Abstract
The region of Lagoa Santa stands out as the cradle of studies on epigean and subterranean ecology in Brazil. The subterranean habitats in the Lagoa Santa Karst area are structurally and biologically diverse, especially due to the wide variety of macro and microhabitats and food resources availability for the fauna. The observed biodiversity is a consequence of the distinct conditions of isolation, atmospheric stability, food availability, and the presence of dry and wet subterranean habitats. In terms of obligate cave fauna, the Lagoa Santa karst tends to present a moderate richness in absolute numbers (41 spp.) but low richness in relative terms (0.48 species per cave). However, such numbers were counted from inventories conducted in approximately 80 out of the 900 caves, so that many other species may still be discovered. The four described troglobites are Brasilomma enigmatica, Tisentnops onix, Charinus taboa, and Spelunconiscus castroi, but 37 other sampled troglomorphic species are still not described. Moreover, there are many human activities causing impacts to the Lagoa Santa Karst, which come from agriculture, cattle ranching, water exploitation, urbanization, deforestation, industries, mining, road construction, etc. The urgency for conservation actions is evident since studies on the population size and ecology of cave fauna are incipient and the low number of conservation units has led these species to an extreme degree of vulnerability and threats. In this perspective, the intensification of biological studies in caves of the Lagoa Santa karst is highly recommended to avoid species loss.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Subterranean Environments in the Lagoa Santa Karst
The high age of the limestone at the Lagoa Santa Karst (Neoproterozoic) and its long exposition to surface weathering contributed to the modeling of landscape in plateaus with different landforms (Berbert-Born 2000; Auler and Piló 2015). Such features influenced directly the hydrological dynamics, the availability of habitats and organic debris availability for the fauna associated with caves and other subterranean environments. Furthermore, rainfall and seasonality favor heterogeneous and peculiar vegetation (Brazilian Savannah) and primarily autogenic hydrology, which forms vertically dissected limestone outcrops with paragenetic caves and frequently short subterranean drainages (Auler and Piló 2015). The elevation ranging from 650 to 900 m asl represents a catchment divide between the basins of Mata Creek and Velhas River (Berbert-Born et al. 1998). Such dynamics and interaction of landscape elements have generated different habitats throughout the time, capable to shelter a specialized fauna occupying subsurface (epikarst) and deep subterranean habitats (dry caves, deep cracks, subterranean rivers, flooded travertines, water table, among others) from millimetric to decametric volumes.
The epikarst can be characterized as the uppermost weathered zone of carbonate rocks with substantially enhanced and more homogeneously distributed permeability and porosity when compared to the bulk rock mass below it (Klimchouk 2004). Despite the fact that this environment is poorly studied for caves of this region, it is plausible to assume that they represent important habitats for several species (e.g., copepods), as happens in several other karstic areas around the world (Culver and Pipan 2009). The meteoric water infiltrating the soil is partially retained in the epikarst and slowly percolates reaching caves via active speleothems even in dry periods of the year. Such waters may transport organic matter dissolved in the soil, therefore representing an important organic resource for microorganisms and small invertebrates, both in epikarstic zones and caves. In this sense, the great variety of mineral deposits (speleothems) present in caves of the region, besides improving the availability of microhabitats for fauna, are good indicators of the intense current and previous hydric dynamics.
Lotic and lentic groundwater bodies certainly comprise essential habitats for several organisms. However, more than 900 caves recorded for the region comprise dry passages with entrances in the lower portion of depressions or in the basis of limestone outcrops. Caves of different extensions that may be associated with the water table represent a minor percentage in this region (Auler 1998), although the occurrence of periodic swallets supplying the phreatic zone is not rare (Berbert-Born et al. 1998).
In these different subterranean habitats, the dynamics of food resource availability for the fauna tend to be complex due to the large territorial extension of the karst submitted to variations on the density of fractures (and caves) at different elevations. Thus, there are several and distinct possibilities of resource input into the caves due to biological (bats, rodents, accidentals) or physical elements (percolation water, rivers, flash flood) (Ferreira and Pompeu 1997; Berbert-Born et al. 1998; Ferreira et al. 2000; Souza-Silva et al. 2005). The input of organic resources (mainly plant debris) may occur gravitationally (via ceiling openings), from the deciduous vegetation present in the top of outcrops, by means of the water movement of flash flood and allogenic drainages, or by organic matter dissolved in percolation water. The vegetation associated with the limestone outcrops of Lagoa Santa karst present many deciduous plant species, which provides a greater accumulation of leaves in the litter during dry periods of the year (see chapter “The Vegetation of Lagoa Santa Karst”, this volume). Part of this material may be carried to the interior of caves mainly during rainy periods, as described for other Brazilian karst regions (Souza-Silva et al. 2011). On the other hand, in permanently dry caves bat guano seems to be predominant trophic resource, comprising an important food source for several groups of invertebrates (Berbert-Born et al. 1998; Ferreira et al. 2000a) and microorganisms (Ferreira et al. 2000b).
In summary, caves in the Lagoa Santa Karst area are structurally and biologically too variable and complex, especially due to the wide variety of macro and microhabitats and food resources for the fauna. Thus, the invertebrates and vertebrates’ communities have variable composition and diversity and were structured based on distinct conditions of isolation, atmospheric stability, food availability, and terrestrial and aquatic habitats.
2 Species Composition
Caves of the karst region of Lagoa Santa shelter a great variety of organisms that have been described since the nineteenth century. Paleontological sites sampled by the naturalist Peter W. Lund in caves of the region revealed living species that possibly used such environments as shelter, besides the extraordinary Pleistocene fauna (see chapter “Cave Paleontology in the Lagoa Santa Karst”, this volume). Among such discoveries, the canids Cerdocyon thous Hamilton Smith 1839 (Berta 1982) and Speothos venaticus Lund 1842 (DeMatteo and Loiselle 2008) stand out, besides some rodent species like Phyllomys brasiliensis Lund 1840 (Emmons et al. 2002), Blarinomys breviceps Winge 1888 (Silva et al. 2003, Machado et al. 2016) and Pseudoryzomys simplex Winge 1887 (Machado et al. 2016). In the same century, members of the expedition Thayer (1865–1866) collected mite specimens, although they were described only 100 years after (Whartonia W. pachywhartoni Vercammen—Grandjean 1966, recently reviewed by Silveira et al. 2015). Although they had sampled components of cave fauna, these first studies involved themes non-related to subterranean biology (e.g., paleontology, photographic inventory).
Later, descriptions of new species sampled in caves of the region were published (ex. Costa-Lima 1940; Wygodzinsky 1950; Schubart 1956) and from 1990 on many works took into account the occurrence of subterranean fauna in the area (ex. Berbert-Born and Horta 1994; Pinto-da-Rocha 1995; Fontanetti 1996; Hermann et al. 1998; Trajano et al. 2000). Currently there are several studies reporting species of Lagoa Santa caves regarding different areas of biology, like ecology (Ferreira and Martins 1999; Gomes et al. 2000; Iniesta et al. 2012; Pellegrini et al. 2016), parasitology (Oliveira et al. 2000; Filho et al. 2007; Coelho-Finamore et al. 2011; Sábio et al. 2015), taxonomy (Gil-Santana et al. 1999; Pena et al. 2005; Mews and Sperber 2008; Campos-Filho et al. 2016), biochemistry (Dantas et al. 2014, 2016), among others. As a result, despite the volume of information being still incipient, such karst region may be considered one of the better sampled areas of Brazil in terms of subterranean fauna, what does not mean it is sufficiently known regarding bioespeleology.
Subterranean arthropods recorded for this karst are beyond those species considered restricted to underground habitats (as indicated in the topic “Troglobites”). Several non-troglobitic species (belonging to different taxonomic groups) were described for those caves, like Amphipoda (Hyalellidae: Hyalella warmingi Stebbing 1899), Gastropoda (Bulimulidae: Thaumastus T. lundi Pena et al. 2005), Pseudoscorpiones (Olpiidae: Progarypus gracilis Mahnert 2001), Diplopoda (Pseudonannolenidae: Pseudonannolene microzoporus Mauries 1987 and Pseudonannolene chaimowiczi Fontanetti 1996; Chelodesmidae: Obiricodesmus rupestris and Sandalodesmus stramineus Schubart 1956), Orthoptera (Phalangopsidae: Endecous cavernicolus Costa-Lima 1940, reviewed by Mews and Sperber 2008), Heteroptera (Reduviidae: Mayemesa lapinhaensis Wygodzinsky 1950, reviewed by Gil-Santana et al. 1999) and Isopoda (Trichorhina pataxosi Campos-Filho et al. 2016). Other species of wide distribution were sampled in some caves, among which some dipterans with medical importance stand out (Psychodidae: Lutzomyia longipalpis Lutz and Neiva 1912, in Oliveira et al. 2000 and Coelho-Finamore et al. 2011; Lutzomyia L. renei Martins, Falcão and Silva 1957, in Sábio et al. 2015; Nyssomyia intermedia Lutz and Neiva 1912 and Nyssomyia neivai Pinto, 1926, in Filho et al. 2007). Besides Diptera, other groups were also recorded, as spiders (Araneidae: Alpaida septemmammata Octavius Pickard-Cambridge 1889, in Hermann et al. 1998; Ctenidae: Enoploctenus maculipes Strand 1909, in Pinto-da-Rocha 1995; Oecobidae: Oecobius annulipes Lucas 1846, in Hermann et al. 1998; Theridiidae: Nesticodes rufipes Lucas 1846, in Hermann et al. 1998; Sicariidae: Loxosceles similis Moenkhaus 1898, in several studies like Pinto-da-Rocha 1995; Hermann et al. 1998; Gomes et al. 2000; Dantas et al. 2014, 2016; Trechaleidae: Trechaleoides keyserlingi Octavius Pickard-Cambridge 1903, in Hermann et al. 1998), scorpions (Buthidae: Tityus serrulatus Lutz and Mello 1922, in Hermann et al. 1998), pseudoscorpions (Chthoniidae: Pseudochthonius strinatii Mahnert 2001, Chernetidae: Spelaeochernes dubius, Spelaeochernes eleonorae and Spelaeochernes gracilipalpus Mahnert 2001), mites (Anystidae: Erythracarus nasutus Otto 1999, in Iniesta et al. 2012; Argasidae: Ornithodoros talaje Guerin-Méneville 1849, in Hermann et al. 1998), gastropods (Subulinidae: Subulina octona Bruguière 1798, in Hermann et al. 1998), bettles (Leiodidae: Dissochaetus murrayi Reitter 1884, in Pinto-da-Rocha 1995), Psocoptera (Psyllipsocidae: Psyllipsocus ramburii Selys-Longchamps 1872, in Lienhard and Ferreira 2015) and Hemiptera (Reduviidae: Zelurus variegatus Costa-Lima 1940, in Hermann et al. 1998). Even components of microbiota were identified in sand flies sheltered in subterranean habitats of the region, like bacteria and protozoa (Stenonotrophomonas maltophilia, Enterobacter cloacae, Pseudomonas putida, Acinetobacter lwoffii, Flavimonas orizihabitans, Klebsiella p. ozaenae, Bacillus thuringiensis in Oliveira et al. 2000; and Leishmania infantum Coelho-Finamore et al. 2011).
These records represent only a small part of specimens observed and/or collected, since many of them were not identified to the species level, and consequently they were not formally described (as indicated in Pinto-da-Rocha 1995; Hermann et al. 1998; Ferreira and Martins 1999; Gomes et al. 2000; Trajano et al. 2000; Mahnert 2001; Iniesta et al. 2012; Pellegrini et al. 2016 and in several other abstracts of meetings and thesis conducted in this karst region). Some species from the caves in the Lagoa Santa Karst are shown in Fig. 1.
Some troglophile and trogloxene species found in the caves of the Lagoa Santa Karst: a Endecous cavernicolus (Phalangopsidae); b Zelurus zikani (Reduviidae); c Diplatyidae earwig (Dermaptera); d Hypena sp. (Noctuidae); e Psyllipsocus falcifer (Psyllipsocidae); f Pseudonannolene taboa (Pseudonannolenidae); g Symphytognatha carstica (Symphytognathidae); h Loxosceles similis (Sicariidae); i Mesabolivar sp. (Pholcidae); j Eusarcus sp. (Gonyleptidae); k Eukoenenia florenciae (Eukoeneniidae); l Progarypus sp. (Olpiidae)
Besides recent (living) species of mammals revealed by excavations of Peter W. Lund, several other vertebrates were recorded in caves of the Lagoa Santa karst. Many representatives of birdlife from Cerrado eventually use the caves entrances to build nests, like João-de-Barro (Furnarius rufus badius Lichtenstein 1823, in Pinto-da-Rocha 1995), Juriti (Leptotila verreauxi decipiens Salvadori 1871, in Pinto-da-Rocha 1995), Saí-Andorinha (Tersina viridis Illiger 1811, in Herman et al. 1998), Graúna (Gnorimopsar chopi Vieillot 1819, in Herman et al. 1998), owls (Tyto furcata Temminck 1827, in Herman et al. 1998), parrots and parakeets (Amazona aestiva Linnaeus 1758, Eupsittula aurea Gmelin 1789; in Pinto-da-Rocha 1995). In relation to anurans, most of the records do not have a taxonomic refinement to the species level, except for the species Rhinella schneideri Werner 1894 (Herman et al. 1998). However, amphibians are frequently found in Brazilian caves and there are occurrences in many studies in the region of Lagoa Santa (ex. Berbert-Born and Horta 1994; Pinto-da-Rocha 1995; Hermann et al. 1998; Matavelli et al. 2015). In relation to fishes, the only identified species comprises the Characidae Astyanax bimaculatus Linnaeus 1758 (Herman et al. 1998), although, as for other groups, there are records of other species identified at higher taxonomic levels (Pinto-da-Rocha 1995; Herman et al. 1998). Among mammals, bats may be considered the most efficient in using caves as shelters (Kunz 1982). Besides performing several ecological functions, they are responsible for the resource input to the hypogean environment (see the topic Ecology). In Lagoa Santa area, studies have indicated the presence of at least 23 species of bats inhabiting caves or cracks in the exokarst (Table 1). Besides the bats, other mammals are also frequently found, like raccoon (Procyon cancrivorous Cuvier 1798) and skunks (Didelphis albiventris Lund 1840).
In this sense, the faunistic composition of Lagoa Santa Karst comprises a great diversity of taxonomic groups that are not exclusive of subterranean environments, although there are species found exclusively in caves. Their adequate management depends on the knowledge on levels of adaptation and uses for different species that compose such communities. Moreover, the knowledge regarding all the present fauna allows distinguishing invasive species in these systems (Mazza et al. 2014). Then, it is increasingly necessary to study communities present in caves of this region for conservation purposes. As the number of inventories increases new species certainly will be described and/or new occurrences will be published for the Lagoa Santa Karst.
3 Troglobites
Different karst areas in Cerrado (Brazilian Savannah) were inventoried and revealed distinct richness of troglobitic species. The Lagoa Santa Karst, when compared to other areas, tends to present a moderate richness of troglobites (in absolute terms), although this richness, in relative terms, may be considered low. Until the present, there are 41 species with troglomorphic traits (potentially troglobitic) recorded for the Lagoa Santa caves. This number was taken from inventories performed in approximately 80 caves, as the mean of troglobites per cave is considerably low (mean of 0.48 troglomorphic species per cave). However, it is important to mention that such inventories were made in a small number of the caves recorded in the area, and then many other species may still be found, as happens for most of the Brazilian karst areas.
For comparison between the troglobites richness of Lagoa Santa Karst and other regions, some studies will be mentioned. A study performed in 15 caves in the karst region of Cordisburgo (adjacent to Lagoa Santa Karst area) revealed 39 troglomorphic species (mean of 2.6 species per cave) (Souza 2012). In the limestone speleological province of Arcos-Pains-Doresópolis, 294 caves were inventoried and 79 troglomorphic species were recorded (mean of 0.27 species per cave) (Zampaulo 2010). Simões (2013) studied 55 caves located in the karst region of Northwest Minas Gerais and found 35 troglomorphic species (mean of 0.84 species per cave). Finally, one study performed in 51 limestone caves from Central-North Minas Gerais revealed 94 troglomorphic species (mean of 2.75 species per cave) (Rabelo 2016).
It is noticeable that the absolute number of troglomorphic species observed for the region of Lagoa Santa Karst (41) is comparable to those found in other regions (such as Cordisburgo—39 species—and Northwest Minas Gerais—35 species). However, the relative number of troglobitic species (0.48 species/cave) is low and only higher to be observed for the region of Arcos-Pains-Doresópolis (0.27 species/cave). It is important to cite that these two previously mentioned areas stand out due to the predominance of small and dry caves, factors that may have contributed to the low relative richness of troglobitic species. Simões et al. (2015) demonstrated that the presence of water bodies and the length of caves are determining factors for the occurrence and richness of troglomorphic species in caves of some karst areas of the Cerrado.
More than 40 troglomorphic (potentially troglobitic) species have been found in the Lagoa Santa Karst, but only 4 have been described: the spiders Brasilomma enigmatica (Prodidomidae—Fig. 2a) and Tisentnops onix (Caponiidae—Fig. 2b), the tailless whip scorpion Charinus taboa (Charinidae—Fig. 2d) and the isopod Spelunconiscus castroi (Styloniscidae). However, there is a larger number of troglomorphic species already identified from caves of this area (37 species). Great part of these species is deposited in the reference collection of the Study Center on Subterranean Biology at the Federal University of Lavras (CEBS/UFLA 2016), and some of them are under the process of description. Such species were collected in the municipality of Confins (palpigrades, isopods, and spiders—Prodidomidae and Oonopidae), Lagoa Santa (mites Labidostomatidae, spiders Nesticus sp and isopods Trichorhina), Matozinhos (pseudoscorpions Chthoniidae, springtails Entomobryiidae, isopods Trichorhina and Styloniscidae and bettles Rizophagidae), Prudente de Morais (spiders Oonopidae, springtails Arrhopalites, millipedes Cryptodesmidae and isopods Trichorhina and Styloniscidae) and in Sete Lagoas (springtails Entomobryomorpha, isopods Trichorhina and Spelunconiscus, harvestmen Spelaeoleptes and millipedes Pyrgodesmidae). Some troglobitic species from the caves in the Lagoa Santa Karst are shown in Fig. 2.
Some troglobitic species found in the caves of the Lagoa Santa Karst: a Brasilomma enigmatica (Prodidomidae); b Tisentnops onix (Caponiidae—photo: Ubirajara Oliveira); c Lygromma sp. (Prodidomidae); d Charinus taboa (Charinidae); e Labidostomatidae mite; f Spaeleoleptes sp. (Escadabiidae); g Pyrgodesmidae millipede; h Trichorhina sp. (Platyarthridae); i Spelunconiscus castroi (Styloniscidae)
Until the present, no exclusively aquatic species has been found in the Lagoa Santa Karst. Considering the 41 known species, 78% are terrestrial and the others are amphibious Styloniscidae (which favor the water but can move along terrestrial environments). Furthermore, most of the troglobitic species present a restricted distribution (occurring in only one cave) and only 5% of them occur in two or more caves. The caves that stand out in the region for their number of troglobitic species are Taboa Cave (6 species), Rei do Mato Cave (4), Lapa Vermelha Cave (4), and Meandro Abismante Cave (3).
In relation to the described species, Charinus taboa (Charinidae) is found in only two relatively close caves (Taboa Cave, 800 m long and BR24 cave, 33.8 m long), both in the municipality of Sete Lagoas, Minas Gerais state. The adults of C. taboa are found in the deeper portions of caves, mainly among speleothems, wall, and ceiling, while juveniles shelter in spaces under the rocks. This species may be considered vulnerable since only 31 specimens were observed in the two caves over years of research. Moreover, the epigean environment around the cave has been altered by human activities. Other species described for Taboa Cave is Tisentnops onix (Caponiidae) through the unique female collected under a rock in aphotic zone (at 15–20 m from the entrance). The occurrence of T. onix in other cave close to Taboa (Cave 64, 58.2 m long) is a strong indicator of the subterranean connectivity of these caves.
The isopod Spelunconiscus castroi (Styloniscidae) was described for the cave MOC32 (Campos-Filho et al. 2014), although nearly no information about this species was presented by the authors (except mentioning that it is an amphibious species). On the other hand, the spider Brasilomma enigmatica occurred in only one cave from Lagoa Santa karst (MOC131) but was also found in other areas (Brescovit et al. 2012). This species presents a wide distribution beyond the Lagoa Santa Karst, also occurring in ferruginous and quartzite caves along with an extension of more than 300 km without apparent subsurface connection with caves of the Lagoa Santa Karst (in this scenario the species was named “enigmatica” alluding to its distribution). The only specimen collected in MOC31 was collected 30 m below the surface level (Brescovit et al. 2012).
Information on the population size and ecology of the described troglobites and the other undescribed troglomorphic species does not exist until the present. Such lack of information associated with the intense human activities in the region and the reduced number of conservation units has led these species to an extreme degree of vulnerability and threats. In this perspective, the intensification of biological studies in caves of the Lagoa Santa karst is highly recommended in order to avoid the species extinction (mainly the troglobites) even before they are formally described (see the topic “Threats”).
4 Ecology
The Lagoa Santa Karst certainly presents an historical relevance regarding biospeleology in Brazil, not only due to the species described from caves (as previously cited) but also especially considering that the first studies on ecology of Brazilian cave communities were performed in this region (e.g., Ferreira and Pompeu 1997; Ferreira et al. 2000b), among which we highlight those regarding ecology of guano communities.
Guano piles are widely considered as an important source of organic matter in caves (Harris 1970; Decu 1986; Ferreira and Martins 1999; Gnaspini 2012). In tropical caves, especially those permanently dry, bat guano may represent the most important source of organic matter, forming the trophic base for the structure of many invertebrate communities (Decu 1986; Ferreira and Martins 1998, 1999). The diversity of bats feeding habits in the tropics is remarkable (Herzig-Straschil and Robinson 1978). The three most common guano types found in the caves of the Lagoa Santa Karst are produced by frugivorous bats (with small undigested seeds), hematophagous bats (with a pasty consistency and reddish color when fresh, becoming black and often powdery when older), and by insectivorous bats (containing small chitinous parts of insects or other arthropods). Although other types also may occur, the most commonly found piles are produced by hematophagous bats (especially Desmodus rotundus), and this certainly has been caused by a historical replacement of the natural forests by pastures (see the topic “Threats”).
Bat guano communities in caves are extremely interesting for ecological studies, especially due to their physical and chemical conditions (that are easily measurable and quite distinguishable when comparing different piles) and the great number of easily defined interactions with the overall cave environment (Fig. 3). However, during decades such communities were considered “isolated” from the cave environment as a whole, but in fact a high number of cave invertebrate groups associate and interact with them (Ferreira et al. 2000a).
Several guano piles traits act structuring the associated communities in the Lagoa Santa Karst (Ferreira et al. 2000b). Ferreira and Pompeu (1997) showed that the distance from the entrance was an important parameter determining the richness of guano communities in Taboa Cave (Sete Lagoas municipality) (Fig. 4b). Other studies performed later found similar (Ferreira et al. 2007) but also distinct results (Ferreira et al. 2000), indicating that the distance from the cave entrance is not always an important factor for the bat guano communities. Since most invertebrates occurring in guano piles are troglophiles (with some rare troglobitic species), the colonization of each deposit may occur from any place inside the cave, and not only from the entrance. On the other hand, the area of guano piles is always important, so that the richest and most diverse communities are always associated to biggest piles (Ferreira and Pompeu 1997; Ferreira and Martins 1998; Ferreira et al. 2000a; Bahia and Ferreira 2005; Ferreira et al. 2007) (Fig. 4c, d). The richness or abundance of species that use faecal deposits tends to be proportional to the availability of this resource (Doube 1986). Furthermore, one would expect some area effect of greater potential microhabitat variability over a larger surface. Hence, in caves of the Lagoa Santa Karst, larger deposits with a greater diversity of microhabitats (like small holes and cracks) have more associated species.
a Similarity values (Renkonen index) between a larger pile (species “source”) and small peripheral piles (species “sink”) according to their distance in relation to the main (larger) pile in Lavoura Cave (Matozinhos municipality); b relation between the distance from the cave entrance and the richness of guano communities in Taboa Cave (Sete Lagoas municipality); c relation between the area, the pH of the guano piles and the richness of guano communities in Lavoura Cave (Matozinhos municipality); d relation between the area, the pH of the guano piles and the diversity of guano communities in Lavoura Cave (Matozinhos municipality)
It is also important to mention that guano communities are structured over some physicochemical parameters, like pH and organic content. The reduction of guano pH over time is well known, since fresh guano is alkaline and becomes acidic due to ammoniac fermentation (Hutchinson 1950). However, pH does not always reflect guano age, since the piles are open systems subject to be affected by chemical (percolation water, floods, etc.) and physical (landslides, sedimentation, etc.) processes occurring in the cave environment (Ferreira et al. 2000a). In the caves of the Lagoa Santa Karst, the richest piles generally present moderate pH (Fig. 4c, d). The relationship between guano pH and richness of the associated communities is probably the result of a preference (by most species of the community) for an optimum and intermediate value of pH (Ferreira et al. 2000a). However, the same authors postulated that if guano pH reflects the piles age, fresh piles (more basic) may not have had enough time to be colonized, while old piles (more acidic) may have been exhausted as a food resource due to consumption or mineralization (Ferreira et al. 2000a). The organic content is also important, as shown by Ferreira et al. (2000a) in Lavoura Cave (Matozinhos municipality) located in the Lagoa Santa Karst. Such authors observed that richer communities were associated with guano piles with higher organic contents, demonstrating the importance of such parameters for these communities.
Finally, there are some evidence that larger piles can act as sources of colonization for smaller ones. In Lavoura Cave (Matozinhos municipality), the community similarities between one big guano pile and smaller piles surrounding it (unpublished data) were correlated. It was observed that smaller piles closer to the biggest ones are more similar to it than piles located far from it (Fig. 4a). This suggests that guano communities in a given cave may act as metacommunities. The concept of metacommunity was first proposed by Gilpin and Hanski (1991) to represent a set of local communities from different sites connected by dispersion of one or more of its components. A metacommunity follows the same dynamics of a metapopulation, and therefore is subject to local extinctions and recolonizations. The low abundance of organisms in some old guano piles enhances local extinctions. As the guano becomes older, its quality as an organic resource decreases, thus the community progressively leaves the pile and colonize others in order to maintain their populations. However, the dynamics of movements between guano piles remain largely unknown, certainly deserving further investigations.
Some studies performed in caves of the Lagoa Santa Karst focused on aspects of population ecology of some species. Ferreira et al. (2005) monitored a cave population of the brown recluse spider (Loxosceles similis) in the Lavoura Cave (located in Matozinhos municipality) and found some patterns especially regarding the displacement of such spiders inside the cave. Loxosceles species are usually considered sedentary having a life area restricted to the vicinities of their webs (Bücherl 1961; Foelix 1987). However, Ferreira et al. (2005) registered a great mobility of such spiders. Many individuals of L. similis were recaptured at distances from 20 to 80 m from the place where they were initially marked (a movement of 40 m was registered in only one week). These distances are much longer than those recorded for some species like L. rufipes (Lucas 1834) and L. intermedia Mello-Leitão, 1934 (around 2 m in one week, what was considered a significant mobility) (Delgado 1966; Fischer 1996). However, the cave environment presents different conditions from those found in external or anthropogenic environments. The scarcity and irregular distribution of food resources in a cave may result in lower prey availability, which may have led the spiders (in the Lavoura Cave) to move longer distances looking for preys.
Another important study performed in the Lagoa Santa Karst presented, for the first time for the tropical region, data regarding the responses of a cave population (of the moth Hypena sp.—Lepidoptera: Noctuidae) to seasonal variations in the epigean environments (Ferreira et al. 2015). Several studies demonstrated the effects of external climatic fluctuations on subterranean populations and communities in the temperate region (Bourne and Cherix 1978; Carchini et al. 1994; Novak et al. 2004; Tobin et al. 2013). Although such effects are expected in areas where external climatic fluctuations are more intense (Mitchell 1969), no studies were historically performed in tropical regions, maybe due to the false impression that both small temperature and humidity fluctuations (typical of the tropics) have little or no effect over the subterranean fauna. In this sense, Ferreira et al. (2015) demonstrated that a cave population responds to seasonal external variations (in this case the Taboa Cave, municipality of Sete Lagoas). During the rainy seasons the population of Hypena sp. was closer to the cave entrance, while during the dry periods, the population peaks occurred slightly deeper inside the cave (Fig. 5). This study is important especially to demonstrate that patterns occurring in temperate caves can also be observed in the tropics. However, further studies are needed to actually understand how cave populations or communities from temperate and tropical regions differ or not, in terms of function and structure.

Modified from Ferreira et al. (2015)
Spatial distribution maps of Hypena sp. in Taboa Cave (Sete Lagoas municipality) demonstrating different densities between seasons. In the dry seasons (17/07/1999 and 10/07/2000) individuals are located far from the cave entrance, while during rainy seasons (16/01/2000 and 19/01/2001) individuals migrate to areas near the entrance. Blue colors indicate low densities while light yellow colors indicate high densities.
Although only few studies regarding general cave communities in the Lagoa Santa Karst were performed (Iniesta et al. 2012), some “patterns” were found, indicating that communities in this region present some similarities. In the Irmãos Piriás Cave (located in Matozinhos municipality), four collection events were conducted between 1999 and 2000. In all events, a reduction of invertebrate richness was observed from the entrance to deeper portions of the cave (Fig. 6a). This entrance-mediated effect was expected, especially in tropical regions, due to the remarkable richness of external communities, which represent an important source of organisms prone to colonize caves. Therefore, cave entrances in the tropics tend to present richer communities when compared to inner portions of the caves. However, although some species can establish themselves in the hypogean habitats, most of them cannot, due to their lack of preadaptive traits. Cave entrances are considered ecotones between epigean and hypogean environments (Prous et al. 2004, 2015). Hence, such regions tend to present intermediate conditions between those habitats. As an example, entrances are more stable than epigean environments (favoring the presence of many species), although they present less organic resources (preventing other species to colonize them). Prous et al. (2015) argued that ecotones in the cave entrances may function as selective permeability membranes acting differently over distinct species. According to Strayer et al. (2003), this “membrane” can act on matter, energy, or species in different ways and can partially transmit, transform, absorb, reflect, amplify them, or act neutrally. Moreover, filters present in these membranes can be biological or physical and the combination of both filters is responsible for the presence and distribution of species in the cave entrances and also within the caves (Prous et al. 2015).
a Relation between the distance from the cave entrance and the invertebrates richness (vertical axis) in different seasons in Irmãos Piriás Cave (Matozinhos municipality); b relation between the distance from the cave entrance and the density of the spider Plato sp. (Theridiosomatidae) in different seasons in Irmãos Piriás Cave; c relation between the distance from the cave entrance and the density of the cave cricket Endecous sp. (Phalangopsidae) in different seasons in Irmãos Piriás Cave
However, although there is a tendency toward the reduction of richness from the entrance to deeper areas in caves, each species inside a cave can present a distinct pattern of distribution. In the Irmãos Piriás Cave, some species presents higher densities in regions closer to the entrance (as the spider Plato sp.—Theridiosomatidae) while for others the higher densities are observed on inner portions of the cave (e.g., the cricket Endecous sp.—Phalangopsidae) (Fig. 6b, c). Furthermore, such patterns of distribution vary not only in space but also throughout time. The community from Irmãos Piriás Cave varied considerably from 1999 to 2001. Richer communities were observed during rainy periods (89 and 94 invertebrate species in January 2000 and January 2001, respectively), while in the dry periods the communities reduced the number of species (44 and 64 invertebrate species in July 1999 and July 2000, respectively). The species turnover between the four sampling events corresponded to 54.18% indicating a considerable species replacement over time. The turnover was calculated using data of presence and absence through the index of Harrison et al. (1992); modified by Whittaker (1960), in order to compare samples of different sizes. βHarrison = {[(S/a) − 1]/(N − 1)}9100), where S = total species richness, a = average richness values, and N = number of samples. This measure ranges from 0 (no turnover) to 100 (each sample has a unique set of species) (Koleff et al. 2003).
In the Taboa Cave (Sete Lagoas Municipality) it was observed a similar pattern to the observed in the Irmãos Piriás Cave. In the rainy periods, not only the richness but also the diversity was higher than in dry seasons. In this cave, in both rainy periods the richness and diversity corresponded to 101 species and H′ = 3.04 (January 2000) and 126 species and H′ = 3.29 (January 2001), while in the dry periods those values corresponded to 35 species and H′ = 2.23 (July 1999) and 75 species and H′ = 2.73 (July 2000). However, the community equitability did not change considerably along the sampling period, corresponding to 0.63 in both dry periods and to 0.66 (January 2000) and 0.68 (January 2001) in the rainy periods. This indicates that although the community can vary along the time in richness, diversity and even in composition, the average distribution of population size is stable, and such “stability” can be the result of the oligotrophic conditions, which represents a limiting pressure upon the distinct populations inhabiting the cave.
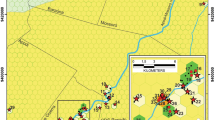
Finally, the epigean landscape can also be important to determine the structure and composition of the cave fauna. It is well known that patterns of biodiversity usually respond to habitat disturbances and different land uses (MacDonald et al. 2000). However, until recently no study had demonstrated spatial-scale influences in subterranean terrestrial communities. In a study conducted in the Lagoa Santa Karst, Pellegrini et al. (2016) analyzed how land use and cave physical structure could influence the terrestrial cave invertebrate species composition. Such authors also determined the influence of different spatial scales under analysis on the composition of invertebrate cave fauna. For that purpose, they determined land uses at three different landscape scales, by gathering data into circular areas of different sizes (50, 100 and 250 m) with centroids in the cave entrances (Fig. 7). The authors found that the best explanatory variable for cave invertebrate composition was the percentage of limestone outcrops covering the surface environments. Furthermore, they confirmed the scale-dependence hypothesis since the models generated during the study (by Distance Based Linear Modeling) became more precise as larger scales were analyzed to explain cave invertebrate composition. According to them, larger scales embrace more precisely more environmental traits that influence the cave fauna composition. In conclusion, Pellegrini et al. (2016) found a strong influence of limestone outcrops structuring cave communities independently of the scale under analysis (although the scale can enhance the precision of the analysis), demonstrating the major importance of preserving those outcrops for the maintenance of the cave fauna composition.

Modified from Pellegrini et al. (2016)
Spatial characterization of the landscape at “Sumidouro State Park”. Different colors represent distinct vegetation cover or land-use types. The black dots indicate the sampled caves, surrounded by the buffers of 50, 100, and 250 m for analyzing the effect of spatial scale on the explanatory power of environmental variables at the cave invertebrate communities.
5 Tourism: Enemy or Ally to the Cave Life?
Several caves around the world present an extraordinary scenic beauty. Such caves historically attracted the attention of general public, which started to use them for touristic purposes. According to Hamilton-Smith (2004) tourist caves can be defined as those attracting the interest of the general people, thus generating some type of financial return, as charge for admission or sale of souvenirs. Several caves have been used worldwide for touristic purposes during decades. Many of these caves receive thousands of visitors every year. Culver and Sket (2002) attested that caves that receive large number of visitors could present a high diversity of species, despite the obvious alterations caused by tourism. Accordingly, the opening of a given cave to tourism does not mean that their biodiversity will necessarily be altered or lost, as in the cases of some show caves, as the Mammoth Cave (Kentucky, USA), Postojna-Planina Cave System (Slovenia), Baget-Sainte Catherine System (France), and Vjetrenica Cave (Bosnia and Herzegovina) (Culver and Sket 2002). However, one cannot assume that touristic use never has deleterious effects in caves, since alterations caused by such activities can affect distinct subterranean species in different ways.
In Brazil, there are many show caves with diverse infrastructure or use. In the Lagoa Santa Karst, two show caves can be highlighted, especially due to the large number of visitors they attract every year. Such caves are Rei do Mato Cave (located in Sete Lagoas municipality) and Lapinha Cave (Located in Lagoa Santa municipality). Both caves were inventoried regarding the fauna and at least in one of them (Lapinha Cave) it was possible to evaluate the effects of the touristic use on the cave communities.
In Rei do Mato Cave (Sete Lagoas municipality), a total of 204 invertebrate species were found during five sampling events (unpublished data from the authors). However, the species richness varied along the time, and richer communities were found during rainy periods, following the general pattern of the cave communities in the area (30 species on average in the dry periods contrasting with 67 species on average in the rainy periods). Most invertebrate species were troglophiles and trogloxenes, and only four species presented troglomorphic traits (two isopods, one springtail, and one harvestman). Furthermore, most species (more than 60%) were concentrated near the entrance during all sampling events, indicating the importance of ecotone communities for this cave, or contrarily, the highly restrictive conditions observed in the inner portions of the cave (caused by natural and/or touristic factors).
In the Lapinha Cave (Lagoa Santa municipality), three sampling events were performed, all during dry periods (unpublished data from the authors). However, the sampling occurred in quite distinct situations. The first sampling (held in June 2002) occurred when the cave was under constant touristic use, since it was opened for this purpose in 1968. However, in 2009, touristic activities were interrupted due to the need of a management plan for the cave, and no tourists were allowed in. The second sampling event was performed in July 2011, 2 years after it was closed for tourism. After the second sampling, tourism was reestablished in the cave. The third sampling occurred in July 2012, one year after the reopening of the cave to tourists. Therefore, the samplings allowed a comparison regarding the direct effects of tourism on the cave communities of this cave. The most significant result observed was an increase in the invertebrate richness of the cave after the tourism ceased in the cave. The first sampling (during touristic use) revealed 58 invertebrate species, while the second event (no tourism in course) revealed 75. Hence, there was an increase of almost 30% in the richness during the period without tourism. The third collection (one year after the reestablishment of tourism) surprisingly revealed again 58 species, thus indicating that the tourism in this cave presented an obvious effect reducing the richness of invertebrate species.
However, diversity did not change considerably comparing the three events. In the first collection (during tourism), it corresponded to 2.3, in the second (after closing the cave) to 2.1, and in the third collection (after the reopening to tourism) it was 2.5. The increase in the diversity after the reestablishment of tourism resulted from a reduction in the dominance in this period (D = 0.14, compared to 0.21 and 0.22 from the first and second samplings, respectively). Furthermore, in general, the abundance tended to increase from the first to the last sampling events, and the highest values were observed after the reopening to tourism (8,141 individuals in the whole cave, compared to 4,966 and 1,210 individuals observed in the first and second samplings, respectively). Some common troglophilic or trogloxene species, as the cave moth Hypena sp. (Noctuidae) and the spiders Mesabolivar sp. (Pholcidae) and Loxosceles sp. (Sicariidae) presented the same pattern (Fig. 8).
Differences observed in population sizes of three species in sampling events conducted during touristic use (June 2002 and July 2012) and during non-touristic use (July 2011), in Lapinha Cave (Lagoa Santa municipality): a Mesabolivar sp. (Pholcidae); b Loxosceles similis (Sicariidae); c Hypena sp. (Noctuidae)
However, it is important to highlight that the reopening of the cave to tourism occurred after the management plan, which indicated the necessity of preserving some areas (as the deeper chamber of the cave) for the fauna, especially due to the presence of a bat colony (Phyllostomus sp.). Accordingly, after the visitation ceased in this chamber, the bat colony increased (as the guano production), allowing more species to establish themselves and to grow their populations. Thus, the deleterious effects of tourism can be reduced by closing some areas to visitors, keeping them preserved and far from the direct effects of the visitation (such as lights, trampling, among others).
According to Hamilton-Smith (2004), the most severe impacts observed in tourist caves usually result from inappropriate use. However, the tourism, per se, can cause several modifications on the cave environment, most of them primarily produced by the introduction of artificial light, which creates a photoperiod in the pristine aphotic areas of the cave. Such photoperiod provides conditions for the growth of photosynthetic organisms (especially algae and moss, but sometimes ferns and other more complex plants) (Fig. 9a–c). In turn, those organisms represent organic resources that were absent in aphotic zones, thus attracting animals that would not be found in such areas. Furthermore, these organisms (Fig. 9c) can be consumed by cave species and the effects of such consumption on the populations (positive or negative) are unknown. Another important alteration caused by the touristic infrastructure in show caves of the Lagoa Santa Karst was the emergence of some fungi that started growing associated to the electrical wiring inside the caves (Fig. 9d). The most important species found in those cables were Fusarium solani, which are also commonly observed in soil. However, since there were no studies on the fungal community before the installation of those cables, it is not possible to evaluate whether those fungi already inhabited the cave soil or were introduced following the emplacement of such infrastructure. The concern lays over the fact that Fusarium species can cause opportunistic mycosis, thus offering risks to the visitors. Furthermore, hundreds of invertebrates (especially barklices and springtails) were observed feeding on those fungi colonies growing on the cabling. Accordingly, the fungi appearance may have caused a disproportional growth of some cave invertebrate populations, besides attracting those species to areas in which they are more exposed to trampling by visitors.
a Algae growing on a speleothem in Rei do Mato Cave (Sete Lagoas municipality); b Psyllipsocus falcifer feeding on such algae (see the greenish color of the specimen’s abdomen); c an angiosperm growing inside the Rei do Mato Cave; d fungi (especially Fusarium solani) growing associated with the electric cabling inside the Rei do Mato Cave
Finally, it is important to highlight the importance of tourism as a powerful tool for environmental education in the karst systems as a whole. Although such activities can produce deleterious effects on the caves and their fauna, such effects can be reduced by management plans and actions. On the other hand, one of the most effective approaches to promote the sustainable use of karst landscapes and caves is education. Show caves, in this context, are extremely important, since they can be used in instructive visits in order to provide a broader view of the environment for the cave visitors and incite more interest for the preservation of those unique and endangered habitats. Accordingly, show caves offer the opportunity to educate the general audience, since during visits technical information can be transmitted, thus providing a subtle inclusion of concepts, expanding the vision, as well as providing human interaction with the subterranean environment.
6 Threats
The Lagoa Santa Karst faces many environmental threats (see chapter “Environmental Problems in the Lagoa Santa Karst”, this volume). The karst region of Lagoa Santa comprises one of the main areas of mineral exploitation of the state of Minas Gerais, with cement production as the main economic activity of the region. This mineral derivative represents an important economic pillar for the state, which is leader on the cement production of Brazil, with more than 14 million tons per year (CBIC 2013). According to Oss and Padovani (2003), for each ton of cement produced approximately 1.4 tons of limestone or similar rock is required. Thus, the accumulation of industries of such segment associated with the limited spatial distribution of Lagoa Santa karst constitutes relevant threats to the local subterranean environment.
The impacts of such economic activity over subterranean environments lead to the complete removal of bedrock for the production of commercial derivatives, alterations in groundwater routes of the local aquifer to access the mineral deposits and water supply of the industrial plant, besides the emission of gases and particulate sediments. The first mentioned impact promotes the complete alteration of landscape in function of the removal and processing of the rock mass. This process may cause the total or partial destruction of cavities and indirectly may interrupt the hydric flow responsible by the genesis and contribution of epigean trophic resources in the karst system. As it has been already demonstrated for other lithologies, the use of water during mining activities alters the balance between the aquifer recharge and the residence time of subterranean waters, limiting the capability of hydric resilience of the system (Gama and Matias 2015). In this sense, the lack of adequate management of the water resources may cause the extinction of many troglobitic amphibious species endemic of this karst area (Trajano and Bichuette 2010; Campos-Filho et al. 2014).
In relation to the emission of gases, studies demonstrate that this industrial sector is responsible for 5–7% of the total anthropic CO2 emitted to the atmosphere (Chen et al. 2010). Additionally, the dust resulting from the process of cement production may accumulate in leaves of different vegetal strata, thus promoting necrosis and chlorosis in plants (Kumar et al. 2008). Considering the Lagoa Santa Karst, this is worrisome since the main trophic resources found in caves are allochthonous (Gomes et al. 2000; Pellegrini et al. 2016; Iniesta et al. 2012). Therefore, alterations on the quality and/or quantity of leaf litter carried into the caves may affect the fauna community, such as the local exclusion of species by inter or intraspecific competition.
The proximity of the Lagoa Santa Karst to a large metropolitan center makes the urban expansion and other anthropic activities possible threats to its conservation. The metropolitan region of Belo Horizonte, in which all the municipalities that compose the karst are included, is the third largest urban agglomeration of Brazil with population over 2.5 million people (IBGE 2016). Environmental impacts of the urban expansion vary from the excessive consumption and alterations of water quality, for modifications of the biodiversity and landscapes at different scales (Larson et al. 2009). According to White et al. (1986), the effects of urbanization over the karst are even more severe. The presence of urban centers affects the natural cycle of water infiltration in the soil, thus generating karst collapse through the increase of hydraulic gradient due to the lowering of the water table and alterations of the rainfall. The contamination of the water table by slurry and other anthropic wastes also occurs in some caves, given the proximity of dumps. One example occurred in a cave located in the municipality of Matozinhos (Meandro Abismante Cave). A faunal inventory conducted in 2002 revealed the presence of troglobitic fauna including an amphibious species of Styloniscidae (probably of the genus Spelunconiscus—Fig. 10b). After this inventory, a technical report was elaborated, which indicated a strong potential of contamination of the aquifer inside the cave given its proximity in relation to the municipal landfill of Matozinhos (Fig. 10a), what would have irreversible consequences for the fauna (especially for the troglobitic isopod that inhabited the small stream in the cave). This report also indicated the need to monitor such population in function of this potential impact. However, unfortunately, no attitude was taken by environmental agencies. In 2005, in a visit to the same cave, the presence of a dense biofilm was observed covering the entire drainage bottom, as well as blackened fragments apparently from the contamination by the landfill (Fig. 10c). No specimen of the troglobitic Styloniscidae was observed during this visit. It is important to highlight that in 2002 dozens of individuals were seen along the accessible stretch of the drainage. Up to now, there is no more information about individuals of this species, and so it is possible that this important species, endemic and new to science, has been extinct even before it was described.
a Landfill of the Matozinhos municipality (red arrows) located closely to the entrance of the Meandro Abismante Cave (white arrow); b Spelunconiscus sp. isopods present in the same cave before contamination; c dense biofilm covering the drainage bottom, as well as blackened fragments apparently from the contamination by the landfill
Furthermore, the economic and social development of the region results in a disordered exploration of natural resources and the substitution of important forest areas of Cerrado by grassland areas for livestock. This is the main threat to this biome, considered one of the hotspots for the conservation of world biodiversity (Myers et al. 2000). Nowadays, pastures for extensive livestock occupy almost 42% of the native area of Cerrado, and deforestation occurs at 30,000 km2/year, a rate superior to those found for the Amazon region (Klink and Machado 2005). A recent study on different land use and its influence on the composition of terrestrial invertebrates from the Lagoa Santa Karst, pastures were present around all the studied caves, representing up to 76% of the buffer area (Pellegrini et al. 2016). In this work, the authors indicate the presence of limestone outcrops as the most determining factor for the composition of the subterranean community, as previously mentioned. Among other factors, this occurs due to the low anthropic influence, since the karst depressions in areas adjacent to the caves and outcrops are frequently used for crops and livestock (Pellegrini et al. 2016). The substitution of the native forests by monocultures generates a series of environmental problems like fragmentation, biodiversity loss, introduction of invasive species, erosion and soil degradation, water pollution, changes in the fire regime (natural impact of this biome), imbalances in the carbon cycle and probable modification of the regional climate (Klink and Machado 2005). Such alterations of epigean landscape may affect directly or indirectly the caves, thus hampering the maintenance of the environmental stability characteristic of subterranean environments.
Since the Lagoa Santa Karst lies close to large metropolitan areas, it presents a high density of roads and highways that makes possible the flow of people and services. Studies demonstrate that the establishment of a road network is among the main anthropic activities that affect directly or indirectly the dynamics of ecological processes, altering the physical, chemical, and biological properties of the environment (Coffin 2007; Forman and Alexander 1998). Direct impacts comprise the death of animals by trampling (Clevenger et al. 2003; Coelho et al. 2008) and isolation of populations (Shepard et al. 2008). Indirectly the roads favor the anthropic access and occupation in contiguous areas (Freitas et al. 2010) and alter the surface water runoff, thus promoting erosion in the soil and carrying the particulate material to other areas. Additionally, the facility of access makes marginal areas more susceptible to the fire (Laurance et al. 2009). The existence of these structures fractioning natural habitats interrupts the genetic flow, what may alter the permanence of populations in fragmented areas (Epps et al. 2005). Despite the absence of studies correlating the presence of roads and the effects of them over the hypogean fauna, it is likely that these anthropic actions may modify the structure of cave communities. Among the possible impacts, one may cite the alteration of the dynamics of trophic resources for the cave, what may severely influence the preservation and maintenance of the fauna. The proximity of roads may still generate constant vibrations in the caves, facilitate the accumulation of garbage and other anthropic wastes, besides altering the adjacent external landscape.
The presented threats represent only part of the risks to the maintenance of fauna and stability of subterranean environments. Since the Lagoa Santa Karst is inserted in an important Brazilian economic region and within the biome most used by agricultural activities, several other impacts may affect the caves and their surroundings (for example, forest fires, land invasion, pesticide contamination, vandalism and graffiti in caves, among others). Due to the social and economic plurality, preventing and mitigating the mentioned impacts is beyond the application of public policies and delimitation of areas for conservation. The awareness of the society and the assignment of value to the subterranean environment through environmental education are essential to guarantee the sustainable management of this karst.
References
Auler AS (1998) Base-level changes inferred from cave paleoflow analysis in the Lagoa Santa Karst, Brazil. J Cave Karst Stud 60(1):58–62
Auler AS and Piló LB (2015) Lagoa Santa Karst: cradle of Brazilian cave studies. In: Vieira BC et al (eds) Landscapes and Landforms of Brazil. World geomorphological landscapes, pp 183–190
Bahia GR, Ferreira RL (2005) Influência das características físico-químicas e da matéria orgânica de depósitos recentes de guano de morcegos na riqueza e diversidade de invertebrados de uma caverna calcária. Revista Brasileira de Zoociências 1(7):165–180
Berbert-Born MLC (2000) The Lagoa Santa karst. Sítios Geológicos e Paleontológicos do Brasil. http://www.unb.br/ig/sigep/sitio015/sitio015.htm. Accessed 25 Oct 2016
Berbert-Born MLC, Horta LCS (1994) Informações básicas para gestão territorial: espeleologia – inventário de cavidades naturais da região Matozinhos-Mocambeiro. Companhia de Pesquisa de Recursos Minerais, Belo Horizonte
Berbert-Born MLC, Horta LCS, Dutra GM (1998) APA Carste de Lagoa Santa: Projeto vida - volume III Levantamento espeleológico. Companhia de Pesquisa de Recursos Minerais. Belo Horizonte
Berta A (1982) Cerdocyon thous. Mamm Species Arch 186:1–4
Bourne JD, Cherix D (1978) Note sur l’écophase souterraine de Triphosa dubitata L. (Lep. Geometridae) et Scoliopteryx libatrix L. (Lep. Noctuidae). Bulletin de la Société Vaudoise des Sciences Naturelles 354(74):147–156. https://doi.org/10.5169/seals-277411
Brescovit AD, Ferreira RL, Souza-Silva M, Rheims C (2012) Brasilomma gen. nov., a new prodidomid genus from Brazil (Araneae, Prodidomidae). Zootaxa (Auckland. Print) (3572):23–32
Bücherl W (1961) Aranhas do gênero Loxosceles e o Loxoscelismo na América. Ciência e Cultura 13(4):213–224
CamposFilho IS, Araujo PB, Bichuette ME, Trajano E, Taiti S (2014) Terrestrial isopods (Crustacea: Isopoda: Oniscidea) from Brazilian caves. Zool J Linn Soc 172(2):360–425
CamposFilho IS, Araujo PB, Bichuette ME, Taiti S (2016) Three new species of terrestrial isopods (Crustacea, Isopoda, Oniscidea) from Brazilian caves. Nauplius 24:e2016001
Carchini G, Rampini M, Sbordoni V (1994) Life cycle and population ecology of the cave cricket Dolichopoda geniculata (Costa) from Valmarino cave (Central Italy). Int J Speleol 23:203–218. https://doi.org/10.5038/1827-806X.23.3.6
CBIC (2013) Câmara Brasileira da Indústria da Construção http://www.cbicdados.com.br/menu/materiais-de-construcao/cimento. Accessed 26 Oct 2016
CEBS-UFLA (2016) Center of studies on subterranean biology from Federal University of Lavras, Minas Gerais, Brazil (www.biologiasubterranea.com.br)
Chen C, Habert G, Bouzidi Y, Jullien A (2010) Environmental impact of cement production: detail of the different processes and cement plant variability evaluation. J Clean Prod 18(5):478–485
Clevenger AP, Chruszcz B, Gunson KE (2003) Spatial patterns and factors influencing small vertebrate fauna road-kill aggregations. Biol Conserv 109(1):15–26
Coelho IP, Kindel A, Coelho AVP (2008) Roadkills of vertebrate species on two highways through the Atlantic Forest Biosphere Reserve, southern Brazil. Eur J Wildl Res 54:689–699
Coelho-Finamore JM, Freitas VC, Assis RR, Melo MN, Novozhilova N, Secundino NF, Pimenta PF, Turco SJ, Soares RP (2011) Leishmania infantum: Lipophosphoglycan intraspecific variation and interaction with vertebrate and invertebrate hosts. Int J Parasitol 41(3):333–342
Coffin AW (2007) From roadkill to road ecology: A review of the ecological effects of roads. J Transp Geogr 15:396–406
Costa-Lima A (1940) Um novo grilo cavernícola de Minas Gerais (Orthoptera: Grylloidea: Phalangositidae). Papéis Avuls Zool 6:43–50
Culver DC, Pipan T (2009) The biology of caves and other subterranean habitats. Oxford University Press, Oxford
Culver DC, Sket B (2002) Biological monitoring in caves. Acta Carsologica 31(1):55–64
Dantas AE, Horta CCR, Martins TM, Carmo AO, Oliveira-Mendes BBR, Goes AM, Kalapothakis E, Gomes DA (2014) Whole venom of Loxosceles similis activates caspases-3,-6,-7, and-9 in human primary skin fibroblasts. Toxicon 84:56–64
Dantas AE, Carmo AO, Horta CCR, Leal HG, Oliveira-Mendes BBR, Martins APV, Chavez-Olortegui C, Kalapothakis E (2016) Description of Loxtox protein family and identification of a new group of Phospholipases D from Loxosceles similis venom gland. Toxicon 120:97–106
Decu V (1986) Some considerations on the bat guano synusia. Travaux de l’Institute de Spéologie “Emile Racovitza” (25):41–51
Delgado A (1966) Investigacion ecológica sobre Loxosceles rufipes (Lucas) en la region costera del Perú. Mem Inst Butantan 33(3):683–688
DeMatteo KE, Loiselle BA (2008) New data on the status and distribution of the bush dog (Speothos venaticus): evaluating its quality of protection and directing research efforts. Biol Conserv 141(10):2494–2505
Doube BM (1986) Spatial and temporal organization in communities associated with dung pads and carcasses. In: Gee JHR, Giller PS (eds) Organization of communities past and present. Blackwell Scientific Publications, Oxford, pp 255–280
Emmons LH, Leite YL, Kock D, Costa LP (2002) A review of the named forms of Phyllomys (Rodentia: Echimyidae) with the description of a new species from coastal Brazil. Am Mus Novit 1–40
Epps CW, Palsbøll PJ, Wehausen JD, Roderick GK, Ramey RR, Mccullough DR (2005) Highways block gene flow and cause a rapid decline in genetic diversity of desert bighorn sheep. Ecol Lett 8(10):1029–1038
Ferreira RL, Martins RP (1998) Diversity and distribution of spiders associated with bat guano piles in Morrinho cave (Bahia State, Brazil). Divers Distrib 4:235–241
Ferreira RL, Martins RP (1999) Trophic structure and natural history of bat guano invertebrate communities, with special reference to Brazilian caves. Trop Zool 12(2):231–252. https://doi.org/10.1080/03946975.1999.10539391
Ferreira RL, Pompeu PS (1997) Fatores que influenciam a riqueza e a diversidade da fauna associada a depósitos de guano na gruta Taboa, Sete Lagoas, Minas Gerais, Brasil. O Carste 2:30–33
Ferreira RL, Prous X, Machado SF, Martins RP (2005) Population dynamics os Loxosceles similis (Moenkhaus, 1898) in a Brazilian dry cave: a new method for evaluation of population size. Revista Brasileira de Zoociências 7(1):129–141
Ferreira RL (2000) Lixeiras de formigueiros: recursos adicionais em sistemas cavernícolas?. O Carste 3(12):154–158
Ferreira RL, Martins RP, Yanega D (2000a) Ecology of bat guano arthropod communities in a Brazilian dry cave. Ecotropica 6:105–116
Ferreira RL, Nonaka E, Rosa C (2000b) Riqueza e abundância de fungos associados a guano de morcegos na gruta da Lavoura. O Carste 12:46–51
Ferreira RL, Prous X, Martins RP (2007) Structure of bat guano communities in a Brazilian dry cave. Trop Zool 20:55–74
Ferreira RL, Martins VM, Paixão EA, Silva MS (2015) Spatial and temporal fluctuations of the abundance of Neotropical cave-dwelling moth Hypena sp. (Noctuidae, Lepidoptera) influenced by temperature and humidity. Subterr Biol 16:47–60
Filho JDA, Galati EAB, Falcão AL (2007) Nyssomyia intermedia (Lutz & Neiva, 1912) and Nyssomyia neivai (Pinto, 1926) (Diptera: Psychodidae: Phlebotominae) geographical distribution and epidemiological importance. Memórias do Instituto Oswaldo Cruz 102(4):481–487
Fischer ML (1996) Biologia e ecologia de Loxosceles intermedia (Mello-Leitão, 1934) (Aranae, Sicariidae), no município de Curitiba, PR. Dissertation, Universidade Federal do Paraná
Foelix RF (1987) Biology of spiders. Harvard University Press, Cambridge
Fontanetti CS (1996) Description of three cave diplopods of Pseudonannolene Silvestri (Diplopoda, Pseudonannolenida, Pseudonannolenidae). Revista Brasileira de Zoologia 13(2):427–433
Forman RT, Alexander LE (1998) Roads and their major ecological effects. Annu Rev Ecol Syst 29:207–231
Freitas SR, Hawbaker TJ, Metzger JP (2010) Effects of roads, topography, and land use on forest cover dynamics in the Brazilian Atlantic Forest. For Ecol Manag 259(3):410–417
Gama EM, Matias GP (2015) Hidrogeologia e os geossistemas ferruginosos. In. Carmo FF, Kamino LHY (eds) Geossistemas ferruginosos do Brasil: Áreas prioritárias para conservação da diversidade geológica e biológica, patrimônio cultural e serviços ambientais. 3i Editora, Belo Horizonte, pp 103–124
Gilpin ME, Hanski IA (1991) Metapopulation dynamics: empirical and theoretical investigations. Academic Press, London
Gil-Santana HR, Zeraik SO, Costa LAA (1999) Sinonimização dos gêneros Amilcaria Wygodzinsky, 1950 e Mayemesa Wygodzinsky, 1945 e descrição do macho de M. lapinhaensis (Wygodzinsky, 1950), Combinação Nova (Hemiptera, Reduviidae, Emesinae) Boletim do Museu Nacional. Zoologia 400:1–10
Gnaspini P (2012) Guano communities. In: White WB, Culver DC (Org.) Encyclopedia of caves, 2 edn. Academic Press/Elsevier, Oxford, Waltham, pp 357–364
Gomes FTMC, Ferreira RL, Jacobi CM (2000) Comunidade de artrópodes de uma caverna calcária em área de mineração: composição e estrutura. Revista Brasileira de Zoociências 3(2):77–96
Grelle CE, Fonseca MT, Moura RT, Aguiar L (1997) Bats from karstic area on Lagoa Santa, Minas Gerais: a preliminary survey. Chiropt Neotrop 3(1):68–70
Guimarães MM, Ferreira RL (2014) Morcegos cavernícolas do Brasil: novos registros e desafios para conservação. Revista Brasileira de Espeleologia 2(4):1–33
Hamilton-Smith E (2004) Tourist caves. In: Gunn J (ed) Encyclopedia of caves and karst science. Taylor and Francis, New York, pp 1554–1561
Harris JA (1970) Bat-guano cave environment. Science 169:1342–1343
Harrison S, Ross SJ, Lawton JH (1992) Beta diversity on geographic gradients in Britain. J Anim Ecol 61:151–158
Hermann G, Kohler HC, Duarte JC Carvalho PGS (1998) APA Carste de Lagoa Santa - Volume III: meio biótico. IBAMA, Belo Horizonte
Herzig-Straschil B, Robinson GA (1978) On the ecology of the fruit bat, Rousettus aegyptiacus leachi (A. Smith, 1829) in the Tsitsikama coastal National Park. Koedoe 21:101–110
Hutchinson P (1950) Vertebrate excretion (cave guano). Bull Am MusM Nat Hist 96:381–485
IBGE - Instituto Brasileiro de Geografia e Estatística (2016) Estimativas populacionais para os municípios brasileiros em 01/07/2016. http://www.ibge.gov.br/home/estatistica/populacao/estimativa2016/estimativa_tcu.shtm. Accessed 25 Apr 2017
Iniesta LFM, Ázara LN, Souza-Silva M, Ferreira RL (2012) Biodiversidade em sete cavernas no Parque Estadual do Sumidouro (Lagoa Santa, MG). Revista Brasileira de Espeleologia 2(2):18–37
Klimchouk A (2004) Towards defining, delimiting and classifying epikarst: its origin, processes and variants of geomorphic evolution. Speleogenesis Evol Karst Aquifers 2(1):1–13
Klink CA, Machado RB (2005) Conservation of the Brazilian Cerrado. Conserv Biol 19(3):707–713
Koleff P, Gaston KJ, Lennon JJ (2003) Measuring beta diversity for presence–absence data. J Anim Ecol 72:367–382
Kumar SS, Singh NA, Kumar V, Sunisha B, Preeti S, Deepali S, Nath SR (2008) Impact of dust emission on plant vegetation in the vicinity of cement plant. Environ Eng Manag J 7(1):31–35
Kunz TH (1982) Roosting ecology of bats. In: Kunz TH (ed) ecology of bats. Plenum Press, New York, pp 1–55
Larson KL, Casagrande D, Harlan SL, Yabiku ST (2009) Residents’ yard choices and rationales in a desert city: social priorities, ecological impacts, and decision tradeoffs. Environ Manag 44(5):921–937
Laurance WF, Goosem M, Laurance SG (2009) Impacts of roads and linear clearings on tropical forests. Trends Ecol Evol 24(12):659–669
Lienhard C, Ferreira RL (2015) Review of Brazilian cave psocids of the families Psyllipsocidae and Prionoglarididae (Psocodea: Psocoptera: Trogiomorpha) with a key to the South American species of these families. Rev Suisse Zool 122(1):121–142
MacDonald D, Crabtree J, Wiesinger G (2000) Agricultural abandonment in mountain areas of Europe: Environmental consequences and policy response. J Environ Manag 59(1):47–69. https://doi.org/10.1006/jema.1999.0335
Machado FS, Lima IJ, Lopes APM, Moura AS, Abreu TCK (2016) New occurrences and biological aspects to four species of rodents (Mammalia: Cricetidae) from Brazil. Revista Agrogeoambiental 8(2):35–51. http://dx.doi.org/10.18406/2316-1817v8n22016800
Mahnert V (2001) Cave-dwelling pseudoscorpions (Arachnida, Pseudoscorpiones). Rev Suisse Zool 108(1):95–148
Matavelli R, Campos AM, Feio RN, Ferreira RL (2015) Occurrence of Anurans in Brazilian caves. Acta Carsologica 44(1):107–120
Mazza G, Reboleira ASPS, Gonçalves F, Aquiloni L, Inghilesi AF, Spigoli D, Stoch F, Taiti S, Gherardi F, Tricarico E (2014) A new threat to groundwater ecosystems: first occurrences of the invasive crayfish Procambarus clarkii (Girard, 1852) in European caves. J Cave Karst Stud 76(1):62–65
Mews CM, Sperber CF (2008) A new species of Endecous Saussure, 1878 and redescription of Endecous cavernicolus Costa-Lima, 1940 (Orthoptera: Grylloidea: Phalangopsidae). Stud Neotrop Fauna Environ 43(2):159–167
Mitchell RW (1969) A comparison of temperate and tropical cave communities. Southwest Nat 14(1):73–88. https://doi.org/10.2307/3669249
Myers N, Mittermeier RA, Mittermeier CG, Fonseca GA, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403(6772):853–858
Novak T, Sambol J, Janžekovič F (2004) Faunal dynamics in the Železna Jama cave. Acta Carsologica 33(2):249–267
Oliveira SMPD, Moraes BAD, Gonçalves CA, Giordano-Dias CM, d’Almeida JM, Asensi MD, Mello RP, Brazil RP (2000) Prevalence of the microbiota in the digestive tract of wild-caught females of Lutzomyia longipalpis (Lutz & Neiva) (Diptera: Psychodidae). Rev Soc Bras Med Trop 33(3):319–322
Oss HG, Padovani AC (2003) Cement manufacture and the environment part II: environmental challenges and opportunities. J Ind Ecol 7(1):93–126
Pellegrini T, Sales LP, Aguiar P, Ferreira RL (2016) Linking spatial scale dependence of land-use descriptors and invertebrate cave community composition. Subterr Biol 18:17–38
Pena MS, Salgado NC, Coelho AC (2005) New species of Thaumastus from Lagoa Santa region, Minas Gerais State, Brazil (Mollusca, Gastropoda, Pulmonata, Bulimuloidea). Iheringia Série Zoologia 95(1):21–24
Pinto-da-Rocha R (1995) Sinopse da fauna cavernícola do Brasil (1907–1994). Papéis avulsos de Zoologia 39:61–73
Prous X, Ferreira RL, Martins RP (2004) Ecotone delimitation: epigean-hypogean transition in cave ecosystems. Austral Ecol 29:374–382
Prous X, Ferreira RL, Jacobi CM (2015) The entrance as a complex ecotone in a Neotropical cave. Int J Speleol 44:177–189
Rabelo LM (2016) Ecologia e conservação de cavernas no eixo Centro-norte de Minas Gerais, Dissertation, Universidade Federal de Lavras
Sábio PB, Andrade AJ Galati EA (2015) Redescription of Lutzomyia (Lutzomyia) renei Martins, Falcão & Silva, 1957 (Diptera: Psychodidae: Phlebotominae). Zootaxa, 3999(4):589–599
Schubart O (1956) “Leptodesmidae” brasileiras II. O gênero Obiricodesmus (Diplopoda, Proterospermophora). Revta Bras Biol 16(3):341–348
Shepard DB, Kuhns AR, Dreslik MJ, Phillips CA (2008) Roads as barriers to animal movement in fragmented landscapes. Anim Conserv 11(4):288–296
Silva CR, Percequillo AR, Ximenes GL, Vivo M (2003) New distributional records of Blarinomys breviceps (Winge, 1888) (Sigmodontinae, Rodentia). Mammalia 67(1):147–152
Silveira PSA, Bernardi, LFO, Pepato AR (2015) New records of the genus Whartonia (Acari, Leeuwenhoekiidae) associated with the bat Carollia perspicillata from southeastern Brazil. Check List 11(6):1793. http://dx.doi.org/10.15560/11.6.1793
Simões MH (2013) Invertebrados cavernícolas: subsídios para a determinação de cavernas e áreas prioritárias para a conservação no noroeste de Minas Gerais. Dissertation, Universidade Federal de Lavras
Simões MH, Souza-Silva M, Ferreira RL (2015) Cave physical attributes influencing the structure of terrestrial invertebrate communities in Neotropics. Subterr Biol 16:103–121
Souza MFVR (2012) Diversidade de invertebrados subterrâneos da região de Cordisburgo, Minas Gerais: subsídios para a definição de cavernas prioritárias para a conservação e para o manejo biológico de cavidades turisticas. Dissertation, Universidade Federal de Lavras
Souza-Silva M, Bernardi LFO, Ferreira RL (2005) Caracterização sistêmica da gruta da Lavoura (Matozinhos, MG): aspectos topoclimáticos, tróficos e biológicos. In: XXVIII Congresso Brasileiro de Espeleologia, Campinas, pp 109–115
Souza-Silva M, Martins RP, Ferreira RL (2011) Trophic dynamics in a neotropical limestone cave. Subterr Biol 9:127–138
Strayer DL, Power ME, Fagan WF, Pickett STA, Belnap J (2003) A classification of ecological boundaries. Bioscience 53(8):723–729
Talamoni SA, Coelho DA, Dias-Silva LH, Amaral AS (2013) Bat assemblages in conservation areas of a metropolitan region in Southeastern Brazil, including an important karst habitat. Braz J Biol 73(2):309–319
Tavares VC, Aguiar LM, Perini FA, Falcão FC, Gregorin R (2010) Bats of the state of Minas Gerais, southeastern Brasil. Chiropt Neotrop 16(1):675–705
Tobin BW, Hutchins BT, Schwartz BF (2013) Spatial and temporal changes in invertebrate assemblage structure from the entrance to deep-cave zone of a temperate marble cave. Int J Speleol 42(3):203–214. https://doi.org/10.5038/1827-806X.42.3.4
Torquetti CG, Assis MAC, Carmo SSA, Talamoni SA (2013) Observations of a diurnal roost of the white-winged vampire bat Diaemus youngi in a karstic area of southeastern Brazil. Chiropt Neotrop 19(1):1147–1150
Trajano E, Bichuette ME (2010) Diversity of Brazilian subterranean invertebrates, with a list of troglomorphic taxa. Subterr Biol 7:1–16
Trajano E, Golovatch SI, Geoffroy JJ, Pinto-da-Rocha R, Fontanetti CS (2000) Synopsis of Brazilian cave-dwelling millipedes (Diplopoda). Papéis Avulsos de Zoologia 41(18):259–287
White EL, Aron G, White WB (1986) The influence of urbanization of sinkhole development in central Pennsylvania. Environ Geol Water Sci 8(1–2):91–97
Whittaker RH (1960) Vegetation of the Siskiyou Mountains, Oregon and California. Ecol Monogr 30:279–338
Wygodzinsky P (1950) Sobre alguns “Emesinae” (Reduviidae, Hemiptera). Revta bras Biol 10(1):73–78
Zampaulo RA (2010) Diversidade de invertebrados cavernícolas na província espeleológica de Arcos, Pains e Doresópolis (MG): subsídios para a determinação de áreas prioritárias para a conservação. Dissertation, Universidade Federal de Lavras
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Ferreira, R.L., de Oliveira, M.P.A., Souza-Silva, M. (2020). Biospeleology of the Lagoa Santa Karst. In: S. Auler, A., Pessoa, P. (eds) Lagoa Santa Karst: Brazil's Iconic Karst Region. Cave and Karst Systems of the World. Springer, Cham. https://doi.org/10.1007/978-3-030-35940-9_10
Download citation
DOI: https://doi.org/10.1007/978-3-030-35940-9_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-35939-3
Online ISBN: 978-3-030-35940-9
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)