Abstract
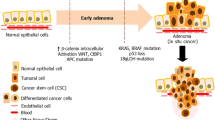
Multiple studies focused on tumor heterogeneity and cellular hierarchies have demonstrated the role of cancer stem cells (CSC) in tumor initiation and recurrence. Colorectal cancer is one of the leading causes of cancer-related death and is hierarchically organized, with the majority of tumor cells descending from a small population of colon cancer stem cells (CCSCs). Such a rare self-renewing population is marked by the acquisition of distinct chromatin regulation and transcriptional programs. Fundamental molecular deviations between CCSCs and bulk tumor cells as well as normal tissues represent a unique therapeutic access to develop novel, selective anticancer therapies.
In this chapter, we describe a methodological pipeline to identify novel molecules to selectively target human CCSC. We present a point-by-point description of a typical phenotypic molecular screening experiment, aiming to identify selective modulators of human CCSCs vs. normal intestinal progenitor cells.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bonnet D, Dick JE (1997) Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 3(7):730–737
Visvader JE, Lindeman GJ (2012) Cancer stem cells: current status and evolving complexities. Cell Stem Cell 10(6):717–728. https://doi.org/10.1016/j.stem.2012.05.007
Kreso A, Dick JE (2014) Evolution of the cancer stem cell model. Cell Stem Cell 14(3):275–291. https://doi.org/10.1016/j.stem.2014.02.006
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66(1):7–30. https://doi.org/10.3322/caac.21332
O'Brien CA, Pollett A, Gallinger S, Dick JE (2007) A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 445(7123):106–110. https://doi.org/10.1038/nature05372
Widschwendter M, Fiegl H, Egle D, Mueller-Holzner E, Spizzo G, Marth C, Weisenberger DJ, Campan M, Young J, Jacobs I, Laird PW (2007) Epigenetic stem cell signature in cancer. Nat Genet 39(2):157–158. https://doi.org/10.1038/ng1941
Suva ML, Riggi N, Bernstein BE (2013) Epigenetic reprogramming in cancer. Science 339(6127):1567–1570. https://doi.org/10.1126/science.1230184
Merlos-Suárez A, Barriga FM, Jung P, Iglesias M, Céspedes MV, Rossell D, Sevillano M, Hernando-Momblona X, da Silva-Diz V, Muñoz P, Clevers H, Sancho E, Mangues R, Batlle E (2011) The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Cell Stem Cell 8(5):511–524. https://doi.org/10.1016/j.stem.2011.02.020
Williams R (2013) Discontinued drugs in 2012: oncology drugs. Expert Opin Investig Drugs 22(12):1627–1644. https://doi.org/10.1517/13543784.2013.847088
Kotz J (2012) Phenotypic screening, take two. Sci BX 5(15). https://doi.org/10.1038/scibx.2012.380
Swinney DC (2013) Phenotypic vs. target-based drug discovery for first-in-class medicines. Clin Pharmacol Ther 93(4):299–301. https://doi.org/10.1038/clpt.2012.236
Swinney DC, Anthony J (2011) How were new medicines discovered? Nat Rev Drug Discov 10(7):507–519. https://doi.org/10.1038/nrd3480
Benoit YD, Guezguez B, Boyd AL, Bhatia M (2014) Molecular pathways: epigenetic modulation of Wnt-glycogen synthase kinase-3 signaling to target human cancer stem cells. Clin Cancer Res 20(21):5372–5378. https://doi.org/10.1158/1078-0432.CCR-13-2491
Kahn M (2014) Can we safely target the WNT pathway? Nat Rev Drug Discov 13(7):513–532. https://doi.org/10.1038/nrd4233
Sachlos E, Risueño RM, Laronde S, Shapovalova Z, Lee JH, Russell J, Malig M, McNicol JD, Fiebig-Comyn A, Graham M, Levadoux-Martin M, Lee JB, Giacomelli AO, Hassell JA, Fischer-Russell D, Trus MR, Foley R, Leber B, Xenocostas A, Brown ED, Collins TJ, Bhatia M (2012) Identification of drugs including a dopamine receptor antagonist that selectively target cancer stem cells. Cell 149(6):1284–1297. https://doi.org/10.1016/j.cell.2012.03.049
Hughes JP, Rees S, Kalindjian SB, Philpott KL (2011) Principles of early drug discovery. Br J Pharmacol 162(6):1239–1249. https://doi.org/10.1111/j.1476-5381.2010.01127.x
Zhang JH, Chung TD, Oldenburg KR (1999) A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J Biomol Screen 4(2):67–73. https://doi.org/10.1177/108705719900400206
Thorne N, Auld DS, Inglese J (2010) Apparent activity in high-throughput screening: origins of compound-dependent assay interference. Curr Opin Chem Biol 14(3):315–324. https://doi.org/10.1016/j.cbpa.2010.03.020
Gupta PB, Onder TT, Jiang G, Tao K, Kuperwasser C, Weinberg RA, Lander ES (2009) Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 138(4):645–659. https://doi.org/10.1016/j.cell.2009.06.034
Yasgar A, Titus SA, Wang Y, Danchik C, Yang SM, Vasiliou V, Jadhav A, Maloney DJ, Simeonov A, Martinez NJ (2017) A high-content assay enables the automated screening and identification of small molecules with specific ALDH1A1-inhibitory activity. PLoS One 12(1):e0170937. https://doi.org/10.1371/journal.pone.0170937
Perreault N, Beaulieu JF (1996) Use of the dissociating enzyme thermolysin to generate viable human normal intestinal epithelial cell cultures. Exp Cell Res 224(2):354–364
Prasetyanti PR, Zimberlin C, De Sousa E, Melo F, Medema JP (2013) Isolation and propagation of colon cancer stem cells. Methods Mol Biol 1035:247–259. https://doi.org/10.1007/978-1-62703-508-8_21
Benoit YD, Lussier C, Ducharme PA, Sivret S, Schnapp LM, Basora N, Beaulieu JF (2009) Integrin alpha8beta1 regulates adhesion, migration and proliferation of human intestinal crypt cells via a predominant RhoA/ROCK-dependent mechanism. Biol Cell 101(12):695–708. https://doi.org/10.1042/BC20090060
Benoit YD, Pare F, Francoeur C, Jean D, Tremblay E, Boudreau F, Escaffit F, Beaulieu JF (2010) Cooperation between HNF-1alpha, Cdx2, and GATA-4 in initiating an enterocytic differentiation program in a normal human intestinal epithelial progenitor cell line. Am J Physiol Gastrointest Liver Physiol 298(4):G504–G517. https://doi.org/10.1152/ajpgi.00265.2009
Sikandar SS, Pate KT, Anderson S, Dizon D, Edwards RA, Waterman ML, Lipkin SM (2010) NOTCH signaling is required for formation and self-renewal of tumor-initiating cells and for repression of secretory cell differentiation in colon cancer. Cancer Res 70(4):1469–1478. https://doi.org/10.1158/0008-5472.CAN-09-2557
Barbaric I, Jones M, Harley DJ, Gokhale PJ, Andrews PW (2011) High-content screening for chemical modulators of embryonal carcinoma cell differentiation and survival. J Biomol Screen 16(6):603–617. https://doi.org/10.1177/1087057111406547
Vermeulen L, De Sousa E, Melo F, van der Heijden M, Cameron K, de Jong JH, Borovski T, Tuynman JB, Todaro M, Merz C, Rodermond H, Sprick MR, Kemper K, Richel DJ, Stassi G, Medema JP (2010) Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat Cell Biol 12(5):468–476. https://doi.org/10.1038/ncb2048
Werbowetski-Ogilvie TE, Bhatia M (2008) Pluripotent human stem cell lines: what we can learn about cancer initiation. Trends Mol Med 14(8):323–332. https://doi.org/10.1016/j.molmed.2008.06.005
Benoit YD, Witherspoon MS, Laursen KB, Guezguez A, Beausejour M, Beaulieu JF, Lipkin SM, Gudas LJ (2013) Pharmacological inhibition of polycomb repressive complex-2 activity induces apoptosis in human colon cancer stem cells. Exp Cell Res 319(10):1463–1470. https://doi.org/10.1016/j.yexcr.2013.04.006
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Benoit, Y.D. (2018). Identification of Novel Molecules Targeting Cancer Stem Cells. In: Beaulieu, JF. (eds) Colorectal Cancer. Methods in Molecular Biology, vol 1765. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7765-9_22
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7765-9_22
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7764-2
Online ISBN: 978-1-4939-7765-9
eBook Packages: Springer Protocols