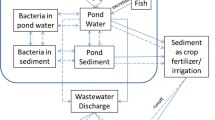
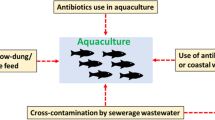
Abstract
Antibiotics are used abusively in humans and agriculture, including aquaculture, making them omnipresent in the environment, posing health risks to aquatic animals and humans. However, how antibiotics affect the anatomy and physiology of fish, including the presence of antibiotic-resistant bacteria (ARB) and antibiotic resistance genes (ARGs) in fish in Africa are currently poorly understood. This chapter synthesized the available literature on the potential risks of antibiotics on cultured fish and human health from Africa. The limited available studies indicate that few antibiotics improve fish growth but do not affect their survival rate. Antibiotics application on fish cause body malformation and damage, which induce hepatotoxicity, nephrotoxicity, leukocytosis, hematopoiesis, and lymphocytosis, suggesting sustained toxic effects. Antibiotics use in fish and other foods like shrimps and vegetables together with antibiotics residues in the aquatic environment have led to an increase in the prevalence of ARB and ARGs, posing a human health risk in various African countries. Limited studies indicate higher residues of antibiotics in cultured fish, which exceed the maximum limits set by the Codex Alimentarius Commission and the World Health Organization. Thus, policies and mechanisms for limiting the use of antibiotics in food animals to protect human health in Africa are urgently needed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
8.1 Introduction
It is globally undoubtedly accepted that antibiotics have saved many lives and eased the suffering of many millions of animals (Byarugaba 2004). However, antibiotics, of either natural or synthetic origin, are used abusively in human, livestock, agriculture, and aquaculture both to prevent proliferation and destroy bacteria (Mehdi et al. 2018). Consequently, antibiotics exist ubiquitously in the environment and are currently deemed as a global pandemic problem posing a health risk to aquatic animals and humans. Unfortunately, the risks caused by antibiotics globally are expected to continue because, between 2000 and 2015 their consumption increased 65% from 21.1 to 34.8 billion defined daily doses (DDDs), and the antibiotic consumption rate increased 39% from 11.3 to 15.7 DDDs per 1000 inhabitants per day (Klein et al. 2018). Astonishingly, the increase in consumption of antibiotics was driven by low- and middle-income countries (LMICs), a characteristic possessed by the majority of African countries.
The African continent poses peculiar features regarding antibiotics consumption. First, most African countries are generally characterized by poverty, ignorance, poor sanitation, hunger and malnutrition, poor and inadequate health care systems, civil conflicts and bad governance (Byarugaba 2004), coupled with an inappropriate prescription as well as self-medication and free sale of antibiotics (Sanou et al. 2018). Secondly, most African countries have weak regulatory agencies and absence/weak regulations concerning antibiotics usage. Accordingly, antibiotics are indiscriminately given as over-the-counter drugs at community pharmacies (Mukonzo et al. 2013), which have been strongly correlated with antibiotic-resistant bacteria (ARB) and antibiotics resistance genes (ARGs) in aquatic animals such as fish and humans in LMICs (Alsan et al. 2015). The lack of regulatory agencies in African countries have caused the indiscriminate use of antibiotics in human for disease treatments and as therapeutic and growth promoters in livestock, agriculture, and aquaculture production. As growth promoters, antibiotics are believed to improve feed conversion, promote animal growth, and reduce mortality and morbidity rates resulting from clinical and subclinical illnesses (Foka et al. 2018).
Moreover, antibiotics applied in fish are poorly absorbed in the intestine, and subsequently are released into the aquatic environments where they selectively cause ARB and ARGs (Fu et al. 2017). Thus, antibiotic resistance in bacteria and genes that cause diseases in man is an issue of significant concern, which is expected to become the leading global cause of death by 2050 (O’Neill 2016). Although the misuse of antibiotics in human medicine is the principal cause of ARB and ARBs in Africa, the use of antibiotics in food animals and their subsequent release into the aquatic environments are contributory factors (Barton 2000; Goutard et al. 2017; Adegoke et al. 2018). Apart from ARB and ARGs, antibiotics also cause human health risk due to their residue amounts in various contaminated foods consumed in Africa (Darwish et al. 2013).
The increasing human population in Africa has led to an increase in reliance on aquaculture to supply safe, reliable, and economical food, contributing 10% of the total global population engaged in fisheries and aquaculture, second only to Asia with 84% (FAO 2018). In some African countries such as Ghana and Sierra Leone, fish contributes or exceeds, 50% of total animal protein intake (FAO 2016). Aquaculture production, mainly from catfish and tilapia, accounted for 17–18% of total fish production in Africa (Fig. 8.1), with a general increasing trend (Fig. 8.2) (FAO 2018). The per capita fish food consumption for Africa was reported as 9.9 kg/year in 2015, partly contributed by unreported data (FAO 2018).
Aquaculture contribution to total fish production (excluding aquatic plants). Source: FAO (2018)
Average annual growth rate of aquaculture production by volume (excluding aquatic plants). Source: FAO (2018)
Although the available information suggests minimal use of antibiotics in aquaculture in African countries, potential contamination of fish from fertilizers used in animals treated with antibiotics is unavoidable (Shah et al. 2012; Wamala et al. 2018), because most farmers fertilize their ponds. However, the effects of antibiotics on fish anatomy and physiology from an African perspective are currently poorly understood, and information on ARB and ARGs in fish and humans due to fish exposure is currently scattered and unfocused. Furthermore, antibiotics are increasingly used in humans and other food animals, with a concomitant prevalence of ARB and ARGs in LMICs (Bernabé et al. 2017). It has been shown that infections caused by ARB may increase health care costs due to patients’ need for more diagnostic tests, more extended hospitalization periods, and poor treatment outcomes (Nyasulu et al. 2012). Despite all these, little attention has been directed towards understanding the antibiotics residues in fish, other food animals, and humans in Africa (Adegoke et al. 2018).
For the first time, this chapter organizes and synthesizes the available information in the literature on the potential risks of antibiotics on cultured fish and human health from Africa. The chapter assesses the effects of antibiotics on fish growth performance, feed utilization, hepatotoxicity and nephrotoxicity, and hematological parameters. It further evaluates the potential human health risks caused by the existence of ARB and ARGs in fish and other consumed foods, in addition to direct risks due to the consumption of fish products containing antibiotics residues. The information generated informs policies to limit the use of antibiotics in food animals by enforcing policies, which regulate their use in Africa to safeguard human health.
8.2 Effects of Antibiotics on Growth Performance and Feed Utilization
Growth performance , feed utilization, survival rate, and body development are important production attributes to fish growers because they affect directly the yield and economics of an aquaculture enterprise. Thus, understanding the effects of antibiotics on these aspects in cultured fish deserves a peculiar consideration. In Africa, very few studies have currently used antibiotics to study growth and related parameters on fish. The literature visited indicated oxytetracycline studied in O. niloticus (El-Sayed et al. 2014), oxytetracycline and florfenicol in O. niloticus ♀ × O. aureus ♂ hybrids (Reda et al. 2013), and chloramphenicol researched in O. niloticus (Shalaby et al. 2006) and African catfish, Clarias gariepinus (Nwani et al. 2014) were the only antibiotics used. Results from these few studies indicated improved growth of treated fish compared to controls. For example, the growth performance of O. niloticus increased significantly with increasing levels of chloramphenicol (Shalaby et al. 2006). Moreover, feeding diets containing oxytetracycline and florfenicol in O. niloticus ♀ × O. aureus ♂ hybrids (Reda et al. 2013) and O. niloticus (El-Sayed et al. 2014) resulted into faster growth performance in treated than control fish.
The precise reasons for the enhanced growth performance of fish after antibiotics administration are subject to scrutiny. Increased growth has been attributed to higher feed consumption and reduced feed conversion ratio. Indeed, the growth rate was increased in O. niloticus treated with chloramphenicol (Shalaby et al. 2006), oxytetracycline and florfenicol (Reda et al. 2013), and oxytetracycline (El-Sayed et al. 2014), in which feed consumption and intake were increased, while feed conversion ratio was reduced. Moreover, apparent protein, lipid, carbohydrate, and energy digestibility were increased in O. niloticus fed on chloramphenicol (Shalaby et al. 2006). These results should be interpreted with caution due to limited studies and the existence of contradicting results elsewhere. It has been recently shown that antibiotics, particularly oxytetracycline, do not cause growth promotion in finfish (Trushenski et al. 2018) and causes multiple effects in Nile tilapia including reduced nutrients digestibility and digestive enzymes (Limbu et al. 2018), growth performance (Limbu et al. 2019a; Limbu et al. (2019b), protein and feed efficiencies (Limbu et al. 2019b).
Like growth performance , studies conducted on the effects of antibiotics on survival rate are also limited. The results obtained in the limited studies do not show any influence of antibiotics on fish survival rate. Exposure to dietary oxytetracycline (El-Sayed et al. 2014) and chloramphenicol (Shalaby et al. 2006) both in O. niloticus and chloramphenicol in C. gariepinus (Nwani et al. 2014) did not significantly influence survival rate of treated fish relative to control. Results detailing the effects of antibiotics on body development are based on C. gariepinus exposed to chloramphenicol baths (Nwani et al. 2014). In this study, treated fish had abnormal behavioral changes at higher concentration of chloramphenicol. The fish swam near the water surface, lost equilibrium, swam erratically, had hyperactivity, and stayed motionless on the bottom of the culture tank. Furthermore, exposed fish had clinical toxic signs such as lightening in skin color of the body surface, erosion of fins and tails, and increased mucus secretions from the whole body. These results indicate that antibiotics application on fish leads to body malformation and damage, which may lead to physiological and metabolic dysfunctions affecting fish health.
8.3 Effects of Antibiotics on Fish Health
8.3.1 Oxidative Stress, Hepatotoxicity, and Nephrotoxicity
Antibiotics used in fish production induce oxidative stress , which affects antioxidant enzymes that protect fish body from reactive oxygen species (ROS) (Limbu et al. 2018). Changes in the activities of antioxidant enzymes indicate an imbalance in the ROS production in the body. Limited studies have been conducted in Africa to assess the antioxidant capacity of fish exposed to antibiotics. A study conducted by Olaniran et al. (2018) indicated reduced glutathione S transferase (GST) and superoxide dismutase (SOD) activities in C. gariepinus exposed to tetracycline. The decreased antioxidants in fish treated with antibiotics may be caused by an excess accumulation of free radicals, such as superoxide anion and hydrogen peroxide beyond the antioxidant capacity to counteract (Yonar et al. 2011; Yonar 2012; Oliveira et al. 2013; Wang et al. 2014). This may oxidize amino acids and cofactors, which may affect the general fish health. Malondialdehyde (MDA) is the main oxidative product of peroxidized polyunsaturated fatty acids and is an important index of lipid peroxidation. The extent of lipid peroxidation is measured in tissues by quantification of thiobarbituric acid reactive substances (TBARS) expressed as MDA concentration (Nunes et al. 2015). Lipid peroxidation is the initial step of cellular membrane damage caused by xenobiotics such antibiotics (Yonar et al. 2011; Yonar 2012). Limited studies have reported on lipid peroxidation using MDA. Reduced MDA level was reported in C. gariepinus exposed to tetracycline, indicating lack of lipid peroxidation (Olaniran et al. 2018).
The liver of fish and other vertebrates is known for its digestive, metabolism, storage, and detoxification functions. The introduction of antibiotics in fish body through medicated feeds may cause liver damage effects that might impair its functions (Dobšíková et al. 2013). Limited studies have reported on the effects of antibiotics on the hepatosomatic index (HSI) as an indicator of hepatotoxicity in fish. The chloramphenicol medicated feeds used in O. niloticus did not cause significant variations in HSI in experimental compared to control fish (Shalaby et al. 2006). Few studies conducted limit the ability to draw logical conclusions. Studies conducted in other parts of the world showed HSI was reduced (Refstie et al. 2006; Limbu et al. 2018) and increased (Topic Popovic et al. 2012; Nakano et al. 2018; Trushenski et al. 2018) in different fish species.
The amount of circulating proteins reflects an organism’s physiology. Plasma proteins and glucose in the circulatory system transport lipids, hormones, vitamins, and minerals and regulate cellular activities, functioning of the immune system, and blood clotting. Imbalances in the plasma protein and glucose counts indicate liver damage, which interferes with its normal functions. A dose-dependent increase in plasma protein levels was reported in O. niloticus exposed to chloramphenicol diet, indicating osmoregulatory dysfunction , hemodilution, or tissue damage surrounding blood vessels (Shalaby et al. 2006). Moreover, plasma glucose increased significantly with increasing levels of chloramphenicol (Shalaby et al. 2006). Although limited, this study indicated that antibiotic medications in cultured fish lead to disturbances in plasma proteins and glucose, which indicate hepatotoxicity .
Except for plasma proteins and glucose, liver dysfunction is manifested by increased levels of specific serum enzymes activities, which signal cellular leakage and impaired liver cell membrane integrity and function. Alanine transaminase (ALT) and aspartate aminotransaminase (AST) are required in the metabolism of amino acids, and their change in activities reflect their leakage into the blood after cytolysis in the liver (Han et al. 2014). Thus, AST and ALT enzymes are commonly used to detect hepatotoxicity due to xenobiotics exposure (Saravanan et al. 2012). Studies from Africa assessing liver damage in cultured fish after antibiotics by using AST and ALT have reported contrasting results. The administration of florfenicol diet in O. niloticus did not alter ALT activity (Reda et al. 2013).
On the contrary, the activities of AST and ALT in plasma decreased significantly with increasing levels of dietary chloramphenicol in O. niloticus (Shalaby et al. 2006). The observed decrease in AST and ALT activities in fish is either due to insufficient detoxification mechanisms to prevent the toxicity action of antibiotics on these enzymes or failure of liver damaged cells to synthesize AST and ALT proteins (Saravanan et al. 2012). On the other hand, oxytetracycline-supplemented diets increased significantly ALT activity in O. niloticus (Reda et al. 2013). Increased ALT activity is due to the ability of antibiotics to accumulate or bind to different cells leading to damage and disintegration of cells, releasing ALT into blood circulation, suggesting impaired liver function.
Histopathological effects provide a quick diagnosis to detect abnormalities in various fish tissues and organs after antibiotics exposure. Antibiotics use indicate species- and antibiotic-specific histopathological effects in the liver and kidney of treated fish. Feeding dietary oxytetracycline and florfenicol in O. niloticus (Reda et al. 2013) induced various pathological alterations in liver and kidney of treated fish. Moreover, both dietary oxytetracycline and florfenicol decreased creatinine in the treated O. niloticus than the control fish (Reda et al. 2013). The existence of several histopathological damages in the liver of treated fish is due to liver degenerations (Reda et al. 2013) and inhibition of somatic cells in mitochondrial protein synthesis by antibiotics resulting in lack of oxidative ATP-generating capacity , which causes proliferation arrest of normal and malignant epithelial cells (Bakke-McKellep et al. 2007). These changes induce hepatotoxicity and nephrotoxicity.
8.3.2 Effects of Antibiotics on Hematological Parameters
Hematological parameters provide essential information on the health of cultured fish after antibiotics application. Results conducted in hematological parameters are still contrasting. Dietary chloramphenicol exposure did not affect mean corpuscular volume (MCV) and mean corpuscular hemoglobin concentration (MCHC) in O. niloticus (Shalaby et al. 2006) and monocytes, eosinophils, and basophils in C. gariepinus (Nwani et al. 2014). Moreover, florfenicol did not show significant differences in immunoglobulin M (IgM) total levels and phagocytic activity in O. niloticus when compared to the control fish (Reda et al. 2013).
However, a concentration- and time-dependent decrease in hemoglobin (Hb), red blood cells (RBC) counts, and MCV were detected in C. gariepinus exposed to chloramphenicol bath (Nwani et al. 2014). The different toxic effects of chloramphenicol bath on various organs caused the observed decrease in Hb, RBC, and MCV in fish. Chloramphenicol suppressed the production of hematological parameters caused by their toxic accumulation in lymphoid organs and pronephros (Nwani et al. 2014). The decreased RBC counts after exposure to antibiotics is due to swelling of RBC, the release of immature erythrocytes , anemia caused by tissues damage, damaged RBC, decrease in erythrocyte life span, and suppressive effects of antibiotics on erythropoietic tissues (Shalaby et al. 2006; Nwani et al. 2014). The deecreased Hb may limit the oxygen-carrying capacity of the fish blood (Nwani et al. 2014) and affect their survival rate. The inhibition of these hematological parameters may lead to sustained toxic effects caused by both dietary and bath exposure to antibiotics, resulting in tissue damage and immunity suppression with possible fatal outcomes.
Notwithstanding the above results, dietary chloramphenicol exposure in O. niloticus increased RBC, Hb, and hematocrit (Shalaby et al. 2006) and its bath elevated WBC, neutrophil count, and lymphocytes in C. gariepinus (Nwani et al. 2014). The use of oxytetracycline diet increased lysozyme activity in O. niloticus (Reda et al. 2013). Increased WBC count and lysozyme activity indicate a protective mechanism of the fish body to antibiotics-induced stress, a condition termed as leukocytosis, which signals a response of damaged tissues and immune system stimulation to counteract antibiotics toxicity (Ambili et al. 2013). An increase in RBC is due to a compensation mechanism for impaired oxygen uptake caused by tissue damages due to the presence of antibiotics in the fish body and high percentage of circulating immature RBC (Ambili et al. 2013). On the other hand, the increased lymphocyte count (lymphocytosis) and the formation of blood cellular components (hematopoiesis) are features of infection due to increased disease-fighting cells after antibiotics exposure in fish. In general, dietary and bath antibiotics exposure in cultured fish cause leukocytosis, hematopoiesis , and lymphocytosis, suggesting sustained toxic effects and compensatory responses to conciliate the fish body to normal health conditions.
8.4 Potential Human Health Risks from Consumption of Antibiotics-Cultured Fish
8.4.1 Antibiotic-Resistant Bacteria and Antibiotic Resistance Genes
The widespread and indiscriminate use of antibiotics in different environmental compartments including fish, agriculture, and human health have led to the development of ARB , ARGs, and transposons. Resistant bacteria and resistance genes may be horizontally or vertically transferred among bacterial communities, the environment, and finally human being via transposons (Biyela et al. 2004). The presence of ARB and ARGs in humans affects the ability of antibiotics to treat diseases and thus compromise their health. Thus, presently, the existence of ARB and ARGs in the environments, particularly those conferring resistance to antibiotics used to treat human diseases, is an issue of major global concern. Although the misuse of antibiotics in human medicine is the principal cause of the problem, ARB and ARGs originating from animals such as fish and agriculture production are also responsible (Barton 2000).
The literature shows that ARB and ARGs pose a human health risk in various African countries contributed by consuming contaminated fish, shrimp, vegetables, and various food sources as well as drinking contaminated water (Table 8.1). The human health risk posed by ARB from fish consumption appears to be widely spread because both cultured and wild fish have been shown to contain them. Various ARB have been isolated in cultured fish from Ghana (Agoba et al. 2017), Tanzania (Shah et al. 2012; Mhongole et al. 2017), and Uganda (Bosco et al. 2012; Wamala et al. 2018). Moreover, wild fish from Uganda (Wamala et al. 2018), Algeria (Dib et al. 2018), South Africa (Fri et al. 2018), and Egypt (Ramadan et al. 2018) were all shown to contain ARB. The ARB contained in fish in the different countries originate from various sources including animal-origin fertilizers (Shah et al. 2012; Omojowo and Omojasola 2013), the aquatic environment (Stenstrom et al. 2016), and possibly fish feeds. It is possible that the ARB from the different compartments are transferred to humans in Africa. Indeed, ARB have been detected in humans from Ethiopia (Kibret and Abera 2014), Ghana (Obeng-Nkrumah et al. 2013), Ivory Coast (Moroh et al. 2014), Libya (Mohammed et al. 2016), and Morocco (El Bouamri et al. 2015). This is an alarming situation because most of the bacteria isolated exhibited high resistance to common antibiotics used for treating frequently occurring diseases in humans in Africa and most of them had multiple antibiotic resistance (MAR) (Bosco et al. 2012; Omojowo and Omojasola 2013; Mohammed et al. 2016; Agoba et al. 2017; Apenteng et al. 2017; Wamala et al. 2018). Although correct and appropriate food cooking procedures may kill bacteria, contamination can occur through improper handling before cooking (Darwish et al. 2013) and possibly through bacteria-human contact because ARB are ubiquitous (Mhongole et al. 2017). Indeed, high levels of antimicrobial resistance (AMR) were obtained in food animals including fish intended for human consumption in Nigeria (Oloso et al. 2018).
Consistent to the existence of ARB in fish from aquaculture and wild environments, their corresponding ARGs also have been detected in cultured fish from Tanzania (Shah et al. 2012) and wild fish from Tanzania (Moremi et al. 2016), Egypt (Ramadan et al. 2018), Algeria (Brahmi et al. 2018; Dib et al. 2018), and South Africa (Fri et al. 2018) (Table 8.2). Coherent to ARB, it is possible that the ARGs detected originate from the aquatic environment (Adesoji and Ogunjobi 2016; Lyimo et al. 2016; Stenstrom et al. 2016). In general, the human health risk associated with ARGs is not only contributed by fish. Reasonably, ARGs have also been found in drinking water in Tanzania (Lyimo et al. 2016) and Nigeria (Adesoji et al. 2015; Adesoji and Ogunjobi 2016), Rhizospheres plants in South Africa (Adegoke and Okoh 2015), various foods in Egypt (Hammad et al. 2018), and wastewater used for urban agriculture in Burkina Faso (Bougnom et al. 2019). Accordingly, ARGs have been detected in the human body in Senegal (Diene et al. 2013).
Similar to ARB, the ARGs detected are those encoding resistance to common antibiotics used for the frequent treatment of human diseases in Africa. Thus, Africans are currently exposed to a double resistance to antibiotics due to the presence of ARB and ARGs. Indeed, transposons and plasmids for transfer of ARGs have been detected in wild fish and water from fish farms in Tanzania (Shah et al. 2012) and South Africa (Fri et al. 2018), various foods in Egypt (Hammad et al. 2018), and wastewater used for agriculture in Burkina Faso (Bougnom et al. 2019). Since antibiotics exist ubiquitously in the environment, Africans are exposed to high health risks due to their close interaction with livestock and the aquatic ecosystem (Wamala et al. 2018), which signifies increased morbidity and mortality (Gyansa-Lutterodt 2013) due to the failure of antibiotics to treat bacterial diseases. The obtained results emphasize the need for policies and mechanisms to limit the use of antibiotics in food animals production in order to protect human health. Moreover, physicians should devise some methods to change patients’ treatment pattern depending on antibiotics susceptibility results. Antibiotics may also pose direct public health effects due to their residuals in different foods consumed by humans as detailed below.
8.4.2 Direct Potential Human Health Risk from Consumption of Fish
Globally, antibiotics residues in foods have attracted much attention in recent years because of growing food safety and public health concerns (Capita and Alonso-Calleja 2011; Landers et al. 2012; Berendonk et al. 2015). Their presence in food animals represent socioeconomic challenges in global trade and consumed animal products (Okocha et al. 2018). In most countries, the use of antibiotics for food animals production requires a withdrawal period before the product can be sold for human consumption. Despite this regulation, most antibiotics are used without observing such a regulation both in fish (Pham et al. 2015) and other animals (Mubito et al. 2014). Consequently, high levels of antibiotics exist in food animals intended for human consumption, which pose a direct human health risk.
Limited studies have been conducted in Africa to detect antibiotics residues in fish and other foods. In Nigeria, Olatoye and Basiru (2013) found oxytetracycline levels in cultured C. gariepinus in the liver and fillets exceeded the Codex Alimentarius Commission established maximum residue limit of 600 and 200 μg/kg, respectively (Table 8.3). Similarly, a study conducted by Olusola et al. (2012) in Nigeria also found tetracycline exceeded international limits of 200 μg/kg from fresh and frozen C. gariepinus and O. niloticus, while chloramphenicol, which has a zero tolerance level, was detected in Officers’ Mess. It has been reported that, in Africa, as in other parts of the world, antibiotic residues in animal-derived foods more commonly exceed the world health organization (WHO) threshold residue levels (Darwish et al. 2013). This further highlights the high human health risk caused by antibiotics because they have also been detected in wastewater treatment plants (WWTPs) in Kenya (Kimosop et al. 2016) and South Africa (Matongo et al. 2015), surface water in Nigeria (Olaniran et al. 2018), hospitals (Kimosop et al. 2016) and river water (K’Oreje et al. 2016; Ngumba et al. 2016) in Kenya, cow’s milk in Tanzania (Ridhiwani 2015) and Algeria (Layada et al. 2016), untreated effluent water from a cow market in Nigeria (Olaniran et al. 2018), and cattle muscle, liver, and kidney (Kimera et al. 2015) in Tanzania.
In practice, the human health risk resulting from antibiotics in Africa may be much higher because of multiple sources. Antibiotics are widely abused by humans for therapy, sometimes without physicians’ prescription, and the quantity of antibiotics prescribed in African countries intended for the treatment of various diseases are high (Adegoke et al. 2018), contributing to elevated levels of residues. The antibiotic residues have been reported to spread rapidly, irrespective of geographical, economic, or legal differences in African countries (Darwish et al. 2013). This represents a serious concern because antibiotics, particularly chronic dietary oxytetracycline used in fish production, have been recently reported to cause direct human health risk in children (Limbu et al. 2018). In general, despite the existence of limited studies on antibiotics residues from cultured fish, the results obtained from C. gariepinus and O. niloticus suggest a widespread human health risk because the two fish species are widely consumed in African countries. Thus, there is an urgent need to control the use of antibiotics in fish intended for human consumption in order to protect human health.
8.5 Conclusion
It is clear that studies on antibiotics used in aquaculture production in Africa are still limited particularly on effects on fish anatomy and physiology. However, the existing limited data highlight toxic effects of antibiotics in the fish body and increasing prevalence of ARB and ARGs coupled with high residues of antibiotics in cultured fish, which pose a significant human health risk. The African countries require coordinated actions to tackle the indiscriminate use of antibiotics in humans, livestock, agriculture, and aquaculture at its grassroots, because currently most of them are characterized by inadequate monitoring, surveillance and weak regulatory systems. Clear policy directions for prohibiting the use of antibiotics on food animals production are urgently needed to protect human health. More studies should be conducted on the potential risks of antibiotics on fish and human health resulting from multiple exposure scenarios.
References
Adegoke AA, Okoh AI (2015) Antibiogram of Stenotrophomonas maltophilia isolated from Nkonkobe Municipality, Eastern Cape Province, South Africa. Jundishapur J Microbiol 8(1):e13975. https://doi.org/10.5812/jjm.13975
Adegoke AA, Faleye AC, Stenstrӧm TA (2018) Residual antibiotics, antibiotic resistant superbugs and antibiotic-resistance genes in surface water catchments: public health impact. Phys Chem Earth Parts A/B/C 105:177–183. https://doi.org/10.1016/j.pce.2018.03.004
Adesoji AT, Ogunjobi AA (2016) Detection of extended-spectrum beta-lactamases resistance genes among bacteria isolated from selected drinking water distribution channels in southwestern Nigeria. Biomed Res Int 2016:9. https://doi.org/10.1155/2016/7149295
Adesoji AT, Ogunjobi AA, Olatoye IO et al (2015) Prevalence of tetracycline resistance genes among multi-drug resistant bacteria from selected water distribution systems in southwestern Nigeria. Ann Clin Microbiol Antimicrob 14(1):35. https://doi.org/10.1186/s12941-015-0093-1
Agoba EE, Adu F, Agyare C et al (2017) Antibiotic resistance patterns of bacterial isolates from hatcheries and selected fish farms in the Ashanti region of Ghana. J Microbiol Antimicrob 9(4):35–46. https://doi.org/10.5897/JMA2017.0387
Alsan M, Schoemaker L, Eggleston K et al (2015) Out-of-pocket health expenditures and antimicrobial resistance in low-income and middle-income countries: an economic analysis. Lancet Infect Dis 15(10):1203–1210. https://doi.org/10.1016/S1473-3099(15)00149-8
Ambili TR, Saravanan M, Ramesh M et al (2013) Toxicological effects of the antibiotic oxytetracycline to an Indian major carp Labeo rohita. Arch Environ Contam Toxicol 64(3):494–503. https://doi.org/10.1007/s00244-012-9836-6
Apenteng JA, Osei-Asare C, Oppong EE et al (2017) Antibiotic sensitivity patterns of microbial isolates from fish ponds: a study in the Greater Accra Region of Ghana. Afr J Pharm Pharmacol 11(28):314–320. https://doi.org/10.5897/AJPP2017.4789
Bakke-McKellep AM, Penn MH, Salas PM et al (2007) Effects of dietary soyabean meal, inulin and oxytetracycline on intestinal microbiota and epithelial cell stress, apoptosis and proliferation in the teleost Atlantic salmon (Salmo salar L.). Br J Nutr 97(4):699–713. https://doi.org/10.1017/s0007114507381397
Barton MD (2000) Antibiotic use in animal feed and its impact on human healt. Nutr Res Rev 13(2):279–299. https://doi.org/10.1079/095442200108729106
Berendonk TU, Manaia CM, Merlin C et al (2015) Tackling antibiotic resistance: the environmental framework. Nat Rev Microbiol 13(5):310–317. https://doi.org/10.1038/nrmicro3439
Bernabé KJ, Langendorf C, Ford N et al (2017) Antimicrobial resistance in West Africa: a systematic review and meta-analysis. Int J Antimicrob Agents 50(5):629–639. https://doi.org/10.1016/j.ijantimicag.2017.07.002
Biyela PT, Lin J, Bezuidenhout CC (2004) The role of aquatic ecosystems as reservoirs of antibiotic resistant bacteria and antibiotic resistance genes. Water Sci Technol 50(1):45–50. https://doi.org/10.2166/wst.2004.0014
Bosco KJ, Kaddumulindwa DH, Asiimwe BB (2012) Antimicrobial drug resistance and plasmid profiles of isolates from humans and foods of animal origin in Uganda. Adv Infect Dis 2(4):151–155. https://doi.org/10.4236/aid.2012.24025
Bougnom BP, Zongo C, McNally A et al (2019) Wastewater used for urban agriculture in West Africa as a reservoir for antibacterial resistance dissemination. Environ Res 168:14–24. https://doi.org/10.1016/j.envres.2018.09.022
Brahmi S, Touati A, Dunyach-Remy C et al (2018) High prevalence of extended-spectrum β-lactamase-producing Enterobacteriaceae in wild fish from the Mediterranean Sea in Algeria. Microb Drug Resist 24(3):290–298. https://doi.org/10.1089/mdr.2017.0149
Byarugaba DK (2004) Antimicrobial resistance in developing countries and responsible risk factors. Int J Antimicrob Agents 24(2):105–110. https://doi.org/10.1016/j.ijantimicag.2004.02.015
Capita R, Alonso-Calleja C (2011) Antibiotic-resistant bacteria: a challenge for the food industry. Crit Rev Food Sci Nutr 53(1):11–48. https://doi.org/10.1080/10408398.2010.519837
Darwish WS, Eldaly EA, El-Abbasy MT et al (2013) Antibiotic residues in food: the African scenario. Jpn J Vet Res 61(Suppl):S13–S22
Dib AL, Agabou A, Chahed A et al (2018) Isolation, molecular characterization and antimicrobial resistance of Enterobacteriaceae isolated from fish and seafood. Food Control 88:54–60. https://doi.org/10.1016/j.foodcont.2018.01.005
Diene SM, Fall B, Kempf M et al (2013) Emergence of the OXA-23 carbapenemase-encoding gene in multidrug-resistant Acinetobacter baumannii clinical isolates from the principal Hospital of Dakar, Senegal. Int J Infect Dis 17(3):e209–e210. https://doi.org/10.1016/j.ijid.2012.09.007
Dobšíková R, Blahová J, Mikulíková I et al (2013) The effect of oyster mushroom β-1.3/1.6-D-glucan and oxytetracycline antibiotic on biometrical, haematological, biochemical, and immunological indices, and histopathological changes in common carp (Cyprinus carpio L.). Fish Shellfish Immunol 35(6):1813–1823. https://doi.org/10.1016/j.fsi.2013.09.006
El Bouamri MC, Arsalane L, El Kamouni Y et al (2015) Antimicrobial susceptibility of urinary Klebsiella pneumoniae and the emergence of carbapenem-resistant strains: a retrospective study from a university hospital in Morocco, North Africa. Afr J Urol 21(1):36–40. https://doi.org/10.1016/j.afju.2014.10.004
El-Sayed SAA, Ahmed SYA, Abdel-Hamid NR (2014) Immunomodulatory and growth performance effects of ginseng extracts as a natural growth promoter in comparison with oxytetracycline in the diets of Nile tilapia (Oreochromis niloticus). Int J Livest Res 4(1):130–142
FAO (2016) The state of world fisheries and aquaculture 2016. Contributing to food security and nutrition for all. Food and Agriculture Organization of the United Nations (FAO), Rome, p 200
FAO (2018) The state of world fisheries and aquaculture 2018—meeting the sustainable development goals. Food and Agriculture Organization of the United Nations (FAO), Rome, p 210
Foka FET, Kumar A, Ateba CN (2018) Emergence of vancomycin-resistant enterococci in South Africa: implications for public health. S Afr J Sci 114(9/10):1–7. https://doi.org/10.17159/sajs.2018/4508
Fri J, Ndip RN, Njom HA et al (2018) Antibiotic susceptibility of non-cholera Vibrios isolated from farmed and wild marine fish (Argyrosomus japonicus), implications for public health. Microb Drug Resist 24(9):1296–1304. https://doi.org/10.1089/mdr.2017.0276
Fu J, Yang D, Jin M et al (2017) Aquatic animals promote antibiotic resistance gene dissemination in water via conjugation: role of different regions within the zebrafish intestinal tract, and impact on fish intestinal microbiota. Mol Ecol 26(19):5318–5333. https://doi.org/10.1111/mec.14255
Goutard FL, Bordier M, Calba C et al (2017) Antimicrobial policy interventions in food animal production in South East Asia. BMJ 358:j3544. https://doi.org/10.1136/bmj.j3544
Gyansa-Lutterodt M (2013) Antibiotic resistance in Ghana. Lancet Infect Dis 13(12):1006–1007. https://doi.org/10.1016/S1473-3099(13)70196-8
Hammad AM, Moustafa A-EH, Mansour MM et al (2018) Molecular and phenotypic analysis of hemolytic Aeromonas strains isolated from food in Egypt revealed clinically important multidrug resistance and virulence profiles. J Food Prot 81(6):1015–1021. https://doi.org/10.4315/0362-028x.Jfp-17-360
Han J, Zhang L, Yang S et al (2014) Detrimental effects of metronidazole on selected innate immunological indicators in common carp (Cyprinus carpio L.). Bull Environ Contam Toxicol 92(2):196–201. https://doi.org/10.1007/s00128-013-1173-6
Kibret M, Abera B (2014) Prevalence and antibiogram of bacterial isolates from urinary tract infections at Dessie Health Research Laboratory, Ethiopia. Asian Pac J Trop Biomed 4(2):164–168. https://doi.org/10.1016/S2221-1691(14)60226-4
Kimera ZI, Mdegela RH, Mhaiki CJN et al (2015) Determination of oxytetracycline residues in cattle meat marketed in the Kilosa district, Tanzania. Onderstepoort J Vet Res 82:01–05. https://doi.org/10.4102/ojvr.v82i1.911
Kimosop SJ, Getenga ZM, Orata F et al (2016) Residue levels and discharge loads of antibiotics in wastewater treatment plants (WWTPs), hospital lagoons, and rivers within Lake Victoria Basin, Kenya. Environ Monit Assess 188(9):532. https://doi.org/10.1007/s10661-016-5534-6
Klein EY, Van Boeckel TP, Martinez EM et al (2018) Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc Natl Acad Sci 115:1–8. https://doi.org/10.1073/pnas.1717295115
K’Oreje KO, Vergeynst L, Ombaka D et al (2016) Occurrence patterns of pharmaceutical residues in wastewater, surface water and groundwater of Nairobi and Kisumu city, Kenya. Chemosphere 149:238–244. https://doi.org/10.1016/j.chemosphere.2016.01.095
Landers TF, Cohen B, Wittum TE et al (2012) A review of antibiotic use in food animals: perspective, policy, and potential. Public Health Rep 127(1):4–22. https://doi.org/10.1177/003335491212700103
Layada S, Benouareth D-E, Coucke W et al (2016) Assessment of antibiotic residues in commercial and farm milk collected in the region of Guelma (Algeria). Int J Food Contam 3(1):19. https://doi.org/10.1186/s40550-016-0042-6
Limbu SM, Zhou L, Sun S-X et al (2018) Chronic exposure to low environmental concentrations and legal aquaculture doses of antibiotics cause systemic adverse effects in Nile tilapia and provoke differential human health risk. Environ Int 115:205–219. https://doi.org/10.1016/j.envint.2018.03.034
Limbu SM, Ma Q, Zhang M-L et al (2019a) High fat diet worsens the adverse effects of antibiotic on intestinal health in juvenile Nile tilapia (Oreochromis niloticus). Sci Total Environ 680:169–180. https://doi.org/10.1016/j.scitotenv.2019.05.067
Limbu SM, Zhang H, Luo Y et al (2019b) High carbohydrate diet partially protects Nile tilapia (Oreochromis niloticus) from oxytetracycline-induced side effects. Environ Pollut 113508. https://doi.org/10.1016/j.envpol.2019.113508
Lyimo B, Buza J, Subbiah M et al (2016) IncF plasmids are commonly carried by antibiotic-resistant Escherichia coli isolated from drinking water sources in northern Tanzania. Int J Microbiol 2016:7. https://doi.org/10.1155/2016/3103672
Matongo S, Birungi G, Moodley B et al (2015) Pharmaceutical residues in water and sediment of Msunduzi River, KwaZulu-Natal, South Africa. Chemosphere 134:133–140. https://doi.org/10.1016/j.chemosphere.2015.03.093
Mehdi Y, Létourneau-Montminy M-P, Gaucher M-L et al (2018) Use of antibiotics in broiler production: global impacts and alternatives. Anim Nutr 4(2):170–178. https://doi.org/10.1016/j.aninu.2018.03.002
Mhongole OJ, Mdegela RH, Kusiluka LJM et al (2017) Characterization of Salmonella spp. from wastewater used for food production in Morogoro, Tanzania. World J Microbiol Biotechnol 33(3):42. https://doi.org/10.1007/s11274-017-2209-6
Mohammed MA, Alnour TMS, Shakurfo OM et al (2016) Prevalence and antimicrobial resistance pattern of bacterial strains isolated from patients with urinary tract infection in Messalata central hospital, Libya. Asian Pac J Trop Med 9(8):771–776. https://doi.org/10.1016/j.apjtm.2016.06.011
Moremi N, Manda EV, Falgenhauer L et al (2016) Predominance of CTX-M-15 among ESBL producers from environment and fish gut from the shores of Lake Victoria in Mwanza, Tanzania. Front Microbiol 7(1862):1–11. https://doi.org/10.3389/fmicb.2016.01862
Moroh JLA, Fleury Y, Tia H et al (2014) Diversity and antibiotic resistance of uropathogenic bacteria from Abidjan. Afr J Urol 20(1):18–24. https://doi.org/10.1016/j.afju.2013.11.005
Mubito EP, Shahada F, Kimanya ME et al (2014) Antimicrobial use in the poultry industry in Dar-es-Salaam, Tanzania and public health implications. Am J Res Comm 2(4):51–63
Mukonzo JK, Namuwenge PM, Okure G et al (2013) Over-the-counter suboptimal dispensing of antibiotics in Uganda. J Multidiscip Healthc 6:303–310. https://doi.org/10.2147/JMDH.S49075
Nakano T, Hayashi S, Nagamine N (2018) Effect of excessive doses of oxytetracycline on stress-related biomarker expression in coho salmon. Environ Sci Pollut Res 25(8):7121–7128. https://doi.org/10.1007/s11356-015-4898-4
Ngumba E, Gachanja A, Tuhkanen T (2016) Occurrence of selected antibiotics and antiretroviral drugs in Nairobi River Basin, Kenya. Sci Total Environ 539:206–213. https://doi.org/10.1016/j.scitotenv.2015.08.139
Nunes B, Antunes SC, Gomes R et al (2015) Acute effects of tetracycline exposure in the freshwater fish Gambusia holbrooki: antioxidant effects, neurotoxicity and histological alterations. Arch Environ Contam Toxicol 68(2):371–381. https://doi.org/10.1007/s00244-014-0101-z
Nwani CD, Mkpadobi BN, Onyishi G et al (2014) Changes in behavior and hematological parameters of freshwater African catfish Clarias gariepinus (Burchell 1822) following sublethal exposure to chloramphenicol. Drug Chem Toxicol 37(1):107–113. https://doi.org/10.3109/01480545.2013.834348
Nyasulu P, Murray J, Perovic O et al (2012) Antimicrobial resistance surveillance among nosocomial pathogens in South Africa: systematic review of published literature. J Exp Clin Med 4(1):8–13. https://doi.org/10.1016/j.jecm.2011.11.002
O’Neill J (2016) Tackling drug-resistant infections globally: final report and recommendations. Review of antimicrobial resistance. HM Government and Wellcome trust, London. https://amr-review.org/sites/default/files/160525_Final%20paper_with%20cover.pdf
Obeng-Nkrumah N, Twum-Danso K, Krogfelt KA et al (2013) High levels of extended-spectrum beta-lactamases in a major teaching hospital in Ghana: the need for regular monitoring and evaluation of antibiotic resistance. Am J Trop Med Hyg 89(5):960–964. https://doi.org/10.4269/ajtmh.12-0642
Okocha RC, Olatoye IO, Adedeji OB (2018) Food safety impacts of antimicrobial use and their residues in aquaculture. Public Health Rev 39:1–22. https://doi.org/10.1186/s40985-018-0099-2
Olaniran EI, Sogbanmu TO, Saliu JKJEM et al (2018) Biomonitoring, physico-chemical, and biomarker evaluations of abattoir effluent discharges into the Ogun River from Kara Market, Ogun State, Nigeria, using Clarias gariepinus. Environ Monit Assess 191(1):44. https://doi.org/10.1007/s10661-018-7168-3
Olatoye IO, Basiru A (2013) Antibiotic usage and oxytetracycline residue in African catfish (Clarias gariepinus in Ibadan, Nigeria). World J Fish Mar Sci 5(3):302–309. https://doi.org/10.5829/idosi.wjfms.2013.05.03.71214
Oliveira R, McDonough S, Ladewig JCL et al (2013) Effects of oxytetracycline and amoxicillin on development and biomarkers activities of zebrafish (Danio rerio). Environ Toxicol Pharmacol 36(3):903–912. https://doi.org/10.1016/j.etap.2013.07.019
Oloso NO, Fagbo S, Garbati M et al (2018) Antimicrobial resistance in food animals and the environment in Nigeria: a review. Int J Environ Res Public Health 15(6):23. https://doi.org/10.3390/ijerph15061284
Olusola AV, Folashade PA, Ayoade OI (2012) Heavy metal (lead, cadmium) and antibiotic (tetracycline and chloramphenicol) residues in fresh and frozen fish types (Clarias gariepinus, Oreochromis niloticus) in Ibadan, Oyo state, Nigeria. Pak J Biol Sci 15(18):895–899. https://doi.org/10.3923/pjbs.2012.895.899
Omojowo F, Omojasola F (2013) Antibiotic resistance pattern of bacterial pathogens isolated from poultry manure used to fertilize fish ponds in new Bussa, Nigeria. Albanian J Agric Sci 12(1):81–85
Pham DK, Chu J, Do NT et al (2015) Monitoring antibiotic use and residue in freshwater aquaculture for domestic use in Vietnam. EcoHealth 12(3):480–489. https://doi.org/10.1007/s10393-014-1006-z
Ramadan H, Ibrahim N, Samir M et al (2018) Aeromonas hydrophila from marketed mullet (Mugil cephalus) in Egypt: PCR characterization of β-lactam resistance and virulence genes. J Appl Microbiol 124(6):1629–1637. https://doi.org/10.1111/jam.13734
Reda RM, Ibrahim RE, E-NG A et al (2013) Effect of oxytetracycline and florfenicol as growth promoters on the health status of cultured Oreochromis niloticus. Egypt J Aquat Res 39(4):241–248. https://doi.org/10.1016/j.ejar.2013.12.001
Refstie S, Bakke-McKellep AM, Penn MH et al (2006) Capacity for digestive hydrolysis and amino acid absorption in Atlantic salmon (Salmo salar) fed diets with soybean meal or inulin with or without addition of antibiotics. Aquaculture 261(1):392–406. https://doi.org/10.1016/j.aquaculture.2006.08.005
Ridhiwani R (2015) Assessment of antibiotic residues in raw cows’ milk produced by small scale dairy farms in Bagamoyo District, Tanzania. Masters dissertation. Sokoine University of Agriculture, Morogoro, Tanzania, p 91
Sanou M, Ky/Ba A, Coulibali P et al (2018) Assessment of the prevalence of extended-spectrum β-lactamase producing gram-negative bacilli at the Charles De Gaulle Paediatric university hospital (CDG-PUH), Ouagadougou, Burkina Faso. Afr J Microbiol Res 12(13):300–306. https://doi.org/10.5897/AJMR2017.8778
Saravanan M, Devi KU, Malarvizhi A et al (2012) Effects of ibuprofen on hematological, biochemical and enzymological parameters of blood in an Indian major carp, Cirrhinus mrigala. Environ Toxicol Pharmacol 34(1):14–22. https://doi.org/10.1016/j.etap.2012.02.005
Shah SQA, Colquhoun DJ, Nikuli HL et al (2012) Prevalence of antibiotic resistance genes in the bacterial flora of integrated fish farming environments of Pakistan and Tanzania. Environ Sci Technol 46(16):8672–8679. https://doi.org/10.1021/es3018607
Shalaby AM, Khattab YA, Abdel Rahman AM (2006) Effects of garlic (Allium sativum) and chloramphenicol on growth performance, physiological parameters and survival of Nile tilapia (Oreochromis niloticus). J Venom Anim Tox Trop Dis 12:172–201
Stenstrom TA, Okoh AI, Adegoke AA (2016) Antibiogram of environmental isolates of Acinetobacter calcoaceticus from Nkonkobe Municipality, South Africa. Fresenius Environ Bull 25(8):3059–3065
Topic Popovic N, Howell T, Babish JG et al (2012) Cross-sectional study of hepatic CYP1A and CYP3A enzymes in hybrid striped bass, channel catfish and Nile tilapia following oxytetracycline treatment. Res Vet Sci 92(2):283–291. https://doi.org/10.1016/j.rvsc.2011.03.003
Trushenski JT, Aardsma MP, Barry KJ et al (2018) Oxytetracycline does not cause growth promotion in finfish1. J Anim Sci 96(5):1667–1677. https://doi.org/10.1093/jas/sky120
Wamala SP, Mugimba KK, Mutoloki S et al (2018) Occurrence and antibiotic susceptibility of fish bacteria isolated from Oreochromis niloticus (Nile tilapia) and Clarias gariepinus (African catfish) in Uganda. Fish Aquat Sci 21(1):6. https://doi.org/10.1186/s41240-017-0080-x
Wang H, Che B, Duan A et al (2014) Toxicity evaluation of β-diketone antibiotics on the development of embryo-larval zebrafish (Danio rerio). Environ Toxicol 29(10):1134–1146. https://doi.org/10.1002/tox.21843
Yonar ME (2012) The effect of lycopene on oxytetracycline-induced oxidative stress and immunosuppression in rainbow trout (Oncorhynchus mykiss, W.). Fish Shellfish Immunol 32(6):994–1001. https://doi.org/10.1016/j.fsi.2012.02.012
Yonar EM, Yonar MS, Sibel S (2011) Protective effect of propolis against oxidative stress and immunosuppression induced by oxytetracycline in rainbow trout (Oncorhynchus mykiss, W.). Fish Shellfish Immunol 31(2):318–325. https://doi.org/10.1016/j.fsi.2011.05.019
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Limbu, S.M. (2020). Antibiotics Use in African Aquaculture: Their Potential Risks on Fish and Human Health. In: Abia, A., Lanza, G. (eds) Current Microbiological Research in Africa. Springer, Cham. https://doi.org/10.1007/978-3-030-35296-7_8
Download citation
DOI: https://doi.org/10.1007/978-3-030-35296-7_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-35295-0
Online ISBN: 978-3-030-35296-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)