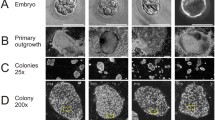
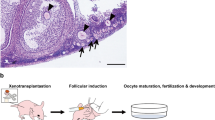
Abstract
The biology of fertility, early development, and pregnancy is variable across mammalian species. In addition, while the physiology and pathophysiology of human diseases can be investigated in other animal models (principally, rodents), differences between human and lower mammals often present limitations in the applicability of physiological processes from rodent models to human biology. Since 1984, when the first live birth from rhesus monkey in vitro fertilization and embryo transfer was reported (Bavister et al., Proc Natl Acad Sci 81:2218–2222, 1984), there has been progress in the implementation of assisted reproductive technologies with several nonhuman primate (NHP) species that play important roles in biomedical research. In recent years, the significance of this progress has been amplified by the development of genomic editing approaches for facile genetic manipulation of the embryo, including methods now applied to NHPs (Liu et al., Cell Stem Cell 14:323–328, 2014; Niu et al., Cell 156:836–843, 2014). In this review, we summarize current protocols and practices for the common marmoset. It is our intention to provide current state-of-the-art protocols for gamete procurement and in vitro fertilization techniques, so that laboratories wishing to implement experimental embryology in marmoset models will have a basic set of tools with which to initiate such studies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Marshall VS, Browne MA, Knowles L, Golos TG, Thomson JA (2003) Ovarian stimulation of marmoset monkeys (Callithrix jacchus) using recombinant human follicle stimulating hormone. J Med Primatol 32:57–66
Grupen CG, Gilchrist RB, Nayudu PL, Barry MF, Schulz SJ, Ritter LJ, Armstrong DT (2007) Effects of ovarian stimulation, with and without human chorionic gonadotrophin, on oocyte meiotic and developmental competence in the marmoset monkey (Callithrix jacchus). Theriogenology 68:861–872
Takahashi T, Hanazawa K, Inoue T, Sato K, Sedohara A, Okahara J, Suemizu H, Yagihashi C, Yamamoto M, Eto T, Konno Y, Okano H et al (2014) Birth of healthy offspring following ICSI in in vitro-matured common marmoset (Callithrix jacchus) oocytes. PLoS One 9:e95560
Gilchrist RB, Nayudu PL, Hodges JK (1997) Maturation, fertilization, and development of marmoset monkey oocytes in vitro. Biol Reprod 56:238–246
Kuederling I, Schneiders A, Sonksen J, Nayudu PL, Hodges JK (2000) Non-invasive collection of ejaculates from the common marmoset (Callithrix jacchus) using penile vibrostimulation. Am J Primatol 52:149–154
Schneiders A, Sonksen J, Hodges JK (2004) Penile vibratory stimulation in the marmoset monkey: a practical alternative to electro-ejaculation, yielding ejaculates of enhanced quality. J Med Primatol 33:98–104
Tomioka I, Takahashi T, Shimada A, Yoshioka K, Sasaki E (2012) Birth of common marmoset (Callithrix jacchus) offspring derived from in vitro-matured oocytes in chemically defined medium. Theriogenology 78:1487–1493
Summers PM, Shephard AM, Taylor CT, Hearn JP (1987) The effects of cryopreservation and transfer on embryonic-development in the common marmoset monkey, Callithrix jacchus. J Reprod Fertil 79:241–250
Marshall VS, Kalishman J, Thomson JA (1997) Nonsurgical embryo transfer in the common marmoset monkey. J Med Primatol 26:241–247
Ishibashi H, Motohashi HH, Kumon M, Yamamoto K, Okada H, Okada T, Seki K (2013) Efficient embryo transfer in the common marmoset monkey (Callithrix jacchus) with a reduced transfer volume: a non-surgical approach with cryopreserved late-stage embryos. Biol Reprod 88:115
Ishibashi H, Motohashi HH, Kumon M, Yamamoto K, Okada H, Okada T, Seki K (2013) Ultrasound-guided non-surgical embryo collection in the common marmoset. Reprod Biol 13:139–144
Kropp J, Di Marzo A, Golos T (2017) Assisted reproductive technologies in the common marmoset: an integral species for developing nonhuman primate models of human diseases. Biol Reprod 96:277–287
Sato K, Oiwa R, Kumita W, Henry R, Sakuma T, Ito R, Nozu R, Inoue T, Katano I, Okahara N, Okahara J, Shimizu Y et al (2016) Generation of a nonhuman primate model of severe combined immunodeficiency using highly efficient genome editing. Cell Stem Cell 19:127–138
Sasaki E, Suemizu H, Shimada A, Hanazawa K, Oiwa R, Kamioka M, Tomioka I, Sotomaru Y, Hirakawa R, Eto T, Shiozawa S, Maeda T et al (2009) Generation of transgenic non-human primates with germline transmission. Nature 459:523–U550
Oerke AK, Einspanier A, Hodges JK (1995) Detection of pregnancy and monitoring patterns of uterine and fetal growth in the marmoset monkey (Callithrix jacchus) by real-time ultrasonography. Am J Primatol 36:1–13
Harlow CR, Gems S, Hodges JK, Hearn JP (1983) The relationship between plasma progesterone and the timing of ovulation and early embryonic-development in the marmoset monkey (Callithrix jacchus). J Zool 201:273–282
Hodges JK, Cottingham PG, Summers PM, Liang YN (1987) Controlled ovulation in the marmoset monkey (Callithrix jacchus) with human chorionic-gonadotropin following prostaglandin-induced luteal regression. Fertil Steril 48:299–305
Summers PM, Wennink CJ, Hodges JK (1985) Cloprostenol-induced luteolysis in the marmoset monkey (Callithrix jacchus). J Reprod Fertil 73:133–138
Wilton LJ, Marshall VS, Piercy EC, Moore HD (1993) In vitro fertilization and embryo development in the marmoset monkey (Callithrix jacchus). J Reprod Fertil 97:481–486
Hernandez-Lopez L, Umland N, Mondragon-Ceballos R, Nayudu PL (2005) Comparison of the effects of Percoll and PureSperm (R) on the common marmoset (Callithrix jacchus) semen. J Med Primatol 34:86–90
Acknowledgments
T.G.G. and J.K.S. would like to thank the veterinary and animal care staff at the Wisconsin National Primate Research Center, particularly Michele Schotzko and Dr. Nancy Schultz-Darken of the Scientific Protocol Implementation Unit, Megan Sosa of the Animal Services Unit, and Drs. Casey Fitz and Kevin Brunner of the Veterinary Services Unit, for assistance in synchronizing donors and collection of gametes to perform marmoset in vitro experiments to generate embryo images. Studies at the University of Wisconsin-Madison were supported by NIH grants R24OD019803 to T.G.G. and P51OD011106 to the WNPRC.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Schmidt, J.K., Golos, T.G. (2019). In Vitro Culture of Embryos from the Common Marmoset (Callithrix jacchus). In: Herrick, J. (eds) Comparative Embryo Culture. Methods in Molecular Biology, vol 2006. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9566-0_21
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9566-0_21
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9565-3
Online ISBN: 978-1-4939-9566-0
eBook Packages: Springer Protocols