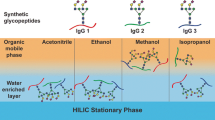
Abstract
Glycoproteins, and in particular glycopeptides, are highly hydrophilic and are often not retained by reversed phase (RP) chromatography. The separation principle of normal phase (NP) is based on hydrophilic interactions, which in many aspects is complementary to RP separations. Hydrophilic interaction liquid chromatography (HILIC) is a fairly new variation of the NP separations used in the 1970s, the major difference being the use of aqueous solvents. HILIC provides a versatile tool for enrichment of glycopeptides before mass spectrometric (MS) analysis, particularly when used for solid phase extraction (SPE), or in combination with other chromatographic resins or ion-pairing reagents. HILIC SPE can be used for glyco-profiling, i.e., for determining the glycan heterogeneity at one specific glycosylation site, for enrichment of glycopeptides from a complex mixture of peptides, as well as for pre-fractionation of complex samples at the protein or peptide level. In this chapter we present a straightforward HILIC SPE enrichment technique and then combine C18 RP and HILIC enrichment for analysis of glycopeptides. Finally, we demonstrate HILIC enrichment using trifluoroacetic acid as an ion-pairing reagent for the enrichment of glycopeptides prior to mass spectrometry analysis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Varki A (1993) Biological roles of oligosaccharides: all of the theories are correct. Glycobiology 3:97–130
Gagneux P, Varki A (1999) Evolutionary considerations in relating oligosaccharide diversity to biological function. Glycobiology 9:747–755
Varki A, Lowe JB (1999) Biological roles of glycans. In: Varki A, Cummings R, Esko J, Freeze H, Hart G, Marth J (eds) Essentials of glycobiology, 2nd edn. Cold Spring Harbor Laboratory Press, Woodbury, NY, pp 57–68
Spiro RG (2002) Protein glycosylation: nature, distribution, enzymatic formation, and disease implications of glycopeptide bonds. Glycobiology 12:43R–56R
Deshpande N, Jensen PH, Packer NH, Kolarich D (2009) GlycoSpectrumScan: fishing glycopeptides from MS spectra of protease digests of human colostrum sIgA. J Proteome Res 9:1063–1075
Larsen MR, Højrup P, Roepstorff P (2005) Characterization of gel-separated glycoproteins using two-step proteolytic digestion combined with sequential microcolumns and mass spectrometry. Mol Cell Proteomics 4:107–119
Mirgorodskaya E, Krogh TN, Roepstorff P (2000) Characterization of protein glycosylation by MALDI-TOFMS. In: Chapman J (ed) Methods in molecular biology: mass spectrometry of proteins and peptides. Humana Press, Totawa, NJ, pp 273–292
Hägglund P, Bunkenborg J, Elortza F, Jensen ON, Roepstorff P (2004) A new strategy for identification of N-glycosylated proteins and unambiguous assignment of their glycosylation sites using HILIC enrichment and partial deglycosylation. J Proteome Res 3:556–566
Kieliszewski MJ, O’Neill M, Leykam J, Orlando R (1995) Tandem mass spectrometry and structural elucidation of glycopeptides from a hydroxyproline-rich plant cell wall glycoprotein indicate that contiguous hydroxyproline residues are the major sites of hydroxyproline O-arabinosylation. J Biol Chem 270:2541–2549
Bunkenborg J, Pilch BJ, Podtelejnikov AV, Wiśniewski JR (2004) Screening for N-glycosylated proteins by liquid chromatography mass spectrometry. Proteomics 4:454–465
Hirabayashi J (2004) Lectin-based structural glycomics: glycoproteomics and glycan profiling. Glycoconj J 21:35–40
Rawn JD, Lienhard GE (1974) Binding of boronic acids to chymotrypsin. Biochemistry 13:3124–3130
Sparbier K, Koch S, Kessler I, Wenzel T, Kostrzewa M (2005) Selective isolation of glycoproteins and glycopeptides for MALDI-TOF MS detection supported by magnetic particles. J Biomol Tech 16:407–413
Alley WR, Mechref Y, Novotny MV (2009) Use of activated graphitized carbon chips for liquid chromatography/mass spectrometric and tandem mass spectrometric analysis of tryptic glycopeptides. Rapid Commun Mass Spectrom 23:495–505
Larsen MR, Jensen SS, Jakobsen LA, Heegaard NHH (2007) Exploring the sialiome using titanium dioxide chromatography and mass spectrometry. Mol Cell Proteomics 6:1778–1787
Calvano CD, Zambonin CG, Jensen ON (2008) Assessment of lectin and HILIC based enrichment protocols for characterization of serum glycoproteins by mass spectrometry. J Proteomics 71:304–317
Kondo A, Miyamoto T, Yonekawa O, Giessing AM, Østerlund EC, Jensen ON (2009) Glycopeptide profiling of beta-2-glycoprotein I by mass spectrometry reveals attenuated sialylation in patients with antiphospholipid syndrome. J Proteomics 73:123–133
Kondo A, Thaysen-Andersen M, Hjernø K, Jensen ON (2010) Characterization of sialylated and fucosylated glycopeptides of β2-glycoprotein I by a combination of HILIC LC and MALDI MS/MS. J Sep Sci 33:891–902
Thaysen-Andersen M, Mysling S, Højrup P (2009) Site-specific glycoprofiling of N-linked glycopeptides using MALDI-TOF MS: strong correlation between signal strength and glycoform quantities. Anal Chem 81:3933–3943
Mysling S, Palmisano G, Højrup P, Thaysen-Andersen M (2010) Utilizing ion-pairing hydrophilic interaction chromatography solid phase extraction for efficient glycopeptide enrichment in glycoproteomics. Anal Chem 82:5598–5609
Stasyk T, Huber LA (2004) Zooming in: fractionation strategies in proteomics. Proteomics 4:3704–3716
Christiansen MN, Kolarich D, Nevalainen H, Packer NH, Jensen PH (2010) Challenges of determining O-glycopeptide heterogeneity: a fungal glucanase model system. Anal Chem 82:3500–3509
Hägglund P, Matthiesen R, Elortza F, Højrup P, Roepstorff P, Jensen ON, Bunkenborg J (2007) An enzymatic deglycosylation scheme enabling identification of core fucosylated N-glycans and O-glycosylation site mapping of human plasma proteins. J Proteome Res 6:3021–3031
Højrup P (2009) Peptide mapping for protein characterization. In: Walker JM (ed) The protein protocols handbook, 3rd edn. Humana Press, Totawa, NJ, pp 965–983
Papac DI, Wong A, Jones AJS (1996) Analysis of acidic oligosaccharides and glycopeptides by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Anal Chem 68:3215–3223
Tsarbopoulos A, Bahr U, Pramanik BN, Karas M (1997) Glycoprotein analysis by delayed extraction and post-source decay MALDI-TOF-MS. Int J Mass Spectrom Ion Process 169–170:251–261
Harvey DJ (1999) Matrix-assisted laser desorption/ionization mass spectrometry of carbohydrates. Mass Spectrom Rev 18:349–450
Yoshida T (2004) Peptide separation by hydrophilic-interaction chromatography: a review. J Biochem Biophys Methods 60:265–280
Wohlgemuth J, Karas M, Eichhorn T, Hendriks R, Andrecht S (2009) Quantitative site-specific analysis of protein glycosylation by LC-MS using different glycopeptide-enrichment strategies. Anal Biochem 395:178–188
Lämmerhofer M, Richter M, Wu J, Nogueira R, Bicker W, Lindner W (2008) Mixed-mode ion-exchangers and their comparative chromatographic characterization in reversed-phase and hydrophilic interaction chromatography elution modes. J Sep Sci 31:2572–2588
Acknowledgments
PHJ was supported by a postdoctoral fellowship from “Annie og Otto Johs. Detlefs’ Almennyttige Fond.”
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Jensen, P.H., Mysling, S., Højrup, P., Jensen, O.N. (2013). Glycopeptide Enrichment for MALDI-TOF Mass Spectrometry Analysis by Hydrophilic Interaction Liquid Chromatography Solid Phase Extraction (HILIC SPE). In: Kohler, J., Patrie, S. (eds) Mass Spectrometry of Glycoproteins. Methods in Molecular Biology, vol 951. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-146-2_10
Download citation
DOI: https://doi.org/10.1007/978-1-62703-146-2_10
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-145-5
Online ISBN: 978-1-62703-146-2
eBook Packages: Springer Protocols