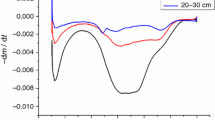
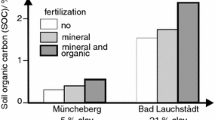
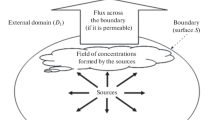
Abstract
Calorimetry measures the heat flow of any chemical, physical, and biological reaction and it is considered an important tool in all those scientific disciplines. Calorimetry evolves and focuses on designing more and more sensitive instruments capable of monitoring the heat rate associated with practically all living systems including soil. To study soil, there are sorts of calorimeters designed as multichannel systems that can monitor up to 24 samples at the same time. Calorimetry detects the heat released by the microbial metabolism, using low quantities of soil for the experimental measurements, ranging now from 0.8 g to 5 g depending on the goal of the study, on the kind of soil, and also on the calorimeter type. Beyond this, calorimeters permit monitoring the soil microbial metabolism directly and continuously, without disturbing the sample during long periods of time, and without the need to culture organisms from the soil or to add radiolabeled or fluorescent substrates.
All these advantages promoted the opening of new research goals in soil science to improve the existing knowledge about soil microbial metabolism, by searching and applying alternative indicators of the soil microbial biochemistry that can be quantified by calorimetry. More recently, and as a consequence of the latest advances in the design of these instruments, studies to monitor the response of the soil microbial population to changing temperature are starting to be considered, due to the direct involvement of soil in the environmental impact of climate change.
This chapter aims to give a specific and detailed description about how to apply calorimetry to study the soil microbial metabolism, responsible for soil biodegradation and deeply involved in the global C cycle, with guidelines going from the experimental design to the application of thermodynamic models to study the soil microbial biochemistry.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Paul EA (ed) (2007) Soil microbiology, ecology and biochemistry, 3rd edn. Academic, Elsevier
Delgado-Baquerizo M, Maestre FT, Reich PB, Jeffries TC, Gaitan JJ, Encinar D, Berdugo M, Campbell CD, Singh BK (2016) Microbial diversity drives multifunctionality in terrestrial ecosystems. Nat Commun 7:10541. doi:10.1038/ncomms10541
Riah-Anglet W, Trinsoutrot-Gattin I, Martin-Laurent F, Laroche-Ajzenberg E, Norini MP, Latour X, Laval K (2015) Soil microbial community structure and function relationships: a heat stress experiment. Appl Soil Ecol 86:121–130
Caldwell BA (2005) Enzyme activities as a component of soil biodiversity: a review. Pedobiologia 49:637–644
Ryan M, Law BE (2005) Interpreting, measuring, and modeling soil respiration. Biogeochem 73:3–27
Lehmann J, Kleber M (2015) The contentious nature of soil organic matter. Nature 528:60–68
Burns RG, De Forest JL, Marxen J, Sinsabaugh RL, Stromberger ME, Wallenstein MD, Weintraub MN, Zoppini A (2013) Soil enzymes in a changing world environment: current knowledge and future directions. Soil Biol Biochem 58:216–234
Blagodatskaya E, Blagodatsky S, Anderson TH, Kuzyakov Y (2014) Microbial growth and carbon use efficiency in the rhizosphere and root-free soil. PLoS One 9(4):e93282. doi:10.1371/journal.pone.0093282
Bölscher T, Wadsö L, Börjesson G, Herrmann AM (2016) Differences in substrate use efficiency: impacts of microbial community composition, land use management, and substrate complexity. Biol Fertil Soils 52:547–559
Forrest WW (1972) Microcalorimetry. In: Norris JR, Ribbons DW (eds) Methods in microcalorimetry, vol 6B. Academic, New York, p 285
Xing-Min R, Qiao-Yun H, Dai-Hua J, Peng C, Wei L (2007) Isothermal Microcalorimetry: a review of applications in soil and environmental sciences. Pedosphere 17(2):137–145
Barros N, Salgado J, Feijoo S (2007) Calorimetry and soil. Thermochim Acta 25:11–17
Poeplau C, Herrmann AM, Kätterer T (2016) Opposing effects of nitrogen and phosphorus on soil microbial metabolism and the implications for soil carbon storage. Soil Biol Biochem 100:83–91
Braissant O, Wirz D, Göpfert B, Daniels AU (2010) Use of isothermal calorimetry to monitor microbial activities. FEMS Microbiol Lett 303:1–8
Bravo D, Braissant O, Cailleau G, Verrecchia E, Junier P (2014) Isolation and characterization of oxalotrophic bacteria from tropical soils. Arch Microbiol 197:65–77
Swallow MJB, Quideau SA (2014) A method for determining community level physiological profiles of organic soil horizons. Soil Sci Soc Am J 79:536–542
Barros N, Feijoo S, Salgado J, Ramajo B, García JR, Hansen L (2008) The dry limit of microbial life in the Atacama desert revealed by calorimetric approaches. Eng Life Sci 8(5):477–486
Renault P, Ben-Sassi M, Bérard A (2013) Improving the MicroResp™ substrate-induced respiration method by a more complete description of CO2 behavior in closed incubation wells. Geoderma 207–208:82–91
Lebuhn M, Heilmann B, Hartmann A (1994) Effects of drying/rewetting stress on microbial auxin production and L-tryptochan catabolism. Biol Fertil Soils 18:302–310
Lauber CL, Zhou N, Gordon JI, Knight R, Fierer N (2010) Effect of storage conditions on the assessment of bacterial community structure in soil and human-associated samples. FEMS Microbiol Lett 307(1):80–86
Núñez-Regueira L, Barros N, Barja I (1994) Effect of storage of soil at 4 °C on the microbial activity studied by microcalorimetry. J Therm Anal Calorim 41:1379–1383
Barros N, Gómez-Orellana I, Feijoo S, Balsa R (1995) The effect of soil moisture on soil microbial activities studied by microcalorimetry. Thermochim Acta 249(1):161–168
Pesaro M, Nicollier G, Zeyer J, Widmer F (2004) Impact of soil drying-rewetting stress on microbial communities and activities and on degradation of two crop protection products. Appl Environ Microbiol 70:2577–2587
Barros N, Feijoo S, Simoni JA, Critter SAM, Airoldi C (2001) Interpretation of the metabolic enthalpy change, ΔHmet, calculated for microbial growth reactions in soils. J Therm Anal Calorim 63:577–588
Herrmann A, Bölscher T (2015) Simultaneous screening of microbial energetics and CO2 respiration in soil samples from different ecosystems. Soil Biol Biochem 83:88–92
Zheng S, Hu J, Chen K, Yao J, Yu Z, Lin X (2009) Soil microbial activity measured by microcalorimetry in response to long term fertilization regimes and available phosphorous on heat evolution. Soil Biol Biochem 41:2094–2099
Nuñez L, Barros N, Barja I (1994) A kinetic analysis of the degradation of glucose by soil microorganisms studied by microcalorimetry. Thermochim Acta 237:73–81
Vor T, Dyckmans J, Flessa H, Beese F (2002) Use of microcalorimetry to study microbial activity during the transition from oxic to anoxic conditions. Biol Fertil Soils 36:66–71
Boling EA, Blanchard GC, Russell WJ (1973) Bacterial identification by microcalorimetry. Nature 241:472–473
Barros N, Salgado J, Rodríguez-Añón JA, Proupín J, Villanueva M, Hansen L (2010) Calorimetric approach to metabolic carbon conversion efficiency in soils: comparison of experimental and theoretical models. J Therm Anal Calorim 99(3):771–777
Herrmann AM, Coucheney E, Nunan N (2014) Isothermal microcalorimetry provides new insight into terrestrial carbon cycle. Environ Sci Technol 48:4344–4352
Sparling GP (1983) Estimation of microbial biomass and activity in soil using microcalorimetry. J Soil Sci 34:381–390
Sesto E, Sigstad E (2011) A new approach to determine soil microbial biomass by calorimetry. J Therm Anal Calorim 104:23–29
Hassan W, Chen W, Huang Q, Mohamed I (2013) Microcalorimetric evaluation of soil microbiological properties under plant residues and dogmatic water gradients in Red soil. JPN Soc Soil Sci Plant Nutr 59(6):858–870
Xu J, Feng Y, Barros N, Zhong L, Cheng R, Liu X (2016) Exploring the potential of microcalorimetry to study the soil microbial metabolic diversity. J Therm Anal Calorim. doi:10.1007/s10973-016-5952-2
Battley EH (1987) Energetics of microbial growth. Wiley, New York
Geyer KM, Kyker-Snowman E, Grandy S, Frey SD (2016) Microbial carbon use efficiency: accounting for population, community, and ecosystem-scale control over the fate of metabolized organic matter. Biogeochem 127:173–188
Kimura T, Takahashi K (1985) Calorimetric studies of soil microbes: quantitative relation between Heat evolution during microbial degradation of glucose and changes in microbial activity. J Gen Microbiol 131:3083–3089
Barros N, Feijoo S (2003) A combined mass and energy balance to provide bioindicators of soil microbiological quality. Biophys Chem 104:561–572
Harris JA, Ritz K, Coucheney E, Grice SM, Lerch TZ, Pawlett M, Herrmann AM (2012) The thermodynamic efficiency of soil microbial communities subject to long-term stress is lower than those under conventional input regimes. Soil Biol Biochem 47:149–157
Braissant O, Keiser J, Meister I, Bachmann A, Wirz D, Göpfert B, Bonkat G, Wadsö I (2015) Isothermal microcaloriemtry accurately detects bacteria, tumorous microtissues and parasitic worms in a label-free well-plate assay. Biotechnol J 10(3):460–468
von Stockar U, Gustafsson L, Larsson C, Marison I, Tissot P, Gnaiger E (1993) Thermodynamic considerations in constructing energy balances for cellular growth. Biochim Biophys Acta 1183:221–240
Wadsö L, Hansen LD (2015) Calorespirometry of terrestrial organisms and ecosystems. Methods 76:11–19
Barros N, Feijoo S, Hansen LD (2011) Calorimetric determination of metabolic heat, CO2 rates and the calorespirometric ratio of soil basal metabolism. Geoderma 160:542–547
Barros N, Hansen LD, Piñeiro V, Perez-Cruzado C, Villanueva M, Proupín J, Rodríguez-Añón JA (2016) Factors influencing the calorespirometric ratios of soil microbial metabolism. Soil Biol Biochem 92:221–229
Hansen L, Mcfarlane C, McKinnon N, Smithv BN, Criddle RS (2004) Use of calorespirometric ratios, heat per CO2 and heat per O2, to quantify metabolic paths and energetics of growing cells. Thermochim Acta 422:55–61
Maskow T, Kemp R, Buchholz F, Schubert T, Kiesel B, Harms H (2010) What heat is telling us about microbial conversions in nature and technology: from chip-to megacalorimetry. Microb Biotechnol 3:269–284
Barros N, Piñeiro V, Hansen L (2015) Calorespirometry: a novel tool to assess the effect of temperature on soil organic matter decomposition. Thermochim Acta 618:15–17
Barros N, Hansen L, Piñeiro V, Vikegard P (2016) Calorimetry measures the response of soil organic matter biodegradation to increasing temperature. J Therm Anal Calorim 123:2397–2403
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Barros Pena, N. (2018). Calorimetry and Soil Biodegradation: Experimental Procedures and Thermodynamic Models. In: Bidoia, E., Montagnolli, R. (eds) Toxicity and Biodegradation Testing. Methods in Pharmacology and Toxicology. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7425-2_7
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7425-2_7
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7424-5
Online ISBN: 978-1-4939-7425-2
eBook Packages: Springer Protocols