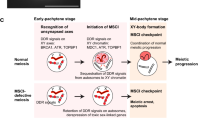
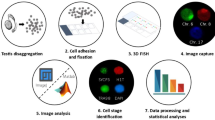
Abstract
A distinct form of X chromosome inactivation takes place during male meiosis, when the male sex chromosomes undergo a phenomenon known as meiotic sex chromosome inactivation (MSCI). MSCI is directed by DNA damage response signaling independent of Xist RNA to silence the transcriptional activity of the sex chromosomes, an essential event in male germ cell development. Here, we present protocols for the preparation and analyses of chromosome spread slides of mouse meiotic spermatocytes, thereby enabling a quick, inexpensive, and powerful cytological method to complement gene expression studies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
McKee BD, Handel MA (1993) Sex chromosomes, recombination, and chromatin conformation. Chromosoma 102(2):71–80
Turner JM (2007) Meiotic sex chromosome inactivation. Development 134(10):1823–1831. https://doi.org/10.1242/dev.000018
Ichijima Y, Sin HS, Namekawa SH (2012) Sex chromosome inactivation in germ cells: emerging roles of DNA damage response pathways. Cell Mol Life Sci 69(15):2559–2572. https://doi.org/10.1007/s00018-012-0941-5
Yan W, McCarrey JR (2009) Sex chromosome inactivation in the male. Epigenetics 4(7):452–456
Inagaki A, Schoenmakers S, Baarends WM (2010) DNA double strand break repair, chromosome synapsis and transcriptional silencing in meiosis. Epigenetics 5(4):255–266
Fernandez-Capetillo O, Mahadevaiah SK, Celeste A, Romanienko PJ, Camerini-Otero RD, Bonner WM, Manova K, Burgoyne P, Nussenzweig A (2003) H2AX is required for chromatin remodeling and inactivation of sex chromosomes in male mouse meiosis. Dev Cell 4(4):497–508
Ichijima Y, Ichijima M, Lou Z, Nussenzweig A, Camerini-Otero RD, Chen J, Andreassen PR, Namekawa SH (2011) MDC1 directs chromosome-wide silencing of the sex chromosomes in male germ cells. Genes Dev 25(9):959–971. https://doi.org/10.1101/gad.2030811
Broering TJ, Alavattam KG, Sadreyev RI, Ichijima Y, Kato Y, Hasegawa K, Camerini-Otero RD, Lee JT, Andreassen PR, Namekawa SH (2014) BRCA1 establishes DNA damage signaling and pericentric heterochromatin of the X chromosome in male meiosis. J Cell Biol 205(5):663–675. https://doi.org/10.1083/jcb.201311050
Namekawa SH, Park PJ, Zhang LF, Shima JE, McCarrey JR, Griswold MD, Lee JT (2006) Postmeiotic sex chromatin in the male germline of mice. Curr Biol 16(7):660–667. https://doi.org/10.1016/j.cub.2006.01.066
Turner JM, Mahadevaiah SK, Ellis PJ, Mitchell MJ, Burgoyne PS (2006) Pachytene asynapsis drives meiotic sex chromosome inactivation and leads to substantial postmeiotic repression in spermatids. Dev Cell 10(4):521–529. https://doi.org/10.1016/j.devcel.2006.02.009
Greaves IK, Rangasamy D, Devoy M, Marshall Graves JA, Tremethick DJ (2006) The X and Y chromosomes assemble into H2A.Z-containing [corrected] facultative heterochromatin [corrected] following meiosis. Mol Cell Biol 26(14):5394–5405. https://doi.org/10.1128/mcb.00519-06
van der Heijden GW, Derijck AA, Posfai E, Giele M, Pelczar P, Ramos L, Wansink DG, van der Vlag J, Peters AH, de Boer P (2007) Chromosome-wide nucleosome replacement and H3.3 incorporation during mammalian meiotic sex chromosome inactivation. Nat Genet 39(2):251–258. https://doi.org/10.1038/ng1949
Sin HS, Barski A, Zhang F, Kartashov AV, Nussenzweig A, Chen J, Andreassen PR, Namekawa SH (2012) RNF8 regulates active epigenetic modifications and escape gene activation from inactive sex chromosomes in post-meiotic spermatids. Genes Dev 26(24):2737–2748. https://doi.org/10.1101/gad.202713.112
Kato Y, Alavattam KG, Sin HS, Meetei A, Pang Q, Andreassen PR, Namekawa SH (2015) FANCB is essential in the male germline and regulates H3K9 methylation on the sex chromosomes during meiosis. Hum Mol Genet 24(18):5234–5249
Hasegawa K, Sin HS, Maezawa S, Broering TJ, Kartashov AV, Alavattam KG, Ichijima Y, Zhang F, Bacon WC, Greis KD, Andreassen PR, Barski A, Namekawa SH (2015) SCML2 establishes the male Germline Epigenome through regulation of histone H2A Ubiquitination. Dev Cell 32(5):574–588. https://doi.org/10.1016/j.devcel.2015.01.014
Alavattam KG, Kato Y, Sin HS, Maezawa S, Kowalski IJ, Zhang F, Pang Q, Andreassen PR, Namekawa SH (2016) Elucidation of the Fanconi Anemia protein network in meiosis and its function in the regulation of histone modifications. Cell Rep 17(4):1141–1157. https://doi.org/10.1016/j.celrep.2016.09.073
Peters AH, Plug AW, van Vugt MJ, de Boer P (1997) A drying-down technique for the spreading of mammalian meiocytes from the male and female germline. Chromosom Res 5(1):66–68
Namekawa SH (2014) Slide preparation method to preserve three-dimensional chromatin architecture of testicular germ cells. J Vis Exp 83:e50819. https://doi.org/10.3791/50819
Namekawa SH, Lee JT (2011) Detection of nascent RNA, single-copy DNA and protein localization by immunoFISH in mouse germ cells and preimplantation embryos. Nat Protoc 6(3):270–284. https://doi.org/10.1038/nprot.2010.195
Bellve AR (1993) Purification, culture, and fractionation of spermatogenic cells. Methods Enzymol 225:84–113
Gaysinskaya V, Bortvin A (2015) Flow cytometry of murine spermatocytes. Curr Protoc Cytom 72:7.44.1–7.4424. https://doi.org/10.1002/0471142956.cy0744s72
Gaysinskaya V, Soh IY, van der Heijden GW, Bortvin A (2014) Optimized flow cytometry isolation of murine spermatocytes. Cytometry A 85(6):556–565. https://doi.org/10.1002/cyto.a.22463
Bastos H, Lassalle B, Chicheportiche A, Riou L, Testart J, Allemand I, Fouchet P (2005) Flow cytometric characterization of viable meiotic and postmeiotic cells by Hoechst 33342 in mouse spermatogenesis. Cytometry A 65(1):40–49. https://doi.org/10.1002/cyto.a.20129
Getun IV, Torres B, Bois PR (2011) Flow cytometry purification of mouse meiotic cells. J Vis Exp (50). https://doi.org/10.3791/2602
Broering TJ, Wang YL, Pandey RN, Hegde RS, Wang SC, Namekawa SH (2015) BAZ1B is dispensable for H2AX phosphorylation on tyrosine 142 during spermatogenesis. Biology Open 4(7):873–884. https://doi.org/10.1242/bio.011734
Muhtadi R, Ahmed EA, Scherthan H (2017) Spermatocyte spreading during meiotic cell preparation is a two step process. J Cytol Histol 8(1000450). https://doi.org/10.4172/2157-7099.1000450
Acknowledgments
We thank past and present members of the Namekawa laboratory for optimizing the experimental conditions. We thank Anna Sosso for reviewing the chapter. This work was supported by the Albert J. Ryan Fellowship to K.G.A. and the NIH Grant GM098605 to S.H.N.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Alavattam, K.G., Abe, H., Sakashita, A., Namekawa, S.H. (2018). Chromosome Spread Analyses of Meiotic Sex Chromosome Inactivation. In: Sado, T. (eds) X-Chromosome Inactivation. Methods in Molecular Biology, vol 1861. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-8766-5_10
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8766-5_10
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-8765-8
Online ISBN: 978-1-4939-8766-5
eBook Packages: Springer Protocols