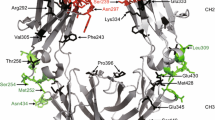
Abstract
Therapeutic antibodies used in the treatment of cancer patients are able to mediate diverse effector mechanisms. Dependent on tumor entity, localization, and tumor burden different effector mechanisms may contribute to the in vivo antitumor activity to a variable degree. Especially Fc-mediated effector functions such as antibody-dependent cell-mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC) have been suggested as being important for the in vivo activity of therapeutic antibodies like rituximab or trastuzumab. In recent years, several strategies have been pursued to further optimize the cytotoxic potential of monoclonal antibodies by modifying their Fc part (Fc engineering) with the ultimate goal to enhance antibody therapy.
Since Fc engineering approaches are applicable to any Fc-containing molecule, strategies to enhance CDC or ADCC activity of full antibodies or scFv-Fc fusion proteins by altering the primary Fc sequence are described.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Nimmerjahn F, Ravetch JV (2005) Divergent immunoglobulin g subclass activity through selective Fc receptor binding. Science 310(5753):1510–1512
Musolino A et al (2008) Immunoglobulin G fragment C receptor polymorphisms and clinical efficacy of trastuzumab-based therapy in patients with HER-2/neu-positive metastatic breast cancer. J Clin Oncol 26(11):1789–1796
Weng WK, Levy R (2003) Two immunoglobulin G fragment C receptor polymorphisms independently predict response to rituximab in patients with follicular lymphoma. J Clin Oncol 21(21):3940–3947
Carter PJ (2006) Potent antibody therapeutics by design. Nat Rev Immunol 6:343–357
Desjarlais JR et al (2007) Optimizing engagement of the immune system by anti-tumor antibodies: an engineer’s perspective. Drug Discov Today 12(21–22):898–910
Jefferis R, Lund J, Goodall M (1995) Recognition sites on human IgG for Fc gamma receptors: the role of glycosylation. Immunol Lett 44(2–3):111–117
Lund J et al (1996) Multiple interactions of IgG with its core oligosaccharide can modulate recognition by complement and human Fc gamma receptor I and influence the synthesis of its oligosaccharide chains. J Immunol 157(11):4963–4969
Jefferis R (2009) Recombinant antibody therapeutics: the impact of glycosylation on mechanisms of action. Trends Pharmacol Sci 30(7):356–362
Shields RL et al (2001) High resolution mapping of the binding site on human IgG1 for Fc gamma RI, Fc gamma RII, Fc gamma RIII, and FcRn and design of IgG1 variants with improved binding to the Fc gamma R. J Biol Chem 276(9):6591–6604
Lazar GA et al (2006) Engineered antibody Fc variants with enhanced effector function. Proc Natl Acad Sci U S A 103(11):4005–4010
Presta LG et al (2002) Engineering therapeutic antibodies for improved function. Biochem Soc Trans 30(4):487–490
Stavenhagen JB et al (2007) Fc optimization of therapeutic antibodies enhances their ability to kill tumor cells in vitro and controls tumor expansion in vivo via low-affinity activating Fcgamma receptors. Cancer Res 67(18):8882–8890
Sazinsky SL et al (2008) Aglycosylated immunoglobulin G1 variants productively engage activating Fc receptors. Proc Natl Acad Sci U S A 105(51):20167–20172
Idusogie EE et al (2001) Engineered antibodies with increased activity to recruit complement. J Immunol 166(4):2571–2575
Repp R et al (2011) Combined Fc-protein- and Fc-glyco-engineering of scFv-Fc fusion proteins synergistically enhances CD16a binding but does not further enhance NK-cell mediated ADCC. J Immunol Methods 373(1–2):67–78
Boel E et al (2000) Functional human monoclonal antibodies of all isotypes constructed from phage display library-derived single-chain Fv antibody fragments. J Immunol Methods 239(1–2):153–166
Derer S et al (2012) Fc engineering: design, expression, and functional characterization of antibody variants with improved effector function. Methods Mol Biol 907:519–536
Clark MR (1997) IgG effector mechanisms. Chem Immunol 65:88–110
Peipp M, van de Winkel JG, Valerius T (2011) Molecular engineering to improve antibodies’ anti-lymphoma activity. Best Pract Res Clin Haematol 24(2):217–229
Jung ST et al (2010) Aglycosylated IgG variants expressed in bacteria that selectively bind FcgammaRI potentiate tumor cell killing by monocyte-dendritic cells. Proc Natl Acad Sci U S A 107(2):604–609
Horton HM et al (2011) Antibody-mediated coengagement of FcgammaRIIb and B cell receptor complex suppresses humoral immunity in systemic lupus erythematosus. J Immunol 186(7):4223–4233
Richards JO et al (2008) Optimization of antibody binding to FcgammaRIIa enhances macrophage phagocytosis of tumor cells. Mol Cancer Ther 7(8):2517–2527
Moore GL et al (2010) Engineered Fc variant antibodies with enhanced ability to recruit complement and mediate effector functions. MAbs 2(2):181–189
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media, New York
About this protocol
Cite this protocol
Derer, S. et al. (2014). Fc Engineering of Antibodies and Antibody Derivatives by Primary Sequence Alteration and Their Functional Characterization. In: Ossipow, V., Fischer, N. (eds) Monoclonal Antibodies. Methods in Molecular Biology, vol 1131. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-992-5_33
Download citation
DOI: https://doi.org/10.1007/978-1-62703-992-5_33
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-991-8
Online ISBN: 978-1-62703-992-5
eBook Packages: Springer Protocols