Abstract
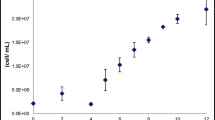
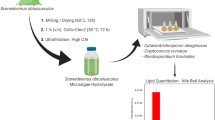
Worldwide, wastewater produced from sewage and industry poses a serious risk to the surrounding environment. As a way to address this problem, an integrated approach for cultivation of oleaginous microorganisms on wastewater leading to effective removal of hazardous components and sustainable production of biodiesel is proposed. Oleaginous yeasts have the unique ability to utilize wastewater as feedstock and accumulate large amounts of triacylglycerols within their cellular compartments at stationary phase (144 h). The lipids stored in an oleaginous microbe can be visualized by fluorescence microscopy and converted into biodiesel through transesterification after extraction. Here, we describe the batch cultivation of oleaginous yeast on sewage and industrial wastewater at 25 °C. High lipid accumulation with efficient removal of toxic chemicals can be achieved by utilizing this integrated method.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Patel A, Arora N, Pruthi V, Pruthi PA (2016) Biological treatment of pulp and paper industry effluent by oleaginous yeast integrated with production of biodiesel as sustainable transportation fuel. J Clean Prod 142:2858–2864
Arora N, Patel A, Sartaj K, Pruthi PA, Pruthi V (2016) Bioremediation of domestic and industrial wastewaters integrated with enhanced biodiesel production using novel oleaginous microalgae. Environ Sci Pollut Res 23:20997–21007
Lamers D, Biezen NV, Martens D, Peters L, Zilver EVD, Dreumel NJ-N, Wijffels RH, Lokman C (2016) Selection of oleaginous yeasts for fatty acid production. BMC Biotechnol 16:45
Beopoulos A, Verbeke J, Bordes F, Guicherd M, Bressy M, Marty A, Nicaud JM (2014) Metabolic engineering for ricinoleic acid production in the oleaginous yeast Yarrowia lipolytica. Appl Microbiol Biotechnol 98:251–262
Deeba F, Pruthi V, Negi YS (2017) Fostering TAG accumulation in novel oleaginous yeast Cryptococcus psychrotolerans IITRFD utilizing groundnut shell for improved biodiesel production. Bioresour Technol. https://doi.org/10.1016/j.biortech.2017.04.001
Deeba F, Pruthi V, Negi YS (2016) Converting paper mill sludge into neutral lipids by oleaginous yeast Cryptococcus vishniaccii for biodiesel production. Bioresour Technol 213:96–102
Patel A, Pravez M, Deeba F, Pruthi V, Singh RP, Pruthi PPA (2014) Boosting accumulation of neutral lipids in Rhodosporidium kratochvilovae HIMPA1 grown on hemp (Cannabis sativa Linn) seed aqueous extract as feedstock for biodiesel production. Bioresour Technol 165:214–222
Vicente G, Bautista LF, Rodriguez R, Gutierrez FJ, Sadaba I, Ruiz-Vazquez RM, Torres-Martinez S, Garre V (2009) Biodiesel production from biomass of an oleaginous fungus. Biochem Eng J 48:22–27
Patel A, Sindhu DK, Arora N, Singh RP, Pruthi V, Pruthi PA (2015) Biodiesel production from non-edible lignocellulosic biomass of Cassia fistula L. fruit pulp using oleaginous yeast Rhodosporidium kratochvilovae HIMPA1. Bioresour Technol 197:91–98
Patel A, Pruthi V, Singh RP, Pruthi PA (2015) Synergistic effect of fermentable and non-fermentable carbon sources enhances TAG accumulation in oleaginous yeast Rhodosporidium kratochvilovae HIMPA1. Bioresour Technol 188:136–144
Li XF, Xu H, Wu QY (2007) Large-scale biodiesel production from microalga Chlorella protothecoides through heterotrophic cultivation in bioreactors. Biotechnol Bioeng 98:764–771
Arora N, Patel A, Pruthi PA, Pruthi V (2016) Boosting TAG accumulation with improved biodiesel production from novel oleaginous microalgae Scenedesmus sp. IITRIND2 utilizing waste sugarcane bagasse aqueous extract (SBAE). Appl Biochem Biotechnol 180:109–121
Arous F, Frikha F, Triantaphyllidou I-E, Aggelis G, Nasri M, Mechichi T (2016) Potential utilization of agro-industrial wastewaters for lipid production by the oleaginous yeast Debaryomyces etchellsii. J Clean Prod 133:899–909
Karakaya A, Laleli Y, Takaç S (2012) Development of process conditions for degradation of raw olive mill wastewater by Rhodotorula glutinis. Int Biodeter Biodegr 75:75–82
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin-phenol reagent. J Biol Chem 193:265–275
Tazeen R, Mular SM, Khan ZH, Khan ND (2017) Comparative analysis of sugar and protein in three different fresh and decaying fruits pomegranate, orange and sweet lime (musambi). IJAR 3:562–564
Bligh EG, Dyer WJ (1959) A rapid method for total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Fei W, Wang H, Fu X, Bielby C, Yang H (2009) Conditions of endoplasmic reticulum stress stimulate lipid droplet formation in Saccharomyces cerevisiae. Biochem J 424:61–67
Morrison WR, Smith LM (1964) Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride–methanol. J Lipid Res 5:600–608
Acknowledgments
This work was supported by the Ministry of Human Resource Development (MHRD), Government of India.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Deeba, F., Pruthi, V., Negi, Y.S. (2019). Production of Oleaginous Organisms or Lipids Using Sewage Water and Industrial Wastewater. In: Balan, V. (eds) Microbial Lipid Production. Methods in Molecular Biology, vol 1995. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9484-7_24
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9484-7_24
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9483-0
Online ISBN: 978-1-4939-9484-7
eBook Packages: Springer Protocols