Abstract
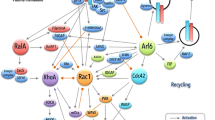
The coordinate modulation of the cellular functions of cadherins and integrins plays an essential role in fundamental physiological and pathological processes, including morphogenesis, tissue differentiation and renewal, wound healing, immune surveillance, inflammatory response, tumor progression, and metastasis. Recent findings state the molecular mechanisms underlying the fine-balanced relationship between cadherins and integrins. In particular, some of the novel results recently obtained raise the possibility of a pivotal role for the small GTPase Rap1 in the functional crosstalk between cadherins and integrins. Considering the importance of the molecular signalling triggered by Rap1, here we provide protocols to study this small GTPase in signalling pathways involving cadherins and integrins.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hynes RO (2002) Integrins: bidirectional, allosteric signaling machines. Cell 110:673–687
Wheelock MJ, Johnson KR (2003) Cadherins as modulators of cellular phenotype. Annu Rev Cell Dev Biol 19:207–235
Peifer M, Yap AS (2003) Traffic control: P120-catenin acts as a gatekeeper to control the fate of classical cadherins in mammalian cells. J Cell Biol 163:437–440
Frame MC (2002) Src in cancer: deregulation and consequences for cell behaviour. Biochim Biophys Acta 1602:114–130
Thiery JP (2002) Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer 2:442–454
Avizienyte E, Wyke AW, Jones RJ et al (2002) Src-induced de-regulation of e-cadherin in colon cancer cells requires integrin signalling. Nat Cell Biol 4:632–638
Gimond C, van Der Flier A, van Delft S et al (1999) Induction of cell scattering by expression of beta1 integrins in beta1-deficient epithelial cells requires activation of members of the rho family of gtpases and downregulation of cadherin and catenin function. J Cell Biol 147:1325–1340
Hintermann E, Yang N, O’Sullivan D et al (2005) Integrin alpha6beta4–erbb2 complex inhibits haptotaxis by up-regulating e-cadherin cell–cell junctions in keratinocytes. J Biol Chem 280:8004–8015
Hodivala KJ, Watt FM (2004) Evidence that cadherins play a role in the downregulation of integrin expression that occurs during keratinocyte terminal differentiation. J Cell Biol 124:589–600
Huttenlocher A, Lakonishok M, Kinder M et al (1998) Integrin and cadherin synergy regulates contact inhibition of migration and motile activity. J Cell Biol 141:515–526
Lu Q, Paredes M, Zhang J et al (1998) Basal extracellular signal-regulated kinase activity modulates cell–cell and cell–matrix interactions. Mol Cell Biol 18:3257–3265
Monier-Gavelle F, Duband JL (1997) Cross talk between adhesion molecules: control of n-cadherin activity by intracellular signals elicited by beta1 and beta3 integrins in migrating neural crest cells. J Cell Biol 137:1663–1681
Nelson CM, Pirone DM, Tan JL et al (2004) Vascular endothelial-cadherin regulates cytoskeletal tension, cell spreading, and focal adhesions by stimulating rhoa. Mol Biol Cell 15:2943–2953
Retta SF, Cassara G, D’Amato M et al (2001) Cross talk between beta(1) and alpha(v) integrins: beta(1) affects beta(3) mrna stability. Mol Biol Cell 12:3126–3138
Schreider C, Peignon G, Thenet S et al (2002) Integrin-mediated functional polarization of caco-2 cells through e-cadherin—actin complexes. J Cell Sci 115:543–552
von Schlippe M, Marshall JF, Perry P et al (2000) Functional interaction between e-cadherin and alphav-containing integrins in carcinoma cells. J Cell Sci 113:425–437
Weber GF, Bjerke MA, DeSimone DW (2011) Integrins and cadherins join forces to form adhesive networks. J Cell Sci 124:1183–1193
Bos JL, de Rooij J, Reedquist KA (2001) Rap1 signalling: adhering to new models. Nat Rev Mol Cell Biol 2:369–377
Ohba Y, Kurokawa K, Matsuda M (2003) Mechanism of the spatio-temporal regulation of ras and rap1. EMBO J 22:859–869
Knox AL, Brown NH (2002) Rap1 gtpase regulation of adherens junction positioning and cell adhesion. Science 295:1285–1288
Bivona TG, Wiener HH, Ahearn IM et al (2004) Rap1 up-regulation and activation on plasma membrane regulates t cell adhesion. J Cell Biol 164:461–470
Caron E (2003) Cellular functions of the rap1 gtp-binding protein: a pattern emerges. J Cell Sci 116:435–440
Dustin ML, Bivona TG, Philips MR (2004) Membranes as messengers in t cell adhesion signaling. Nat Immunol 5:363–372
Katagiri K, Maeda A, Shimonaka M et al (2003) Rapl, a rap1-binding molecule that mediates rap1-induced adhesion through spatial regulation of lfa-1. Nat Immunol 4:741–748
Hogan C, Serpente N, Cogram P et al (2004) Rap1 regulates the formation of e-cadherin-based cell–cell contacts. Mol Cell Biol 24:6690–6700
Price LS, Hajdo-Milasinovic A, Zhao J (2004) Rap1 regulates e-cadherin-mediated cell–cell adhesion. J Biol Chem 279:35127–35132
Yajnik V, Paulding C, Sordella R et al (2003) Dock4, a gtpase activator, is disrupted during tumorigenesis. Cell 112:673–684
Hattori M, Minato N (2003) Rap1 gtpase: functions, regulation, and malignancy. J Biochem 134:479–484
Ricono JM, Huang M, Barnes LA et al (2009) Specific cross-talk between epidermal growth factor receptor and integrin alphavbeta5 promotes carcinoma cell invasion and metastasis. Cancer Res 69:1383–1391
Balzac F, Avolio M, Degani S et al (2005) E-cadherin endocytosis regulates the activity of rap1: a traffic light gtpase at the crossroads between cadherin and integrin function. J Cell Sci 118:4765–4783
Chrzanowska-Wodnicka M (2010) Regulation of angiogenesis by a small gtpase rap1. Vascul Pharmacol 53:1–10
Ferro E, Goitre L, Retta SF et al (2012) The interplay between ros and ras gtpases: physiological and pathological implications. J Signal Transduct 2012:365769. doi:10.1155/2012/365769
Goitre L, Pergolizzi B, Ferro E et al (2012) Molecular crosstalk between integrins and cadherins: do reactive oxygen species set the talk? J Signal Transduct 2012:807682. doi:10.1155/2012/807682
Retta SF, Balzac F, Avolio M (2006) Rap1: a turnabout for the crosstalk between cadherins and integrins. Eur J Cell Biol 85:283–293
Ambesi-Impiombato FS, Parks LA, Coon HG (1980) Culture of hormone-dependent functional epithelial cells from rat thyroids. Proc Natl Acad Sci U S A 77:3455–3459
Franke B, van Triest M, de Bruijn KM et al (2000) Sequential regulation of the small gtpase rap1 in human platelets. Mol Cell Biol 20:779–785
Rottner K, Krause M, Gimona M et al (2001) Zyxin is not colocalized with vasodilator-stimulated phosphoprotein (vasp) at lamellipodial tips and exhibits different dynamics to vinculin, paxillin, and vasp in focal adhesions. Mol Biol Cell 12:3103–3113
Zaidel-Bar R, Ballestrem C, Kam Z et al (2003) Early molecular events in the assembly of matrix adhesions at the leading edge of migrating cells. J Cell Sci 116:4605–4613
Zaidel-Bar R, Cohen M, Addadi L et al (2004) Hierarchical assembly of cell-matrix adhesion complexes. Biochem Soc Trans 32:416–420
Retta SF, Barry ST, Critchley DR et al (1996) Focal adhesion and stress fiber formation is regulated by tyrosine phosphatase activity. Exp Cell Res 229:307–317
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Goitre, L., Retta, S.F. (2014). Combined Pulldown and Time-Lapse Microscopy Studies for Determining the Role of Rap1 in the Crosstalk Between Integrins and Cadherins. In: Trabalzini, L., Retta, S. (eds) Ras Signaling. Methods in Molecular Biology, vol 1120. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-791-4_12
Download citation
DOI: https://doi.org/10.1007/978-1-62703-791-4_12
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-790-7
Online ISBN: 978-1-62703-791-4
eBook Packages: Springer Protocols