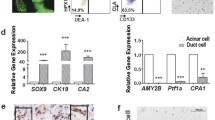
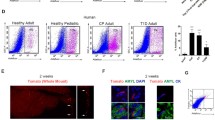
Abstract
Acinar-to-ductal metaplasia may play important roles in the development of various pancreatic diseases. Here, we describe a method to induce ADM in primary human cells in 3D culture. We developed a flow cytometry lineage tracing strategy to identify and sort viable acinar, ductal, and acinar-derived ductal-like cells for further molecular and functional analysis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Guney MA, Gannon M (2009) Pancreas cell fate. Birth Defects Res Part C Embryo Today Rev 87:232–248
Shih HP, Wang A, Sander M (2013) Pancreas organogenesis: from lineage determination to morphogenesis. Annu Rev Cell Dev Biol 29:81–105
Longnecker D (2014) Anatomy and histology of the pancreas. Pancreapedia. https://doi.org/10.3998/panc.2014.3
Bailey JM, DelGiorno KE, Crawford HC (2014) The secret origins and surprising fates of pancreas tumors. Carcinogenesis 35:1436–1440
Logsdon CD, Arumugam T, Ramachandran V (2015) Animal Models of Gastrointestinal and Liver Diseases. The difficulty of animal modeling of pancreatic cancer for preclinical evaluation of therapeutics. Am J Physiol Gastrointest Liver Physiol 309:G283–G291
De La OJ-P, Emerson LL, Goodman JL et al (2008) Notch and Kras reprogram pancreatic acinar cells to ductal intraepithelial neoplasia. Proc Natl Acad Sci U S A 105:18907–18912
Kopp JL, Grompe M, Sander M (2016) Stem cells versus plasticity in liver and pancreas regeneration. Nat Cell Biol 18:238–245
Shi G, DiRenzo D, Qu C et al (2013) Maintenance of acinar cell organization is critical to preventing Kras-induced acinar-ductal metaplasia. Oncogene 32:1950–1958
Kopp JL, von Figura G, Mayes E et al (2012) Identification of Sox9-dependent acinar-to-ductal reprogramming as the principal mechanism for initiation of pancreatic ductal adenocarcinoma. Cancer Cell 22:737–750
Means AL, Meszoely IM, Suzuki K et al (2005) Pancreatic epithelial plasticity mediated by acinar cell transdifferentiation and generation of nestin-positive intermediates. Development 132:3767–3776
Liou G-Y, Döppler H, Necela B et al (2013) Macrophage-secreted cytokines drive pancreatic acinar-to-ductal metaplasia through NF-κB and MMPs. J Cell Biol 202:563–577
Liou G-Y, Döppler H, Braun UB et al (2015) Protein kinase D1 drives pancreatic acinar cell reprogramming and progression to intraepithelial neoplasia. Nat Commun 6:6200
Chen N-M, Singh G, Koenig A et al (2015) NFATc1 Links EGFR signaling to induction of Sox9 transcription and acinar-ductal transdifferentiation in the pancreas. Gastroenterology 148:1024–1034.e9
Houbracken I, de Waele E, Lardon J et al (2011) Lineage tracing evidence for transdifferentiation of acinar to duct cells and plasticity of human pancreas. Gastroenterology 141:731–741
Lee J, Sugiyama T, Liu Y et al (2013) Expansion and conversion of human pancreatic ductal cells into insulin-secreting endocrine cells. Elife 2:e00940. https://doi.org/10.7554/eLife.00940
Liu J, Akanuma N, Liu C et al (2016) TGF-β1 promotes acinar to ductal metaplasia of human pancreatic acinar cells. Sci Rep 6:30904
Akanuma N, Liu J, Liou GY et al (2017) Paracrine secretion of transforming growth factor β by ductal cells promotes acinar-to-ductal metaplasia in cultured human exocrine pancreas tissues. Pancreas 46:1202–1207. https://doi.org/10.1097/MPA.0000000000000913
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Liu, J., Wang, P. (2019). Lineage Tracing of Primary Human Pancreatic Acinar and Ductal Cells for Studying Acinar-to-Ductal Metaplasia. In: Su, G. (eds) Pancreatic Cancer. Methods in Molecular Biology, vol 1882. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8879-2_5
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8879-2_5
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8878-5
Online ISBN: 978-1-4939-8879-2
eBook Packages: Springer Protocols