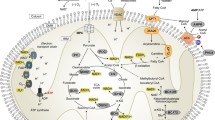
Abstract
Metabolic reprogramming has been associated to a plethora of diseases, and there has been increased demand for methodologies able to determine the metabolic alterations that characterize the pathological states and help developing metabolically centered therapies. In this chapter, methodologies for monitoring TCA cycle turnover and its interaction with pyruvate cycling and anaplerotic reactions will be presented. These methodologies are based in the application of stable 13C isotope “tracers”/substrates and 13C–NMR isotopomer analysis of metabolic intermediates. These methodologies can be applied at several organizational levels, ranging from isolated organelles and organs to whole organisms/humans. For the sake of simplicity, only very simple and well-defined models will be presented, including isolated heart mitochondria and isolated perfused hearts and livers.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Chance EM, Seeholzer SH, Kobayashi K, Williamson JR (1983) Mathematical analysis of isotope labeling in the citric acid cycle with applications to 13C NMR studies in perfused rat hearts. J Biol Chem 258:13785–13794
Malloy CR, Sherry AD, Jeffrey FM (1987) Carbon flux through citric acid cycle pathways in perfused heart by 13C NMR spectroscopy. FEBS Lett 212(1):58–62
Malloy CR, Sherry AD, Jeffrey FM (1990) Analysis of tricarboxylic acid cycle of the heart using 13C isotope isomers. Am J Phys 259(3 Pt 2):H987–H995
Weiss RG, Gloth ST, Kalil-Filho R, Chacko VP, Stern MD, Gerstenblith G (1992) Indexing tricarboxylic acid cycle flux in intact hearts by carbon-13 nuclear magnetic resonance. Circ Res 70:392–408
Yu X, White LT, Doumen C, Damico LA, LaNoue KF, Alpert NM, Lewandowski ED (1995) Kinetic analysis of dynamic 13C NMR spectra: metabolic flux, regulation, and compartmentation in hearts. Biophys J 69(5):2090–2102
Mason GF, Gruetter R, Rothman DL, Behar KL, Shulman RG, Novotny EJ (1995) Simultaneous determination of the rates of the TCA cycle, glucose utilization, alpha-ketoglutarate/glutamate exchange, and glutamine synthesis in human brain by NMR. J Cereb Blood Flow Metab 15:12–25
Gruetter R, Seaquist ER, Ugurbil K (2001) A mathematical model of compartmentalized neurotransmitter metabolism in the human brain. Am J Physiol Endocrinol Metab 281:E100–E112
Henry P-G, Öz G, Provencher S, Gruetter R (2003) Toward dynamic isotopomer analysis in the rat brain in vivo: automatic quantitation of 13C NMR spectra using LCModel. NMR Biomed 16:400–412
Des Rosiers C, Lloyd S, Comte B, Chatham JC (2004) A critical perspective of the use of 13C-isotopomer analysis by GCMS and NMR as applied to cardiac metabolism. Metab Eng 6(1):44–58
Malloy CR, Thompson JR, Jeffrey FM, Sherry AD (1990) Contribution of exogenous substrates to acetyl coenzyme a: measurement by 13C NMR under non-steady-state conditions. Biochemistry 29(29):6756–6761
Jessen ME, Kovarik TE, Jeffrey FM, Sherry AD, Storey CJ, Chao RY, Ring WS, Malloy CR (1993) Effects of amino acids on substrate selection, anaplerosis, and left ventricular function in the ischemic reperfused rat heart. J Clin Invest 92(2):831–839
Carvalho RA, Sousa RP, Cadete VJ, Lopaschuk GD, Palmeira CM, Bjork JA, Wallace KB (2010) Metabolic remodeling associated with subchronic doxorubicin cardiomyopathy. Toxicology 270:92–98
Ragavan M, Kirpich A, Fu X, Burgess SC, McIntyre LM, Merritt ME (2017) A comprehensive analysis of myocardial substrate preference emphasizes the need for a synchronized fluxomic/metabolomic research design. Am J Physiol Heart Circ Physiol 312(6):H1215–H1223
Jeffrey FM, Reshetov A, Storey CJ, Carvalho RA, Sherry AD, Malloy CR (1999) Use of a single 13C NMR resonance of glutamate for measuring oxygen consumption in tissue. Am J Phys 277:E1111–E1121
Carvalho RA, Zhao P, Wiegers CB, Jeffrey FM, Malloy CR, Sherry AD (2001) TCA cycle kinetics in the rat heart by analysis of 13C isotopomers using indirect 1H{13C}detection. Am J Physiol Heart Circ Physiol 281:H1413–H1421
Carvalho RA, Rodrigues TB, Zhao P, Jeffrey FM, Malloy CR, Sherry AD (2004) A 13C isotopomer kinetic analysis of cardiac metabolism: influence of altered cytosolic redox and [Ca2+]o. Am J Physiol Heart Circ Physiol 287(2):H889–H895
Lewandowski ED, Yu X, LaNoue KF, White LT, Doumen C, O'Donnell JM (1997) Altered metabolite exchange between subcellular compartments in intact postischemic rabbit hearts. Circ Res 81:165–175
O'Donnell JM, Doumen C, LaNoue KF, White LT, Yu X, Alpert NM, Lewandowski ED (1998) Dehydrogenase regulation of metabolite oxidation and efflux from mitochondria in intact hearts. Am J Phys 274(2 Pt 2):H467–H476
Jin ES, Jones JG, Merritt M, Burgess SC, Malloy CR, Sherry AD (2004) Glucose production, gluconeogenesis, and hepatic tricarboxylic acid cycle fluxes measured by nuclear magnetic resonance analysis of a single glucose derivative. Anal Biochem 327(2):149–155
Darley-Usmar VM, Rickwood D, Wilson MT (1987) Mitochondria - a practical approach. IRL Press Limited, England
Oliveira PJ, Rolo AP, Sardão VA, Coxito PM, Palmeira CM, Moreno AJ (2001) Carvedilol in heart mitochondria: protonophore or opener of the mitochondrial K(ATP) channels? Life Sci 69(2):123–132
Nunes PM, Jones JG, Rolo AP, Palmeira CM, Carvalho RA (2011) Ursodeoxycholic acid treatment of hepatic steatosis: a 13C-NMR metabolic study. NMR Biomed 24(9):1145–1158
Bartosek I, Guaitani A, Miller LL (1973) Isolated liver perfusion and its applications. Raven Press, New York
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Carvalho, R.A., Jarak, I. (2018). Mitochondrial Bioenergetics by 13C–NMR Isotopomer Analysis. In: Palmeira, C., Moreno, A. (eds) Mitochondrial Bioenergetics. Methods in Molecular Biology, vol 1782. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7831-1_13
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7831-1_13
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7830-4
Online ISBN: 978-1-4939-7831-1
eBook Packages: Springer Protocols