Abstract
The surgical treatment of thoracic aneurysms or acute or chronic aortic dissections involving the aortic arch is still challenging despite significant advances in strategies and perioperative management. Even today, aortic arch surgery is associated with substantial risks for patients as reflected in the high mortality and morbidity rates. Conventional open repair techniques of the aortic arch are nowadays challenged by the introduction and wide adoption of hybrid endovascular prostheses, with the frozen elephant trunk (FET) procedure being the most important. This modern hybrid aortic arch repair combined with or without debranching of the supra-aortic vessels provides in certain pathologies a complete one-stage repair of the aortic arch and descending aorta. If necessary, the FET technique provides a secure landing zone for a subsequent thoracic endovascular aortic repair (TEVAR) of the remaining descending aorta. This chapter describes the indications and surgical technique of hybrid aortic arch repair and provides a summary of clinical outcomes and common complications.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Debranching of supra-aortic vessels
- Frozen elephant trunk
- Hybrid aortic arch repair
- Thoracic endovascular aortic repair
- Total endo-prosthetic arch repair
-
Conventional, open arch surgery is nowadays challenged by the introduction of hybrid aortic arch repair strategies including the frozen elephant trunk (FET) procedure.
-
Modern hybrid arch procedures reduce the invasiveness of arch surgery and facilitate a single-staged approach in patients with extensive disease of the aortic arch and descending aorta.
-
Various hybrid stent grafts combined with branched supra-aortic vessel grafts and debranching procedures have been introduced and are applied depending on the underlying aortic pathology, extent of the disease and surgeon’s preference.
-
Hybrid aortic arch surgery using the FET technique requires the use of cardiopulmonary bypass and hypothermic circulatory arrest with selective brain perfusion.
-
Clinical results of hybrid aortic surgery are currently based on data from large registries and single center reports and support the efficacy and safety of this approach.
-
Total endo-prosthetic arch repair is currently under clinical investigation as hybrid technologies and devices improve further.
Introduction
Surgical treatment of the aortic arch through a median sternotomy has been performed for over five decades and has nowadays become an integral part of aortic surgery [1,2,3,4]. Depending on the extent of the disease and repair, the so-called “hemiarch” or “full arch” replacement with common vascular grafts and reinsertion of the supra-aortic vessels is routinely performed under hypothermic circulatory arrest and selective cerebral perfusion with excellent results (Fig. 58.1). Since the first description by Borst et al. in 1983 the introduction of the classical elephant trunk technique (ET) allowed not only to address the aortic arch pathology but also the jeopardized descending aorta [5]. The classical ET technique consisted of a vascular graft extending from the distal aortic arch into the proximal descending aorta, giving rise to the term “elephant trunk”, and, thereby, preparing a second stage surgical intervention for repair of the descending aorta through a thoracotomy (Fig. 58.2a) [4, 5].
The “classical” (a) and “frozen” elephant trunk (b) prosthesis for aortic arch repair. One of the first available FET prosthesis, the Chavan-Haverich endograft from Hannover (c), consisted of a Dacron vascular prosthesis with stainless-steel stents affixed at its distal end. With kind approval by Professor Dr. A. Haverich
This technique was later modified by Svensson and coworkers [6], but it was again the group from Hannover that advanced the well-established classical elephant trunk to the frozen elephant trunk (FET) technique into clinical practice by combining the concepts of the classical elephant trunk with endovascular stenting of the descending aorta [7]. In their publication from 1993, Karck and coworkers [7] introduced a hybrid stented graft prosthesis that combined a covered stent at its distal portion sutured to the proximal end of a conventional tube graft. Thus, stenting of the descending aorta was made feasible by expansive radical forces of the frozen elephant trunk while enabling a classical full arch replacement at the same time (Fig. 58.2b, c).
Later, a commercially available hybrid graft was introduced by Jakob from Essen in Germany [8, 9]. This non-branched, hybrid graft enabled the treatment of patients with complex aortic pathologies and allowed a single-staged treatment of certain pathologies within the descending aorta without the need for a subsequent procedure. It was Kazui and later Shrestra et al. that reported the use of a modified four-branched hybrid prosthesis for supra-aortic vessel revascularization and arterial body perfusion during cardiopulmonary bypass [10, 11].
Since the commercial introduction of various grafts (Fig. 58.3), the hybrid aortic arch repair (FET) has become increasingly popular as a valuable alternative to the classical elephant trunk technique. The concept of hybrid repair includes complete replacment of the diseased aortic arch, restoring the supra-aortic vessel perfusion and extending the aortic repair into the diseased descending aorta and, thereby, achieving a satisfactory sealing of the diseased aorta with the hybrid stent graft. Thus, similar to the conventional elephant trunk technique, the FET technique enables in certain pathologies a single-staged procedure or paves the way for a two-staged repair by providing a secure and easy landing zone for a subsequent thoracic endovascular aortic repair (TEVAR) of the remaining descending aorta. For that reason, treatment of the aortic arch is currently evolving towards the “new frontier” of hybrid arch repair.
The two most widely used grafts in Europe for hybrid arch repair. The E-vita OPEN PLUS® Hybrid stent-graft prosthesis is a straight tube graft without branches and a Z-shaped nitinol stent (a). With kind approval of Jotec, Hechingen, Germany. The Vascutek Thoraflex® Hybrid stent-graft (b) has four branches for selective anastomosis of the supra-aortic vessels and an arterial inflow graft that enables faster body perfusion after completion of the distal anastomosis and reduces circulatory arrest time. With kind approval from Vascutek Inchinnan, Scotland
Indications and Patient Selection
The indications for the use of the FET technique are currently under constant evaluation but commonly include acute or chronic type A or B dissections with the primary entry in the arch or proximal descending aorta, complex aneurysms of the arch and descending aorta, or penetrating aortic ulcers [12,13,14].
The FET procedure in aortic dissections with intimal tear or reentry in the aortic arch or proximal descending aorta offers beyond a complete aortic arch replacement, a high rate of early (<3 months) and late (>6 months) false lumen thrombosis in over 80% of patients, thereby, promoting aortic remodeling and reducing distal malperfusion syndromes [14]. Residual dissection flaps and/or perfused false lumen of the downstream aorta are frequently observed in the arch or descending thoracic aorta after conventional, mostly limited, aortic arch repair that may subsequently lead to a distal malperfusion syndrome. Later on, a sequela of false lumen dilatation, aneurysm formation or even rupture can be observed. Thus, hybrid aortic arch surgery using the FET is recommended in this setting in the latest guidelines of the European Society of Cardiology for the treatment of Stanford type A acute aortic dissection with malperfusion (Table 58.1) and is also endorsed in the position paper of the Vascular Domain of European Association of Cardiothoracic Surgery [13, 14].
Additional indications for the hybrid FET approach include post-dissection aneurysmal formation of the arch after repair of Type A dissection, retrograde type A dissections after TEVAR and any type of complex aortic aneurysms that would otherwise require a two-staged approach.
Similarly, the currently proposed contraindications for the use of the FET procedure are not absolute and also under clinical investigation. As the experience with the FET procedure is constantly increasing, certain pitfalls or technical limitations have been recognized or solved that may interfere with the performance of a safe and efficient FET procedure [14, 15]. Severe aortic kinking or a tortuous aorta, older age, multiple comorbidities and major neurological deficits especially in acute aortic dissection, can be considered as relative contraindications [14,15,16,17].
Operative Technique
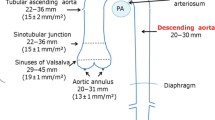
Detailed analysis of the underlying aortic pathology is crucial for adopting the adequate surgical strategy. Preprocedural examination including coronary angiography, echocardiography, thoracic X-ray and Doppler sonography of the carotids should be performed in all elective patients. A high-resolution computed tomography (CT) scan of the complete thoracoabdominal aorta is required to verify the extent of the aortic disease and/or to detect the location of the primary entry site and potential re-entries, true and false lumen perfusion and the supra-aortic or distal vessel perfusion in acute aortic dissection. The proper sizing and distal landing zone of the FET can be evaluated intraoperatively by angioscopy, but also in preoperative CT scans by measuring the aortic diameter at the distal landing zone of the FET. This can be measured from the site of the distal anastomosis of the hybrid graft downwards to the descending aorta [18]. The length of the current FET prosthesis ranges between 10 and 16 cm. Oversizing is usually not recommended in patients with aortic dissections to avoid new intimal tears or even vessel perforation but can be tolerated in some cases of extensive aneurysms [18].
The techniques of hybrid aortic arch repair are numerous and highly variable depending on the underlying aortic pathology as well as surgeons’ experience and preference [1, 4, 9, 14]. The strategy ranges from total endovascular arch repair after previous debranching of the supra-aortic vessel to the FET procedure with or without multibranched grafts for supra-aortic revascularization or extraanatomical bypass grafts to the left subclavian artery or axillary artery. The underlying principle in all procedures is to create a safe proximal aortic anastomosis of the FET and, if needed, to facilitate a landing zone for a subsequent TEVAR procedure.
The Criado classification has gained wide acceptance in order to classify the different hybrid techniques of aortic arch repair [19]. Four different zones (Fig. 58.4) within the aortic arch are described that can be used based on the surgeons’ strategy, extent of disease and patient’s anatomy. Zone 0 is located proximal to the innominate artery, zone 1 proximal to the left common carotid artery, zone 2 between the left carotid and subclavian artery and zone 3 distal to the left subclavian artery and extending into the descending aorta. Depending on the chosen proximal landing zone of the FET, revascularization of the supra-aortic vessels is usually performed either by a direct implantation or via seperate revascularization with a multibranched hybrid aortic stent graft (zone 3) or multibranched/single vessel grafts (zone 0–2).
The zones of the aortic arch adopted from the classification by Criado et al. [19]. Zone 0 – before the innominate artery; Zone 1 – between the innominate artery and left common carotid artery; Zone 2 – between the left common carotid and subclavian artery; Zone 3 – distal to the left subclavian artery
The following steps briefly describe our approach of a Zone 3 FET procedure using an E-vita Jotec hybrid prosthesis (Jotec, Hechingen, Germany) with direct “en bloc” reimplantation of the supra-aortic vessels. As aforementioned the preferred cannulation site, type of cerebral or myocardial protection and temperature of hypothermic arrest is highly variable in the literature and is discussed elsewhere.
-
1.
Cardiopulmonary bypass (CPB) and cardiocerebral protection: The right axillary artery is used in most cases after previously excluding a dissection of the vessel. It offers the advantage of antegrade perfusion of the aorta and right-lateral cerebral perfusion during circulatory arrest. We prefer the direct cannulation of the axillary artery using a purse string suture and Seldinger technique (Fig. 58.5a). Venous cannulation is usually performed after sternotomy through the right atrium. Alternative arterial access routes include the carotid artery, innominate artery, the femoral artery or direct aortic cannulation. After CPB is established cooling is initiated to moderate hypothermia with a target nasopharyngeal temperature of 25–28 °C.
-
2.
The aortic arch and the origin of the supra-aortic vessels are then dissected (Fig. 58.5b). After reaching the target body temperature the aorta is clamped and cold blood cardioplegic cardiac arrest established either through the antegrade, selective or retrograde route. Cardioplegic delivery is repeated every 20 min to maintain arrest.
-
3.
Unilateral cerebral perfusion is initiated after clamping of the innominate artery. Then the aortic arch is opened and bilateral, selective, antegrade cerebral perfusion employed after direct catheterization of the left carotid artery. The aortic arch is resected to the plane of the distal anastomosis (Zone 3) and the supra-aortic vessel island prepared for the “en bloc” reinsertion using the “island” technique.
-
4.
We usually reinforce the wall of the aortic stump with felt stripes and/or U-shaped pledged sutures, especially in acute or chronic dissections (Fig. 58.6a). Then the stent graft is inserted into the “true” lumen and the stent is released in a stepwise fashion. Care should be taken to avoid any dislocation of the collar of the prosthesis from the plane of the distal anastomosis. A transfemoral wire can be used to ensure proper positioning of the hybrid graft in the true lumen [20, 21]. Optimal positioning in the distal landing zone can also be ensured by angioscopy of the aorta before deploying the stent graft [20].
-
5.
After stent release the distal anastomosis is performed between the collar of the hybrid prosthesis and the aorta either with a running 3–0 Prolene suture or the the U-shaped sutures and the proximal part of the hybrid graft is retrieved (Fig. 58.6b). This anastomosis becomes easier when a more proximal landing zone (zone 0–2) is chosen. Then single or multibranched grafts must be used for revascularization of the respective supra-aortic vessels, usually the left subclavian artery.
-
6.
Arch replacement is completed with the proximal part of the FET tube graft by “en-bloc” reinsertion of the supra-aortic vessel with the island technique when a non-branched prosthesis is used (E-vita, Jotec, Fig. 58.7a). In case of a multi-branched prosthesis (Thoraflex, Vascutek, Inchinnan, Scotland), the supra-aortic vessels are revascularized separately with the predefined branches. This approach offers the advantage of early low body reperfusion via the arterial inflow side branch of the hybrid graft after completion of the left subclavian artery anastomosis. Finally, the proximal tube graft is sutured to the ascending aorta (Fig. 58.7b).
Direct cannulation of the right axillary artery using a pursue string suture with 5–0 Prolene and introducing a flexible arterial cannula with the Seldinger technique (a). Venous cannulation is performed after sternotomy through the right atrium. A left ventricular vent is placed and a retrograde cardioplegia catheter introduced into the coronary sinus. Then the ascending aorta, the aortic arch and the origin of the supra-aortic vessels are dissected (b)
The aortic wall for the distal anastomosis is reinforced with felt stripes and/or U-shaped pledged sutures, especially in aortic dissections (a). Then the stent graft is introduced, and the stent released while taking care that the collar of the hybrid prosthesis remains in the plane of the distal anastomosis. After completing the distal anastomosis the proximal part of the hybrid graft is retrieved and unfolded (b)
Clinical Outcomes
The clinical benefits of FET procedure for treatment of aortic arch disease in comparison to the classical ET are controversially discussed in the literature [4, 22]. However, the FET procedure offers the advantage of avoiding a second-stage procedure in certain diseases and false lumen thrombosis is often observed at early and late follow-up after treatment of aortic dissections [14]. Although the experience with the FET hybrid procedures is constantly growing, the evidence is still based on data from large registries or single center reports and not randomized trials [4, 14]. In a recent systematic review of over 841 hybrid procedures Chakos and coworkers [22] reported an 8% hospital mortality, 7% stroke rate and 4% spinal cord injury rate (Table 58.2). In other systematic reviews early mortality ranged between 8% and 11%, cerebrovascular events between 4% and 7% and spinal injury between 4% and 7% [23, 24]. In addition, long-term survival was up to 75% at 7 years after FET implantation [22]. In the international E-vita OPEN-Plus registry the FET procedure was associated with 18% mortality in 350 patients with aortic dissection and 13% mortality in 159 patients with thoracic aneurysms. Stroke rate was 8% and spinal cord injury occurred in 8% of all patients, especially in patients with a distal landing zone of the FET lower than T10 [25]. Similarly, five-year survival after FET implantation was 75% in another single center series with a total of 286 patients [26].
Despite the potential benefits of the FET technique over the classical ET technique for aortic arch repair, it is still associated with some significant perioperative complications (Table 58.2), such as spinal cord injury, recurrent nerve palsy or endoleaks [14, 16, 23,24,25]. Consequently, deliberate use of this innovative procedure should be avoided since the clinical outcomes reported so far are not clearly superior to the conventional ET technique [4, 14, 22].
Conclusion
The FET technique is an intriguing and innovative hybrid procedure for aortic arch repair that has gained wide acceptance in the cardiovascular community. Latest advances in hybrid technology aiming at a total endovascular arch repair strategy are under clinical investigation and will further reshape the future of aortic arch repair.
References
Shrestha M, Bachet J, Bavaria J, Carrel TP, De Paulis R, Di Bartolomeo R, Etz CD, Grabenwöger M, Grimm M, Haverich A, Jakob H, Martens A, Mestres CA, Pacini D, Resch T, Schepens M, Urbanski PP, Czerny M. Current status and recommendations for use of the frozen elephant trunk technique: a position paper by the vascular domain of EACTS. Eur J Cardiothorac Surg. 2015;47:759–69.
Hagl C, Pichlmaier M, Khaladj N. Elephant trunks in aortic surgery: fresh and frozen. J Thorac Cardiovasc Surg. 2013;145(3 Suppl):S98–102.
Tian DH, Wan B, Di Eusanio M, Black D, Yan TD. A systematic review and meta-analysis on the safety and efficacy of the frozen elephant trunk technique in aortic arch surgery. Ann Cardiothorac Surg. 2013;2:581–91.
Bachet J. Open repair techniques in the aortic arch are still superior. Ann Cardiothorac Surg. 2018;7:328–44.
Borst HG, Walterbusch G, Schaps D. Extensive aortic replacement using “elephant trunk” prosthesis. Thorac Cardiovasc Surg. 1983;31:37–40.
Svensson LG. Rationale and technique for replacement of the ascending aorta, arch and distal aorta using a modified elpahnt trunk procedure. J Card Surg. 1992;7:301–12.
Karck M, Chavan A, Hagl C, Friedrich H, Galanski M, Haverich A. The frozen elephant trunk technique: a new treatment for thoracic aortic aneurysms. J Thorac Cardiovasc Surg. 2003;125:1550–3.
Jakob H, Tsagakis K, Pacini D, Di Bartolomeo R, Mestres C, Mohr F, et al. The International E-vita open registry: data sets of 274 patients. J Cardiovasc Surg. 2011;52:717–23.
Jakob H, Dohle D, Benedik J, Jánosi RA, Schlosser T, Wendt D, Thielmann M, Erbel R, Tsagakis K. Long-term experience with the E-vita open hybrid graft in complex thoracic aortic disease. Eur J Cardiothorac Surg. 2017;51:329–38.
Kazui T, Watanabe A, Inoue N, Inaoka M, Komatsu S. Total aortic arch graft replacement using a prosthetic graft with three branches for acute type A aortic dissection. Kyobu Geka. 1992;45:851–6.
Shrestha M, Pichlmaier M, Martens A, Hagl C, Khaladj N, Haverich A. Total aortic arch replacement with a novel four-branched frozen elephant trunk graft: first-in-man results. Eur J Cardiothorac Surg. 2013;43:406–10.
Grabenwoger M, Alfonso F, Bachet J, Bonser R, Czerny M, Eggebrecht H, et al. Thoracic Endovascular Aortic Repair (TEVAR) for the treatment of aortic diseases: a position statement from the European Association for Cardio-Thoracic Surgery (EACTS) and the European Society of Cardiology (ESC), in collaboration with the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J. 2012;33:1558–63.
Erbel R, Aboyans V, Boileau C, Bossone E, Bartolomeo RD, Eggebrecht H, et al. 2014 ESC guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2014;35:2873–926.
Shrestha M, Bachet J, Bavaria J, Carrel TP, De Paulis R, Di Bartolomeo R, et al. Current status and recommendations for use of the frozen elephant trunk technique: a position paper by the vascular domain of EACTS. Eur J Cardiothorac Surg. 2015;47:759–69.
Haverich A. Aortic arch replacement with frozen elephant trunk-when not to use it. Ann Cardiothorac Surg. 2013;2:592–6.
Damberg A, Schalte G, Autschbach R, Hoffman A. Safety and pitfalls in frozen elephant trunk implantation. Ann Cardiothorac Surg. 2013;2:669–76.
Chiu P, Miller DC. Evolution of surgical therapy for Stanford acute type A aortic dissection. Ann Cardiothorac Surg. 2016;5:275–95.
Hoffman A, Damberg ALM, Schälte G, Mahnken AH, Raweh A, Autschbach R. Thoracic stent graft sizing for frozen elephant trunk repair in acute type A dissection. J Thorac Cardiovasc Surg. 2013;145:964–9.
Criado FJ, Barnatan MF, Rizk Y, et al. Technical strategies to expand stent-graft applicability in the aortic arch and proximal descending thoracic aorta. J Endovasc Ther. 2002;9:II32–8.
Tsagakis K. Angioscopy as a supplement to frozen elephant trunk treatment. Ann Cardiothorac Surg. 2013;2:653–5.
Di Bartolomeo R, Murana G, Di Marco L, Alfonsi J, Gliozzi G, Amodio C, Leone A, Pacini D. Is the frozen elephant trunk frozen? Gen Thorac Cardiovasc Surg. 2019;67:111–7. https://doi.org/10.1007/s11748-018-0911-4.
Chakos A, Jbara J, Yan TD, Tian DH. Long-term survival and related outcomes for the hybrid versus the traditional arch repair – a meta-analysis. Ann Cardiothorac Surg. 2018;7:319–27.
Koullias GJ, Wheatley GH III. State-of-the-art of hybrid procedures for the aortic arch: a meta-analysis. Ann Thorac Surg. 2010;90:689–97.
Cao P, De Rango P, Czerny M, et al. Systematic review of clinical outcomes in hybrid procedures for aortic arch dissections and other arch diseases. J Thorac Cardiovasc Surg. 2012;144:1286–300, 1300.e1–2
Leontyev S, Tsagakis K, Pacini D, Di Bartolomeo R, Mohr FW, Weiss G, Grabenwoeger M, Mascaro JG, Iafrancesco M, Franke UF, Göbel N, Sioris T, Widenka K, Mestres CA, Jakob H. Impact of clinical factors and surgical techniques on early outcome of patients treated with frozen elephant trunk technique by using EVITA open stent-graft: results of a multicentre study. Eur J Cardiothorac Surg. 2016;49:660–6.
Tsagakis K, Wendt D, Dimitriou AM, Thielmann M, Shehada SE, El Gabry M, Jakob HG. The frozen elephant trunk treatment is the operation of choice for all kinds of arch disease. J Cardiovasc Surg. 2018;59:540–6.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Liakopoulos, O.J., Merkle, J., Wahlers, T.C.W. (2020). Hybrid Aortic Arch Repair. In: Raja, S. (eds) Cardiac Surgery. Springer, Cham. https://doi.org/10.1007/978-3-030-24174-2_58
Download citation
DOI: https://doi.org/10.1007/978-3-030-24174-2_58
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-24173-5
Online ISBN: 978-3-030-24174-2
eBook Packages: MedicineMedicine (R0)