Abstract
Radiologic imaging has proven invaluable in confirming clinically suspected pathology, discovering coexistent or unsuspected pathology, and providing a road map for surgical planning. MRI is especially useful for evaluating large and small blending musculotendinous stabilizers in patients with complex injuries or difficult clinical exams. The intent of this article is to review the magnetic resonance imaging (MRI) appearance of the normal musculotendinous anatomy and the appearance of injury.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Posteromedial corner
- Medial collateral ligament (MCL)
- Lateral collateral ligament (LCL)
- Magnetic resonance (MR) imaging (MRI)
- Posterolateral corner
- Radiology
- Knee
- Ligament
- Injury
- Cruciate
1 Introduction
Clinical assessment for ligamentous injury can be imprecise, particularly in certain subsets of patients. More difficult clinical exams include obese patients, patients with pain and guarding, and those with complex injuries (e.g., multiligament injuries). The consequences of an inaccurate evaluation and misdiagnosis may be severe as missed ligamentous injuries have been implicated in accelerated secondary osteoarthritis [1,2,3] and may contribute to cruciate graft failure [4,5,6]. MRI is not without its own limitations, which include artifacts and interobserver variation. MRI is most accurate when performed in the acute to subacute time period (days after the injury). MRI is less accurate and should be used cautiously in cases of chronic injuries as a previously torn ligament with interval scarring may at times appear morphologically intact although physiologically incompetent. The combination of accurate clinical exam with high-quality imaging and interpretation provides the best opportunity for successful treatment outcome. With this in mind, the following chapter will highlight pearls and pitfalls of knee MRI focusing on the normal appearance and injuries to the central, medial, lateral, posteromedial corner, and posterolateral corner (PLC) stabilizers.
1.1 Image Quality
The intent of this article is not to review MRI imaging protocols and equipment, but it is imperative to briefly touch on the subject of image quality. Image quality is dependent upon a number of factors, including imaging equipment and how well the imaging equipment is utilized. The primary factor leading to the varying quality from one MRI to the next is based on the “magnetic field strength” or the strength of the magnet in the MRI. Low field strength MRI ranges from 0.3 to 1.0 T and high field strength MRI ranges between 1.5 and 3.0 T. Higher magnetic field strength results in higher image quality. “Open MRI” usually operates with low field strength and hence results in lower quality images. Thus, it is imperative that both the clinician and patient are aware of the large discrepancy in image quality between low field imaging systems versus those obtained with high field imaging. Without high-quality imaging and appropriate imaging protocols, subtle and sometimes glaring pathology may be missed by even the most imaging astute interpreting physician. Thus, clinicians should be knowledgeable of the equipment and protocols employed by surrounding imaging centers so that they may make educated recommendations to their patients.
1.2 Plain Radiographs
Although plain radiographs cannot directly evaluate ligament injury, they are commonly performed in the setting of traumatic injury and there are several signs or fractures that are known to be associated with ligament injuries. Examples include the “arcuate sign” and Segond fracture, which are discussed later in greater detail. As such, having the plain radiographs available when interpreting MRI is useful and can help direct attention to potential sites of injury.
2 Central Stabilizers: Normal Anatomy and Injury
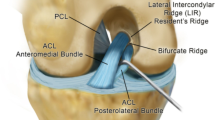
To determine the integrity of the ACL, both its signal and morphology must be closely scrutinized. The sagittal plane of imaging is often utilized as the sequence, which lays out the ligament from its femoral to its tibial attachment. The axial plane should be used together with the sagittal plane, as it best shows the femoral attachment (Fig. 7.1). In the sagittal plane, the normal morphology of the ACL is appreciated as it parallels the roof of the intercondylar notch, following Blumensaat’s line [7]. The normal ACL signal intensity is predominantly hypointense on both T1 and T2 sequences, but the ligament almost always demonstrates internal striations that should not be confused with the injury. There are two functional bundles of the ACL named based on their relative attachments to the tibia: the anteromedial bundle (AMB) and the posterolateral bundle (PLB) [8, 9]. The anteromedial and PLBs of the ACL are not always separated as distinct structures on every MRI. However, they may be seen in the axial and coronal planes.
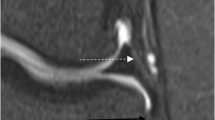
ACL tears most commonly take place in the midsubstance, but can occur anywhere throughout the course of the ligament [7]. Findings suggesting ACL tear include nonvisualization, discontinuity, or abnormal slope or tilt of the ligament [10]. Figure 7.2 demonstrates classic ACL tears. While classic ACL tears are readily apparent on MRI, a tear at the femoral attachment can be subtle and thus overlooked (Fig. 7.3). This femoral avulsion type of tear may not be well depicted in the sagittal plane and is exceedingly difficult to see on open or low field MRI scanners. Therefore, the axial and coronal imaging planes should be employed in one’s search pattern [11]. Despite the lack of clinical instability and characteristic MR appearance, ACL ganglions and mucoid degeneration may at times be confused with an ACL tear on MRI [12] (Fig. 7.4). Finally, MRI should be used with caution in diagnosing chronic ligament tears of any type. A scarred but incompetent ACL may appear intact on MRI and may even scar down to the PCL rather than the femur after an injury (Fig. 7.5).
Coronal PD and axial T2 images in a patient with arthroscopically proven femoral avulsion of the ACL. In a there is increased signal at the femoral attachment of the ACL and the attachment itself is nonvisualized (circle). In b the femoral attachment of the ACL is absent (circle). Compare Fig. 7.3b with Fig. 7.1b, which shows the normal femoral attachment
Demonstrates ACL tear mimics. a Demonstrates an intact ACL with mild diffuse mucoid degeneration (arrows). In a ACL is thickened with T2 bright striations, but the slope is normal, there is no focal discontinuity, and the femoral attachment is intact. b Demonstrates an intact ACL with ACL ganglion (arrow)
Classic bone contusion patterns seen on MRI should raise suspicion of, but are not diagnostic of, ACL tear. When present, the ACL must be scrutinized for injury. The most common pattern is the “kissing contusion” pattern seen with pivot shift injury, which shows contusions in the posterior lateral tibial plateau and lateral femoral condyle [7, 13]. The pivot shift pattern is often accompanied by contrecoup contusion in the posteromedial tibial plateau [14] (Fig. 7.6). Less common bruising patterns with ACL tears include hyperextension (Fig. 7.7) and dashboard (i.e., pretibial impaction in flexion) contusion patterns, the latter almost always seen with multiligamentous injuries [7].
The normal appearance of the PCL is quite different than that of the ACL. The PCL is homogeneously low in signal on all MRI sequences and is not taut but is normally curved from its femoral to its tibial attachment (Fig. 7.8). Unlike the ACL, the classic PCL tear may be more subtle since it rarely demonstrates complete discontinuity. Both the completely torn and the more common partially torn PCL are both well seen in the sagittal plane. The latter is denoted by thickening and intrasubstance fluid bright signal with areas of partial discontinuity [10] (Fig. 7.9). Of note, isolated ACL and PCL injuries are the exception, and when present the posteromedial corner, PLC, and menisci should be double checked for injury [10, 15].
3 Cruciate Grafts: Normal Appearance and Injury
MRI evaluation of cruciate grafts can be challenging for a variety of reasons. In addition to the standard limitations of MRI (i.e., motion), the postoperative images are often hindered by susceptibility artifact and poor fat suppression. The normal MRI appearance of mature ACL and PCL grafts is uniformly low signal on all MRI sequences (Fig. 7.10). This allows one to utilize the same signal, morphology, and orientation changes seen with native cruciate to diagnose ACL and PCL graft tears (Figs. 7.11, 7.12, and 7.13). However, the MRI appearance of an uninjured ACL or PCL graft can be variable depending on the age and type of graft. For example, there can be nonpathologic signal changes in a maturing patellar tendon ACL graft for up to 4 years [16]. Focal or segmental increased signal within the graft on fluid-sensitive sequences can be seen with partial or single-bundle graft tear, fluid between the two bundles, or signal changes from normal graft maturation [17, 18]. The brightness of the signal and the orientation of the signal can be helpful in distinguishing tear from a normal maturing graft. Intermediate intensity (rather than fluid bright) signal alteration that decreases on follow-up exams is typical of graft maturation [16]. Fluid bright signal changes are more concerning for partial thickness graft tear [2]. Secondary findings supporting graft tear include pivot shift bone contusions or signs of graft impingement, the latter which is often due to poor tunnel placement [18,19,20]. Graft impingement manifests on MRI as focal anterior signal changes in the graft and/or bowing of the graft as it contacts the intercondylar roof [18, 20, 21].
a and b Demonstrate two different patients with midsubstance ACL graft tears, as denoted by arrows and circle, respectively. Compare to normal graft in Fig. 7.10a
A double-bundle PCL graft with single-bundle tear. The tear involves the more anterior bundle near the femoral attachment with an intact posterior bundle (circle). Compare the attenuated PCL graft proximally at site of tear (circle) to the normal graft thickness distally where both bundles are intact (arrow)
4 Medial and Lateral Stabilizers
Numerous interdigitating structural layers stabilize both the medial and lateral knee, including both the posteromedial and PLCs. The terminology for these structures is inconsistent in the literature, with numerous names given to the same structures. Because of this, it is important to make sure there is a clear understanding of the terminology utilized in the radiologic report between the musculoskeletal radiologist and orthopedist. In daily practice as well as in the surgical and radiologic literature, these structures are often collectively called the medial collateral ligament (MCL) and lateral collateral ligament (LCL). However, this antiquated terminology undermines the complexity and importance of the individual structures. More recent improved understanding of the intricate anatomy and function of these stabilizers suggests that injuries to these structures should be distinguished rather than lumped together.
5 Medial Stabilizers: Normal Anatomy
Warren initially introduced the layered approach in describing the anatomy stabilizing the middle third of the knee before these structures blend with others as they extend into the anterior and posterior thirds of the knee [22]. These layers, from superficial to deep, are as follows: layer I [crural or sartorius fascia], layer II [tibial collateral ligament (TCL) or superficial MCL (sMCL)], and layer III [deep MCL or middle third capsular ligament]. All three layers are consistently demonstrated on MRI (Fig. 7.14). The deep MCL is a thick condensation of the joint capsule that underlies the TCL and can be broken down into a long thin meniscofemoral ligament and a short thick meniscotibial (coronary) ligament [22, 23]. Of the three layers, this innermost layer may sometimes be challenging to image, and often its components cannot be followed on a single image as they extend from their meniscal to their respective bony attachments. The soft tissue edema that accompanies injury to the deep layer helps to separate these thin structures from the overlying TCL.
The normal medial stabilizer anatomy in the middle third of the knee. Note the thick low signal superficial MCL (thick white arrows). The underlying deep MCL ligament has meniscofemoral (thin black arrow) and meniscotibial components (thin white arrow), which tether the meniscus in place. Also note bucket handle tear (circle)
6 Posteromedial Corner: Normal Anatomy
Further posterior, the deep MCL blends with and reinforces one of the components of the posteromedial corner, the posterior oblique ligament (POL) [22,23,24]. The POL itself is actually three blending ligaments, but on MRI, it can be conceptualized as a single ligament that contributes in forming the posteromedial capsule (Fig. 7.15). The POL, like the deep MCL, has meniscofemoral (MF) and meniscotibial (MT) components. Also like the deep MCL, they attach to the posterior horn of the medial meniscus helping to tether the medial meniscus in place. On MRI, the POL and TCL can be differentiated from one another based on relative location (posterior vs. anterior) and respective tibial attachments (proximal vs. distal) [11].
The second component of the posteromedial corner, the semimembranosus tendon, fans out and attaches to the tibia posteromedially. The two major arms of the semimembranosus, the anterior and direct arms, are well seen on MRI and rarely appear injured. The anterior arm inserts to the medial aspect of the tibia at the level of the joint line, and the direct arm inserts to the posteromedial tibia just below the joint line. Fascial extensions from both arms of this tendon also help to form and reinforce the joint capsule [22,23,24].
7 Medial and Posteromedial Structures: Injury and Pitfalls
Clinical grading of injury to the medial stabilizers is similar to that of most other ligaments [25, 26]. Correlating the clinical MCL injury grade with imaging grade has proven difficult, particularly in the setting of multiple injuries, because of the tendency of overlap and interobserver variation on both the radiologic and surgical sides [27]. Generally speaking, the same imaging criteria utilized for all other ligamentous injuries are also used for the medial stabilizers [28, 29]. Grade 1 injuries demonstrate periligamentous signal changes (edema and/or hemorrhage) on MRI without internal signal changes or areas of discontinuity. Grade 2 injuries demonstrate intrasubstance signal changes in addition to periligamentous signal changes, sometimes with areas of partial discontinuity. Grade 3 tears demonstrate complete discontinuity, often exemplified by wavy ligament. Figures 7.16, 7.17, 7.18, 7.19, 7.20 and 7.21 show varying degrees of injuries to the medial and posteromedial corner stabilizers.
A grade 3 injury to the medial stabilizers and posteromedial stabilizers (POL). a Shows a wavy proximal superficial MCL (large white arrows) with focal discontinuity at the tibial attachment (white circle). The deep MCL is nonvisualized and was torn as well. b Demonstrates nonvisualization of the meniscofemoral (thin black arrow) and meniscotibial (thin white arrow) components of the POL
It is important to be aware of imaging pitfalls in diagnosing injuries to the medial stabilizers. First, periligamentous edema is not diagnostic of “MCL sprain” because it also may accompany meniscal tears, osteoarthritis [30, 31], or edema tracking from ruptured Baker’s cyst. Another common pitfall is misdiagnosing MCL sprain in the setting of patella dislocation [11]. In this instance, edema often tracks superficial to the MCL from the adjacent injury. The classic bone contusions present on MRI with patella dislocation in the medial patellar facet and anterolateral femoral condyle can readily distinguish the two entities in those instances when the clinical picture is confusing.
8 Posterolateral Corner
The large lateral and posterolateral stabilizers including the iliotibial band (ITB), biceps femoris tendon, and fibular collateral ligament (FCL) are well assessed on MRI. However, the evaluation of the smaller ligaments is more challenging because they vary in their configuration anatomically, are inconsistently present, and are obliquely oriented [11]. Despite the above difficulties, evaluation of the PLC can be accomplished with a thorough understanding of the anatomy while correlating all three imaging planes to avoid confusion.
Prior to discussing MRI findings, two important X-ray signs of PLC injury should be noted. A classic X-ray sign of PLC injury is a bony avulsion fracture from the fibular head termed the “arcuate sign” [32] (Fig. 7.22). This type of fracture may indicate injury to any combination of the posterolateral stabilizers including the “arcuate complex” (popliteofibular, arcuate, and fabellofibular ligaments) and/or the conjoined tendon insertion [33, 34]. Unlike the arcuate sign, the Segond fracture is not a direct sign of PLC injury but is highly associated with cruciate tears and PLC injuries [33, 34]. This thin cortical avulsion fracture typically occurs where the anterior aponeurotic extension of the FCL (termed the anterior oblique band) blends with the thin posterior fibers of the ITB to form and reinforce the capsule as it attaches to the lateral tibial rim [35]. The Segond fracture is subtle on X-rays, but the low signal intensity sliver of the avulsed cortex is even more inconspicuous on MRI (Fig. 7.23).
An X-ray with MRI correlation demonstrating a lateral/posterolateral corner injury. a Is an AP radiograph, which shows two superimposed avulsed bone fragments from the fibular head (circle). The smaller and more posterior and medial of the two fragments are the fibular head. This bony avulsion has been termed the “arcuate sign.” The larger and more lateral fragment is Gerdy’s tubercle. b Shows bony avulsion of the iliotibial band insertion onto Gerdy’s tubercle (white arrow). c Shows osseous avulsion of the fibular head, which includes the conjoined tendon (black arrow)
8.1 Lateral and PLC: Anatomy and Injury
The ITB is the terminal extension of the tensor fascia latae, which has five blending layers that insert onto Gerdy’s tubercle [36]. The distinct layers of the ITB are not consistently separated on MRI with standard imaging [11]. The ITB is uncommonly injured, and both the normal and injured ITB are best visualized in the coronal plane.
The large but obliquely oriented FCL and biceps femoris are both well evaluated on sequential coronal MR images (Fig. 7.24). The femoral attachment of the FCL is approximately 2 cm above the joint line, which abuts and is just anterior to the lateral gastrocnemius origin on the lateral femoral epicondyle. The “conjoined insertion” of the FCL is with the biceps femoris tendon onto the head of the fibula far laterally [37, 38].
A coronal MRI demonstrating normal posterolateral corner anatomy. The white arrow demonstrates the normal appearance of the fibular collateral ligament (FCL) (white arrows) from its femoral attachment to its fibular attachment. The conjoined attachment (circle) with the partially imaged biceps femoris (black arrow) can be appreciated. It is abnormal to see the entire FCL on one slice because it is normally obliquely oriented. If seen, as on this image, this is either due to an anteriorly translated tibia from ACL tear or due to technologist error (incorrect obliquely oriented coronal images)
The popliteus complex is made up of a number of structures including the popliteus tendon, popliteofibular ligament, and the popliteal meniscal fascicles. The origin of the popliteus tendon is intra-articular from a sulcus on the lateral femoral condyle, inferior, and anterior to the proximal attachment of the FCL [37, 38] (Fig. 7.25). As the intra-articular portion of the popliteus wraps posteromedially, it gives off a thin anteroinferior fascicle and a thicker posterosuperior popliteomeniscal fascicle, both which help tether the lateral meniscus in place [39]. Tear of these fascicles has been correlated with lateral meniscus tear [40]. The popliteomeniscal fascicles are best seen in the sagittal plane and commonly in the coronal plane (Fig. 7.26). These two fascicles envelope the popliteus tendon as it wraps posteromedially, forming the floor and roof of the popliteus hiatus, respectively [38, 39, 41, 42]. The popliteal hiatus is boundary between the intra- and extra-articular components of the popliteus tendon [43] (Fig. 7.27).
A moderate strain of the popliteus at the proximal myotendinous junction (black arrows). The soft tissue edema nicely delineates the posterior capsule/medial limb of the arcuate ligament (white arrows) as the popliteus exits the joint at the popliteal hiatus. A small portion of the intact biceps femoris tendon can be seen (thin white arrows)
As the popliteus tendon exits the hiatus, it becomes extra-articular, and shortly afterward it gives off its fibular attachment, known as the popliteofibular ligament (Fig. 7.28), which arises laterally from the popliteus at its myotendinous junction. It inserts medial to the attachments of the fabellofibular ligament and arcuate ligament far posterior on the fibular styloid [37, 38, 44]. The thick but short and obliquely oriented popliteofibular ligament is notoriously difficult to image [37, 45] despite being nearly always present on anatomic dissection [46]. The popliteofibular ligament is most commonly a single band, but extensive anatomic variation including multiple bands that differ in their obliquity has been described [37, 38, 41, 47].
The fabellofibular and arcuate ligaments help to form and stabilize the posterolateral knee joint capsule. They are not consistently present in dissection, vary in size and thickness, and can be present alone or in combination [38, 46, 48,49,50]. When present, the fabellofibular ligament arises from the fabella and inserts distally into the lateral base of the fibular head just anterolateral to the popliteofibular ligament [37] (Fig. 7.29). The arcuate ligament has medial and lateral limbs, which ascend as a single ligament from the fibular head just anterior to the fabellofibular ligament (when present together) [37, 38]. The medial and lateral limbs then separate in the form of a Y, with the thicker lateral limb coursing straight proximally and attaching to the lateral femoral condyle in reinforcing the lateral joint capsule [38]. The medial limb courses medial and superficial to the popliteal tendon and then blends with fibers of the popliteal oblique ligament in helping to reinforce the posterior joint capsule [38]. The arcuate ligament, most commonly the medial limb, is usually only seen when thickened or when it is highlighted by edema (see Figs. 7.27, 7.31 and 7.32).
Injuries to the ITB, FCL, biceps femoris, popliteus complex, and capsular structures are rarely in isolation and may occur in various combinations. Attempts should be made to identify injuries to each specific structure, although missed injury to the smallest capsular structures is less consequential than the larger stabilizers like the FCL, conjoined tendon, and popliteofibular ligament. On MRI, the coronal plane best depicts the variety of injuries occurring to the PLC stabilizers (Figs. 7.30, 7.31, 7.32, 7.33 and 7.34). Like the pivot shift contusion pattern with ACL tear, fibular head edema is highly suggestive of PLC injury, and when present, the PLC structures should be closely scrutinized for injury (Fig. 7.35). Knowledge of the PLC insertional relationship to one another on the fibular head (attachments from medial to lateral) may help one to determine which specific structure is injured [43]. For example, edema medially is suggestive of an arcuate complex or popliteofibular ligament injury rather than injury to the more laterally inserting conjoined tendon. For the best chance at accurate diagnosis and as not to confuse these structures with one another, correlation with all three imaging planes is suggested. Despite this, even in the best of circumstances, it may at times be difficult to distinguish specific injuries and also between nonvisualization from injury versus absence due to anatomic variability [11]. In such instances, the radiologic report may convey the high suspicion for a PLC injury. Although the specific ligamentous injury is not specified, the purpose is to alert the surgeon that the PLC needs to be closely evaluated clinically, perhaps under anesthesia at time of surgery.
A moderate strain to the popliteus (black arrows), a grade 1–2 sprain of the popliteofibular ligament at its fibular attachment (large white arrow). Minimal linear signal at conjoined tendon insertion is within normal limits (thin white arrows). Note how edema highlights a portion of the intact arcuate ligament (small black arrow), which is situated between the popliteofibular ligament and conjoined tendon
Fibular head edema at the popliteofibular ligament attachment (circle). There is a grade 2 injury to the popliteofibular ligament (white arrow) and a grade 3 injury to the conjoined tendon insertion (thin white arrows). The extra-articular popliteus at the myotendinous junction was intact (black arrows)
9 Conclusion
When interpreting complex knee injuries on MRI, it should now be apparent that a thorough understanding of the complex anatomy of the knee, high-quality imaging, and a meticulous search pattern are vital to accurate diagnosis. In the acute setting, knee MRI is extremely valuable for presurgical planning, given its high accuracy in diagnosing the structure injured, the degree of the injury, and the specific location of tear within the involved ligament or tendon.
References
Kannus P. Nonoperative treatment of grade II and III sprains of the lateral ligament compartment of the knee. Am J Sports Med. 1989;17(1):83–8.
Recht MP, Parker RD, Irizarry JM. Second time around: evaluating the postoperative anterior cruciate ligament. Magn Reson Imaging Clin N Am. 2000;8(2):285–97.
Hughston JC, Andrews JR, Cross MJ, Moschi A. Classification of knee ligament injuries. Part II. The lateral compartment. J Bone Joint Surg Am. 1976;58:173–9.
Fleming RE Jr, Blatz DJ, McCarroll JR. Posterior problems in the knee. Posterior cruciate insufficiency and posterolateral rotatory insufficiency. Am J Sports Med. 1981;9(2):107–13.
Hughston JC, Jacobson KE. Chronic posterolateral rotatory instability of the knee. J Bone Joint Surg Am. 1985;67(3):351–9.
O’Brien SJ, Warren RF, Pavlov H, Panariello R, Wickiewicz TL. Reconstruction of the chronically insufficient anterior cruciate ligament with the central third of the patellar ligament. J Bone Joint Surg Am. 1991;73(2):278–86.
Sanders TG, Medynski MA, Feller JF, Lawhorn KW. Bone contusion patterns of the knee at MR imaging: footprint of the mechanism of injury. Radiographics. 2000;20:S135–51.
Colombet P, Robinson J, Christel P, et al. Morphology of anterior cruciate ligament attachments for anatomic reconstruction: a cadaveric dissection and radiographic study. Arthroscopy. 2006;22(9):984–92.
Duthon VB, Barea C, Abrassart S, Fasel JH, Fritschy D, Menetrey J. Anatomy of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc. 2006;14(3):204–13.
Roberts CC, Towers JD, Spangehl MJ, Carrino JA, Morrison WB. Advanced MR imaging of the cruciate ligaments. Magn Reson Imaging Clin N Am. 2007;15(1):73–86.
Malone WJ, Verde F, Weiss D, Fanelli GC. MR imaging of knee instability. Magn Reson Imaging Clin N Am. 2009;17:697–724.
Bergin D, Morrison WB, Carrino JA, Nallamshetty SN, Bartolozzi AR. Anterior cruciate ligament ganglia and mucoid degeneration: coexistence and clinical correlation. AJR Am J Roentgenol. 2004;182(5):1283–7.
Murphy BJ, Smith RL, Uribe JW, Janecki CJ, Hechtman KS, Mangasarian RA. Bone signal abnormalities in the posterolateral tibia and lateral femoral condyle in complete tears of the anterior cruciate ligament: a specific sign? Radiology. 1992;182(1):221–4.
Kaplan PA, Gehl RH, Dussault RG, Anderson MW, Diduch DR. Bone contusions of the posterior lip of the medial tibial plateau (contrecoup injury) and associated internal derangements of the knee at MR imaging. Radiology. 1999;211(3):747–53.
Sonin AH, Fitzgerald SW, Hoff FL, Friedman H, Bresler ME. MR imaging of the posterior cruciate ligament: normal, abnormal, and associated injury patterns. Radiographics. 1995;15(3):551–61.
Saupe N, White LM, Chiavaras MM, et al. Anterior cruciate ligament reconstruction grafts: MR imaging features at long-term follow-up–correlation with functional and clinical evaluation. Radiology. 2008;249(2):581–90.
Steiner ME, Hecker AT, Brown CHJ, Hayes WC. Anterior cruciate ligament graft fixation. Comparison of hamstring and patellar tendon grafts. Am J Sports Med. 1994;22(2):240–6.
Bencardino JT, Beltran J, Feldman MI, Rose DJ. MR imaging of complications of anterior cruciate ligament graft reconstruction. Radiographics. 2009;29:2115–26.
Streich NA, Friedrich K, Gotterbarm T, Schmitt H. Reconstruction of the ACL with a semitendinosus tendon graft: a prospective randomized single blinded comparison of double-bundle versus single-bundle technique in male athletes. Knee Surg Sports Traumatol Arthrosc. 2008;16(3):232–8.
Papakonstantinou O, Chung CB, Chanchairujira K, Resnick DL. Complications of anterior cruciate ligament reconstruction: MR imaging. Eur Radiol. 2003;13(5):1106–17.
Howell SM. Principles for placing the tibial tunnel and avoiding roof impingement during reconstruction of a torn anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc. 1998;6(Suppl 1):S49–55.
Warren LF, Marshall JL. The supporting structures and layers on the medial side of the knee: an anatomical analysis. J Bone Joint Surg Am. 1979;61(1):56–62.
LaPrade RF, Engebretsen AH, Ly TV, Johansen S, Wentorf FA, Engebretsen L. The anatomy of the medial part of the knee. J Bone Joint Surg Am. 2007;89(9):2000–10.
Hughston JC, Eilers AF. The role of the posterior oblique ligament in repairs of acute medial (collateral) ligament tears of the knee. J Bone Joint Surg Am. 1973;55(5):923–40.
Cham DM, Hsu CYC. Cartilage and ligament injuries in sports injuries. London: Oxford University Press; 1993. p. 54–70.
Cheryne S. Disorders of the knee. In: Dee R, Mango M, Hurst LC, editors. Principles of orthopaedic practice, vol. 2. New York: McGraw-Hill; 1989. p. 1283.
Reid DC. Sports injury assessment and rehabilitation. New York: Churchill Livingstone; 1992. p. 502–7.
Schweitzer ME, Tran D, Deely DM, Hume EL. Medial collateral ligament injuries: evaluation of multiple signs, prevalence and location of associated bone bruises, and assessment with MR imaging. Radiology. 1995;194(3):825–9.
Stoller DW. Magnetic resonance imaging in orthopaedics and sports medicine. Vol. 1. 3rd ed. Baltimore, MD: Lippincott Williams & Wilkins; 2007. p. 553–4.
Bergin D, Keogh C, O’Connell M, et al. Atraumatic medial collateral ligament oedema in medial compartment knee osteoarthritis. Skeletal Radiol. 2002;31:14–8.
Bergin D, Hochberg H, Zoga AC, Qazi N, Parker L, Morrison WB. Indirect soft-tissue and osseous signs on knee MRI of surgically proven meniscal tears. AJR Am J Roentgenol. 2008;191(1):86–92.
Shindell R, Walsh WM, Connolly JF. Avulsion fracture of the fibula: the “arcuate sign” of posterolateral knee instability. Nebr Med J. 1984;69(11):369–71.
Huang G, Yu JS, Munshi M, et al. Avulsion fracture of the head of the fibula (the “arcuate” sign): MR imaging findings predictive of injuries to the posterolateral ligaments and posterior cruciate ligament. AJR Am J Roentgenol. 2003;180(2):381–7.
Juhng S, Lee JK, Choi S, Yoon K, Roh B, Won J. MR evaluation of the “arcuate” sign of posterolateral knee instability. AJR Am J Roentgenol. 2003;180(2):381–7.
Campos JC, Chung CB, Lektrakul N, et al. Pathogenesis of the Segond fracture: anatomic and MR imaging evidence of an iliotibial tract or anterior oblique band avulsion. Radiology. 2001;219(2):381–6.
Terry GC, Hughston JC, Norwood LA. The anatomy of the iliopatellar band and iliotibial tract. Am J Sports Med. 1986;14(1):39–45.
Munshi M, Pretterklieber ML, Kwak S, Antonio GE, Trudell DJ, Resnick D. MR imaging, MR arthrography, and specimen correlation of the posterolateral corner of the knee: an anatomic study. AJR Am J Roentgenol. 2003;180(4):1095–101.
Diamantopoulos A, Tokis A, Tzurbakis M, Patsopoulos I, Georgoulis A. The posterolateral corner of the knee: evaluation under microsurgical dissection. Arthroscopy. 2005;21(7):826–33.
Peduto AJ, Nguyen A, Trudell DJ, Resnick DL. Popliteomeniscal fascicles: anatomic considerations using MR arthrography in cadavers. AJR Am J Roentgenol. 2008;190(2):442–8.
De Smet AA, Asinger DA, Johnson RL. Abnormal superior popliteomeniscal fascicle and posterior pericapsular edema: indirect MR imaging signs of a lateral meniscal tear. AJR Am J Roentgenol. 2001;176(1):63–6.
Staubli HU, Birrer S. The popliteus tendon and its fascicles at the popliteal hiatus: gross anatomy and functional arthroscopic evaluation with and without anterior cruciate ligament deficiency. Arthroscopy. 1990;6(3):209–20.
Cohn AK, Mains DB. Popliteal hiatus of the lateral meniscus. Anatomy and measurement at dissection of 10 specimens. Am J Sports Med. 1979;7(4):221–6.
Lee J, Papakonstantinou O, Brookenthal KR, Trudell D, Resnick DL. Arcuate sign of posterolateral knee injuries: anatomic, radiographic, and MR imaging data related to patterns of injury. Skeletal Radiol. 2003;32(11):619–27.
Brinkman J, Schwering PJA, Blankevoort L, Kooloos JG, Luites J, Wymenga AB. The insertion geometry of the posterolateral corner of the knee. J Bone Joint Surg Br. 2005;87(10):1364–8.
De Maeseneer M, Shahabpour M, Vanderdood K, De Ridder F, Van Roy F, Osteaux M. Posterolateral supporting structures of the knee: findings on anatomic dissection, anatomic slices and MR images. Eur Radiol. 2001;11(11):2170–7.
Watanabe Y, Moriya H, Takahashi K, et al. Functional anatomy of the posterolateral structures of the knee. Arthroscopy. 1993;9(1):57–62.
Ullrich K, Krudwig WK, Witzel U. Posterolateral aspect and stability of the knee joint. I. Anatomy and function of the popliteus muscle-tendon unit: an anatomical and biomechanical study. Knee Surg Sports Traumatol Arthrosc. 2002;10(2):86–90.
Maynard MJ, Deng X, Wickiewicz TL, Warren RF. The popliteofibular ligament. Rediscovery of a key element in posterolateral stability. Am J Sports Med. 1996;24(3):311–6.
Sudasna S, Harnsiriwattanagit K. The ligamentous structures of the posterolateral aspect of the knee. Bull Hosp Jt Dis Orthop Inst. 1990;50(1):35–40.
Seebacher JR, Inglis AE, Marshall JL, Warren RF. The structure of the posterolateral aspect of the knee. J Bone Joint Surg Am. 1982;64(4):536–41.
Acknowledgements
Special thanks to prior contributors Faiz Syed MD and Peter Bolos MD.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
James Malone, W., Hostmeyer, S.C. (2019). MRI Imaging in the Multiple-Ligament-Injured Knee. In: Fanelli, G. (eds) The Multiple Ligament Injured Knee. Springer, Cham. https://doi.org/10.1007/978-3-030-05396-3_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-05396-3_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-05395-6
Online ISBN: 978-3-030-05396-3
eBook Packages: MedicineMedicine (R0)