerster et al. 2002). However, these correlations between hormone levels and behaviour do not demonstrate that testosterone is a direct modulator of singing. Moreover, other experiments have failed to observe an effect of testosterone implants on dawn singing characteristics (Kunc et al. 2006; Shimmura and Yoshimura 2013).
Hypothesis 1: Circadian Hormone Cycles
The possibility that dawn singing is an epiphenomenon of circadian fluctuations in hormones was proposed by Staicer et al. (1996). Since androgens play an important role in both the development and the expression of singing behaviour in birds and other taxa (Alward et al. 2018), testosterone was the first hormone that was proposed as candidate for this role. Although there is ample evidence showing that testosterone and singing behaviour are connected in songbirds (see above), the issue here is not whether testosterone promotes singing, but rather whether circadian variation in testosterone levels is responsible for the dawn chorus.
There is not much information on how androgen levels vary through the day, but the existing data do not support this hypothesis at all. Some studies have shown that levels of testosterone and luteinizing hormone (which stimulates testosterone production) are higher at night and decrease in the early morning (Laucht et al. 2011; Needham et al. 2017; Ottinger and Follett 1987; Rattner et al. 1982). This evidence would imply that the daily peak of testosterone does not coincide with the dawn chorus. Furthermore, a study in Japanese quail (Coturnix japonica) showed that normal daily cycles of crowing were preserved regardless of whether males had been castrated or not (Wada 1986). In addition, the dawn chorus of the silver-beaked tanager (Ramphocelus carbo) in Amazonia shows a strong seasonality that is not connected to an increase in testosterone levels (Quispe et al. 2016).
A more promising candidate for circadian regulation of behaviour is melatonin (Hau et al. 2017). As mentioned above, there is experimental evidence for a role of this hormone in affecting circadian and seasonal singing patterns (Cassone et al. 2008; Wang et al. 2012). However, documenting circadian rhythms of melatonin is extremely difficult given the rapid fluctuations in concentration that occur during the day and night, as well as the individual variation in cycling patterns (Seltmann et al. 2016). A common melatonin curve typically involves basal levels during the day with a sudden peak soon before sleep, a maintained plateau for some hours and a dramatic decrease some hours before dawn (Seltmann et al. 2016). Thus, melatonin can explain why birds are awake before dawn, but this does not tell us much about why they are singing at this time. Although it is possible that species-specific differences in dawn timing are connected to sleep cycles and melatonin secretion patterns, little data are available on this respect. In addition, research shows that links between melatonin and behaviour are extraordinarily plastic (Daan and Aschoff 1975). Thus, it would seem naive to assume that the dawn chorus is a mere epiphenomenon of variation in this hormone. On the contrary, research suggests that selection for early activation may drive changes in melatonin secretion (Dominoni et al. 2013a).
An even more basal explanation of dawn and dusk choruses in birds could be based on the fact that a whole suite of physiological and activity patterns show a bimodal peak (early and late in the day), even in complete isolation from external stimuli (Aschoff 1966; Refinetti 2016). Basic physiological processes related to thermoregulation, metabolism or food intake are often described by a bimodal pattern (Aschoff and Meyer-Lohmann 1955; Brandt and Cresswell 2009). These circadian rhythms are similar to those found in bird singing and other signalling routines (Cuthill and Macdonald 1990; Ord 2008). In that sense, it could be argued that the most parsimonious explanation for the dawn chorus should be sought with reference to basic organic functions, given the whole suite of physiological and behavioural components that are affected in the same manner (Aschoff 1966). However, it seems unlikely that the diversity of patterns observed in dawn chorusing could be simply explained as non-functional consequences of circadian cycles (Hau 2007). The patterns that we describe in this chapter show large individual and species differences, suggesting that ecological factors largely modulate this behaviour.
Hypothesis 2: Self-stimulation of Androgen Production
This hypothesis was proposed in a previous review (Staicer et al. 1996) and, to our knowledge, it has not yet been tested. According to this hypothesis, by singing at dawn males would auto-stimulate their own gonadal hormone production, and this would be advantageous later on in their daily social interactions. This suggestion is based on studies that show that vocalizing influences ovulation patterns in female African collared doves (Streptopelia roseogrisea) (Cheng 1992), or the bird’s own androgen levels in budgerigars (Melopsittacus undulatus) (Brockway 1967). However, a role for this function in dawn singing remains highly speculative.
3.4.2 Breeding Cycle
A good way to understand the function of song is to examine the link between breeding stages and singing behaviour (Catchpole 1973). Following this idea, analyses on how dawn singing varies with breeding stage have led to hypotheses about its possible functions (e.g. mate attraction, mate guarding, territory defence, etc.).
Surprisingly there is no comparative analysis examining how song timing with respect to sunrise changes throughout the breeding season for different species (Fig. 3.3). Available data show that in temperate areas, most species sing gradually earlier with respect to sunrise as the reproductive season starts, and tend to sing later when reproduction wanes (Allard 1930; Da Silva et al. 2014; Hasan 2011; Keast 1994a; Lein 2007; Leopold and Eynon 1961; Scheer 1952). This pattern is by no means general and there are strong species- and population-specific differences. In a study conducted in Norway, Amrhein et al. (2008) compared the singing behaviour of blue and great tits across the season. They found that both species sang later relative to sunrise as the season progressed and that, whereas single-brooded blue tits stopped dawn singing after the laying period, great tits continued singing, possibly preparing for a second brood (Amrhein et al. 2008). In contrast, other populations of blue tits sing increasingly early as the season progresses (Da Silva et al. 2014; Poesel et al. 2001).
Changes throughout the year in the time of the first song (white dots) or the first call (black dots) in the song sparrow (Melospiza melodia) in relation to sunrise (dashed curve) and civil twilight (solid curve). Reprinted from Fisler (1962), by permission of Oxford University Press
These data suggest that earlier than average dawn singing is characteristic of the first part of the reproductive season, which includes the pairing stage and the female fertility period. Singing early at the time of pairing is to be expected if song earliness conveys reliable information about male quality (see Hypothesis 5), since it would pay males to show their capacity at this time. However, the period of female fertility is a key phase of the reproductive season that has been specifically linked to the dawn chorus.
Hypothesis 3: Manipulation of Female Mating Behaviour Around the Fertilisation Window
The fertile period of female birds typically lasts from some days before to a few days after laying of the first egg (Birkhead and Møller 1998). Within this period, some authors have assumed that females would be particularly fertile at dawn, right after laying (Birkhead and Møller 1992; Sheldon and Burke 1994). Ruth Mace (1987a) showed that the duration of male song at dawn in the great tit was closely connected to fertility of the male’s own female. In her study, the dawn chorus appeared only before laying, increased in duration at the female’s fertile period and shortened progressively until stopping altogether a week after the last egg was laid (Fig. 3.4a). This coincidence in time (both the period of the season and the time of the day) led Mace to suggest that the function of dawn singing would be to: a) protect paternity at a time when sneaker males could approach fertile females, and b) to increase female receptivity to copulations by the singing male.
Contrasting patterns in dawn song in relation to female fertility: (a) song rate showing an increase around the fertility period in the great tit (Parus major). Reprinted from Mace (1987a); (b) dawn song, but not day-time song, was almost absent when a male’s female was fertile in the chipping sparrow (Spizella passerine; M,B = mating or copulations, nest-building) in contrast to unpaired males (U), just paired (P), egg-laying (L), incubation (I), nestling (N) or fledgling (F) periods. Reprinted from Liu and Kroodsma (2007), by permission of Oxford University Press
A similar hypothesis was later proposed to explain high singing rates of paired males in general (Møller 1991). These two hypotheses assume that song is an honest signal of male quality (Gil and Gahr 2002), and that a high rate of song at dawn is disproportionally costly for low quality males (see hypothesis 5 further down for evidence of dawn song acting as a handicap).
Additional evidence for a link between female fertility and dawn song has been found in many other species, including the yellowhammer (Emberiza citrinella) (Møller 1988), two species of Ficedula flycatchers (Pärt 1991; Vabishchevich 2011), the Eastern phoebe (Sayornis phoebe) (Bruni and Foote 2014), the Eastern kingbird (Tyrannus tyrannus) (Sexton et al. 2007), the Eurasian blackbird (Cuthill and Macdonald 1990), the dusky warbler Phylloscopus fuscatus (Forstmeier and Balsby 2002), and the European starling Sturnus vulgaris (Pinxten and Eens 1998). In these cases, fertility is linked with increases in the duration or advances in singing at dawn, but males generally show a dawn chorus before and after female fertility has stopped, suggesting that this behaviour is not exclusively directed to the male’s own partner.
Additional behavioural patterns have been observed during the female fertility period, implying an important role of male dawn song at this time for some species, but with alternative functions than those proposed previously. Thus, in several species of Paridae, males sang for longer at dawn when their females were experimentally removed, and stop singing as soon as they appeared again (Mace 1986; Otter and Ratcliffe 1993; Welling et al. 1997a). A similar pattern was found in the collared flycatcher (Ficedula albicollis): males stopped dawn singing as soon as their females appeared in sight (Pärt 1991). In addition, song characteristics or singing behaviour at this particular time of the breeding season show important modifications in some species, supporting a role of dawn song in female guarding and extra-pair mating. For instance, a study in a Dutch great tit population showed that males produced songs with slightly lower-frequency on the day before the first egg was laid, and that females appear to use differences between males in these songs to choose extra-pair partners (Halfwerk et al. 2011). In the blacked-capped chickadee (Poecile atricapillus), males sing at lower rates when their females are fertile than at other times of the breeding season (Foote et al. 2008b).
However, this hypothesis has met several hurdles that limit its value. Firstly, although further studies on the great tit have also found a link of early dawn song with female fertility, none has documented a complete cessation of dawn song after laying (Amrhein et al. 2008; Naguib et al. 2016), suggesting that the extreme dependency of dawn song with fertility shown by Mace was fortuitous. In addition, there are strong differences between species in whether males sing or not while their females are fertile (Gil et al. 1999b). Examples of birds in which their dawn chorus is completely unrelated to female fertility include the American robin (Turdus migratorius) (Slagsvold 1996), the common nightingale (Luscinia megarhynchos) (Kunc et al. 2005) and two different Phylloscopus warblers (Gil et al. 1999b; Rodrigues 1996). In an even more contrasting fashion, chipping sparrow males (Spizella passerina) do not sing at all when females are fertile (Liu 2004), whereas they sing for the remainder of the season (Fig. 3.4b).
A second hurdle to the hypothesis is the disputed evidence for a fertility window after laying (Birkhead et al. 1996). Birkhead et al. (1996) reanalysed existing data and concluded that fertility is in fact reduced after laying. This point of view has been criticised for being exclusively based on evidence from artificial insemination studies in non-passeriformes, whereas further studies in the wild do show increased mating rates in the early morning in some species (Drachmann et al. 1997). Also, important rates of early-morning mating have been found in the nightingale, a species in which females look for males and mate before dawn (Roth et al. 2009) and in the highly promiscuous superb fairy-wren (Malurus cyaneus) (Double and Cockburn 2000), species in which females perform extra-pair forays at night. A particularly strong case for the hypothesis is provided by the European starling Sturnus vulgaris, a species that does not lay eggs at dawn but in the late morning. In agreement with the fertility insurance hypothesis, this species shows a peak of singing and mating in the late mornings of the fertile period around the time of egg laying (Pinxten and Eens 1998).
At least two additional reasons for dawn singing in relation to female fertility can be proposed (Staicer et al. 1996). Firstly, irrespectively of the existence of a fertilisation window, if females are more likely to look for extra-pair copulations at dawn to escape their mate’s guarding (Double and Cockburn 2000; Roth et al. 2009), it would make sense for males to show a good performance at this time in order to attract these females. This would explain why males keep on singing when their own partner is no longer fertile. In other words, the selective pressure would not just relate to ensuring the male’s paternity with his own female (Mace 1987a; Møller 1991), but to increase the probability of fertilising other females. A prediction from this hypothesis is that the dawn chorus should be longer (seasonally speaking) with increasing levels of extra-pair paternity and breeding asynchrony in the population. An experimental study in which male great tit sleep was prolonged with melatonin led to a decrease in paternity (Greives et al. 2015), in agreement with data showing that early singing males are more successful at fathering extra-pair offspring in this species (Kempenaers et al. 2010).
A second reason for singing early around the female’s fertile period may be to increase female egg allocation (Biard et al. 2009; Gil 2008). Females can modify their investment in eggs (in terms of egg size or of contents, such as carotenoids or hormones) as a function of male attractiveness (Cunningham and Russell 2000; Gil et al. 1999a, 2004). This process should lead males to show off their ornaments not just before pairing, but also during egg production, since females may adaptively regulate their investment as a function of male quality. If singing early represents a handicap (see hypothesis 5), it would pay males to show their quality by singing as early as possible at the time when the female is producing eggs (i.e. a few days before egg laying).
3.4.3 Social Interactions
Singing behaviour takes place in several types of social interactions, from mate attraction to male-male counter-singing. Important cues about the intended receivers of the dawn chorus could be given by the particular characteristics of the song at this time (e.g. singing style or song repertoire), to the extent that song directed to males may differ from that directed to females (Collins et al. 2009). However, since the two main functions of song may entail honest signalling (Berglund et al. 1996), differences in repertoire size or song rate between dawn and other times of the day may not tell us much about the likely target of the dawn chorus (Dalziell and Cockburn 2008). Instead, more subtle characteristics, such as singing patterns (song matching, overlapping, singing site within the territory, etc.), may be more informative.
Singing location. Studies on several species have shown that dawn song is often uttered at territory boundaries, suggesting that it is directed to neighbouring territories (Bolsinger 2000; Lein 2007; Liu and Kroodsma 2007). For instance, in the chestnut-sided warbler (Setophaga pensylvanica), the use of unaccented songs at dawn, predominantly used in male-male interactions, takes place at territory borders (Byers 1996). However, studies in other species offer a contrasting view. Thus, in great tits and collared flycatchers, males sing near the nest (Mace 1986; Pärt 1991), or in the centre of the territory in the case of the yellowhammer (Møller 1988). Conflicting demands of foraging (Atienza and Illera 1997) and avoiding predation (Møller 2011) are known to determine choice of song posts. Given that both foraging substrate and predation risk change with time of day, analysing changes in song post location seems a promising source of evidence on the function of song and constraints on singing.
Song characteristics. In addition to daily differences in singing sites, several studies have found that songs at dawn sound particularly loud in some species. For instance, in the banded wren (Thryophilus pleurostictus), dawn songs are louder and more complex than those sung later in the day, and they are reminiscent of those that birds use in agonistic interactions (Trillo and Vehrencamp 2005). In the chipping sparrow, dawn songs are shorter but sung at very high rates (Liu and Kroodsma 2007). The intensity of some dawn performances can also be assessed by how song is matched to neighbours. Thus, in the black-capped chickadee, the levels of song matching are particularly high at this time (Foote et al. 2008a). Several researchers suggest that these singing styles reflect a dynamic network of communication between territory holders (Brunner and Pasinelli 2010; Foote et al. 2008a; Naguib et al. 1999; Trillo and Vehrencamp 2005). It would be interesting to know by means of playback experiments whether dawn songs elicit stronger responses than songs used during the daytime.
Special dawn songs. Ever since the first observations on dawn singing, researchers have noted differences between dawn song and songs that are used later in the day (Fig. 3.5). In most species studied so far, dawn songs tend to be more complex and elaborate than daytime songs (Staicer et al. 1996). Early authors already remarked that the Eastern kingbird and the Eastern wood pewee (Contopus virens) use a particular song before sunrise, which changes drastically after that (Allard 1930; Allen 1913). Similar patterns have been described in many species of the Americas (for a comprehensive list see: Staicer et al. 1996), mostly in the Tyrannidae and Parulidae (Kroodsma et al. 1989; Morse 1989; Spector 1992) and in some Emberizidae (Nelson and Croner 1991). Less is known about species that do not change repertoire in a dramatic way. Some examples show that differences may be subtle, with changes in general song composition. For instance, dawn repertoires are more complex and variable than daytime repertoires in Golden-cheeked warblers (Setophaga chrysoparia) (Bolsinger 2000), Canada warblers (Cardellina canadensis) (Demko et al. 2013) or field sparrows (Spizella pusilla) (Nelson and Croner 1991). Interestingly, we could not find examples among European species, with the exception of the common reed bunting (Emberiza schoeniclus) in which males sing at dawn a particular repertoire of long, highly versatile songs with short between-song intervals (Brunner and Pasinelli 2010), and the Eurasian blackbird, whose dawn song sounds louder than daytime song to human ears (Dabelsteen 1992).
Hypothesis 4: Territory Defence and Social Dynamics
One of the first explanations proposed for the dawn chorus was based on the idea that there should be a higher number of vacant territories in the morning than at other times of the day, because of overnight mortality, thus increasing territorial instability (Kacelnik and Krebs 1983). In that context, it would pay territorial birds to reinforce border defence at this time since new males would try to settle in these vacancies. Although territory turnover may be generally too low to explain the strength and ubiquity of the dawn chorus, some data do suggest that invasion rates in the early morning are higher than later in the day (Amrhein et al. 2004a; Kacelnik and Krebs 1983). Floaters in some species actively seek territories at this time (Amrhein et al. 2004a; Dalziell and Cockburn 2008), perhaps because all males are singing and it is easier to detect vacant areas than later on in the day (Amrhein et al. 2004a). In this sense, the hypothesis could generalise to the fact that dawn is a time of high risk for territory owners.
The hypothesis does not require a higher than average night mortality. Since no territories are taken up at night, there will always be a higher number of vacancies at dawn than in any other time of the day, even if mortality rate is constant through the day (Kacelnik and Krebs 1983). The realisation that the night represents a break from territory defence and signalling for most songbirds suggests to us an additional twist to this hypothesis. Even if no territorial vacancies appear, social relationships among territory holders have a dynamic nature (Møller 1990), and need to be continuously refreshed throughout the breeding season (Burt and Vehrencamp 2005; Staicer et al. 1996). This necessity may explain why birds sing through the day in most temperate songbird species, although singing rates are lower after dawn (Slagsvold 1977). This process is interrupted by the night and, thus, dawn would emerge as the first window of time to resettle hierarchies, territory borders and dominance relationships. Even if no territory vacancies have appeared during the night, the absence of communication during this period could select for a renewal on information exchanges that may transmit information on resource holding capacity, body condition, etc., since the probability of a change is higher given the extent of time that has passed.
Several sources of evidence support the importance of the dawn chorus for resettling territory and hierarchy structures (Foote et al. 2010). Firstly, mated males of many species show dawn song throughout the breeding season, well after female attraction and fertile periods, suggesting strong selection for signalling territory occupancy (Amrhein et al. 2004b; Liu and Kroodsma 2007). Secondly, we have seen before that in many species dawn songs are particularly loud, aggressive and directed to territory boundaries, often associated with a high degree of counter singing and song matching. For instance, a detailed study of singing interactions in the banded wren has revealed a broad interactive network at this time, with complex matching patterns among many birds (Burt and Vehrencamp 2005). A third line of evidence comes from experimental manipulations of intruder presence that lead to changes in the dawn chorus in the morning following the manipulation (Amrhein and Erne 2006; Erne and Amrhein 2008; Foote et al. 2011; Liu 2004; Sexton et al. 2007; Xia et al. 2014). A clever experiment compared the next-morning song response of Eurasian wrens (Troglodytes troglodytes), depending on whether they had been exposed to a normal playback or a “scary” playback which involved several changes in loudspeaker position, simulating a particularly challenging intruder (Amrhein and Lerch 2010). The results showed that the wrens that had received the challenging playback started to sing earlier at dawn, and sang more and longer songs than those exposed to normal playback (Fig. 3.6).
Differences in the response of Eurasian wrens (Troglodytes troglodytes) depending on whether they received a stationary playback (black dots, solid lines) or a playback simulating an intruder changing song posts (white dots, dashed lines): (a) shows mean (± SE) dawn song output (PC1 scores loaded by song output and song earliness) shortly before (first day) and one day after (second day) the playback; and (b) shows number of songs per minute over 6 min in response to the two types of playbacks. Reprinted from Amrhein and Lerch (2010)
3.4.4 Individual Differences
Following the suggestion that dawn singing could represent a handicap (Gil and Gahr 2002; Hutchinson et al. 1993; Montgomerie 1985), several studies have examined whether there are differences in dawn song timing, song rate or song length in relation to individual variation in fitness-related traits (e.g. quality, condition, social status, etc.). We should stress that, as far as the question of “why do birds sing at dawn” is concerned, the individual difference of interest is timing of singing, i.e. is there a relationship between male quality and time of first song at dawn? Timing considered as a sexually selected trait has been formalised by Hau et al. (2017), who propose that daily timing of trait expression is linked with circadian clock genes and can be subjected to sexual selection in birds and mammals.
Researchers have found repeatable differences between individuals in dawn song timing in several species (Amrhein and Erne 2006; Dunnett and Hinde 1953; Foote et al. 2011; Snijders et al. 2015). A remarkable study in the Eastern kingbird found timing to be a repeatable trait between different years (Fig. 3.7) (Murphy et al. 2008). This implies that males are consistent in how early they sing, a prediction from sexual selection models that propose a relationship between variation on this trait and individual condition or resource holding capacity (see hypothesis 5 below). Similarly, multiple parameters of sleep behaviour in the blue tit, such as sleep duration or awakening time, also show substantial individual repeatability across years (Steinmeyer et al. 2010).
Between-year repeatability for dawn song time (a) and rate (b) in Eastern kingbirds (Tyrannus tyrannus). Reprinted from Murphy et al. (2008), with permission from Elsevier
Several studies have found that song timing can predict traits related to individual quality or condition. For instance, early singing males are larger, and possibly older in Eastern kingbirds (Murphy et al. 2008). Dominant black-capped chickadees sing earlier than subordinates (Otter et al. 1997). Also, older blue tits sing earlier than young birds (Poesel et al. 2006), although a non-significant tendency in the opposite direction was found for great tits (Snijders et al. 2015). Finally, early-singing willow tits (Poecile montanus) had stronger anti-predatory responses and were more likely to survive than late-singers (Welling et al. 1997b). Further evidence on the relationship between song timing and condition is provided by experiments that have found earlier song in food-supplemented birds (Barnett and Briskie 2007; Cuthill and Macdonald 1990; Grava et al. 2009; Montgomerie 1985; but see: Saggese et al. 2011; Thomas 1999; Thomas and Cuthill 2002).
An experiment on domestic roosters (G. gallus domesticus) has shown that dominant birds crow earlier, and that experimentally-induced changes in hierarchy lead to a correlated effect on timing; i.e. beta males sang earlier when the alpha male had been removed (Shimmura et al. 2015). This elegant experiment confirms observations on red jungle-fowl (G. gallus murghi) in the wild that link early crowing time to higher positions within the group hierarchy (Collias and Collias 1967). Although this example on a highly social and hierarchical species may not translate easily into the typically passerine-dominated dawn chorus, such a strong link between dominance and timing does suggest that males do signal something relevant by singing earlier than others.
A more definite test of the fitness advantage of early singing is provided by extra-pair paternity (EPP) studies. Several studies have linked the earliness of singing with higher success at siring extra-pair offspring. Eastern kingbirds that sang earlier had higher numbers of EPP chicks, and cuckolders sang earlier than cuckoldees (Dolan et al. 2007). Taking advantage of differences in dawn singing timing caused by artificial lighting of a part of the forest, Kempenaers et al. (2010) found that early blue tits, i.e. those exposed to artificial light, were more successful at obtaining EPP than late birds. This study replicated previous results in the same species, in which natural variation in dawn song was also identified as a good predictor of EPP success (Poesel et al. 2006). Similarly, male great tits treated with melatonin woke later and lost more paternity than control birds (Greives et al. 2015). It is not clear whether the mating advantages accrued by early birds was a consequence of males using the period before dawn for pursuing extra-pair mating, or was mediated by female choice of early singing birds. Evidence for the latter possibility is provided by a study showing that female Lincoln’s sparrows (Melospiza lincolnii) preferentially chose songs that they had heard at very cold temperatures (Beaulieu and Sockman 2012), a situation associated with dawn song in this species.
Despite the previous examples, rather few studies have measured individual differences in dawn singing time, possibly because of the inherent difficulty of obtaining these data. The recent development of cheap automatic recording units (ARUs) and miniaturised sound-recording data loggers will improve the situation in the coming years.
Hypothesis 5: Early Singing as a Handicap
One of the leading hypotheses to account for the evolution and maintenance of male ornaments is Zahavi’s handicap hypothesis and variations on it (Biernaskie et al. 2018; Grafen 1990; Kokko et al. 2006; Zahavi 1977). The hypothesis states that ornaments should be costly and that the cost should disproportionally increase with increasing expression of the signals. The suggestion that early singing in the dawn chorus could act as a handicap was proposed by Montgomerie (1985), on the assumption that singing at dawn is costlier than at other times of the day. It is by no means clear why this should be so, although some possibilities may be: temperature-dependent singing costs (Ward and Slater 2005) in the case of temperate-climate species, higher risk of predation by nocturnal birds of prey (Schmidt and Belinsky 2013), or costs of sleep deprivation (Cirelli and Tononi 2008).
However, as we saw above, several lines of evidence suggest that early singing, at least in some species, fulfils some of the requirements for handicaps: (1) there is individual repeatability in dawn singing time (Murphy et al. 2008); (2) some experiments and correlative studies show positive relationships between age, dominance, access to supplemental food and time of the dawn chorus (Cuthill and Macdonald 1990; Murphy et al. 2008; Otter et al. 1997; Poesel et al. 2006; Shimmura et al. 2015); (3) early singers may obtain a disproportional share of extra-pair fertilisations (Dolan et al. 2007; Kempenaers et al. 2010); and (4) predation by nocturnal predators may limit early singing (Lima 2009, but see Sect. 3.4.7).
Despite these suggestive findings, a true demonstration of a handicap requires some extra evidence both on the signal-production side (Cotton et al. 2004), and on the female response to it (e.g. Ballentine et al. 2004). A good test of this hypothesis would require experimental modifications of singing time, which are difficult to induce. Even if tampering with melatonin levels could modify singing time (Dominoni et al. 2013a), there are many correlated effects of this hormone that would render the results difficult to interpret (Greives et al. 2012).
3.4.5 Foraging
The role of food is central to several hypotheses about the dawn chorus (the inefficient foraging, unpredictable conditions and handicap hypotheses), and hence estimating the relationship between song and food availability is a crucial step to test these alternatives. Foraging also shows diel cycles, being constrained by both abiotic and biotic factors. Dawn does not appear to be a good time for a bird to forage, since low temperatures and dim light levels affect foraging success. It has been shown that the activity of arthropods is greatly reduced at low temperatures (Avery and Krebs 1984), decreasing the ability of birds to locate food. In addition, low light levels constrain vision, decreasing searching ability and foraging profitability (Kacelnik 1979), probably for both insectivorous and granivorous birds (Fig. 3.8).
Foraging profitability increases as a function of light levels in the great tit (Parus major). Shaded in grey is the time of dawn chorus, showing that light conditions are not ideal for foraging at this time. Reprinted from Kacelnik and Krebs (1983)
Species differences in dawn chorus timing in relation to the type of diet were suggested early on by Armstrong (1963), who remarked that vermivorous and insectivorous species sing earlier than granivorous birds, and speculated that differences in vision and in the behaviour of prey may explain this pattern. However, we have not been able to find a proper test of this hypothesis. Most of the recent comparative analyses of dawn chorus timing dwell on differences in relative eye size with little mention of food guild (Berg et al. 2006; Gil et al. 2015; Thomas et al. 2002). Since eye size is related to food, i.e. birds that forage on mobile prey have relatively larger eyes than seed-eaters (Garamszegi et al. 2002), it seems that vision and feeding are so closely correlated that teasing apart these factors to explain song timing may be challenging (Chen et al. 2015).
Although some behavioural and ecological adaptations in relation to food guild have been studied (e.g. Falk et al. 1992; Wiens and Johnston 1977), we could find surprisingly little comparative information on how type of food affects behavioural patterns such as time-budgets or food search routines. In addition to differences in predictability between insects and seeds, important variation in metabolizable energy provided by arthropods, seeds or fruit (Karasov 1990) probably imposes differential constraints on time-budgets.
Hypothesis 6: Inefficient Foraging at Dawn
This hypothesis posits that dawn is a time when, because of low insect activity and poor visual conditions, it is not profitable to forage (Kacelnik 1979; Kacelnik and Krebs 1983). The lack of interference with feeding activities would favour birds that use this time for singing and communicating. Inefficient foraging is a clever hypothesis, but it has some difficulties. For instance, it has been found that birds breeding in continuous light in the Arctic also show dawn choruses (Staicer et al. 1996). This finding however would not necessarily refute the hypothesis since it is possible that foraging profitability in these birds may also change daily in response to variations in temperature or more subtle light differences.
An experimental test of this hypothesis in captivity failed to support one of its major predictions: birds did not modify their dawn singing activity in response to a dramatic change in food availability (Mace 1989). A possible line of research here could involve comparing male and female feeding time budgets in species in which females do not sing. Such a comparison could give us evidence as to whether males are using free time to sing, or else are trading off precious feeding time for singing (Greig-Smith 1983). The hypothesis has however received some theoretical support from a game-theory model, which predicts the occurrence of both dawn and dusk chorus by simply allowing feeding profitability to vary as originally suggested by Kacelnik (Hutchinson 2002). See section “Unpredictable overnight conditions” below for a combined discussion of these two hypotheses.
The inefficient foraging hypothesis also touches an important point as it suggests that dawn is “spare” time within the bird’s time budget. For this to be true, it means that sleep in birds should require less time than the night duration. Several lines of evidence suggest that this is the case. Firstly, studies analysing differences in activity across latitudes show that birds are remarkably flexible in their sleep requirements, and that low light levels are a constraint in activity (Daan and Aschoff 1975). Secondly, it has been shown that late-singing species spend more time awake waiting to sing than early species (Scheer 1951). Although this latter evidence is largely anecdotal, it supports the idea that the duration of night in spring actually exceeds the requirements for sleeping time. Thirdly, studies of migratory restlessness show prolonged activity over night (Berthold 1973), or round-the-clock singing and mating patterns in nightingales and pectoral sandpipers (Calidris melanotos) (Lesku et al. 2012; Roth et al. 2012). Thus, if birds do not need to sleep for the whole night at the breeding period, it makes sense that dawn is used for an activity not requiring acute vision such as song (Siegel 2009). An alternatively view would propose that singing earlier may uses precious sleeping time (Cirelli and Tononi 2008), and that time of dawn song is a sexually-selected character (see handicap hypothesis above). This would suggest that early waking-up in birds may be sexually selected (Amrhein et al. 2002; Greives et al. 2015), although it is not clear what kind of costs are paid. It is possible that an increase in predation rather than costs of reduced sleep is the true currency of this mechanism. The study of comparative sleep in birds is in its infancy in comparison to mammals, and evidence so far shows that bird sleep has some important peculiarities (Roth et al. 2006). The ongoing development of small sleep dataloggers should allow researchers in the next decade to provide important insights on this topic (e.g. Lesku et al. 2012; Steinmeyer et al. 2010).
Hypothesis 7: Unpredictable Overnight Conditions
Stochastic-dynamic programming (SDP) models allow the prediction of optimal routines given quite realistic assumptions about foraging success, energy use and singing rewards (McNamara et al. 1987). Using this technique, a pioneering study by McNamara et al. (1987) showed that a dawn chorus could simply arise as a consequence of adaptive foraging decisions. The key aspect lies in the unpredictability of overnight costs. Briefly, since there is always some unpredictability of how demanding the night may be in terms of energy requirements, birds must go to sleep with an extra amount of energy. As dawn approaches, and the probability of overnight starvation decreases, birds can use that surplus of energy for singing at dawn. Interestingly, the model also predicts a smaller peak of singing at dusk, which is in agreement with many singing cycles in passerines (Cuthill and Macdonald 1990; Møller 1988; Morse 1989).
One of the most useful methods for testing the effect of energy managing on dawn chorus timing is by providing supplementary food. Experiments in four species have shown that individual males provided with supplementary food tend to sing earlier and at higher rates (Barnett and Briskie 2007; Cuthill and Macdonald 1990; Grava et al. 2009; Thomas 1999). In contrast to this finding, a study with great tits showed the opposite effect, with supplemented birds singing later than controls (Saggese et al. 2011). The main difference of the latter study is that it involved long-term instead of short-term modifications.
Although assumptions of the SDP models are somehow simplistic, they do make some interesting predictions on how variance in overnight requirements and foraging profitability may affect singing (McNamara et al. 1987). One key prediction is that the dawn chorus should be longer with increasing unpredictability of overnight requirements (Hutchinson et al. 1993; McNamara et al. 1987). In our opinion, the most relevant test of this model should focus on different populations of the same species facing contrasting environmental conditions, or on different species that differ in weather sensitivity. However, to our knowledge, no study so far has taken this approach.
At the individual level, an experiment in European robins (Erithacus rubecula) found that birds lost more weight when overnight temperature was lower, but they did not adjust their singing in relation to temperature (Thomas and Cuthill 2002). However, in this case it is possible that food supplementation may have altered the normal patterns of foraging and singing. Only one study that we know of has experimentally tested the prediction for a negative relationship between energy predictability and dawn song duration (Thomas 1999). In this experiment, Thomas provided supplemental feeding to territorial robins under two different treatments: predictable and unpredictable. His findings show that robins sang relatively less at dawn (and hence relatively more at dusk) after they have been fed in an unpredictable fashion, whereas the contrary was true when food supplementation was constant (Thomas 1999). The results supported the SDP model’s prediction and imply that the high unpredictability of night time temperature together with food predictability combine to adjust singing times between dawn and dusk. It is regrettable that this hypothesis has not been tested further (Hutchinson and McNamara 2000), not just at the individual level, but also within a comparative perspective.
Further developments with this type of models have introduced more complex situations, such as differences in male quality and in singing costs across the time of day (Hutchinson et al. 1993). The latter effectively assumed that dawn song works as a strategic handicap (see hypothesis 5) within the framework of SDP predictions. By using SDP models, Hutchinson (2002) pitched Kacelnik’s inefficient foraging hypothesis (see hypothesis 6) against hypotheses based on the stochasticity of overnight energy requirements, and found that both models were equally satisfactory in predicting dawn and dusk choruses.
Although these models are useful in generating ideas and predictions, in practice they are extraordinarily challenging to test (Hutchinson and McNamara 2000). For instance, they require a precise estimate of song costs, while the literature on this respect show ambiguous findings (Gaunt et al. 1996; Thomas 2002; Ward and Slater 2005; Ward et al. 2003). Generally speaking, data suggest that the greatest cost is that of lost foraging time, rather than a of singing per se (Gil and Gahr 2002). Similarly, foraging profitability across the day depends on so many factors, and data are so scant on this, that some predictions are extremely difficult to test empirically.
3.4.6 Predation
Predation is a key factor that permeates every single aspect of bird behaviour and ecology, but its role in shaping dawn chorus is seldom considered (Lima 2009). This is regrettable because experiments have shown that predators do use acoustic signals to locate prey in a variety of taxa (Haskell 1994; Krams 2001; Zuk and Kolluru 1998), influencing the evolution of singing behaviour (e.g. Tuttle and Ryan 1981). It is not clear whether predation is higher at dawn or not: some researchers have predicted higher than average predation risk at dawn (Lima 2009), while others have proposed the opposite (Staicer et al. 1996).
Research shows that birds modify behaviour in response to predation risk, and that this can vary with changes in light or noise levels (Fernandez-Juricic and Tran 2007; Klett-Mingo et al. 2016). Since dawn is characterised by low light levels, foraging at this time could be risky if predator sight in these conditions is better than their prey’s (Fernandez-Juricic and Tran 2007). It has been found that choice of perch during the day can be explained by predation risk (Møller 1991), and a study has shown that blue tits choose comparably safer perches from which to sing at dawn than later on in the day (Parker and Tillin 2006). However, we have not been able to find any experiment that has directly tested the influence of predation risk on singing at dawn. Nonetheless, a couple of studies on dusk singing show an influence of predation risk perception. Thus, a playback experiment with male veeries (Catharus fuscescens) shows reduced levels of dusk song in places where owl calls have been previously broadcast (Schmidt and Belinsky 2013). Similarly, a correlative study in tawny owls (Strix aluco) shows that dusk calling rates are reduced in areas with high calling rates of Eurasian eagle owls (Bubo bubo), a natural predator of the tawny owl (Lourenço et al. 2013).
Hypothesis 8: Low Predation at Dawn
It has been proposed that the dawn chorus is advantageous because of a presumably lower risk of predation at dawn (Staicer et al. 1996). However, as we have seen above, although there are some studies showing that the behaviour of singing birds is affected by predator risk, no study that we know of has analysed whether indeed dawn is less risky than other times of the day. Lower predation may also be a consequence of a dilution effect, since many birds singing simultaneously would reduce the chances of being located and predated (Hamilton 1971; Lima 2009). However, the dilution effect would not explain why dawn is the best time to sing, it would simply select birds to sing synchronously.
One of the predictions of this hypothesis, namely that birds should use more exposed perches at dawn than later on in the day (Staicer et al. 1996), was actually found to be the contrary in a study in blue tits (Parker and Tillin 2006). It is important to empirically establish the relationship between predation risk and time of day, because if predation is higher at dawn (Lima 2009), contrary to the predictions of this hypothesis, we would face a handicap situation (see hypothesis 5) with very different predictions. In agreement with the latter idea, Lima (2009) proposed that the relationship between relative eye size and time of singing at dawn could be explained by differences in predation risk, in addition to the more common explanations based on foraging profitability or unpredictable overnight conditions. Following this suggestion, the prediction would be that earliest-singing species, those with larger eyes, should excel at detecting predators at dawn.
3.4.7 Weather
Weather is also a pervasive factor influencing almost all aspects of bird ecology and behaviour. Most studies find that “bad” weather in general (rain, clouds, storms or low temperature) tends to delay the onset of the dawn chorus, although not all species respond similarly to these weather phenomena (Bruni et al. 2014; Keast 1994b). Several studies in northern temperate areas find a positive correlation between overnight temperature and duration of song, often implying earlier choruses (Bruni et al. 2014; Garson and Hunter 1979).
However, to the extent that bad weather limits singing in general (Curio 1959), this evidence does not shed much light on the reasons behind the dawn chorus. In other words, given that singing interferes with foraging, we expect a positive correlation between temperature (proxy of food availability, at least for insectivores) and singing activity irrespective of the time of day. Also, since bad weather typically implies a decrease in light levels, it is possible that the reasons for this relationship can also be explained by the effect of light (see Sect. 3.4.8).
Predictions about how bad weather should affect dawn singing are complex, and may depend on the particular aspect of weather (clouds vs. temperature) and the particular theory that is tested (Hutchinson 2002). A study in great tits found that the correlation between overnight temperature and dawn singing was lower (and non- significant) than that with overall morning singing (Garson and Hunter 1979). This pattern was explained as evidence of low foraging profitability at dawn: since profitability is low at dawn it would pay birds to sing irrespectively of temperature, whereas a finer adjustment would make sense later on in the morning, when feeding time is more rewarding and would trade-off with time devoted to sing.
Hypothesis 9: Better Sound Transmission Conditions at Dawn
A widely cited hypothesis for explaining the dawn chorus proposes that this particular time of the day is the best time for sound transmission (Henwood and Fabrick 1979). This proposal draws on the logic that sound attenuation and degradation are a function of the habitat and the atmospheric conditions in which the sound is produced and perceived (Morton 1975; Richards and Wiley 1980). Several studies have shown that atmospheric conditions before sunrise, characterised by low temperatures and absence of air turbulence, allow better sound transmission than those found later in the day, leading to a longer transmission distance and a larger active space (Brenowitz 1982; Henwood and Fabrick 1979; Larom et al. 1997). However, not all studies have been successful at finding better transmission efficiency at dawn, suggesting that this is by no means a general advantage (Brown and Handford 2003; Dabelsteen and Mathevon 2002). A refinement of this hypothesis proposed that dawn offers not better, but a more consistent sound transmission than other times of day (Brown and Handford 2003).
The sound transmission hypothesis has an important limitation (Mace 1987b): even if we accept that atmospheric conditions are on average best at dawn (Henwood and Fabrick 1979), or at least more constant (Brown and Handford 2003), the high number of birds singing at that time should impose huge perception costs (Langemann et al. 1998; Poesel et al. 2007; Pohl et al. 2009) that would most likely reduce this advantage (Mace 1987b). Indeed the dawn chorus can be so noisy that it has been suggested that switching singing to a dusk chorus could be a good strategy to reduce overlapping of song at dawn (Belinsky et al. 2012). In general, we believe that sound transmission does not offer a good explanation for the maintenance of the bird dawn chorus.
3.4.8 Light
From very early on, observers have found that higher light levels lead to earlier choruses and that, on cloudy mornings, birds sang later than usual (Allard 1930; Scheer 1952). From a comparative perspective, species with relatively large eyes for their size also start to sing earlier (Berg et al. 2006; Gil et al. 2015; Thomas et al. 2002), thus providing additional evidence for a role of light in song timing (see Sect. 3.2).
Studies that have tracked song timing with respect to the moon phase have shown that a full moon leads to earlier dawn choruses in several species (Bruni et al. 2014; York et al. 2014). In a study on the white-browed sparrow weaver (Plocepasser mahali), York et al. (2014) found that the effect of the moon phase was only detected when the moon was not hidden by clouds. In other words, it was the actual increase in light and not the lunar cycle that was responsible for the change in behaviour.
Recent interest on the effects of artificial lighting in bird behaviour has led to numerous studies that have shown earlier choruses in urban areas (Bergen and Abs 1997; Miller 2006). It is important to underline that correlative studies, simply comparing areas that differ in light levels, may be affected by other confounding factors, such as noise (see Sect. 3.4.9), and these may be difficult to disentangle statistically (Fuller et al. 2007; Nordt and Klenke 2013). In addition to effects on the dawn chorus, correlative and experimental data show that artificial light at night alters also the timing of activity (Dominoni et al. 2013a), so that earlier song could follow from earlier wake-up time. Furthermore, artificial light also advanced reproductive cycles, leading to earlier gonad maturation, singing and breeding (Da Silva et al. 2015; Dominoni et al. 2013b; Gwinner and Brandstatter 2001).
A major study investigating the effects of night light pollution on avian behaviour found that four out of five species considered showed an advance in singing in suburban areas with artificial light (Kempenaers et al. 2010). This advance differed between species: earlier singers showed larger advances than late singers (Fig. 3.9); indeed, the last singer of the group, the common chaffinch (Fringilla coelebs), did not show an advance at all (Kempenaers et al. 2010). These patterns were partially replicated in an experiment in the wild in which light levels were manipulated, leading to immediate advances in singing for all studied species (Da Silva et al. 2016). Although the experimental results did not fully mirror the correlational data, the general pattern found in this and additional studies is that the earlier singing species are also those in which night light shows a stronger effect on song timing (Da Silva and Kempenaers 2017; Da Silva et al. 2014; Kempenaers et al. 2010). However, another similar experimental study testing different light colours found no effects of night lighting on singing, suggesting that fast timing plasticity is not present in all populations, and that previous selection may prime the response of birds to increased lighting (Da Silva et al. 2017).
Mean time of first songs in 5 European bird species in 6 control areas (dark blue dots) and 6 artificially illuminated sites (light blue dots) across 19 consecutive spring days. Reprinted from Kempenaers et al. (2010), with permission from Elsevier
The fact that birds sing earlier when nocturnal light levels are higher (due to moon lighting or artificial light pollution) does not explain why birds use dawn to sing rather other times of the day. However, to the extent that the size of the response varies between different species, this implies that the balance of costs and benefits also differs. In this respect is it important to consider the relationship of light with feeding. It is reasonable to think that effects may differ depending on type of food, since, for instance, arthropods relying on light may also show differences in behaviour. Some studies have found that birds take advantage of artificial light for feeding for a longer time (Russ et al. 2015), and that light is an important factor in the foraging/vigilance trade-off (Fernandez-Juricic and Tran 2007). Together, this evidence backs Kacelnik’s suggestion (1979) that the dawn chorus occurs at a time when foraging is not profitable.
3.4.9 Noise
Background noise is a limiting factor in all types of communication and, in the case of acoustic signalling, it may lead to a reduction in the distance over which sounds can be detected and recognized, also increasing the likelihood of false alarms and missed positives (Klump 1996; Langemann et al. 1998; Pohl et al. 2009). As noise is predictable in some environments, many researchers have examined whether birds modify their singing regime with respect to the occurrence of noise around dawn.
Anthropogenic Noise
Since noise in urban environments is reduced at night and in the first hours of the day, it makes sense for birds to try to reduce the overlap with urban noise by singing earlier than they would in the natural environment. This would lead to an earlier dawn chorus. The first study to detect this pattern in three common woodland species was not able to separate the contribution of light and that of noise pollution to the advance (Bergen and Abs 1997). However, research on three different species has shown that earlier dawn choruses in cities are due to increased noise levels after controlling for the effect of light (Dorado-Correa et al. 2016; Fuller et al. 2007; Nordt and Klenke 2013). This advance is considered advantageous, as it gives birds a window of communication at a time when noise levels are lower than later in the day (Fuller et al. 2007).
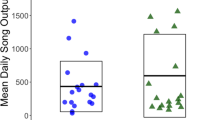
In these studies, it is important to demonstrate that earlier singing is not caused by noise disturbance, but rather that birds anticipate noise and sing earlier than that. An experiment in which noise was experimentally broadcast before dawn resulted in some species singing earlier, but it is unclear whether they were actually woken up by the sound rather than singing in anticipation of it (Arroyo-Solis et al. 2013). In contrast, true anticipation has been documented in studies that examined song timing near airports, in which birds were found to sing earlier than in control areas, thus anticipating the noisy start of airport operations (Dominoni et al. 2016; Gil et al. 2015; Sierro et al. 2017). Interestingly, in one of these studies, Gil et al. (2015) found that the effect size of the advance was inversely proportional to how early birds took part in the chorus. In other words, the advance was greater for the late birds than for the early birds. This made adaptive sense because the overlap with aircraft noise was greater for the late birds. On the same line, a study with blackbirds found that the advance was only detectable when airport timing overlapped with the dawn chorus (Sierro et al. 2017). In striking contrast to these effects, advances in timing due to light pollution are found in early, and not in late singers (Kempenaers et al. 2010).
Biotic Noise
In tropical environments, insects are a major source of noise for singing birds (Kirschel et al. 2009), and two different studies have examined whether birds avoid singing at the same time as cicadas. The first of these studies showed short-term avoidance, by which birds reduced the number of vocalizations or stopped singing when cicada song was present (Hart et al. 2015). The other study, conducted on Barro Colorado Island (Stanley et al. 2016), reports avoidance of insect-noise and also shows that birds whose songs overlap the frequency of the cicadas delayed their timing to avoid masking. However, the latter result may suffer from a basic methodological problem, as data collection was based on acoustic and not on visual observations, and singing birds could have lower detectability because of higher sound masking.
Although bird species start singing at different times, suggesting a division of the twilight-to-sunrise period, the truth is that, for a large part of the time, a respectable proportion of the bird community is singing simultaneously (although in some species there are strict slots seldom overlapped in time: Luther 2008). This realisation has led researchers to ask whether some form of time-sharing is at work during the dawn chorus, particularly in species-rich communities and among species that overlap extensively in acoustic frequency.
Avoidance of song overlap has been documented for several pairs of species coexisting in the same community (Brumm 2006; Cody and Brown 1969; Popp et al. 1985), but these findings only explain short-term adjustments that do not alter the general timing of singing activity. However, within a dawn chorus context, with many birds singing at the same time, it is relevant to ask if the avoidance of song overlap is related to the degree of song similarity between species. Similar songs lead to a greater degree of masking, and a higher probability of a potential receiver not detecting the signal (Klump 1996). The three studies that so far have tackled this question, all within Amazonian bird assemblages, have not found a common answer, possibly due to differences in methodology (Luther 2009; Planque and Slabbekoorn 2008; Tobias et al. 2014). A first study documented temporal avoidance between certain pairs of species, but not necessarily those with the largest overlap in frequency (Planqué and Slabbekoorn 2008). In contrast, working in a community with a high number of species, Luther (2009) found that birds that sang together within 30-min intervals at the same height in the forest showed lower overlap in frequency characteristics than birds picked at random (Luther 2009). This study suggested that species that sing at the same place and time reduce interference by shifting singing times (Luther 2009). In striking contrast, a more recent study by Tobias and co-authors (2014) found that species that sang within 10-min chorus sections had higher than expected song similarity and phylogenetic relatedness (Tobias et al. 2014). This finding could be explained by interspecific competition for resources leading to higher song similarity (Tobias et al. 2014), but the grounds for such claim are slightly speculative at this point.
This last study is one of the few examples pointing to interspecific communication in the dawn chorus. Earlier, Møller (1992) found increased singing activity of five different species after the playback of the song of the black wheatear (Oenanthe leucura), and suggested social facilitation as the underlying mechanism. Other attempts to test this hypothesis have failed to find support. For instance, a study in two species of thrushes (Turdidae) has shown that heterospecific playbacks do not lead to earlier choruses on the following days, whereas this is the case for species-specific playbacks (Hodgson et al. 2018).
In contrast to these minor temporal changes, the question of whether species-specific timing varies in response to competition between species for an acoustic space has received less attention. It is likely that the best answer to this question will come from comparative, rather than single-community studies. We are only aware of one study (as yet unpublished) that has compared whether differences in species richness affect the way that dawn chorus time is shared. Comparing dawn chorus patterns across the globe, Pollard (2009) found in her PhD thesis that, at sites with high species richness, the onset of dawn song was more variable than at sites of low richness. This pattern suggests that competition between species may indeed promote wider separation between the singing times of species singing in the same chorus. A limitation of this study is that most data on choruses came from the Northern Hemisphere, with a very small sample size for the Equator and the Southern Hemisphere (Pollard 2009).
3.5 Conclusions
Despite important efforts in the study of the dawn chorus in the last few decades, this review shows that there is still a good degree of confusion, and that much work is still needed. Four out of the nine hypotheses proposed to explain this phenomenon have not been empirically tested, while others present equivocal support as general theories. In Table 3.1, we have listed the key findings for each hypothesis, as well as our personal evaluation of the evidence supporting them and specific suggestions for further research.
In spite of this plethora of explanations, most hypotheses are not incompatible with each other. Their explanatory value changes depending on the species, and at any rate we should not expect a single explanation (Kacelnik and Krebs 1983; Mace 1987b). Although there might be common patterns in dawn song across bird species, the double function of male song (male vs. female receivers) is likely to be responsible for the contrasting patterns of dawn choruses of (for instance) great tits (Mace 1987a) and chipping sparrows (Liu and Kroodsma 2007) (Fig. 3.3). More importantly, the function of song may change across the season, and thus we can expect different functions at different times of the reproductive cycle for the same species.
Moreover, there is an interesting historical distinction among these hypotheses. Many of them arose from an optimality standpoint, and postulated that early singing is selected for reasons that should benefit all signalling males (low predation, good sound transmission, use of spare time and energy, etc.). In contrast, the handicap hypothesis proposed a distinct view, suggesting that singing early is costly and reflects male quality. This implies a shift from optimality to costliness. These two views are not incompatible, since sexual selection can permeate processes of signal production, modifying optimal patterns and shaping individual variation. In the case of bird song, we expect that whatever general selective advantage is accrued by singing early, between-individual variation in performance based on condition can provide a way to assess male quality for individuals of both sexes (Gil and Gahr 2002). In other words, while some hypotheses explain early singing, the handicap hypothesis aims to explain earlier-than-average singing.
Our personal view is that the best-supported hypotheses for early singing can be gathered around three complementary lines of explanation. Firstly, energetic constraints (inefficient foraging at dawn and unpredictable overnight conditions—hypotheses 6 and 7) allow a window of time in which singing has a low cost. Secondly, and depending on the species, it pays males to sing early to manipulate female mating (hypothesis 3) or settle territory boundaries and assert ownership (hypothesis 4). And, thirdly, costs of early singing (hypothesis 5) should push song timing earlier than optimal through a handicap mechanism that prevents dishonest signalling (but see Hutchinson et al. 1993).
One interesting possibility is that a main selective pressure on dawn singing is synchronicity in signalling. This would not explain why it pays to sing at dawn, but rather that there is strong selection for signalling in synchrony. Synchronous signalling results in a dilution of predation risk and a decrease in invasion rate (Kacelnik and Krebs 1983), allows ritualization and comparison among signallers (West-Eberhard 1979; Zahavi 1980), as well as favours the development of useful communication networks (Burt and Vehrencamp 2005; McGregor 2005; Mennill et al. 2003). We believe that this possibility has not been sufficiently explored.
In the near future, the development of new techniques for experimentally manipulating hormone levels, as well as remote-sensing devices for assessing singing performance (Sugai et al. 2019) or studies integrating singing with spatial movements of conspecifics (Amy et al. 2010), may offer suitable methods to test hypotheses that are still poorly explored (e.g., circadian cycles and handicap hypotheses). Further studies will also require detailed data at the individual scale and comparative approaches, which can provide more precision and a more relevant baseline to disentangle factors determining species- and population-specific patterns.
Ernst Mayr (1997) concluded that most, if not all, biological processes are probabilistic, the result of several factors with a high degree of randomness. This pluralism naturally leads to multiple hypotheses being correct at the same time, often including the traditional bimodal perspective in biology (i.e. proximal vs. evolutionary explanations). We believe that research on the bird dawn chorus represents a good example of this pluralism of factors and illustrates how several hypotheses can coexist simultaneously. We hope that this review may help future research to refine our knowledge of this puzzling phenomenon.
References
Allard HA (1930) The first morning song of some birds of Washington, DC: its relation to light. Am Nat 64:436–469
Allen FH (1913) More notes on the morning awakening. Auk 30:229–235
Alward BA, Cornil CA, Balthazart J, Ball GF (2018) The regulation of birdsong by testosterone: multiple time-scales and multiple sites of action. Horm Behav 104:32–40
Amrhein V, Erne N (2006) Dawn singing reflects past territorial challenges in the winter wren. Anim Behav 71:1075–1080
Amrhein V, Lerch S (2010) Differential effects of moving versus stationary territorial intruders on territory defence in a songbird. J Anim Ecol 79:82–87
Amrhein V, Korner P, Naguib M (2002) Nocturnal and diurnal singing activity in the nightingale: correlations with mating status and breeding cycle. Anim Behav 64:939–944
Amrhein V, Kunc HP, Naguib M (2004a) Non-territorial nightingales prospect territories during the dawn chorus. Proc R Soc B 271:S167–S169
Amrhein V, Kunc HP, Naguib M (2004b) Seasonal patterns of singing activity vary with time of day in the nightingale (Luscinia megarhynchos). Auk 121:110–117
Amrhein V, Johannessen LE, Kristiansen L, Slagsvold T (2008) Reproductive strategy and singing activity: blue tit and great tit compared. Behav Ecol Sociobiol 62:1633–1641
Amy M, Sprau P, de Goede P, Naguib M (2010) Effects of personality on territory defence in communication networks: a playback experiment with radio-tagged great tits. Proc R Soc B 277:3685–3692
Armstrong EA (1963) A study of bird song. Oxford University Press, London
Arroyo-Solis A, Castillo JM, Figueroa E, Lopez-Sanchez JL, Slabbekoorn H (2013) Experimental evidence for an impact of anthropogenic noise on dawn chorus timing in urban birds. J Avian Biol 44:288–296
Aschoff J (1966) Circadian activity pattern with two peaks. Ecology 47:657–662
Aschoff J, Meyer-Lohmann J (1955) Die Aktivität gekäfigter Grünfinken im 24-Stunden-Tag bei unterschiedlich langer Lichtzeit mit und ohne Dämmerung. Z Tierpsychol 12:254–265
Atienza JC, Illera JC (1997) Tree species selection to perform singing and foraging behaviour by Great and Blue Tits: a trade-off between food gathering and territorial behaviour? Bird Study 44:117–119
Au WW, Mobley J, Burgess WC, Lammers MO, Nachtigall PE (2000) Seasonal and diurnal trends of chorusing humpback whales wintering in waters off western Maui. Mar Mamm Sci 16:530–544
Avery MI, Krebs JR (1984) Temperature and foraging success of great tits (Parus major) hunting for spiders. Ibis 126:33–38
Ballentine B, Hyman J, Nowicki S (2004) Vocal performance influences female response to male bird song: an experimental test. Behav Ecol 15:163–168
Barnett CA, Briskie JV (2007) Energetic state and the performance of dawn chorus in silvereyes (Zosterops lateralis). Behav Ecol Sociobiol 61:579–587
Beaulieu M, Sockman KW (2012) Song in the cold is ‘hot’: memory of and preference for sexual signals perceived under thermal challenge. Biol Lett 8:751–753
Belinsky KL, Hogle JL, Schmidt KA (2012) Veeries experience more varied acoustic competition at dawn than at dusk. Wilson J Ornithol 124:265–269
Bentley GE (2001) Unraveling the enigma: the role of melatonin in seasonal processes in birds. Microsc Res Tech 53:63–71
Berg KS, Brumfield RT, Apanius V (2006) Phylogenetic and ecological determinants of the neotropical dawn chorus. Proc R Soc B 273:999–1005
Bergen F, Abs M (1997) Verhaltensökologie Studie zur Gesangsaktivität von Blaumeise (Parus caeruleus), Kohlmeise (Parus major) und Buchfink (Fringilla coelebs) in einer Grosstadt. J Ornithol 138:451–467
Berglund A, Bisazza A, Pilastro A (1996) Armaments and ornaments: an evolutionary explanation of traits of dual utility. Biol J Linn Soc 58:385–399
Berthold P (1973) Relationships between migratory restlessness and migration distance in six Sylvia species. Ibis 115:594–599
Biard C, Gil D, Karadas F, Saino N, Spottiswoode CN, Surai PF, Moller AP (2009) Maternal effects mediated by antioxidants and the evolution of carotenoid-based signals in birds. Am Nat 174:696–708
Biernaskie JM, Perry JC, Grafen A (2018) A general model of biological signals, from cues to handicaps. Evol Lett 2:201–209
Birkhead TR, Møller AP (1992) Sperm competition in birds: evolutionary causes and consequences. Academic, London
Birkhead TR, Møller AP (1998) Sperm competition and sexual selection. Academic, London
Birkhead TR, Cunningham EJA, Cheng KM (1996) The insemination window provides a distorted view of sperm competition in birds. Proc R Soc B 263:1187–1192
Bolsinger JS (2000) Use of two song categories by golden-cheeked Warblers. Condor 102:539–552
Brandt MJ, Cresswell W (2009) Diurnal foraging routines in a tropical bird, the rock finch Lagonosticta sanguinodorsalis: how important is predation risk? J Avian Biol 40:90–94
Brenowitz EA (1982) The active space of red-winged blackbird song. J Comp Physiol 147:511–522
Brockway BF (1967) The influence of vocal behavior on the performer’s testicular activity in budgerigars (Melopsittacus undulatus). Wilson Bull 79:328–334
Brown TJ, Handford P (2003) Why birds sing at dawn: the role of consistent song transmission. Ibis 145:120–129
Brumm H (2006) Signalling through acoustic windows: nightingales avoid interspecific competition by short-term adjustment of song timing. J Comp Physiol A 192:1279–1285
Bruni A, Foote JR (2014) Dawn singing of eastern phoebes varies with breeding stage and brood number. Wilson J Ornithol 126:500–507
Bruni A, Mennill DJ, Foote JR (2014) Dawn chorus start time variation in a temperate bird community: relationships with seasonality, weather, and ambient light. J Ornithol 155:877–890
Brunner P, Pasinelli G (2010) Variation in singing style use in the reed bunting Emberiza schoeniclus: influencing factors and possible functions. J Avian Biol 41:388–397
Burt JM, Vehrencamp SL (2005) Dawn chorus as an interactive communication network. In: McGregor PK (ed) Animal communication networks. Cambridge University Press, Cambridge, pp 320–343
Byers BE (1996) Messages encoded in the songs of chestnut-sided warblers. Anim Behav 52:691–705
Cassone VM, Westneat DF (2012) The bird of time: cognition and the avian biological clock. Front Mol Neurosci 5:32
Cassone VM, Bartell PA, Earnest BJ, Kumar V (2008) Duration of melatonin regulates seasonal changes in song control nuclei of the House Sparrow, Passer domesticus: independence from gonads and circadian entrainment. J Biol Rhythms 23:49–58
Catchpole CK (1973) The functions of advertising song in the sedge warbler (Acrocephalus schoenobaenus) and the reed warbler (A. scirpaceus). Behaviour 46:300–319
Catchpole CK (1982) The evolution of bird sounds in relation to mating and spacing behaviour. In: Kroodsma DE, Miller EH (eds) Acoustic communication in birds. Vol 1. Production, perception and design features of sounds. Academic, New York, pp 253–295
Catchpole CK, Slater PJB (2008) Bird song: biological themes and variations, 2nd edn. Cambridge University Press, Cambridge
Chen W-M, Lee Y-F, Tsai C-F, Yao C-T, Chen Y-H, Li S-H, Kuo Y-M (2015) Dawn chorus variation in East-Asian tropical montane forest birds and its ecological and morphological correlates. Contrib Zool 84:255–265
Cheng M-F (1992) For whom does the female dove coo? A case for the role of vocal self-stimulation. Anim Behav 43:1035–1044
Cirelli C, Tononi G (2008) Is sleep essential? PLoS Biol 6:e216
Cody ML, Brown JH (1969) Song asynchrony in neighbouring bird species. Nature 222:778
Collias NE, Collias EC (1967) A field study of the red jungle fowl in north-central India. Condor 69:360–386
Collins SA, de Kort SR, Perez-Tris J, Telleria JL (2009) Migration strategy and divergent sexual selection on bird song. Proc R Soc B 276:585–590
Cotton S, Fowler K, Pomiankowski A (2004) Do sexual ornaments demonstrate heightened condition-dependent expression as predicted by the handicap hypothesis? Proc R Soc B 271:771–783
Cresswell W (1994) Song as a pursuit deterrent signal, and its occurrence relative to other anti-predation behaviors of skylark (Alauda arvensis) on attack by merlins (Falco columbarius). Behav Ecol Sociobiol 34:217–223
Cunningham EJA, Russell AF (2000) Egg investment is influenced by male attractiveness in the mallard. Nature 404:74–77
Curio E (1959) Verhaltensstudien am Trauerschnäpper: Beiträge zur Ethologie und Ökologie von Musicapa h. hypoleuca Pallas. Z Tierpsychol (Beiheft) 3:1–118
Cuthill IC, Macdonald WA (1990) Experimental manipulation of the dawn and dusk chorus in the blackbird Turdus merula. Behav Ecol Sociobiol 26:209–216
D’Spain G, Batchelor H (2006) Observations of biological choruses in the Southern California Bight: a chorus at midfrequencies. J Acoust Soc Am 120:1942–1955
Da Silva A, Kempenaers B (2017) Singing from North to South: latitudinal variation in timing of dawn singing under natural and artificial light conditions. J Anim Ecol 86:1286–1297
Da Silva A, Samplonius JM, Schlicht E, Valcu M, Kempenaers B (2014) Artificial night lighting rather than traffic noise affects the daily timing of dawn and dusk singing in common European songbirds. Behav Ecol 25:1037–1047
Da Silva A, Valcu M, Kempenaers B (2015) Light pollution alters the phenology of dawn and dusk singing in common European songbirds. Philos Trans R Soc Lond B Biol Sci 20140126:370
Da Silva A, Valcu M, Kempenaers B (2016) Behavioural plasticity in the onset of dawn song under intermittent experimental night lighting. Anim Behav 117:155–165
Da Silva A, de Jong M, van Grunsven RHA, Visser ME, Kempenaers B, Spoelstra K (2017) Experimental illumination of a forest: no effects of lights of different colours on the onset of the dawn chorus in songbirds. R Soc Open Sci 4:16063
Daan S, Aschoff J (1975) Circadian rhythms of locomotor activity in captive birds and mammals: their variations with season and latitude. Oecologia 18:269–316
Dabelsteen T (1992) Interactive playback: a finely tuned response. In: McGregor PK (ed) Playback and studies of animal communication. Springer, New York, pp 97–110
Dabelsteen T, Mathevon N (2002) Why do songbirds sing intensively at dawn? Acta Ethol 4:65–72
Dalziell AH, Cockburn A (2008) Dawn song in superb fairy-wrens: a bird that seeks extrapair copulations during the dawn chorus. Anim Behav 75:489–500
Demko AD, Reitsma LR, Staicer CA (2013) Two song categories in the canada warbler (Cardellina canadensis). Auk 130:609–616
Doggett F (1974) Romanticism’s singing bird. Stud Engl Lit 1500–1900(14):547–561
Dolan AC, Murphy MT, Redmond LJ, Sexton K, Duffield D (2007) Extrapair paternity and the opportunity for sexual selection in a socially monogamous passerine. Behav Ecol 18:985–993
Dominoni DM, Goymann W, Helm B, Partecke J (2013a) Urban-like night illumination reduces melatonin release in European blackbirds (Turdus merula): implications of city life for biological time-keeping of songbirds. Front Zool 10:1–10
Dominoni DM, Helm B, Lehmann M, Dowse HB, Partecke J (2013b) Clocks for the city: circadian differences between forest and city songbirds. Proc R Soc B 280:20130593
Dominoni DM, Greif S, Nemeth E, Brumm H (2016) Airport noise predicts song timing of European birds. Ecol Evol 6:6151–6159
Dorado-Correa AM, Rodriguez-Rocha M, Brumm H (2016) Anthropogenic noise, but not artificial light levels predicts song behaviour in an equatorial bird. R Soc Open Sci 3:160231–160231
Double M, Cockburn A (2000) Pre-dawn infidelity: females control extra-pair mating in superb fairy-wrens. Proc R Soc B 267:465–470
Drachmann J, Komdeur J, Boomsma JJ (1997) Copulation behaviour in the linnet Carduelis cannabina and the insemination window hypothesis. J Avian Biol 28:191–196
Dunnett GE, Hinde RA (1953) The winter roosting and awakening behaviour of captive Great Tits. Br J Anim Behav 1:91–95
Eriksson D, Wallin L (1986) Male bird song attracts females: a field experiment. Behav Ecol Sociobiol 19:297–299
Erne N, Amrhein V (2008) Long-term influence of simulated territorial intrusions on dawn and dusk singing in the Winter Wren: spring versus autumn. J Ornithol 149:479–486
Falk H, Biebach H, Krebs JR (1992) Learning a time-place pattern of food availability: a comparison between an insectivorous and a granivorous weaver species (Ploceus bicolor and Euplectes hordeaceus). Behav Ecol Sociobiol 31:9–15
Falls JB (1988) Does song deter territorial intrusion in White-throated Sparrows (Zonotrichia albicollis)? Can J Zool 66:206–211
Farina A, Ceraulo M, Bobryk C, Pieretti N, Quinci E, Lattanzi E (2015) Spatial and temporal variation of bird dawn chorus and successive acoustic morning activity in a Mediterranean landscape. Bioacoustics 24:269–288
Fernandez-Juricic E, Tran E (2007) Changes in vigilance and foraging behaviour with light intensity and their effects on food intake and predator detection in house finches. Anim Behav 74:1381–1390
Fisler GF (1962) Variation in the morning awakening time of some birds in south-central Michigan. Condor 64:184–198
Foerster K, Poesel A, Kunc H, Kempenaers B (2002) The natural plasma testosterone profile of male blue tits during the breeding season and its relation to song output. J Avian Biol 33:269–275
Foote JR, Fitzsimmons LP, Mennill DJ, Ratcliffe LM (2008a) Male chickadees match neighbors interactively at dawn: support for the social dynamics hypothesis. Behav Ecol 19:1192–1199
Foote JR, Fitzsimmons LP, Mennill DJ, Ratcliffe LM (2008b) Tied to the nest: male black-capped chickadees decrease dawn chorus movement behaviour when their mate is fertile. Anim Behav 76:1227–1233
Foote JR, Fitzsimmons LP, Mennill DJ, Ratcliffe LM (2010) Black-capped chickadee dawn choruses are interactive communication networks. Behaviour 147:1219–1248
Foote JR, Fitzsimmons LP, Mennill DJ, Ratcliffe LM (2011) Male black-capped chickadees begin dawn chorusing earlier in response to simulated territorial insertions. Anim Behav 81:871–877
Forstmeier W, Balsby TJ (2002) Why mated dusky warblers sing so much: territory guarding and male quality announcement. Behaviour 139:89–111
Fuller RA, Warren PH, Gaston KJ (2007) Daytime noise predicts nocturnal singing in urban robins. Biol Lett 3:368–370
Garamszegi LZ, Moller AP, Erritzoe J (2002) Coevolving avian eye size and brain size in relation to prey capture and nocturnality. Proc R Soc B 269:961–967
Garson PJ, Hunter ML (1979) Effects of temperature and time of year on the singing behaviour of wrens Troglodytes troglodytes and great tits Parus major. Ibis 121:481–487
Gaunt AS, Bucher TL, Gaunt SLL, Baptista LF (1996) Is singing costly? Auk 113:718–721
Gil D (2008) Hormones in avian eggs: physiology, ecology and behavior. Adv Stud Behav 38:337–398
Gil D, Gahr M (2002) The honesty of bird song: multiple constraints for multiple traits. Trends Ecol Evol 17:133–141
Gil D, Graves JA, Hazon N, Wells A (1999a) Male attractiveness and differential testosterone investment in zebra finch eggs. Science 286:126–128
Gil D, Graves JA, Slater PJB (1999b) Seasonal patterns of singing in the willow warbler: evidence against the fertility announcement hypothesis. Anim Behav 58:995–1000
Gil D, Leboucher G, Lacroix A, Cue R, Kreutzer M (2004) Female canaries produce eggs with greater amounts of testosterone when exposed to preferred male song. Horm Behav 45:64–70
Gil D, Honarmand M, Pascual J, Pérez-Mena E, Macías-García C (2015) Birds living near airports advance their dawn-chorus and reduce overlap with aircraft noise. Behav Ecol 26:435–443
Grafen A (1990) Biological signals as handicaps. J Theor Biol 144:517–546
Grava T, Grava A, Otter KA (2009) Supplemental feeding and dawn singing in black-capped chickadees. Condor 111:560–564
Green AJ (1990) Determinants of chorus participation and the effects of size, weight and competition on advertisement calling in the tungara frog, Physalaemus pustulosus (Leptodactylidae). Anim Behav 39:620–638
Greenfield MD (2015) Signal interactions and interference in insect choruses: singing and listening in the social environment. J Comp Physiol A 201:143–154
Greig-Smith P (1983) Use of perches as vantage points during foraging by male and female stonechats Saxicola torquata. Behaviour 86:215–236
Greives TJ, Kingma SA, Beltrami G, Hau M (2012) Melatonin delays clutch initiation in a wild songbird. Biol Lett 8:330–332
Greives TJ et al (2015) Costs of sleeping in: circadian rhythms influence cuckoldry risk in a songbird. Funct Ecol 29:1300–1307
Gwinner E, Brandstatter R (2001) Complex bird clocks. Philos Trans R Soc Lond B Biol Sci 356:1801–1810
Haldar C, Singh SS (2001) Seasonal changes in melatonin and immunological adaptations in birds. J Endocrinol Reprod 5:13–24
Halfwerk W, Bot S, Buikx J, van der Velde M, Komdeur J, ten Cate C, Slabbekoorn H (2011) Low-frequency songs lose their potency in noisy urban conditions. Proc Natl Acad Sci U S A 108:14549–14554
Hamilton WD (1971) Geometry for the selfish herd. J Theor Biol 31:295–311
Hart PJ, Hall R, Ray W, Beck A, Zook J (2015) Cicadas impact bird communication in a noisy tropical rainforest. Behav Ecol 26:839–842
Hasan NM (2011) Effect of seasonal variations, altitude and geographical location on the onset of dawn chorus in three bird species in the Middle East. Open Ornithol J 4:30–34
Haskell D (1994) Experimental evidence that nestling begging behaviour incurs a cost due to nest predation. Proc R Soc B 257:161–164
Hau M (2007) Regulation of male traits by testosterone: implications for the evolution of vertebrate life histories. Bioessays 29:133–144
Hau M et al (2017) Timing as a sexually selected trait: the right mate at the right moment. Philos Trans R Soc Lond B Biol Sci 372:20160249
Helm B, Visser ME (2010) Heritable circadian period length in a wild bird population. Proc R Soc B 277:3335–3342
Henwood K, Fabrick A (1979) Quantitative-analysis of the dawn chorus: temporal selection for communicatory optimization. Am Nat 114:260–274
Hodgson L, Waas JR, Foote JR (2018) Early singers attend to conspecific but not heterospecific behavioural cues at dawn. J Avian Biol 49:e01749
Hutchinson JMC (2002) Two explanations of the dawn chorus compared: how monotonically changing light levels favour a short break from singing. Anim Behav 64:527–539
Hutchinson JMC, McNamara JM (2000) Ways to test stochastic dynamic programming models empirically. Anim Behav 59:665–676
Hutchinson JMC, McNamara JM, Cuthill IC (1993) Song, sexual selection, starvation and strategic handicaps. Anim Behav 45:1153–1177
Kacelnik A (1979) Foraging efficiency of great tits (Parus major L.) in relation to light-intensity. Anim Behav 27:237–241
Kacelnik A, Krebs JR (1983) The dawn chorus in the great tit (Parus major): proximate and ultimate causes. Behaviour 83:287–309
Karasov WH (1990) Digestion in birds: chemical and physiological determinants and ecological implications. In: Morrison ML, Ralph CJ, Verner J, Jehl JR (eds) Avian foraging: theory, methodology, and applications. Studies in avian biology, vol 13. Cooper Ornithological Society, Lawrence, pp 391–415
Keast A (1994a) The dawn chorus in a eucalypt forest bird community, seasonal shifts in timing and contribution of individual species. Corella 18:133–140
Keast A (1994b) Temporal vocalization patterns in members of a eucalypt forest bird community: the effects of weather on song production. Emu 94:172–180
Kempenaers B, Borgstroem P, Loes P, Schlicht E, Valcu M (2010) Artificial night lighting affects dawn song, etra-pair siring success, and lay date in songbirds. Curr Biol 20:1735–1739
Kirschel ANG, Blumstein DT, Cohen RE, Buermann W, Smith TB, Slabbekoorn H (2009) Birdsong tuned to the environment: green hylia song varies with elevation, tree cover, and noise. Behav Ecol 20:1089–1095
Klett-Mingo JI, Pavón I, Gil D (2016) Great tits (Parus major) increase vigilance time and reduce feeding effort during peaks of aircraft noise. Anim Behav 115:29–34
Klump GM (1996) Bird communication in the noisy world. In: Kroodsma DE, Miller EH (eds) Ecology and evolution of acoustic communication in birds. Cornell University, Ithaca, pp 321–338
Kokko H, Jennions MD, Brooks R (2006) Unifying and testing models of sexual selection. Annu Rev Ecol Evol Syst 37:43–66
Krams I (2001) Communication in crested tits and the risk of predation. Anim Behav 61:1065–1068
Krebs JR, Ashcroft R, Webber M (1978) Song repertoires and territory defence in the Great Tit. Nature 271:539–542
Kroodsma DE, Byers BE (1991) The function(s) of bird song. Am Zool 31:318–328
Kroodsma DE, Bereson RC, Byers BE, Minear E (1989) Use of song types by the Chestnut-sided Warbler: evidence for both intra- and inter-sexual functions. Can J Zool 67:447–456
Kunc HP, Amrhein V, Naguib M (2005) Seasonal variation in dawn song characteristics in the common nightingale. Anim Behav 70:1265–1271
Kunc HP, Foerster K, Vermeirssen ELM, Kempenaers B (2006) Experimentally elevated plasma testosterone levels do not influence singing behaviour of male blue tits (Parus caeruleus) during the early breeding season. Ethology 112:984–992
Lambrechts M, Dhondt AA (1986) Male quality, reproduction, and survival in the great tit (Parus major). Behav Ecol Sociobiol 19:57–63
Langemann U, Gauger B, Klump GM (1998) Auditory sensitivity in the great tit: perception of signals in the presence and absence of noise. Anim Behav 56:763–769
Langmore NE (1998) Functions of duet and solo songs of female birds. Trends Ecol Evol 13:136–140
Larom D, Garstang M, Payne K, Raspet R, Lindeque M (1997) The influence of surface atmospheric conditions on the range and area reached by animal vocalizations. J Exp Biol 200:421–431
Laucht S, Dale J, Mutzel A, Kempenaers B (2011) Individual variation in plasma testosterone levels and its relation to badge size in house sparrows Passer domesticus: it’s a night-and-day difference. Gen Comp Endocrinol 170:501–508
Lein MR (2007) Patterns of dawn singing by Buff-breasted Flycatchers. J Field Ornithol 78:343–351
Leopold A, Eynon AE (1961) Avian daybreak and evening song in relation to light intensity. Condor 63:269–293
Lesku JA et al (2012) Adaptive sleep loss in polygynous pectoral sandpipers. Science 337:1654–1658
Lima SL (2009) Predators and the breeding bird: behavioral and reproductive flexibility under the risk of predation. Biol Rev 84:485–513
Liu W-C (2004) The effect of neighbours and females on dawn and daytime singing behaviours by male chipping sparrows. Anim Behav 68:39–44
Liu W-C, Kroodsma DE (2007) Dawn and daytime singing behavior of chipping sparrows (Spizella passerina). Auk 124:44–52
Lourenço R, Goytre F, del Mar Delgado M, Thornton M, Rabaça JE, Penteriani V (2013) Tawny owl vocal activity is constrained by predation risk. J Avian Biol 44:461–468
Luther DA (2008) Signaller: receiver coordination and the timing of communication in Amazonian birds. Biol Lett 4:651–654
Luther D (2009) The influence of the acoustic community on songs of birds in a neotropical rain forest. Behav Ecol 20:864–871
Mace R (1986) Importance of female behavior in the dawn chorus. Anim Behav 34:621–622
Mace R (1987a) The dawn chorus in the great tit Parus major is directly related to female fertility. Nature 330:745–746
Mace R (1987b) Why do birds sing at dawn? Ardea 75:123–132
Mace R (1989) The relationship between daily routines of singing and foraging: an experiment on captive great tits Parus major. Ibis 131:415–420
Martin GR (1993) Producing the image. In: Zeiger HP, Bischof HJ (eds) Vision, brain and behaviour in birds. MIT Press, Cambridge, pp 5–24
Martínez-Ortega C, Santos ESA, Gil D (2014) Species-specific differences in relative eye size are related to patterns of edge avoidance in an Amazonian rainforest bird community. Ecol Evol 19:3736–3745
Mayr E (1997) This is biology: the science of the living world. Cambridge University Press, Cambridge
McCabe RA (1951) The song and song-flight of the Alder Flycatcher. Wilson Bull 63:89–98
McDonald MV (1989) Function of song in Scott’s seaside sparrow Ammodramus maritimus peninsulae. Anim Behav 38:468–485
McGregor PK (2005) Animal communication networks. Cambridge University Press, Cambridge
McNamara JM, Mace RH, Houston AI (1987) Optimal daily routines of singing and foraging in a bird singing to attract a mate. Behav Ecol Sociobiol 20:399–405
Mennill DJ, Boag PT, Ratcliffe LM (2003) The reproductive choices of eavesdropping female black-capped chickadees, Poecile atricapillus. Naturwissenschaften 90:577–582
Miller MW (2006) Apparent effects of light pollution on singing behavior of American robins. Condor 108:130–139
Møller AP (1988) Spatial and temporal distribution of song in the yellowhammer Emberiza citrinella. Ethology 78:321–331
Møller AP (1990) Changes in the size of avian breeding territories in relation to the nesting cycle. Anim Behav 40:1070–1079
Møller AP (1991) Why mated songbirds sing so much: mate guarding and male announcement of mate fertility status. Am Nat 138:994–1014
Møller AP (1992) Interspecific response to playback of bird song. Ethology 90:315–320
Møller AP (2011) Song post height in relation to predator diversity and urbanization. Ethology 117:529–538
Møller AP, Erritzøe J (2010) Flight distance and eye size in birds. Ethology 116:458–465
Montgomerie RD (1985) Why do birds sing at dawn? In: Proceedings of the 19th International Ethol Conference, p 242
Morse DH (1989) Song patterns of warblers at dawn and dusk. Wilson Bull 101:26–35
Morton ES (1975) Ecological sources of selection on avian sounds. Am Nat 109:17–34
Mountjoy DJ, Lemon RE (1991) Song as an attractant for male and female European starlings, and the influence of song complexity on their response. Behav Ecol Sociobiol 28:97–100
Murphy MT, Sexton K, Dolan AC, Redmond LJ (2008) Dawn song of the eastern kingbird: an honest signal of male quality? Anim Behav 75:1075–1084
Naguib M, Fichtel C, Todt D (1999) Nightingales respond more strongly to vocal leaders of simulated dyadic interactions. Proc Biol Sci 266:537–543
Naguib M, van Rooij EP, Snijders L, Van Oers K (2016) To sing or not to sing: seasonal changes in singing vary with personality in wild great tits. Behav Ecol 27:932–938
Needham KB, Dochtermann NA, Greives TJ (2017) Consistent individual variation in day, night, and GnRH-induced testosterone concentrations in house sparrows (Passer domesticus). Gen Comp Endocrinol 246:211–217
Nelson DA, Croner LJ (1991) Song categories and their functions in the field sparrow (Spizella pusilla). Auk 108:42–52
Nordt A, Klenke R (2013) Sleepless in town: drivers of the temporal shift in dawn song in urban European blackbirds. PLoS One 8:e71476
Odom KJ, Hall ML, Riebel K, Omland KE, Langmore NE (2014) Female song is widespread and ancestral in songbirds. Nat Commun 5:3379
Ord TJ (2008) Dawn and dusk “chorus” in visually communicating Jamaican anole lizards. Am Nat 172:585–592
Otter K, Ratcliffe L (1993) Changes in singing behavior of male black-capped chickadees (Parus atricapillus) following mate removal. Behav Ecol Sociobiol 33:409–414
Otter K, Chruszcz B, Ratcliffe L (1997) Honest advertisement and song output during the dawn chorus of black-capped chickadees. Behav Ecol 8:167–173
Ottinger MA, Follett BK (1987) Circulating levels of luteinizing hormone and androgen in mature male Japanese Quail over 24 hours. Poult Sci 66:1411–1413
Parker TH, Tillin H (2006) Male Blue Tits Cyanistes caeruleus choose early-leafing tree species during spring dawn chorus. Bird Study 53:253–257
Pärt T (1991) Is dawn singing related to paternity insurance: the case of the collared flycatcher. Anim Behav 41:451–456
Peters J, Lebrasseur O, Deng H, Larson G (2016) Holocene cultural history of Red jungle fowl (Gallus gallus) and its domestic descendant in East Asia. Quat Sci Rev 142:102–119
Phelps SM, Rand AS, Ryan MJ (2006) The mixed-species chorus as public information: túngara frogs eavesdrop on a heterospecific. Behav Ecol 18:108–114
Pinxten R, Eens M (1998) Male starlings sing most in the late morning, following egg-laying: a strategy to protect their paternity? Behaviour 135:1197–1211
Planqué R, Slabbekoorn H (2008) Spectral overlap in songs and temporal avoidance in a Peruvian bird assemblage. Ethology 114:262–271
Poesel A, Foerster K, Kempenaers B (2001) The dawn song of the blue tit Parus caeruleus and its role in sexual selection. Ethology 107:521–531
Poesel A, Kunc HP, Foerster K, Johnsen A, Kempenaers B (2006) Early birds are sexy: male age, dawn song and extrapair paternity in blue tits, Cyanistes (formerly Parus) caeruleus. Anim Behav 72:531–538
Poesel A, Dabelsteen T, Pedersen SB (2007) Implications of conspecific background noise for features of blue tit, Cyanistes caeruleus, communication networks at dawn. J Ornithol 148:123–128
Pohl NU, Slabbekoorn H, Klump GM, Langemann U (2009) Effects of signal features and environmental noise on signal detection in the great tit, Parus major. Anim Behav 78:1293–1300
Pollard AL (2009) Visual constraints upon avian behaviour. PhD thesis. School of Biosciences, Cardiff University
Popp JW, Ficken RW, Reinartz JA (1985) Short-term temporal avoidance of interspecific acoustic interference among forest birds. Auk 102:744–748
Quispe R, Sèbe F, Da Silva ML, Gahr M (2016) Dawn-song onset coincides with increased HVC androgen receptor expression but is decoupled from high circulating testosterone in an equatorial songbird. Physiol Behav 156:1–7
Rattner BA, Sileo L, Scanes CG (1982) Oviposition and the plasma concentrations of LH, progesterone and corticosterone in bobwhite quail (Colinus virginianus) fed parathion. J Reprod Fertil 66:147–155
Refinetti R (2016) Circadian physiology. CRC, Boca Raton
Richards DG, Wiley RH (1980) Reverberations and amplitude fluctuations in the propagation of sound in a forest: implications for animal communication. Am Nat 115:381–399
Riebel K, Hall ML, Langmore NE (2005) Female songbirds still struggling to be heard. Trends Ecol Evol 20:419–420
Rodrigues M (1996) Song activity in the chiffchaff: territorial defence or mate guarding? Anim Behav 51:709–716
Rodríguez AB, Terrón MP, Durán J, Ortega E, Barriga C (2001) Physiological concentrations of melatonin and corticosterone affect phagocytosis and oxidative metabolism of ring dove heterophils. J Pineal Res 31:31–38
Roth TC, Lesku JA, Amlaner CJ, Lima SL (2006) A phylogenetic analysis of the correlates of sleep in birds. J Sleep Res 15:395–402
Roth T, Sprau P, Schmidt R, Naguib M, Amrhein V (2009) Sex-specific timing of mate searching and territory prospecting in the nightingale: nocturnal life of females. Proc R Soc B 276:2045–2050
Roth T, Sprau P, Naguib M, Amrhein V (2012) Sexually selected signaling in birds: a case for Bayesian change-point analysis of behavioral routines. Auk 129:660–669
Rusak B (2013) Vertebrate behavioral rhythms. In: Aschoff J (ed) Biological rhythms. Plenum, New York
Russ A, Rueger A, Klenke R (2015) Seize the night: European Blackbirds (Turdus merula) extend their foraging activity under artificial illumination. J Ornithol 156:123–131
Saggese K, Korner-Nievergelt F, Slagsvold T, Amrhein V (2011) Wild bird feeding delays start of dawn singing in the great tit. Anim Behav 81:361–365
Scheer G (1951) Uber die zeitliche Differenz zwischen Erwachen und Gesangsbeginn. Die Vogelwarte 16:13–15
Scheer G (1952) Beobachtungen und Untersuchungen über die Abhangigkeit des Frühgesangs der Vogel von inneren und äußeren Faktoren. Biol Abhandlungen 3(4):1–68
Schmidt KA, Belinsky KL (2013) Voices in the dark: predation risk by owls influences dusk singing in a diurnal passerine. Behav Ecol Sociobiol 67:1837–1843
Seltmann S, Trost L, Ter Maat A, Gahr M (2016) Natural melatonin fluctuation and its minimally invasive simulation in the zebra finch. Peer J 4:e1939
Sexton K, Murphy MT, Redmond LJ, Dolan AC (2007) Dawn song of eastern kingbirds: intrapopulation variability and sociobiological correlates. Behaviour 144:1273–1295
Shakespeare W (1597) An excellent conceited tragedie of Romeo and Juliet. John Danter, London
Sheldon BC, Burke T (1994) Copulation behavior and paternity in the chaffinch. Behav Ecol Sociobiol 1994:149–156
Shimmura T, Yoshimura T (2013) Circadian clock determines the timing of rooster crowing. Curr Biol 23:R231–R233
Shimmura T, Ohashi S, Yoshimura T (2015) The highest-ranking rooster has priority to announce the break of dawn. Sci Rep 5:11683
Siegel JM (2009) Sleep viewed as a state of adaptive inactivity. Nat Rev Neurosci 10:747
Sierro J, Schloesing E, Pavón I, Gil D (2017) European blackbirds exposed to aircraft noise advance their chorus, modify their song and spend more time singing. Front Ecol Evol 5:68
Slagsvold T (1977) Bird song activity in relation to breeding cycle, spring weather, and environmental phenology. Ornis Scand 8:197–222
Slagsvold T (1996) Dawn and dusk singing of male American Robins in relation to female behavior. Wilson Bull 108:507–515
Snijders L, van Rooij EP, Henskens MFA, van Oers K, Naguib M (2015) Dawn song predicts behaviour during territory conflicts in personality-typed great tits. Anim Behav 109:45–52
Spector DA (1992) Wood-warbler song systems. In: Power DM (ed) Current Ornithology, vol 9. Springer, Boston, MA, pp 199–238
Staicer CE, Spector DA, Horn AG (1996) The dawn chorus and other diel patterns in acoustic signalling. In: Kroodsma DE, Miller EH (eds) Ecology and evolution of acoustic communication in birds. Cornell University Press, Ithaca, pp 426–453
Stanley CQ, Walter MH, Venkatraman MX, Wilkinson GS (2016) Insect noise avoidance in the dawn chorus of Neotropical birds. Anim Behav 112:255–265
Steinmeyer C, Schielzeth H, Mueller JC, Kempenaers B (2010) Variation in sleep behaviour in free-living blue tits, Cyanistes caeruleus: effects of sex, age and environment. Anim Behav 80:853–864
Sugai LSM, Silva TSF, Ribeiro JW Jr, Llusia D (2019) Terrestrial passive acoustic monitoring: review and perspectives. Bioscience 69:15–25
Thomas RJ (1999) The effect of variability in the food supply on the daily singing routines of European robins: a test of a stochastic dynamic programming model. Anim Behav 57:365–369
Thomas RJ (2002) The costs of singing in nightingales. Anim Behav 63:959–966
Thomas RJ, Cuthill IC (2002) Body mass regulation and the daily singing routines of European robins. Anim Behav 63:285–292
Thomas RJ, Szekely T, Cuthill IC, Harper DGC, Newson SE, Frayling TD, Wallis PD (2002) Eye size in birds and the timing of song at dawn. Proc R Soc B 269:831–837
Tobias JA, Planque R, Cram DL, Seddon N (2014) Species interactions and the structure of complex communication networks. Proc Natl Acad Sci U S A 111:1020–1025
Trillo PA, Vehrencamp SL (2005) Song types and their structural features are associated with specific contexts in the banded wren. Anim Behav 70:921–935
Tuttle MD, Ryan MJ (1981) Bat predation and the evolution of frog vocalizations in the neotropics. Science 214:677–678
Vabishchevich AP (2011) Dawn singing in pied flycatchers: mated males sing highly versatile songs in the early morning. Ann Zool Fenn 48:376–380
Van Duyse E, Pinxten R, Eens M (2003) Seasonal fluctuations in plasma testosterone levels and diurnal song activity in free-living male great tits. General Comp Endocrino 134:1–9
Van Duyse E, Pinxten R, Snoeijs T, Eens M (2005) Simultaneous treatment with an aromatase inhibitor and an anti-androgen decreases the likelihood of dawn song in free-living male great tits, Parus major. Horm Behav 48:243–251
Wada M (1986) Circadian rhythms of testosterone-dependent behaviors, crowing and locomotor activity, in male Japanese quail. J Comp Physiol A 158:17–25
Wang G, Harpole CE, Trivedi AK, Cassone VM (2012) Circadian regulation of bird song, call, and locomotor behavior by pineal melatonin in the zebra finch. J Biol Rhythm 27:145–155
Ward S, Slater PJ (2005) Raised thermoregulatory costs at exposed song posts increase the energetic cost of singing for willow warblers Phylloscopus trochilus. J Avian Biol 36:280–286
Ward S, Speakman JR, Slater PJ (2003) The energy cost of song in the canary, Serinus canaria. Anim Behav 66:893–902
Welling P, Koivula K, Orell M (1997a) Dawn chorus and female behaviour in the Willow Tit Parus montanus. Ibis 139:1–3
Welling PP, Rytkonen SO, Koivula KT, Orell MI (1997b) Song rate correlates with paternal care and survival in willow tits: advertisement of male quality? Behaviour 134:891–904
West-Eberhard MJ (1979) Sexual selection, social competition, and evolution. Proc Am Philos Soc 123:222–234
Wiens J, Johnston R (1977) Adaptive correlates of granivory in birds. In: Pinowski J, Kendeigh SC (eds) Granivorous birds in ecosystems: their evolution, populations, energetics, adaptations, impact and control. Cambridge University Press, Cambridge, pp 301–340
Wright HW (1912) Morning awakening and even-song. Auk 29:307–327
Xia C, Wei C, Lloyd H, Liu J, Wu Q, Zhang Y (2014) Dawn singing intensity of the male brownish-flanked bush warbler: effects of territorial insertions and number of neighbors. Ethology 120:324–330
York JE, Young AJ, Radford AN (2014) Singing in the moonlight: dawn song performance of a diurnal bird varies with lunar phase. Biol Lett 10:20130970
Zahavi A (1977) The cost of honesty: further remarks on the handicap principle. J Theor Biol 67:603–615
Zahavi A (1980) Ritualization and the evolution of movement signals. Behaviour 72:77–80
Zuk M, Kolluru GR (1998) Exploitation of sexual signals by predators and parasitoids. Q Rev Biol 73:415–438
Acknowledgments
We wish to thank Peter Slater, Barbara Helm, Marc Naguib, John Hutchinson, Valentin Amrhein, Constantino Macías, Thierry Aubin and Nicolas Mathevon for their insightful comments on previous versions of this review. Thiago Bicudo kindly provided the Brazilian data for Fig. 3.1. DL acknowledges post-doctoral study grants by the Fondation Fyssen (France) and the Comunidad de Madrid (Atracción de Talento Investigador, 2016-T2/AMB-1722, CAM, Spain). DG research was supported during the writing of this review by research grant CGL2017-83843-C2-1-P from the Spanish Ministry for Science, and DL research by grant CGL2017-88764-R from the Spanish Ministry for Science.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Gil, D., Llusia, D. (2020). The Bird Dawn Chorus Revisited. In: Aubin, T., Mathevon, N. (eds) Coding Strategies in Vertebrate Acoustic Communication. Animal Signals and Communication, vol 7. Springer, Cham. https://doi.org/10.1007/978-3-030-39200-0_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-39200-0_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-39199-7
Online ISBN: 978-3-030-39200-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)