Abstract
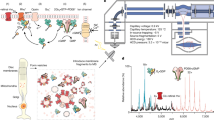
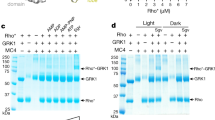
G protein-coupled receptors (GPCRs) comprise the largest family of transmembrane receptors and are targets for over 30% of all drugs on the market. Structural information of GPCRs and more importantly that of the complex between GPCRs and their signaling partner heterotrimeric G proteins is of great importance. Here we present a method for the large-scale purification of the rhodopsin–transducin complex, the GPCR–G protein signaling complex in visual phototransduction, directly from their native retinal membrane using native proteins purified from bovine retinae. Formation of the complex on native membrane is orchestrated in part by the proper engagement of lipid-modified rhodopsin and transducin (i.e., palmitoylation of the rhodopsin C-terminus, myristoylation and farnesylation of the αT and γ1, respectively). The resulting complex is of high purity and stability and has proved suitable for further biophysical and structural studies. The methods described here should be applicable to other recombinantly expressed receptors from insect cells or mamalian cells by forming stable, functional complexes directly on purified cell membranes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bjarnadóttir TK et al (2006) Comprehensive repertoire and phylogenetic analysis of the G protein-coupled receptors in human and mouse. Genomics 88:263–273
Santos R et al (2017) A comprehensive map of molecular drug targets. Nat Rev Drug Discov 16:19–34
Fredriksson R, Lagerstrom MC, Lundin LG, Schiöth HB (2003) The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol Pharmacol 63:1256–1272
Simon MI, Strathmann MP, Gautam N (1991) Diversity of G proteins in signal transduction. Science 252:802–808
Downes GB, Gautam N (1999) The G protein subunit gene families. Genomics 62:544–552
Li F, De Godoy M, Rattan S (2004) Role of adenylate and guanylate cyclases in β1-, β2-, and β3-adrenoceptor-mediated relaxation of internal anal sphincter smooth muscle. J Pharmacol Exp Ther 308:1111–1120
Rosenbaum DM, Cherezov V, Hanson MA, Rasmussen SG, Thian FS, Kobilka TS, Choi HJ, Yao XJ, Weis WI, Stevens RC, Kobilka BK (2007) GPCR engineering yields high-resolution structural insights into β2-adrenergic receptor function. Science 318:1266–1273
Caffrey M, Cherezov V (2009) Crystallizing membrane proteins using lipidic mesophases. Nat Protoc 4:706–731
Xu S, Fischetti RF (2007) Design and Performance of a Compact Collimator on GM/CA-CAT At the Advanced Photon Source. Proc SPIE 6665:66650X1–66650X8
Bai XC, McMullan G, Scheres SH (2015) How cryo-EM is revolutionizing structural biology. Trends Biochem Sci 40:49–57
Rasmussen SG, DeVree BT, Zou Y, Kruse AC, Chung KY, Kobilka TS, Thian FS, Chae PS, Pardon E, Calinski D, Mathiesen JM, Shah ST, Lyons JA, Caffrey M, Gellman SH, Steyaert J, Skiniotis G, Weis WI, Sunahara RK, Kobilka BK (2011) Crystal structure of the β2 adrenergic receptor-Gs protein complex. Nature 477:549–555
Liang Y-L, Khoshouei M, Radjainia M, Zhang Y et al (2017) Phase-plate cryo-EM structure of a class B GPCR–G-protein complex. Nature 546(7656):118–123
Zhang Y et al (2017) Cryo-EM structure of the activated GLP-1 receptor in complex with a G protein. Nature 546:248–253
Stryer L (1991) Visual excitation and recovery. J Biol Chem 266:10711–10714
Palczewski K, Kumasaka T, Hori T, Behnke CA, Motoshima H, Fox BA, Le Trong I, Teller DC, Okada T, Stenkamp RE, Yamamoto M, Miyano M (2000) Crystal structure of rhodopsin: a G protein-coupled receptor. Science 289:739–745
Li J, Edwards PC, Burghammer M, Villa C, Schertler GFX (2004) Structure of bovine rhodopsin in a trigonal crystal form. J Mol Biol 343:1409–1436
Park JH, Scheerer P, Hofmann KP, Choe HW, Ernst OP (2008) Crystal structure of the ligand-free G-protein-coupled receptor opsin. Nature 454:183–187
Choe HW, Kim YJ, Park JH, Morizumi T, Pai EF, Krauss N, Hofmann KP, Scheerer P, Ernst OP (2011) Crystal structure of metarhodopsin II. Nature 471:651–655
Scheerer P, Park JH, Hildebrand PW, Kim YJ, Krauss N, Choe HW, Hofmann KP, Ernst OP (2008) Crystal structure of opsin in its G-protein-interacting conformation. Nature 455:497–502
Gao Y et al (2017) Isolation and structure-function characterization of a signaling-active rhodopsin-G protein complex. J Biol Chem 292:14280–14289
Skiba NP, Bae H, Hamm HE (1996) Mapping of effector binding sites of transducin alpha-subunit using G alpha t/G alpha i1 chimeras. J Biol Chem 271:413–424
Ballesteros JA, Shi L, Javitch JA (2001) Structural mimicry in G protein-coupled receptors: implications of the high-resolution structure of rhodopsin for structure–function analysis of rhodopsin-like receptors. Mol Pharmacol 60:1–19
Zhang FL, Casey PJ (1996) Protein prenylation: molecular mechanisms and functional consequences. Annu Rev Biochem 65:241–269
Min KC, Gravina SA, Sakmar TP (2000) Reconstitution of the vertebrate visual cascade using recombinant transducin purified from Sf9 cells. Protein Expr Purif 20:514–526
Ramachandran S, Cerione RA (2011) A dominant-negative Gα mutant that traps a stable rhodopsin-Gα-GTP-βγ complex. J Biol Chem 286:12702–12711
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Gao, Y., Erickson, J.W., Cerione, R.A., Ramachandran, S. (2019). Purification of the Rhodopsin–Transducin Complex for Structural Studies. In: Linder, M. (eds) Protein Lipidation. Methods in Molecular Biology, vol 2009. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9532-5_23
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9532-5_23
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9531-8
Online ISBN: 978-1-4939-9532-5
eBook Packages: Springer Protocols