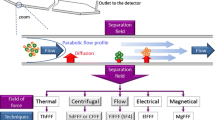
Abstract
Field-flow fractionation is an analytical technique that allows the separation of particles over a size range, from a few nanometers to several microns in diameter. The separation takes place under mild conditions and is suited for the analysis of neutral or charged particles. A single measurement yields the size and concentration of each component of a mixture. However, developing a suitable fractionation method can be tedious and time-consuming. In this chapter, we present asymmetrical flow field-flow fractionation (AF4) conditions that have proven their reliability for the analysis of quantum dots and other nanoparticles in the 5–50 nm size range. Common pitfalls are emphasized together with strategies to overcome them.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Giddings JC (1966) A new separation concept based on a coupling of concentration and flow nonuniformities. Separation Sci 1(1):123–125
Giddings JC (1989) Field-flow fractionation of macromolecules. J Chromatogr 470(2):327–35
Giddings JC et al (1980) Analysis of biological macromolecules and particles by field-flow fractionation. Methods Biochem Anal 26:79–136
Schimpf ME, Caldwell K, Giddings JC (2000) Field flow fractionation handbook, vol xviii. Wiley-Interscience, New York, p 592
Thompson GH, Myers MN, Giddings JC (1969) Thermal field-flow fractionation of polystyrene samples. Anal Chem 41(10):1219–1222
Giddings JC, Yang FJF, Myers MN (1974) Sedimentation field-flow fractionation. Anal Chem 46(13):1917–1924
Caldwell KD, Gao YS (1993) Electrical field-flow fractionation in particle separation. 1. Monodisperse standards. Anal Chem 65(13):1764–1772
Giddings JC, Yang FJ, Myers MN (1976) Flow-field-flow fractionation: a versatile new separation method. Science 193(4259):1244–1245
Vickrey TM, Garcia-ramirez JA (1980) Magnetic field-flow fractionation: theoretical basis. Sep Sci Technol 15(6):1297–1304
Wahlund KG, Giddings JC (1987) Properties of an asymmetrical flow field-flow fractionation channel having one permeable wall. Anal Chem 59(9):1332–1339
Wahlund KG, Litzen A (1989) Application of an asymmetrical flow field-flow fractionation channel to the separation and characterization of proteins, plasmids, plasmid fragments, polysaccharides and unicellular algae. J Chromatogr 461:73–87
Fraunhofer W, Winter G (2004) The use of asymmetrical flow field-flow fractionation in pharmaceutics and biopharmaceutics. Eur J Pharm Biopharm 58(2):369–383
Weers JG, Arlauskas RA (2004) Particle size analysis of perfluorocarbon emulsions in a complex whole blood matrix by sedimentation field-flow fractionation. Colloids Surf B Biointerfaces 33(3–4):265–269
Pinaud F et al (2010) Probing cellular events, one quantum dot at a time. Nat Methods 7(4):275–285
Maysinger D, Lovric J (2007) Quantum dots and other fluorescent nanoparticles: quo vadis in the cell? Adv Exp Med Biol 620:156–167
Pierobon P, Cappello G (2011) Quantum dots to tail single bio-molecules inside living cells. Adv Drug Deliv Rev 64(2):167–178
Huang HC et al (2011) Inorganic nanoparticles for cancer imaging and therapy. J Control Release 155(3):344–357
Mahmoudi M, Serpooshan V, Laurent SS (2011) Engineered nanoparticles for biomolecular imaging. Nanoscale 3(8):3007–3026
Algar WR et al (2011) The controlled display of biomolecules on nanoparticles: a challenge suited to bioorthogonal chemistry. Bioconjug Chem 22(5):825–858
Rosenthal SJ et al (2011) Biocompatible quantum dots for biological applications. Chem Biol 18(1):10–24
Choi HS, Frangioni JV (2010) Nanoparticles for biomedical imaging: fundamentals of clinical translation. Mol Imaging 9(6):291–310
Hutter E, Maysinger D (2011) Gold nanoparticles and quantum dots for bioimaging. Microsc Res Tech 74(7):592–604
Gaponik N et al (2010) Progress in the light emission of colloidal semiconductor nanocrystals. Small 6(13):1364–1378
Smith MH et al (2010) Monitoring the erosion of hydrolytically-degradable nanogels via multiangle light scattering coupled to asymmetrical flow field-flow fractionation. Anal Chem 82(2):523–530
Giddings JC (1993) Field-flow fractionation: analysis of macromolecular, colloidal, and particulate materials. Science 260(5113):1456–1465
Pons T et al (2009) Synthesis of near-infrared-emitting, water-soluble CdTeSe/CdZnS core/shell quantum dots. Chem Mater 21(8):1418–1424
Bouby M, Geckeis H, Geyer FW (2008) Application of asymmetric flow field-flow fractionation (AsFlFFF) coupled to inductively coupled plasma mass spectrometry (ICPMS) to the quantitative characterization of natural colloids and synthetic nanoparticles. Anal Bioanal Chem 392(7–8):1447–1457
Hassellov M et al (2008) Nanoparticle analysis and characterization methodologies in environmental risk assessment of engineered nanoparticles. Ecotoxicology 17(5):344–361
Rameshwar T et al (2006) Determination of the size of water-soluble nanoparticles and quantum dots by field-flow fractionation. J Nanosci Nanotechnol 6(8):2461–2467
Al Hajaj et al. (2011) ACS Nano, 5(6): 4909–4918
Zattoni A et al (2009) Asymmetrical flow field-flow fractionation with multi-angle light scattering detection for the analysis of structured nanoparticles. J Chromatogr A 1216(52):9106–9112
Hupfeld S et al (2010) Liposome fractionation and size analysis by asymmetrical flow field-flow fractionation/multi-angle light scattering: influence of ionic strength and osmotic pressure of the carrier liquid. Chem Phys Lipids 163(2):141–147
Ricq L et al (1997) Electrokinetic characterization of polyethersulfone UF membranes. Desalination 109(3):253–261
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this protocol
Cite this protocol
Moquin, A., Winnik, F.M., Maysinger, D. (2013). Separation Science: Principles and Applications for the Analysis of Bionanoparticles by Asymmetrical Flow Field-Flow Fractionation (AF4). In: Weissig, V., Elbayoumi, T., Olsen, M. (eds) Cellular and Subcellular Nanotechnology. Methods in Molecular Biology, vol 991. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-336-7_30
Download citation
DOI: https://doi.org/10.1007/978-1-62703-336-7_30
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-335-0
Online ISBN: 978-1-62703-336-7
eBook Packages: Springer Protocols