Abstract
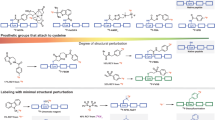
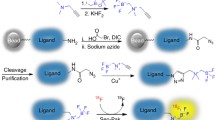
There has been significant and rapid growth in the development of amino acid-based molecular imaging agents (e.g., peptides, proteins, and antibody constructs) largely due to facile library preparation and high throughput screening. Positron-emitting fluorine-18 (half-life = 109.7 min) has a unique set of properties that match well with the pharmacokinetics of smaller sized constructs. Several indirect fluorine-18 labeling approaches have been developed yet only a few have advanced to human trials. Enzymatically catalyzed radiofluorination utilizing lipoic acid ligase shows promise as a mild site-specific method for coupling fluorine-18-labeled carboxylate substrates with biomolecules. Methods for preparation of two [18F]fluorocarboxylates and their ligation to a specific peptide sequence (LAP peptide) are presented herein.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Fani M, Maecke HR, Okarvi SM (2012) Radiolabeled peptides: valuable tools for the detection and treatment of cancer. Theranostics 2:481–501
Jackson IM, Scott PJH, Thompson S (2017) Clinical applications of radiolabeled peptides for PET. Semin Nucl Med 47:493–523
Mullard A (2016) FDA approvals for the first 6 months of 2016. Nat Rev Drug Disc 15:523
Vaidyanathan G, Zalutsky MR (2006) Synthesis of N-succinimidyl 4-[18F]fluorobenzoate, an agent for labeling proteins and peptides with 18F. Nat Protoc 1:1655–1661
Vaidyanathan G, Zalutsky MR (1992) Labeling proteins with fluorine-18 using N-succinimidyl 4-[18F]fluorobenzoate. Nucl Med Biol 19:275–281
Berndt M, Pietzsch J, Wuest F (2007) Labeling of low-density lipoproteins using the 18F-labeled thiol-reactive reagent N-[6-(4[18F]fluorobenzylidene)aminooxy-hexyl]maleimide. Nuc Med Biol 34:5–15
Gill HS, Tinianow JN, Ogasawara A et al (2009) A modular platform for the rapid site-specific radiolabeling of proteins with 18F exemplified by quantitative positron emission tomography of human epidermal growth factor receptor 2. J Med Chem 52:5816–5825
Jeon J, Shen B, Xiong L et al (2012) Efficient method for site-specific 18F-labeling of biomolecules using the rapid condensation reaction between 2-cyanobenzothiazole and cysteine. Bioconjug Chem 23:1902–1908
Hausner SH, Marik J, Gagnon MKJ, Sutcliffe JL (2008) In vivo positron emission tomography (PET) imaging with an αvβ6 specific peptide radiolabeled using 18F-“click” chemistry: evaluation and comparison with the corresponding 4-[18F]Fluorobenzoyl- and 2-[18F]Fluoropropionyl- peptides. J Med Chem 51:5901–5904
Liu S, Hassink M, Selvaraj R et al (2013) Efficient 18F labeling of cysteine-containing peptides and proteins using tetrazine-trans-cyclooctene ligation. Mol Imag 12:121–128
Marik J, Sutcliffe JL (2006) Click for PET: rapid preparation of [F-18]fluoropeptides using Cu-I catalyzed 1,3-dipolar cycloaddition. Tet Lett 47:6681–6684
Wängler C, Niedermoser S, Chin J et al (2012) One-step 18F-labeling of peptides for positron emission tomography imaging using the SiFA methodology. Nat Protoc 7:1946–1955
Wängler C, Waser B, Alke A et al (2010) One-step 8F-labeling of carbohydrate-conjugated octreotate-derivatives containing a silicon-fluoride-acceptor (SiFA): in vitro and in vivo evaluation as tumor imaging agents for positron emission tomography (PET). Bioconjug Chem 21:2289–2296
Fernández-Suárez M, Baruah H, Martínez-Hernández L et al (2007) Redirecting lipoic acid ligase for cell surface protein labeling with small-molecule probes. Nat Biotech 25:1483–1487
Uttamapinant C, White KA, Baruah H et al (2010) A fluorophore ligase for site-specific protein labeling inside living cells. PNAS 107:10914–10919
Puthenveetil S, Liu DS, White KA et al (2009) Yeast display evolution of a kinetically efficient 13-amino acid substrate for lipoic acid ligase. J Am Chem Soc 131:16430–16438
Cohen JD, Zou P, Ting AY (2012) Site-specific protein modification using lipoic acid ligase and bis-aryl hydrazone formation. Chembiochem 13:888–894
Cohen JD, Thompson S, Ting AY (2011) Structure-guided engineering of a Pacific blue fluorophore ligase for specific protein imaging in living cells. Biochemist 50:8221–8225
Drake CR, Sevillano N, Truillet C et al (2016) Site-specific radiofluorination of biomolecules with 8-[18F]-Fluorooctanoic acid catalyzed by Lipoic acid ligase. ACS Chem Biol 11:1587–1594
Drake CR, Sevillano N, Blecha J, et al (Submitted) Second-generation 18F-labeled prosthetic for automated enzymatically catalyzed radiolabeling of proteins on the ELIXYS platform.
Nagatsugi F, Sasaki S, Maeda M (1994) 8-[F-18] Fluorooctanoic acid and its Beta-substituted derivatives as potential agents for cerebral fatty-acid studies—synthesis and biodistribution. Nucl Med Biol 21:809–817
Acknowledgments
This work was supported by a seed grant from the UCSF Department of Radiology and Biomedical Imaging, the UCSF Resource Allocation Program, the US Department of Energy (DOE DE-SC002061), and NIH National Institute of Biomedical Imaging and Bioengineering SBIR (R43EB023782). Additional support for C.R.D. was received from the National Cancer Institute (HHSN261201400041C) and National Institute of Mental Health (2R44MH097271). M.J.E. was supported by the National Institute of Mental Health (R01MH115043) and the National Institute of Biomedical Imaging and Bioengineering (R01EB025207).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Drake, C.R., Evans, M.J., VanBrocklin, H.F. (2019). Enzymatically Catalyzed Radiofluorination of Biomolecules. In: Massa, S., Devoogdt, N. (eds) Bioconjugation. Methods in Molecular Biology, vol 2033. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9654-4_13
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9654-4_13
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9653-7
Online ISBN: 978-1-4939-9654-4
eBook Packages: Springer Protocols