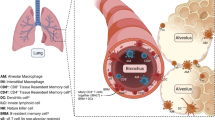
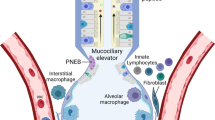
Abstract
The nasal passages, conducting airways and gas-exchange surfaces of the lung, are constantly exposed to substances contained in the air that we breathe. While many of these suspended substances are relatively harmless, some, for example, pathogenic microbes, noxious pollutants, and aspirated gastric contents can be harmful. The innate immune system, lungs and conducting airways have evolved specialized mechanisms to protect the respiratory system not only from these harmful inhaled substances but also from the overly exuberant innate immune activation that can arise during the host response to harmful inhaled substances. Herein, we discuss the cell types that contribute to lung innate immunity and inflammation and how their activities are coordinated to promote lung health.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bals R et al (1998) Human beta-defensin 2 is a salt-sensitive peptide antibiotic expressed in human lung. J Clin Invest 102(5):874–880
Bals R et al (1998) The peptide antibiotic LL-37/hCAP-18 is expressed in epithelia of the human lung where it has broad antimicrobial activity at the airway surface. Proc Natl Acad Sci U S A 95(16):9541–9546
Garcia JR et al (2001) Human beta-defensin 4: a novel inducible peptide with a specific salt-sensitive spectrum of antimicrobial activity. FASEB J 15(10):1819–1821
Goldman MJ et al (1997) Human beta-defensin-1 is a salt-sensitive antibiotic in lung that is inactivated in cystic fibrosis. Cell 88(4):553–560
Jia HP et al (2001) Discovery of new human beta-defensins using a genomics-based approach. Gene 263(1-2):211–218
Singh PK et al (1998) Production of beta-defensins by human airway epithelia. Proc Natl Acad Sci U S A 95(25):14,961–14,966
Alekseeva L et al (2009) Inducible expression of beta defensins by human respiratory epithelial cells exposed to Aspergillus fumigatus organisms. BMC microbiology 9:33
Beisswenger C, Bals R (2005) Functions of antimicrobial peptides in host defense and immunity. Curr Protein Pept Sci 6(3):255–264
Harder J et al (2000) Mucoid pseudomonas aeruginosa, TNF-alpha, and IL-1beta, but not IL-6, induce human beta-defensin-2 in respiratory epithelia. Am J Respir Cell Mol Biol 22(6):714–721
Thompson AB et al (1990) Lower respiratory tract lactoferrin and lysozyme arise primarily in the airways and are elevated in association with chronic bronchitis. J Lab Clin Med 115(2):148–158
Zhao YX et al (2000) Secretion of complement components of the alternative pathway (C3 and factor B) by the human alveolar type II epithelial cell line A549. Int J Mol Med 5(4):415–419
Becker MN et al (2000) CD14-dependent lipopolysaccharide-induced beta-defensin-2 expression in human tracheobronchial epithelium. J Biol Chem 275(38):29731–29736
Diamond G, Legarda D, Ryan LK (2000) The innate immune response of the respiratory epithelium. Immunol Rev 173:27–38
Jany B, Betz R, Schreck R (1995) Activation of the transcription factor NF-kappa B in human tracheobronchial epithelial cells by inflammatory stimuli. Eur Respir J 8(3):387–391
Jaspers I, Flescher E, Chen LC (1997) Ozone-induced IL-8 expression and transcription factor binding in respiratory epithelial cells. Am J Physiol 272(3 Pt 1):L504–L511
Janssen YM et al (1995) Asbestos induces nuclear factor kappa B (NF-kappa B) DNA-binding activity and NF-kappa B-dependent gene expression in tracheal epithelial cells. Proc Natl Acad Sci U S A 92(18):8458–8462
Takizawa H et al (1999) Diesel exhaust particles induce NF-kappa B activation in human bronchial epithelial cells in vitro: importance in cytokine transcription. J Immunol 162(8):4705–4711
Quay JL et al (1998) Air pollution particles induce IL-6 gene expression in human airway epithelial cells via NF-kappaB activation. Am J Respir Cell Mol Biol 19(1):98–106
Platz J et al (2004) Microbial DNA induces a host defense reaction of human respiratory epithelial cells. J Immunol 173(2):1219–1223
Skerrett SJ et al (2004) Respiratory epithelial cells regulate lung inflammation in response to inhaled endotoxin. Am J Physiol Lung Cell Mol Physiol 287(1):L143–L152
Hajjar AM et al (2005) An essential role for non-bone marrow-derived cells in control of Pseudomonas aeruginosa pneumonia. Am J Respir Cell Mol Biol 33(5):470–475
Cowburn AS et al (2008) Advances in neutrophil biology: clinical implications. Chest 134(3):606–612
Nathan C (2006) Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol 6(3):173–182
Wang Q, Doerschuk CM, Mizgerd JP (2004) Neutrophils in innate immunity. Semin Respir Crit Care Med 25(1):33–41
Sittipunt C et al (2001) Nitric oxide and nitrotyrosine in the lungs of patients with acute respiratory distress syndrome. Am J Respir Crit Care Med 163(2):503–510
Oppenheim JJ et al (2007) Alarmins initiate host defense. Adv Exp Med Biol 601:185–194
Wang J et al (2017) Visualizing the function and fate of neutrophils in sterile injury and repair. Science 358(6359):111–116
Furze RC, Rankin SM (2008) The role of the bone marrow in neutrophil clearance under homeostatic conditions in the mouse. FASEB J 22(9):3111–3119
Suratt BT et al (2001) Neutrophil maturation and activation determine anatomic site of clearance from circulation. Am J Physiol Lung Cell Mol Physiol 281(4):L913–L921
Furze RC, Rankin SM (2008) Neutrophil mobilization and clearance in the bone marrow. Immunology 125(3):281–288
Doherty DE et al (1992) Prolonged monocyte accumulation in the lung during bleomycin-induced pulmonary fibrosis. A noninvasive assessment of monocyte kinetics by scintigraphy. Lab Invest 66(2):231–242
Harmsen AG et al (1985) The role of macrophages in particle translocation from lungs to lymph nodes. Science 230(4731):1277–1280
Kitamura T et al (1999) Idiopathic pulmonary alveolar proteinosis as an autoimmune disease with neutralizing antibody against granulocyte/macrophage colony-stimulating factor. J Exp Med 190(6):875–880
Martin RJ et al (1980) Pulmonary alveolar proteinosis: the diagnosis by segmental lavage. Am Rev Respir Dis 121(5):819–825
Nugent KM, Pesanti EL (1983) Macrophage function in pulmonary alveolar proteinosis. Am Rev Respir Dis 127(6):780–781
Holt PG (1978) Inhibitory activity of unstimulated alveolar macrophages on T-lymphocyte blastogenic response. The American review of respiratory disease 118(4):791–793
Toews GB et al (1984) The accessory cell function of human alveolar macrophages in specific T cell proliferation. Journal of immunology 132(1):181–186
Gardai SJ et al (2003) By binding SIRPalpha or calreticulin/CD91, lung collectins act as dual function surveillance molecules to suppress or enhance inflammation. Cell 115(1):13–23
Morris DG et al (2003) Loss of integrin alpha(v)beta6-mediated TGF-beta activation causes Mmp12-dependent emphysema. Nature 422(6928):169–173
Munger JS et al (1999) The integrin alpha v beta 6 binds and activates latent TGF beta 1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96(3):319–328
Kawabe T et al (1992) Immunosuppressive activity induced by nitric oxide in culture supernatant of activated rat alveolar macrophages. Immunology 76(1):72–78
Roth MD, Golub SH (1993) Human pulmonary macrophages utilize prostaglandins and transforming growth factor beta 1 to suppress lymphocyte activation. Journal of leukocyte biology 53(4):366–371
Bilyk N, Holt PG (1993) Inhibition of the immunosuppressive activity of resident pulmonary alveolar macrophages by granulocyte/macrophage colony-stimulating factor. J Exp Med 177(6):1773–1777
Bromley SK, Mempel TR, Luster AD (2008) Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat Immunol 9(9):970–980
Springer TA (1994) Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell 76(2):301–314
Mould KJ et al (2017) Cell origin dictates programming of resident versus recruited macrophages during acute lung injury. Am J Respir Cell Mol Biol 57(3):294–306
Vignais PV (2002) The superoxide-generating NADPH oxidase: structural aspects and activation mechanism. Cellular and molecular life sciences : CMLS 59(9):1428–1459
Chang YC et al (1998) Virulence of catalase-deficient aspergillus nidulans in p47(phox)−/− mice. Implications for fungal pathogenicity and host defense in chronic granulomatous disease. J Clin Invest 101(9):1843–1850
Jackson SH, Gallin JI, Holland SM (1995) The p47phox mouse knock-out model of chronic granulomatous disease. J Exp Med 182(3):751–758
Kelly JK et al (1986) Fatal Aspergillus pneumonia in chronic granulomatous disease. Am J Clin Pathol 86(2):235–240
Tauber AI et al (1983) Chronic granulomatous disease: a syndrome of phagocyte oxidase deficiencies. Medicine 62(5):286–309
Castranova V (1994) Generation of oxygen radicals and mechanisms of injury prevention. Environ Health Perspect 102(Suppl 10):65–68
Fireman E et al (1989) Suppressive activity of alveolar macrophages and blood monocytes from interstitial lung diseases: role of released soluble factors. Int J Immunopharmacol 11(7):751–760
Gossart S et al (1996) Reactive oxygen intermediates as regulators of TNF-alpha production in rat lung inflammation induced by silica. J Immunol 156(4):1540–1548
Kondo T et al (1994) Current smoking of elderly men reduces antioxidants in alveolar macrophages. American J Respir Crit Care Med 149(1):178–182
Lake FR et al (1994) Functional switching of macrophage responses to tumor necrosis factor-alpha (TNF alpha) by interferons. Implications for the pleiotropic activities of TNF alpha. J Clin Invest 93(4):1661–1669
Laszlo DJ et al (1993) Development of functional diversity in mouse macrophages. Mutual exclusion of two phenotypic states. Am J Pathol 143(2):587–597
Noble PW et al (1993) Hyaluronate activation of CD44 induces insulin-like growth factor-1 expression by a tumor necrosis factor-alpha-dependent mechanism in murine macrophages. J Clin Invest 91(6):2368–2377
Riches DW et al (1988) Differential regulation of gene expression during macrophage activation with a polyribonucleotide. The role of endogenously derived IFN. J Immunol 141(1):180–188
Stout RD et al (2005) Macrophages sequentially change their functional phenotype in response to changes in microenvironmental influences. J Immunol 175(1):342–349
Gordon S (2003) Alternative activation of macrophages. Nat Rev Immunol 3(1):23–35
Riches DW (1995) Signalling heterogeneity as a contributing factor in macrophage functional diversity. Semin Cell Biol 6(6):377–384
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8(12):958–969
Mantovani A et al (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23(11):549–555
Mantovani A et al (2013) Macrophage plasticity and polarization in tissue repair and remodelling. J Pathol 229(2):176–185
Ramachandran P et al (2012) Differential Ly-6C expression identifies the recruited macrophage phenotype, which orchestrates the regression of murine liver fibrosis. Proc Natl Acad Sci U S A 109(46):E3186–E3195
Martinez FO et al (2006) Transcriptional profiling of the human monocyte-to-macrophage differentiation and polarization: new molecules and patterns of gene expression. J Immunol 177(10):7303–7311
Murray PJ, Wynn TA (2011) Obstacles and opportunities for understanding macrophage polarization. J Leukoc Biol 89(4):557–563
Sica A, Mantovani A (2012) Macrophage plasticity and polarization: in vivo veritas. J Clin Invest 122(3):787–795
Lawrence T, Natoli G (2011) Transcriptional regulation of macrophage polarization: enabling diversity with identity. Nat Rev Immunol 11(11):750–761
Banerjee S et al (2013) MicroRNA let-7c regulates macrophage polarization. J Immunol 190(12):6542–6549
Graff JW et al (2012) Identifying functional microRNAs in macrophages with polarized phenotypes. J Biol Chem 287(26):21816–21825
Liu G, Abraham E (2013) MicroRNAs in immune response and macrophage polarization. Arterioscler Thromb Vasc Biol 33(2):170–177
Shibata Y et al (2001) Alveolar macrophage deficiency in osteopetrotic mice deficient in macrophage colony-stimulating factor is spontaneously corrected with age and associated with matrix metalloproteinase expression and emphysema. Blood 98(9):2845–2852
Thepen T, Van Rooijen N, Kraal G (1989) Alveolar macrophage elimination in vivo is associated with an increase in pulmonary immune response in mice. J Exp Med 170(2):499–509
Conflicts of Interest
DWHR has no conflicts of interest to declare. TRM has no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Riches, D.W.H., Martin, T.R. (2018). Overview of Innate Lung Immunity and Inflammation. In: Alper, S., Janssen, W. (eds) Lung Innate Immunity and Inflammation. Methods in Molecular Biology, vol 1809. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8570-8_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8570-8_2
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8569-2
Online ISBN: 978-1-4939-8570-8
eBook Packages: Springer Protocols