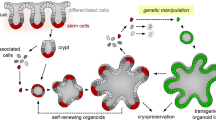
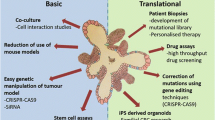
Abstract
The development of intestinal organoid technology has greatly accelerated research in the field of colorectal cancer. Contrary to traditional cancer cell lines, organoids are composed of multiple cell types arranged in 3D structures highly reminiscent of their native tissues. Thus, organoids provide a near-physiological and readily accessible model to study tissue morphogenesis, adult stem cell behavior and tumorigenesis. Here, we provide protocols for establishing intestinal organoid cultures from genetically modified mouse lines and describe methods to overexpress and knockout genes of interest using lentiviral-based approaches.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Holland JD, Klaus A, Garratt AN, Birchmeier W (2013) Wnt signaling in stem and cancer stem cells. Curr Opin Cell Biol 25(2):254–264. https://doi.org/10.1016/j.ceb.2013.01.004
Krausova M, Korinek V (2014) Wnt signaling in adult intestinal stem cells and cancer. Cell Signal 26(3):570–579. https://doi.org/10.1016/j.cellsig.2013.11.032
Fearon ER (2011) Molecular genetics of colorectal cancer. Annu Rev Pathol 6:479–507. https://doi.org/10.1146/annurev-pathol-011110-130235
Jackstadt R, Sansom OJ (2016) Mouse models of intestinal cancer. J Pathol 238(2):141–151. https://doi.org/10.1002/path.4645
Sansom OJ, Griffiths DF, Reed KR, Winton DJ, Clarke AR (2005) Apc deficiency predisposes to renal carcinoma in the mouse. Oncogene 24(55):8205–8210. https://doi.org/10.1038/sj.onc.1208956
Ashton GH, Morton JP, Myant K, Phesse TJ, Ridgway RA, Marsh V, Wilkins JA, Athineos D, Muncan V, Kemp R, Neufeld K, Clevers H, Brunton V, Winton DJ, Wang X, Sears RC, Clarke AR, Frame MC, Sansom OJ (2010) Focal adhesion kinase is required for intestinal regeneration and tumorigenesis downstream of Wnt/c-Myc signaling. Dev Cell 19(2):259–269. https://doi.org/10.1016/j.devcel.2010.07.015
Buller NV, Rosekrans SL, Metcalfe C, Heijmans J, van Dop WA, Fessler E, Jansen M, Ahn C, Vermeulen JL, Westendorp BF, Robanus-Maandag EC, Offerhaus GJ, Medema JP, D'Haens GR, Wildenberg ME, de Sauvage FJ, Muncan V, van den Brink GR (2015) Stromal Indian hedgehog signaling is required for intestinal adenoma formation in mice. Gastroenterology 148(1):170–180 e176. https://doi.org/10.1053/j.gastro.2014.10.006
Cordero JB, Ridgway RA, Valeri N, Nixon C, Frame MC, Muller WJ, Vidal M, Sansom OJ (2014) c-Src drives intestinal regeneration and transformation. EMBO J 33(13):1474–1491. https://doi.org/10.1002/embj.201387454
Marsh V, Winton DJ, Williams GT, Dubois N, Trumpp A, Sansom OJ, Clarke AR (2008) Epithelial Pten is dispensable for intestinal homeostasis but suppresses adenoma development and progression after Apc mutation. Nat Genet 40(12):1436–1444. https://doi.org/10.1038/ng.256
Sansom OJ, Meniel VS, Muncan V, Phesse TJ, Wilkins JA, Reed KR, Vass JK, Athineos D, Clevers H, Clarke AR (2007) Myc deletion rescues Apc deficiency in the small intestine. Nature 446(7136):676–679. https://doi.org/10.1038/nature05674
Gregorieff A, Liu Y, Inanlou MR, Khomchuk Y, Wrana JL (2015) Yap-dependent reprogramming of Lgr5(+) stem cells drives intestinal regeneration and cancer. Nature 526(7575):715–718. https://doi.org/10.1038/nature15382
Schepers AG, Snippert HJ, Stange DE, van den Born M, van Es JH, van de Wetering M, Clevers H (2012) Lineage tracing reveals Lgr5+ stem cell activity in mouse intestinal adenomas. Science 337(6095):730–735. https://doi.org/10.1126/science.1224676
Vermeulen L, Morrissey E, van der Heijden M, Nicholson AM, Sottoriva A, Buczacki S, Kemp R, Tavare S, Winton DJ (2013) Defining stem cell dynamics in models of intestinal tumor initiation. Science 342(6161):995–998. https://doi.org/10.1126/science.1243148
Date S, Sato T (2015) Mini-gut organoids: reconstitution of the stem cell niche. Annu Rev Cell Dev Biol 31:269–289. https://doi.org/10.1146/annurev-cellbio-100814-125218
Leushacke M, Barker N (2014) Ex vivo culture of the intestinal epithelium: strategies and applications. Gut 63(8):1345–1354. https://doi.org/10.1136/gutjnl-2014-307204
Drost J, van Jaarsveld RH, Ponsioen B, Zimberlin C, van Boxtel R, Buijs A, Sachs N, Overmeer RM, Offerhaus GJ, Begthel H, Korving J, van de Wetering M, Schwank G, Logtenberg M, Cuppen E, Snippert HJ, Medema JP, Kops GJ, Clevers H (2015) Sequential cancer mutations in cultured human intestinal stem cells. Nature 521(7550):43–47. https://doi.org/10.1038/nature14415
Matano M, Date S, Shimokawa M, Takano A, Fujii M, Ohta Y, Watanabe T, Kanai T, Sato T (2015) Modeling colorectal cancer using CRISPR-Cas9-mediated engineering of human intestinal organoids. Nat Med 21(3):256–262. https://doi.org/10.1038/nm.3802
Doench JG, Fusi N, Sullender M, Hegde M, Vaimberg EW, Donovan KF, Smith I, Tothova Z, Wilen C, Orchard R, Virgin HW, Listgarten J, Root DE (2016) Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR-Cas9. Nat Biotechnol 34(2):184–191. https://doi.org/10.1038/nbt.3437
Koo BK, Stange DE, Sato T, Karthaus W, Farin HF, Huch M, van Es JH, Clevers H (2011) Controlled gene expression in primary Lgr5 organoid cultures. Nat Methods 9(1):81–83. https://doi.org/10.1038/nmeth.1802
Sanjana NE, Shalem O, Zhang F (2014) Improved vectors and genome-wide libraries for CRISPR screening. Nat Methods 11(8):783–784. https://doi.org/10.1038/nmeth.3047
Shalem O, Sanjana NE, Hartenian E, Shi X, Scott DA, Mikkelsen TS, Heckl D, Ebert BL, Root DE, Doench JG, Zhang F (2014) Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 343(6166):84–87. https://doi.org/10.1126/science.1247005
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Oatway, C., Hirsch, C.L., Gregorieff, A. (2018). Use of Organoids to Characterize Signaling Pathways in Cancer Initiation. In: Beaulieu, JF. (eds) Colorectal Cancer. Methods in Molecular Biology, vol 1765. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7765-9_21
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7765-9_21
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7764-2
Online ISBN: 978-1-4939-7765-9
eBook Packages: Springer Protocols