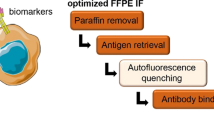
Abstract
Histomorphology has significantly changed over the last decades due to technological achievements in immunohistochemistry (IHC) for the visualization of specific proteins and in molecular pathology, particularly in the field of in situ hybridization of small oligonucleotides and amplification of DNA and RNA amplicons. With an increased availability of suitable methods, the demands regarding the observer of histomorphological slides were the supply of complex quantitative data as well as more information about protein expression and cell-cell interactions in tissue sections. Advances in fluorescence-based multiplexed IHC techniques, such as multispectral imaging (MSI), allow the quantification of multiple proteins at the same tissue section. In histopathology, it is a well-known technique for over a decade yet harboring serious problems concerning quantitative preciseness and tissue autofluorescence of multicolor staining when using formalin-fixed, paraffin-embedded (FFPE) tissue specimen. In recent years, milestones in tissue preparation, fluorescent dyes, hardware imaging, and software analysis were achieved including automated tissue segmentation (e.g., tumor vs. stroma) as well as in cellular and subcellular multiparameter analysis.
This chapter covers the role that MSI plays in anatomic pathology for the analysis of FFPE tissue sections, discusses the technical aspects of MSI, and provides a review of its application in the characterization of immune cell infiltrates and beyond regarding its prognostic and predictive value and its use for guidance of clinical decisions for immunotherapeutic strategies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Engels B, Engelhard VH, Sidney J et al (2013) Relapse or eradication of cancer is predicted by peptide-major histocompatibility complex affinity. Cancer Cell 23(4):516–526
Gajewski TF, Schreiber H, Fu Y-X (2013) Innate and adaptive immune cells in the tumor microenvironment. Nat Immunol 14(10):1014–1022
Becht E, de Reyniès A, Giraldo NA et al (2016) Immune and stromal classification of colorectal cancer is associated with molecular subtypes and relevant for precision immunotherapy. Clin Cancer Res 22(12):4057–4066
Pages F, Berger A, Camus M et al (2005) Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med 353(14):2654–2666
Galon J, Costes A, Sanchez-Cabo F et al (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(11):1960–1964
Mlecnik B, Tosolini M, Kirilovsky A et al (2011) Histopathologic-based prognostic factors of colorectal cancers are associated with the state of the local immune reaction. J Clin Oncol 29(7):610–618
Galon J, Pagès F, Marincola FM et al (2012) Cancer classification using the immunoscore: a worldwide task force. J Transl Med 10:205. https://doi.org/10.1186/s12967-017-1310-9
Abel EJ, Bauman TM, Weiker M et al (2014) Analysis and validation of tissue biomarkers for renal cell carcinoma using automated high-throughput evaluation of protein expression. Hum Pathol 45(5):1092–1099
van Zwieten A (2013) Tissue microarray technology and findings for diagnostic immunohistochemistry. Pathology 45(1):71–79
Gorris MAJ, Halilovic A, Rabold K et al (2018) Eight-color multiplex immunohistochemistry for simultaneous detection of multiple immune checkpoint molecules within the tumor microenvironment. J Immunol 200(17):347–354
Lichtman JW, Conchello JA (2005) Fluorescence microscopy. Nat Methods 2(12):910–919
Toth ZE, Mezey E (2007) Simultaneous visualization of multiple antigens with tyramide signal amplification using antibodies from the same species. J Histochem Cytochem 55(6):545–554
Stack EC, Wang C, Roman KA et al (2014) Multiplexed immunohistochemistry, imaging, and quantitation: a review, with an assessment of Tyramide signal amplification, multispectral imaging and multiplex analysis. Methods 70(1):46–58
Mezheyeuski A, Bergsland CH, Backman M et al (2018) Multispectral imaging for quantitative and compartment-specific immune infiltrates reveals distinct immune profiles that classify lung cancer patients. J Pathol 244(4):421–431. https://doi.org/10.1002/path.5026
Ying L, Yan F, Meng Q et al (2017) Understanding immune phenotypes in human gastric disease tissues by multiplexed immunohistochemistry. J Transl Med 15:206–210. https://doi.org/10.1186/s12967-017-1311-8
Feng Z, Shawn M, Jensen SM et al (2016) Multispectral Imaging of T and B Cells in Murine Spleen and Tumor. J Immunol 198(4):3943–3950
Ng SSM, Nagy BA, Jensen SM et al (2017) Heterodimeric IL15 treatment enhances tumor infiltration, persistence, and effector functions of adoptively transferred tumor-specific T cells in the absence of lymphodepletion. Clin Cancer Res 23(17):2817–2830
Feng Z, Puri S, Moudgil T et al (2015) Multispectral imaging of formalin-fixed tissue predicts ability to generate tumor-infiltrating lymphocytes from melanoma. J Immunother Cancer 3:47. https://doi.org/10.1186/s40425-015-0091-z
Feng Z, Bethmann D, Kappler M et al (2017) Multiparametric immune profiling in HPV- oral squamous cell cancer. JCI Insight 2(14):1–17
Yang L, Liu Z, Tan J et al (2018) Multispectral imaging reveals hyper active TGF-β signaling in colorectal cancer. Cancer Biol Ther 19(2):105–112
Acknowledgments
Claudia Wickenhauser, Daniel Bethmann, Zipei Feng, and Shawn M. Jensen contributed equally to this work.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Wickenhauser, C. et al. (2019). Multispectral Fluorescence Imaging Allows for Distinctive Topographic Assessment and Subclassification of Tumor-Infiltrating and Surrounding Immune Cells. In: Pico de Coaña, Y. (eds) Immune Checkpoint Blockade. Methods in Molecular Biology, vol 1913. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8979-9_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8979-9_2
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8978-2
Online ISBN: 978-1-4939-8979-9
eBook Packages: Springer Protocols