Abstract
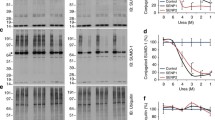
Studies on the regulation of nuclear receptors, such as the peroxisome proliferator-activated receptors (PPARs), are important to enhance our understanding of their molecular, cellular, and physiological behavior. A decade ago, it was shown that the SUMOylation pathway plays a very important role in the regulation of transcription factor activity. The SUMOylation process involves the covalent binding of SUMO protein to the target protein. However, experimental procedures to demonstrate that low-expressed proteins, such as PPARs, are SUMOylated, remain tricky, and require specific optimization for each protein.
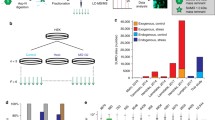
Here, we provide a simple and useful experimental method to investigate the SUMOylation of PPARs in a cellular context. The procedure for studying SUMOylation in living cells is based on the purification under denaturating conditions of total SUMOylated proteins followed by the specific detection of the PPAR proteins. For that purpose, cells are transfected with both 6xHistidine-tagged SUMO and PPAR expression vectors. Since the polyHistidine tag binds to nickel cationic ion-linked agarose matrix (Ni-NTA matrix), His-tagged SUMO proteins covalently linked to the protein substrate can be specifically precipitated and separated from the unSUMOylated proteins. The SUMO-modified PPAR proteins can subsequently be visualized by western blotting using anti-PPAR antibodies. Many questions relative to the regulation of PPAR SUMOylation can be appropriately addressed by adapting this protocol.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hochstrasser M (2009) Origin and function of ubiquitin-like proteins. Nature 458:422–429
Gareau JR, Lima CD (2010) The SUMO pathway: emerging mechanisms that shape specificity, conjugation and recognition. Nat Rev Mol Cell Biol 11:861–871
Geiss-Friedlander R, Melchior F (2007) Concepts in sumoylation: a decade on. Nat Rev Mol Cell Biol 8:947–956
Hay RT (2005) SUMO: a history of modification. Mol Cell 18:1–12
Kim JH, Baek SH (2009) Emerging roles of desumoylating enzymes. Biochim Biophys Acta 1792:155–162
Zhao J (2007) Sumoylation regulates diverse biological processes. Cell Mol Life Sci 64:3017–3033
Treuter E, Venteclef N (2010) Transcriptional control of metabolic and inflammatory pathways by nuclear receptor SUMOylation. Biochim Biophys Acta 1812(8):909–918
Pourcet B, Fuchart J, Staels B et al (2006) Selective PPAR modulators, dual and pan PPAR agonists: multimodal drugs for the treatment of type 2 diabetes and atherosclerosis. Expert Opin Emerg Drugs 11:379–401
Nolte RT, Wisely G, Westin S et al (1998) Ligand binding and co-activator assembly of the peroxisome proliferator-activated receptor-gamma. Nature 395:137–143
Xu HE, Lambert M, Montana V et al (1999) Molecular recognition of fatty acids by peroxisome proliferator-activated receptors. Mol Cell 3:397–403
Blanquart C, Barbier O, Fruchart J et al (2003) Peroxisome proliferator-activated receptors: regulation of transcriptional activities and roles in inflammation. J Steroid Biochem Mol Biol 85:267–273
Ghisletti S, Huang W, Ogawa S et al (2007) Parallel SUMOylation-dependent pathways mediate gene- and signal-specific transrepression by LXRs and PPARgamma. Mol Cell 25:57–70
Ohshima T, Koga H, Shimotohno K (2004) Transcriptional activity of peroxisome proliferator-activated receptor gamma is modulated by SUMO-1 modification. J Biol Chem 279:29551–29557
Pascual G, Fong A, Ogawa S et al (2005) A SUMOylation-dependent pathway mediates transrepression of inflammatory response genes by PPAR-gamma. Nature 437:759–763
Pourcet B, Pineda-Torra I, Derudas B et al (2010) SUMOylation of human peroxisome proliferator-activated receptor alpha inhibits its trans-activity through the recruitment of the nuclear corepressor NCoR. J Biol Chem 285:5983–5992
Blomster HA, Imanishi S, Siimes J et al (2010) In vivo identification of sumoylation sites by a signature tag and cysteine-targeted affinity purification. J Biol Chem 285:19324–19329
Bruderer R, Tatham M, Plechanovova A et al (2011) Purification and identification of endogenous polySUMO conjugates. EMBO Rep 12:142–148
Ganesan AK, Kho Y, Kim S et al (2007) Broad spectrum identification of SUMO substrates in melanoma cells. Proteomics 7:2216–2221
Golebiowski F, Matic I, Tatham M et al (2009) System-wide changes to SUMO modifications in response to heat shock. Sci Signal 2:ra24
Miller MJ, Barrett-Witt G, Hua Z et al (2010) Proteomic analyses identify a diverse array of nuclear processes affected by small ubiquitin-like modifier conjugation in Arabidopsis. Proc Natl Acad Sci USA 107:16512–16517
Choi SJ, Chung S, Rho E et al (2006) Negative modulation of RXRalpha transcriptional activity by small ubiquitin-related modifier (SUMO) modification and its reversal by SUMO-specific protease SUSP1. J Biol Chem 281:30669–30677
Kaul S, Blackford J Jr, Cho S et al (2002) Ubc9 is a novel modulator of the induction properties of glucocorticoid receptors. J Biol Chem 277:12541–12549
Poukka H, Aarnisalo P, Karvonen U et al (1999) Ubc9 interacts with the androgen receptor and activates receptor-dependent transcription. J Biol Chem 274:19441–19446
Tatham MH, Jaffray E, Vaughan O et al (2001) Polymeric chains of SUMO-2 and SUMO-3 are conjugated to protein substrates by SAE1/SAE2 and Ubc9. J Biol Chem 276:35368–35374
Leuenberger N, Pradervand S, Wahli W (2009) Sumoylated PPARalpha mediates sex-specific gene repression and protects the liver from estrogen-induced toxicity in mice. J Clin Invest 119:3138–3148
Yamashita D, Yamaguchi T, Shimizu M et al (2004) The transactivating function of peroxisome proliferator-activated receptor gamma is negatively regulated by SUMO conjugation in the amino-terminal domain. Genes Cells 9:1017–1029
Tatham MH, Rodriguez M, Xirodimas D et al (2009) Detection of protein SUMOylation in vivo. Nat Protoc 4:1363–1371
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Nakabayashi H, Taketa K, Miyano K et al (1982) Growth of human hepatoma cells lines with differentiated functions in chemically defined medium. Cancer Res 42:3858–3863
Staels B, Koenig W, Habib A et al (1998) Activation of human aortic smooth-muscle cells is inhibited by PPARalpha but not by PPARgamma activators. Nature 393:790–793
Degerny C, Monte D, Beaudoin C et al (2005) SUMO modification of the Ets-related transcription factor ERM inhibits its transcriptional activity. J Biol Chem 280:24330–24338
Sentis S, Le Romancer M, Bianchin C et al (2005) Sumoylation of the estrogen receptor alpha hinge region regulates its transcriptional activity. Mol Endocrinol 19:2671–2684
Hwang EJ, Lee J, Jeong J et al (2009) SUMOylation of RORalpha potentiates transcriptional activation function. Biochem Biophys Res Commun 378:513–517
Liu B, Shuai K (2008) Regulation of the sumoylation system in gene expression. Curr Opin Cell Biol 20:288–293
Poukka H, Karvonen U, Janne O et al (2000) Covalent modification of the androgen receptor by small ubiquitin-like modifier 1 (SUMO-1). Proc Natl Acad Sci USA 97:14145–14150
Tojo M, Matsuzaki K, Minami T et al (2002) The aryl hydrocarbon receptor nuclear transporter is modulated by the SUMO-1 conjugation system. J Biol Chem 277:46576–46585
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this protocol
Cite this protocol
Pourcet, B., Staels, B., Glineur, C. (2013). PPAR SUMOylation: Some Useful Experimental Tips. In: Badr, M., Youssef, J. (eds) Peroxisome Proliferator-Activated Receptors (PPARs). Methods in Molecular Biology, vol 952. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-155-4_10
Download citation
DOI: https://doi.org/10.1007/978-1-62703-155-4_10
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-154-7
Online ISBN: 978-1-62703-155-4
eBook Packages: Springer Protocols