Abstract
This paper presents the results of a series of studies aimed at investigating the reference material (RM) of a multicomponent solution. An algorithm for estimating the uncertainty induced by the chemical element content in the RM is proposed taking into account the mass of the measurand in each component of the mixture. The results calculated according to the preparation procedure using the proposed algorithm were confirmed by the Monte Carlo method, while the elemental content in the mixture was confirmed via inductively coupled plasma optical emission spectroscopy. The applicability of weighted mean estimates for characterizing an RM of a multicomponent solution is shown.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
It is well-known that reference materials (RMs) perform the role of measurement standards in chemistry. RMs of chemical element solutions have been widely used in modern quantitative chemical analysis in constructing calibration dependences of the output signals of, e.g., spectral measurement instruments on the amount of substance. The ease-of-use of RM multicomponent solutions is characterized by the versatility of modern instrumental methods of analysis, which allows calibration of measurement instruments for simultaneous determination of several elements by one or several signals.
2 Literature Review
Over the past eight years, the Ural Scientific Research Institute for Metrology (UNIIM) has accumulated a repository of pure substances, comprising over 70 compounds of 55 individual elements. Having undergone a full purity assessment, these substances are candidates for comparison measurement standards [1]. Such comparison measurement standards in international practice are called primary reference materials [2], primary calibration substances (materials) [3], primary standards for elemental determination [4], etc. The work [5] shows that high-purity materials may act as the material realization of a mole unit in the International System of Units (SI); their gravimetrically prepared solutions are primary calibration solutions and can be used in a variety of methods for quantitative chemical analysis. Having a sufficiently large collection of pure substances, it is reasonable to use their solutions (primary calibration solution [5]) to characterize the RMs of element solutions (secondary calibration solution [5]) using high-performance inductively-coupled plasma spectrometry with high precision [6]. In this case, considering that the relative expanded uncertainty of a mass fraction of the basic component in a solution of comparison measurement standard will be in the order of 0.05%, the relative expanded uncertainty of this component mass fraction in the RM will be in the order of (0.1–0.5)% depending on its stability or the assigned shelf life. Thus, a scheme for ensuring the traceability of RMs of mono-element solutions can be implemented.
As for the production of an RM of a multicomponent solution, it is apparent that the simplest way is to mix mono-element solutions with a well-known analyte content. For the estimation of the uncertainty induced by the preparation method, a method set out in ISO Guide 35: 2017 and based on the use of weighted-mean estimates [7, 9.3.4] is proposed, where the content of a measured component in a mixture is determined using the formulas:
where
-
\( \bar{x}_{i} \) is the weighted mean of the content of the i-th component in the mixture;
-
\( x_{ij} \) is the content of the i-th component in the j-th component in the mixture;
-
\( w_{j} \) is weight coefficients;
-
\( m_{j} \) is the mass of the j-th component in the mixture.
ISO Guide 35: 2017 also offers a formula for estimating the standard uncertainty due to characterization using the gravimetric preparation procedure of a binary mixture [7, (13)], for the case when N = 2.
However, within the framework of the GUM methodology [8], the work [9] shows that the square of the combined standard uncertainty of the output value \( x_{j} \) can be represented for an unlimited number of components \( (N < \infty ) \) in general:
where
-
\( u_{{x_{ij} }}^{{}} \) is the standard uncertainty of the content of the j-th component in the mixture;
-
\( u_{{m_{j} }}^{{}} \) is the standard measurement uncertainty of the mass of the j-th component in the mixture.
3 Materials and Methods
In order to test the proposed approach, a multicomponent RM was prepared, consisting of a mix of solutions of chemical elements (V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Cd) in a matrix of 5% nitric acid (blank). Mono-element solutions were gravimetrically prepared from pure metals corrected for the air buoyancy in weighing the metal sample intake and final solution. For the preparation of solutions, containers of high density polyethylene were used, purified using non-boiling distillation of nitric and hydrofluoric acids and deionized water. The mass fraction of the main component in the basic material was determined according to the scheme “100% minus combined impurities” using the results of measurements by the mass spectrometry method with inductively coupled plasma (ICP MS, PerkinElmer Inc. NexION 300D). For the determination of gas-forming impurities (N, O, H), the reducing melting method (CGHE, Eltra GmbH ELEMENTRAC ONH-p) was used. The evaluation of this value and associated uncertainty was tested by the following formulas:
where
-
uc(w) is the combined standard uncertainty of the content of the main component in the starting material;
-
\( z_{i} \) is the mass fraction of the i-th detected impurity;
-
n is the number of detected impurities;
-
\( y_{j} \) is the detection limit established for the j-th impurity according to the 3σ criterion;
-
r is the number of detection limits;
-
\( u(z_{i} ) \) is the standard measurement uncertainty of the j-th impurity;
-
\( u(y_{j} ) = y_{j} /2 \) is the standard uncertainty caused by the establishment of a detection limit in the j-th impurity.
When n + r = 91, the most complete assessment of purity in the characterization of comparison standards is achieved.
The mass fractions of the target impurity components in each solution were determined in the same way using ICP MS and the mass fraction of the major component according to the formula:
where
-
\( K_{stab} ,K_{homo} ,K_{evap} \) are coefficients associated with temporal stability, homogeneity and possible evaporation of the solution during the preparation process, respectively (in the calculations they are taken to be equal to 1, and their uncertainties are determined experimentally);
-
\( w_{0} \) is the mass fraction of the main component in the form (in the example below, the value is taken to be equal to 0 with the uncertainty expressed in the form of the detection limit);
-
\( m_{n} \) is the mass of the starting metal sample intake (pure metal);
-
\( m_{r} \) is the mass of the solution;
-
\( w \) is the mass fraction of the main component in the starting material;
-
\( \rho_{a} ,\rho_{r} ,\rho_{n} \) are the densities of air, solution and starting material, respectively.
The characteristic budget of uncertainty for the analyte content in such a solution is assembled in Table 1. It can be seen that the relative expanded uncertainty of the element mass fraction in such a solution is lower than 0.05%.
The original data of the mixture preparation are presented in Table 2.
For the estimation of the uncertainty of the mass fraction of the target components in the mixture by formula (3), the standard mass measurement uncertainty was taken to be 0.0005 g, and the uncertainties of the mass fractions of the components listed in Table 3 were used. The values of the standard uncertainties of the main component mass fractions in each of the solutions are taken to be equal to 0.1%, since such tasks commonly imply the use of the so-called secondary calibration solutions, rather than reference solutions.
4 Results
The proposed approach was validated using the Monte Carlo simulation (MMC) [10] for 1 × 106 values of the content of each element having a normal distribution of input values, with the standard deviation being numerically equal to their total standard uncertainty. The output value has a distribution close to normal with a coverage factor of about 1.96, depending on the input data (Fig. 1).
Table 4 displays the results of calculations using formulas (1–3), the averaged data obtained by MMC and the relative deviation of these assessments.
The comparison showed the compatibility of the obtained estimates and, consequently, the correctness of the weighted means of the components in the mixture and calculation of their standard uncertainty according to (1–3). At the same time, it is shown that the most significant contribution to the uncertainty of certified values is made by the uncertainty associated with the mass fractions of the certified components in their initial solutions (the relative contribution is more than 90%). Thus, the uncertainty of the certified values is composed of uncertainties associated with the gravimetric preparation of pure substance solutions calculated by the “100% minus combined impurities” scheme and those induced by the stability of these solutions and the results of comparative measurements using high-performance inductively coupled plasma optical emission spectroscopy (ICP OES, PerkinElmer Inc. OPTIMA 7300DV).
Then, if the mass of mixture components is determined sufficiently accurately, in order to avoid loss of precision of the final results, the sum of the impurity of the target element in the components of the mixture should not significantly exceed the value of the standard uncertainty of the target component content in the main solution. In this case, the accuracy of determining the composition of the impurity is not significant. Conversely, when the minimum accumulation of the target component impurities is impossible to achieve, the uncertainty of the combined content of impurities must not exceed ½ of the value of the standard uncertainty of the content of the target component in its main solution.
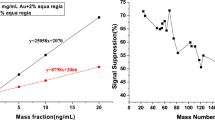
Estimates of components received by the preparation procedure are confirmed by measurements of these quantities by the ICP OES method (\( x_{l} \), L = 2) (Fig. 2).
5 Discussion and Conclusions
It is worth noting that the question remains what to consider as the RM certified value: (1) the results obtained according to the preparation procedure; (2) the results of ICP OES measurements; (3) their mean or weighted mean.
There exists a widespread opinion that the uncertainty of the certified RM value cannot be better than the uncertainty associated with the measurement of this quantity, which can be demonstrated by the producer of that RM. We believe the approach, according to which the results of direct measurements, rather than the calculated data, are taken into account in calculations, to be rather unreasonable. The thing is that any additional knowledge about the measured (certified) value can bring the researcher closer to more reliable estimates. Therefore, such information should never be wasted.
It seems sensible to consider the final result to be the weighted mean of the abovementioned estimates \( (x_{r} ) \) with weighting coefficients associated with their standard uncertainties \( (u_{l} ) \) and the square of the combined standard uncertainty of the quantity \( x_{r} \) as a component of the uncertainty of the certified RM value due to characterization:
This approach is generally similar to that used in the characterization of mono-element RMs [11, 12], where the certified value is obtained as the weighted mean of the results of gravimetric preparation of a solution from a high-purity compound (with the established content of the main component) and those obtained using ICP OES (the spectrometer being calibrated by four primary standards from a high-purity compound with the established content of the main component).
Abbreviations
- RM:
-
Reference material
- GUM:
-
Guide to the expression of uncertainty in measurement
- ICP-MS:
-
Inductively coupled plasma mass spectrometry
- CGHE:
-
Carrier gas hot extraction
- MMC:
-
Monte Carlo method
- ICP OES:
-
Inductively coupled plasma optical emission spectrometry
References
Database “Reference standards in the form of high-purity substances”. Ural Research Institute for Metrology, Ekaterinburg. https://www.uniim.ru/db_etalons_ref
Medvedevskih SV, Sobina EP, Migal PV, Goryaeva LI, Gorbunova EM, Tabatchikova TN, Sobina AV, Firsanov VA, Medvedevskikh MYu, Krasheninina MP (2014) On the use of pure inorganic substances in metrology of analytical measurements. Ref Mater 3:58–67 (in Russ)
Matschat R, Czerwensky M, Pattberg S, Heinrich HJ, Tutschku S (2002) High purity metals as primary calibration materials for elemental analysis—their importance and their certification. Mater Trans 43(2):90–97. https://doi.org/10.2320/matertrans.43.90
Kipphardt H, Matschat R, Vogl J, Gusarova T, Czerwensky M, Heinrich HJ, Hioki A, Konopelko LA, Methven B, Miura T et al (2010) Purity determination as needed for the realization of primary standards for elemental determination—status of international comparability. Accred Qual Assur 15(1):29–37. https://doi.org/10.1007/s00769-009-0557-0
Vogl J et al (2018) Establishing comparability and compatibility in the purity assessment of high purity zinc as demonstrated by the CCQM-P149 intercomparison. Metrologia 211–221. https://doi.org/10.1088/1681-7575/aaa677
Salit ML, Turk GC, Lindstrom AP, Butler TA, Beck CM II, Norman B (2001) Single-element solution comparisons with a high-performance inductively coupled plasma optical emission spectrometric method. Anal Chem 73:4821–4829. https://doi.org/10.1021/ac0155097
ISO Guide 35:2017 Reference materials—guidance for characterization and assessment of homogeneity and stability
JCGM 100:2008 Evaluation of measurement data—guide to the expression of uncertainty in measurement
Medvedevskih SV, Firsanov VA (2018) Determination of the integral content of the components of substances in the control, recording, and monitoring of production and generation of waste products in industrial quantities. Meas Tech 61:182–191. https://doi.org/10.1007/s11018-018-1407-4
JCGM 101:2008. Evaluation of measurement data—supplement 1 to the “Guide to the expression of uncertainty in measurement”—propagation of distributions using a Monte Carlo method
Standard reference materials. Spectrometry, single element standard solutions: NIST. https://www.nist.gov/srmors/viewTable.cfm?tableid=39
NIST (2000) NIST Special Publication 260-136. https://www.nist.gov/sites/default/files/documents/srm/SP260-136.PDF
Acknowledgements
We are grateful to Tatiana N. Tabatchikova, the leading engineer of UNIIM, for providing measurement results obtained using the ICP MS method.
Author Contribution Statement
Migal P. V.: Identification of the paper concept and metrology, data analysis, collection of literature data, obtainment of experimental data, preparation of the manuscript draft, computer manuscript preparation, preparation of the final manuscript.
Medvedevskikh S. V.: Concept and initiation of the research, methodological support, critical analysis and text processing.
Firsanov V. A.: Methodical and mathematical support for the works described in the article.
Conflict of Interest
One of the authors is a member of the Editorial Board of these proceedings.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this paper
Cite this paper
Migal, P.V., Medvedevskikh, S.V., Firsanov, V.A. (2020). A Method for Estimating the Certified Value Uncertainty of a Multicomponent Reference Material. In: Medvedevskikh, S., Kremleva, O., Vasil’eva, I., Sobina, E. (eds) Reference Materials in Measurement and Technology. RMMT 2018. Springer, Cham. https://doi.org/10.1007/978-3-030-32534-3_17
Download citation
DOI: https://doi.org/10.1007/978-3-030-32534-3_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-32533-6
Online ISBN: 978-3-030-32534-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)