Abstract
In retinal disease, despite the loss of light sensitivity as photoreceptors die, many retinal interneurons survive in a physiologically and metabolically functional state for long periods. This provides an opportunity for treatment by genetically adding a light sensitive function to these cells. Optogenetic therapies are in development, but, to date, they have suffered from low light sensitivity and narrow dynamic response range of microbial opsins. Expression of light-sensitive G protein coupled receptors (GPCRs), such as vertebrate rhodopsin , can increase sensitivity by signal amplification , as shown by several groups. Here, we describe the methods to (1) express light gated GPCRs in retinal neurons, (2) record light responses in retinal explants in vitro, (3) record cortical light responses in vivo, and (4) test visually guided behavior in treated mice.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
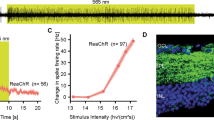
Similar content being viewed by others
References
Henriksen BS, Marc RE, Bernstein PS (2014) Optogenetics for retinal disorders. J Ophthalmic Vis Res 9:374–382
Marc R, Pfeiffer R, Jones B (2014) Retinal prosthetics, optogenetics, and chemical photoswitches. ACS Chem Neurosci 5:895–901
Busskamp V, Duebel J, Balya D et al (2010) Genetic reactivation of cone photoreceptors restores visual responses in retinitis pigmentosa. Science 329:413–417
Busskamp V, Picaud S, Sahel JA et al (2012) Optogenetic therapy for retinitis pigmentosa. Gene Ther 19:169–175
Nirenberg S, Pandarinath C (2012) Retinal prosthetic strategy with the capacity to restore normal vision. Proc Natl Acad Sci U S A 109:15012–15017
Doroudchi MM, Greenberg KP, Zorzos AN et al (2011) Towards optogenetic sensory replacement. Conf Proc IEEE Eng Med Biol Soc 2011:3139–3141
Doroudchi MM, Greenberg KP, Liu J et al (2011) Virally delivered channelrhodopsin-2 safely and effectively restores visual function in multiple mouse models of blindness. Mol Ther 19:1220–1229
Thyagarajan S, van Wyk M, Lehmann K et al (2010) Visual function in mice with photoreceptor degeneration and transgenic expression of channelrhodopsin 2 in ganglion cells. J Neurosci 30:8745–8758
Lagali PS, Balya D, Awatramani GB et al (2008) Light-activated channels targeted to ON bipolar cells restore visual function in retinal degeneration. Nat Neurosci 11:667–675
Bi A, Cui J, Ma YP et al (2006) Ectopic expression of a microbial-type rhodopsin restores visual responses in mice with photoreceptor degeneration. Neuron 50:23–33
Berger W, Kloeckener-Gruissem B, Neidhardt J (2010) The molecular basis of human retinal and vitreoretinal diseases. Prog Retin Eye Res 29:335–375
Shintani K, Shechtman DL, Gurwood AS (2009) Review and update: current treatment trends for patients with retinitis pigmentosa. Optometry 80:384–401
Curcio CA, Owsley C, Jackson GR (2000) Spare the rods, save the cones in aging and age-related maculopathy. Invest Ophthalmol Vis Sci 41:2015–2018
Ho AC, Humayun MS, Dorn JD et al (2015) Long-term results from an epiretinal prosthesis to restore sight to the blind. Ophthalmology 122:1547–1554
Mazzoni F, Novelli E, Strettoi E (2008) Retinal ganglion cells survive and maintain normal dendritic morphology in a mouse model of inherited photoreceptor degeneration. J Neurosci 28:14282–14292
Haverkamp S, Michalakis S, Claes E et al (2006) Synaptic plasticity in CNGA3(−/−) mice: cone bipolar cells react on the missing cone input and form ectopic synapses with rods. J Neurosci 26:5248–5255
RetroSense Therapeutics Phase I/II Clinical Trial for RST-001; trial #NCT02556736. www.clinicaltrials.gov.
Grossman N, Nikolic K, Grubb MS et al (2011) High-frequency limit of neural stimulation with ChR2. Conf Proc IEEE Eng Med Biol Soc 2011:4167–4170
Grossman N, Nikolic K, Toumazou C et al (2011) Modeling study of the light stimulation of a neuron cell with channelrhodopsin-2 mutants. IEEE Trans Biomed Eng 58:1742–1751
Cehajic-Kapetanovic J, Eleftheriou C, Allen AE et al (2015) Restoration of vision with ectopic expression of human rod opsin. Curr Biol 25:2111–2122
http://www.acucela.com/Read-About-Us/Press-Releases/160404_RP
Gaub BM, Berry MH, Holt AE (2015) Optogenetic vision restoration using rhodopsin for enhanced sensitivity. Mol Ther 23:1562–1571
Westenskow PD, Kurihara T, Bravo S et al (2015) Performing subretinal injections in rodents to deliver retinal pigment epithelium cells in suspension. J Vis Exp 95:52247
Flannery JG, Visel M (2013) Adeno-associated viral vectors for gene therapy of inherited retinal degenerations. Methods Mol Biol 935:351–369
Dalkara D, Byrne LC, Klimczak RR et al (2013) In vivo-directed evolution of a new adeno-associated virus for therapeutic outer retinal gene delivery from the vitreous. Sci Transl Med 5:189ra176
Petrs-Silva H, Dinculescu A, Li Q et al (2011) Novel properties of tyrosine-mutant AAV2 vectors in the mouse retina. Mol Ther 19:293–301
Mutter M, Benkner B, Münch T (2017) Optogenetik als mögliche Therapie bei degenerativen Netzhauterkrankungen. Med Genet 29:239–247
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Gaub, B.M., Berry, M.H., Visel, M., Holt, A., Isacoff, E.Y., Flannery, J.G. (2018). Optogenetic Retinal Gene Therapy with the Light Gated GPCR Vertebrate Rhodopsin. In: Boon, C., Wijnholds, J. (eds) Retinal Gene Therapy. Methods in Molecular Biology, vol 1715. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7522-8_12
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7522-8_12
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7521-1
Online ISBN: 978-1-4939-7522-8
eBook Packages: Springer Protocols