Abstract
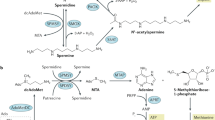
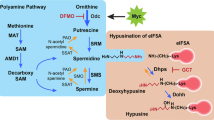
This chapter provides an overview of how the polyamine pathway has been exploited as a target for the treatment and prevention of multiple forms of cancer, since this pathway is disrupted in all cancers. It is divided into three main sections. The first explores how the polyamine pathway has been targeted for chemotherapy, starting from the first drug to target it, difluoromethylornithine (DFMO) to the large variety of polyamine analogues that have been synthesised and tested throughout the years with all their potentials and pitfalls. The second section focuses on the use of polyamines as vectors for drug delivery. Knowing that the polyamine transport system is upregulated in cancers and that polyamines naturally bind to DNA, a range of polyamine analogues and polyamine-like structures have been synthesised to target epigenetic regulators, with encouraging results. Furthermore, the use of polyamines as transport vectors to introduce toxic/bioactive/fluorescent agents more selectively to the intended target in cancer cells is discussed. The last section concentrates on chemoprevention, where the different strategies that have been undertaken to interfere with polyamine metabolism and function for antiproliferative intervention are outlined and discussed.
Similar content being viewed by others
References
Russell DH, Levy CC, Schimpff SC et al (1971) Urinary polyamines in cancer patients. Cancer Res 31:1555–1558
Kingsnorth AN, Wallace HM, Bundred NJ et al (1984) Polyamines in breast cancer. Br J Surg 71:352–356
Kingsnorth AN, Lumsden AB, Wallace HM (1984) Polyamines in colorectal cancer. Br J Surg 71:791–794
Giardiello FM, Hamilton SR, Hylind LM et al (1997) Ornithine decarboxylase and polyamines in familial adenomatous polyposis. Cancer Res 57:199–201
Manni A, Grove R, Kunselman S et al (1995) Involvement of the polyamine pathway in breast cancer progression. Cancer Lett 92:49–57
Bello-Fernandez C, Packham G, Cleveland JL (1993) The ornithine decarboxylase gene is a transcriptional target of c-Myc. Proc Natl Acad Sci U S A 90:7804–7808
Shantz LM, Levin VA (2007) Regulation of ornithine decarboxylase during oncogenic transformation: mechanisms and therapeutic potential. Amino Acids 33:213–223
Prakash NJ, Schechter PJ, Grove J et al (1978) Effect of alpha-difluoromethylornithine, an enzyme-activated irreversible inhibitor of ornithine decarboxylase, on L1210 leukemia in mice. Cancer Res 38:3059–3062
Meyskens FL Jr, Gerner EW (1999) Development of difluoromethylornithine (DFMO) as a chemoprevention agent. Clin Cancer Res 5:945–951
Abeloff MD, Rosen ST, Luk GD et al (1986) Phase II trials of alpha-difluoromethylornithine, an inhibitor of polyamine synthesis, in advanced small cell lung cancer and colon cancer. Cancer Treat Rep 70:843–845
Horn Y, Schechter PJ, Marton LJ (1987) Phase I–II clinical trial with alpha-difluoromethylornithine—an inhibitor of polyamine biosynthesis. Eur J Cancer Clin Oncol 23:1103–1107
Levin VA, Prados MD, Yung WK et al (1992) Treatment of recurrent gliomas with eflornithine. J Natl Cancer Inst 84:1432–1437
Prados MD, Wara WM, Sneed PK et al (2001) Phase III trial of accelerated hyperfractionation with or without difluromethylornithine (DFMO) versus standard fractionated radiotherapy with or without DFMO for newly diagnosed patients with glioblastoma multiforme. Int J Radiat Oncol Biol Phys 49:71–77
Meyskens FL Jr, Gerner EW, Emerson S et al (1998) Effect of alpha-difluoromethylornithine on rectal mucosal levels of polyamines in a randomized, double-blinded trial for colon cancer prevention. J Natl Cancer Inst 90:1212–1218
Williams-Ashman HG, Schenone A (1972) Methyl glyoxal bis(guanylhydrazone) as a potent inhibitor of mammalian and yeast S-adenosylmethionine decarboxylases. Biochem Biophys Res Commun 46:288–295
Nass MM (1984) Analysis of methylglyoxal bis(guanylhydrazone)-induced alterations of hamster tumor mitochondria by correlated studies of selective rhodamine binding, ultrastructural damage, DNA replication, and reversibility. Cancer Res 44:2677–2688
Regenass U, Mett H, Stanek J et al (1994) CGP 48664, a new S-adenosylmethionine decarboxylase inhibitor with broad spectrum antiproliferative and antitumor activity. Cancer Res 54:3210–3217
Pless M, Belhadj K, Menssen HD et al (2004) Clinical efficacy, tolerability, and safety of SAM486A, a novel polyamine biosynthesis inhibitor, in patients with relapsed or refractory non-Hodgkin’s lymphoma: results from a phase II multicenter study. Clin Cancer Res 10:1299–1305
van Zuylen L, Bridgewater J, Sparreboom A et al (2004) Phase I and pharmacokinetic study of the polyamine synthesis inhibitor SAM486A in combination with 5-fluorouracil/leucovorin in metastatic colorectal cancer. Clin Cancer Res 10:1949–1955
Wallace HM, Niiranen K (2007) Polyamine analogues – an update. Amino Acids 33:261–265
Wallace HM, Fraser AV (2003) Polyamine analogues as anticancer drugs. Biochem Soc Trans 31:393–396
Casero RA Jr, Marton LJ (2007) Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases. Nat Rev Drug Discov 6:373–390
Porter CW, Bergeron RJ (1988) Regulation of polyamine biosynthetic activity by spermidine and spermine analogs—a novel antiproliferative strategy. Adv Exp Med Biol 250:677–690
Porter CW, Ganis B, Libby PR et al (1991) Correlations between polyamine analogue-induced increases in spermidine/spermine N1-acetyltransferase activity, polyamine pool depletion, and growth inhibition in human melanoma cell lines. Cancer Res 51:3715–3720
Wallace HM, Mackarel AJ (1998) Regulation of polyamine acetylation and efflux in human cancer cells. Biochem Soc Trans 26:571–575
Pledgie-Tracy A, Billam M, Hacker A et al (2010) The role of the polyamine catabolic enzymes SSAT and SMO in the synergistic effects of standard chemotherapeutic agents with a polyamine analogue in human breast cancer cell lines. Cancer Chemother Pharmacol 65:1067–1081
Casero RA Jr, Celano P, Ervin SJ et al (1989) Differential induction of spermidine/spermine N1-acetyltransferase in human lung cancer cells by the bis(ethyl)polyamine analogues. Cancer Res 49:3829–3833
Wolff AC, Armstrong DK, Fetting JH et al (2003) A Phase II study of the polyamine analog N1,N11-diethylnorspermine (DENSpm) daily for five days every 21 days in patients with previously treated metastatic breast cancer. Clin Cancer Res 9:5922–5928
Streiff RR, Bender JF (2001) Phase 1 study of N1-N11-diethylnorspermine (DENSPM) administered TID for 6 days in patients with advanced malignancies. Investig New Drugs 19:29–39
Hector S, Porter CW, Kramer DL et al (2004) Polyamine catabolism in platinum drug action: Interactions between oxaliplatin and the polyamine analogue N1,N11-diethylnorspermine at the level of spermidine/spermine N1-acetyltransferase. Mol Cancer Ther 3:813–822
Choi W, Gerner EW, Ramdas L et al (2005) Combination of 5-fluorouracil and N1,N11-diethylnorspermine markedly activates spermidine/spermine N1-acetyltransferase expression, depletes polyamines, and synergistically induces apoptosis in colon carcinoma cells. J Biol Chem 280:3295–3304
Casero RA Jr, Mank AR, Saab NH et al (1995) Growth and biochemical effects of unsymmetrically substituted polyamine analogues in human lung tumor cells 1. Cancer Chemother Pharmacol 36:69–74
Hacker A, Marton LJ, Sobolewski M et al (2008) In vitro and in vivo effects of the conformationally restricted polyamine analogue CGC-11047 on small cell and non-small cell lung cancer cells. Cancer Chemother Pharmacol 63:45–53
Saab NH, West EE, Bieszk NC et al (1993) Synthesis and evaluation of unsymmetrically substituted polyamine analogues as modulators of human spermidine/spermine-N1-acetyltransferase (SSAT) and as potential antitumor agents. J Med Chem 36:2998–3004
Fraser AV, Woster PM, Wallace HM (2002) Induction of apoptosis in human leukaemic cells by IPENSpm, a novel polyamine analogue and anti-metabolite. Biochem J 367:307–312
Dredge K, Kink JA, Johnson RM et al (2009) The polyamine analog PG11047 potentiates the antitumor activity of cisplatin and bevacizumab in preclinical models of lung and prostate cancer. Cancer Chemother Pharmacol 65:191–195
Carew JS, Nawrocki ST, Reddy VK et al (2008) The novel polyamine analogue CGC-11093 enhances the antimyeloma activity of bortezomib. Cancer Res 68:4783–4790
Huang Y, Hager ER, Phillips DL et al (2003) A novel polyamine analog inhibits growth and induces apoptosis in human breast cancer cells. Clin Cancer Res 9(7):2769–2777
Huang Y, Keen JC, Pledgie A et al (2006) Polyamine analogues down-regulate estrogen receptor alpha expression in human breast cancer cells. J Biol Chem 281:19055–19063
Shi Y, Lan F, Matson C et al (2004) Histone demethylation mediated by the nuclear amine oxidase homolog LSD1. Cell 119:941–953
Zheng YC, Ma J, Wang Z et al (2015) A systematic review of histone lysine-specific demethylase 1 and its inhibitors. Med Res Rev 35:1032–1071
Huang Y, Stewart TM, Wu Y et al (2009) Novel oligoamine analogues inhibit lysine-specific demethylase 1 and induce reexpression of epigenetically silenced genes. Clin Cancer Res 15:7217–7228
Zhu Q, Huang Y, Marton LJ et al (2012) Polyamine analogs modulate gene expression by inhibiting lysine-specific demethylase 1 (LSD1) and altering chromatin structure in human breast cancer cells. Amino Acids 42:887–898
Wu Y, Steinbergs N, Murray-Stewart T et al (2012) Oligoamine analogues in combination with 2-difluoromethylornithine synergistically induce re-expression of aberrantly silenced tumour-suppressor genes. Biochem J 442:693–701
Nowotarski SL, Pachaiyappan B, Holshouser SL et al (2015) Structure-activity study for (bis)ureidopropyl- and (bis)thioureidopropyldiamine LSD1 inhibitors with 3-5-3 and 3-6-3 carbon backbone architectures. Bioorg Med Chem 23:1601–1612
Hazeldine S, Pachaiyappan B, Steinbergs N et al (2012) Low molecular weight amidoximes that act as potent inhibitors of lysine-specific demethylase 1. J Med Chem 55:7378–7391
Milovica V, Turchanowa L, Khomutov AR et al (2001) Hydroxylamine-containing inhibitors of polyamine biosynthesis and impairment of colon cancer cell growth. Biochem Pharmacol 61:199–206
Liang F, Wan S, Li Z et al (2006) Medical applications of macrocyclic polyamines. Curr Med Chem 13:711–727
Pasini A, Caldarera CM, Giordano E (2014) Chromatin remodeling by polyamines and polyamine analogs. Amino Acids 46:595–603
Varghese S, Gupta D, Baran T et al (2005) Alkyl-substituted polyaminohydroxamic acids: a novel class of targeted histone deacetylase inhibitors. J Med Chem 48:6350–6365
Palmer AJ, Wallace HM (2010) The polyamine transport system as a target for anticancer drug development. Amino Acids 38:415–422
Aldana-Masangkay GI, Sakamoto KM (2011) The role of HDAC6 in cancer. J Biomed Biotechnol 2011:875824
Varghese S, Senanayake T, Murray-Stewart T et al (2008) Polyaminohydroxamic acids and polyaminobenzamides as isoform selective histone deacetylase inhibitors. J Med Chem 51:2447–2456
Sharma SK, Hazeldine S, Crowley ML et al (2012) Polyamine-based small molecule epigenetic modulators. Medchemcomm 3:14–21
Bandyopadhyay K, Baneres JL, Martin A et al (2009) Spermidinyl-CoA-based HAT inhibitors block DNA repair and provide cancer-specific chemo- and radiosensitization. Cell Cycle 8:2779–2788
Phanstiel O 4th, Kaur N, Delcros JG (2007) Structure-activity investigations of polyamine-anthracene conjugates and their uptake via the polyamine transporter. Amino Acids 33:305–313
Xie S, Wang J, Zhang Y et al (2010) Antitumor conjugates with polyamine vectors and their molecular mechanisms. Expert Opin Drug Deliv 7:1049–1061
Thibault B, Clement E, Zorza G et al (2016) F14512, a polyamine-vectorized inhibitor of topoisomerase II, exhibits a marked anti-tumor activity in ovarian cancer. Cancer Lett 370:10–18
Annereau JP, Brel V, Dumontet C et al (2010) A fluorescent biomarker of the polyamine transport system to select patients with AML for F14512 treatment. Leuk Res 34:1383–1389
Kruczynski A, Pillon A, Creancier L et al (2013) F14512, a polyamine-vectorized anti-cancer drug, currently in clinical trials exhibits a marked preclinical anti-leukemic activity. Leukemia 27:2139–2148
Traquete R, Ghani RA, Phanstiel O et al (2013) Ant 4,4, a polyamine-anthracene conjugate, induces cell death and recovery in human promyelogenous leukemia cells (HL-60). Amino Acids 44:1193–1203
Tian ZY, Xie SQ, Du YW et al (2009) Synthesis, cytotoxicity and apoptosis of naphthalimide polyamine conjugates as antitumor agents. Eur J Med Chem 44:393–399
Li M, Li Q, Zhang YH et al (2013) Antitumor effects and preliminary systemic toxicity of ANISpm in vivo and in vitro. Anti-Cancer Drugs 24:32–42
Magoulas G, Papaioannou D, Papadimou E et al (2009) Preparation of spermine conjugates with acidic retinoids with potent ribonuclease P inhibitory activity. Eur J Med Chem 44:2689–2695
Tang XH, Gudas LJ (2011) Retinoids, retinoic acid receptors, and cancer. Annu Rev Pathol 6:345–364
Vourtsis D, Lamprou M, Sadikoglou E et al (2013) Effect of an all-trans-retinoic acid conjugate with spermine on viability of human prostate cancer and endothelial cells in vitro and angiogenesis in vivo. Eur J Pharmacol 698:122–130
Agostinelli E, Vianello F, Magliulo G et al (2015) Nanoparticle strategies for cancer therapeutics: nucleic acids, polyamines, bovine serum amine oxidase and iron oxide nanoparticles (review). Int J Oncol 46:5–16
Cui PF, Xing L, Qiao JB et al (2016) Polyamine metabolism-based dual functional gene delivery system to synergistically inhibit the proliferation of cancer. Int J Pharm 506:79–86
Jeter JM, Alberts DS (2012) Difluoromethylornithine: the proof is in the polyamines. Cancer Prev Res (Phila) 5:1341–1344
Shapiro J, Lui H (2001) Vaniqa—eflornithine 13.9% cream. Skin Therapy Lett 6:1–3. 5
Gerner EW, Meyskens FL Jr (2009) Combination chemoprevention for colon cancer targeting polyamine synthesis and inflammation. Clin Cancer Res 15:758–761
Burke CA, Dekker E, Samadder NJ et al (2016) Efficacy and safety of eflornithine (CPP-1X)/sulindac combination therapy versus each as monotherapy in patients with familial adenomatous polyposis (FAP): design and rationale of a randomized, double-blind, Phase III trial. BMC Gastroenterol 16:87
Witherspoon M, Chen Q, Kopelovich L et al (2013) Unbiased metabolite profiling indicates that a diminished thymidine pool is the underlying mechanism of colon cancer chemoprevention by alpha-difluoromethylornithine. Cancer Discov 3:1072–1081
Zell JA, McLaren CE, Chen WP et al (2010) Ornithine decarboxylase-1 polymorphism, chemoprevention with eflornithine and sulindac, and outcomes among colorectal adenoma patients. J Natl Cancer Inst 102:1513–1516
Bassiri H, Benavides A, Haber M et al (2015) Translational development of difluoromethylornithine (DFMO) for the treatment of neuroblastoma. Transl Pediatr 4:226–238
Westermann F, Muth D, Benner A et al (2008) Distinct transcriptional MYCN/c-MYC activities are associated with spontaneous regression or malignant progression in neuroblastomas. Genome Biol 9:R150
Wallick CJ, Gamper I, Thorne M et al (2005) Key role for p27Kip1, retinoblastoma protein Rb, and MYCN in polyamine inhibitor-induced G1 cell cycle arrest in MYCN-amplified human neuroblastoma cells. Oncogene 24:5606–5618
Yco LP, Geerts D, Mocz G et al (2015) Effect of sulfasalazine on human neuroblastoma: analysis of sepiapterin reductase (SPR) as a new therapeutic target. BMC Cancer 15:477–488
Saulnier Sholler GL, Gerner EW, Bergendahl G et al (2015) A phase I trial of DFMO targeting polyamine addiction in patients with relapsed/refractory neuroblastoma. PLoS One 10:e0127246
Alberts DS, Dorr RT, Einspahr JG et al (2000) Chemoprevention of human actinic keratoses by topical 2-(difluoromethyl)-dl-ornithine. Cancer Epidemiol Biomark Prev 9:1281–1286
Kreul SM, Havighurst T, Kim K et al (2012) A phase III skin cancer chemoprevention study of DFMO: long-term follow-up of skin cancer events and toxicity. Cancer Prev Res (Phila) 5:1368–1374
Jeter JM, Curiel-Lewandrowski C, Stratton SP et al (2016) Phase IIB randomized study of topical difluoromethylornithine and topical diclofenac on sun-damaged skin of the forearm. Cancer Prev Res (Phila) 9:128–134
Elmets CA, Athar M (2010) Targeting ornithine decarboxylase for the prevention of nonmelanoma skin cancer in humans. Cancer Prev Res (Phila) 3:8–11
Meyskens FL Jr, Simoneau AR, Gerner EW (2014) Chemoprevention of prostate cancer with the polyamine synthesis inhibitor difluoromethylornithine. Recent Results Cancer Res 202:115–120
Sinicrope FA, Broaddus R, Joshi N et al (2011) Evaluation of difluoromethylornithine for the chemoprevention of Barrett’s esophagus and mucosal dysplasia. Cancer Prev Res (Phila) 4:829–839
Vlastos AT, West LA, Atkinson EN et al (2005) Results of a phase II double-blinded randomized clinical trial of difluoromethylornithine for cervical intraepithelial neoplasia grades 2 to 3. Clin Cancer Res 11:390–396
Messing E, Kim KM, Sharkey F et al (2006) Randomized prospective phase III trial of difluoromethylornithine vs placebo in preventing recurrence of completely resected low risk superficial bladder cancer. J Urol 176:500–504
Fabian CJ, Kimler BF, Brady DA et al (2002) A phase II breast cancer chemoprevention trial of oral alpha-difluoromethylornithine: breast tissue, imaging, and serum and urine biomarkers. Clin Cancer Res 8:3105–3117
Babbar N, Gerner EW, Casero RA Jr (2006) Induction of spermidine/spermine N1-acetyltransferase (SSAT) by aspirin in Caco-2 colon cancer cells. Biochem J 394:317–324
Xu H, Chaturvedi R, Cheng Y et al (2004) Spermine oxidation induced by Helicobacter pylori results in apoptosis and DNA damage: implications for gastric carcinogenesis. Cancer Res 64:8521–8525
Chaturvedi R, de Sablet T, Peek RM et al (2012) Spermine oxidase, a polyamine catabolic enzyme that links Helicobacter pylori CagA and gastric cancer risk. Gut Microbes 3:48–56
Chaturvedi R, de Sablet T, Asim M et al (2015) Increased Helicobacter pylori-associated gastric cancer risk in the Andean region of Colombia is mediated by spermine oxidase. Oncogene 34:3429–3440
Murray-Stewart T, Sierra JC, Piazuelo MB et al (2016) Epigenetic silencing of miR-124 prevents spermine oxidase regulation: implications for Helicobacter pylori-induced gastric cancer. Oncogene 35:5480–5488
Goodwin AC, Destefano Shields CE, Wu S et al (2011) Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc Natl Acad Sci U S A 108:15354–15359
O’Hagan HM, Wang W, Sen S et al (2011) Oxidative damage targets complexes containing DNA methyltransferases, SIRT1, and polycomb members to promoter CpG Islands. Cancer Cell 20:606–619
Hong SK, Chaturvedi R, Piazuelo MB et al (2010) Increased expression and cellular localization of spermine oxidase in ulcerative colitis and relationship to disease activity. Inflamm Bowel Dis 16:1557–1566
Goodwin AC, Jadallah S, Toubaji A et al (2008) Increased spermine oxidase expression in human prostate cancer and prostatic intraepithelial neoplasia tissues. Prostate 68:766–772
Tucker JM, Murphy JT, Kisiel N et al (2005) Potent modulation of intestinal tumorigenesis in Apcmin/+ mice by the polyamine catabolic enzyme spermidine/spermine N1-acetyltransferase. Cancer Res 65:5390–5398
Wang X, Feith DJ, Welsh P et al (2007) Studies of the mechanism by which increased spermidine/spermine N1-acetyltransferase activity increases susceptibility to skin carcinogenesis. Carcinogenesis 28:2404–2411
Hughes A, Smith NI, Wallace HM (2003) Polyamines reverse non-steroidal anti-inflammatory drug-induced toxicity in human colorectal cancer cells. Biochem J 374:481–488
Saunders FR, Wallace HM (2007) Polyamine metabolism and cancer prevention. Biochem Soc Trans 35:364–368
Babbar N, Gerner EW (2011) Targeting polyamines and inflammation for cancer prevention. Recent Results Cancer Res 188:49–64
Xie SQ, Zhang YH, Li Q et al (2012) COX-2-independent induction of apoptosis by celecoxib and polyamine naphthalimide conjugate mediated by polyamine depression in colorectal cancer cell lines. Int J Color Dis 27:861–868
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Damiani, E., Wallace, H.M. (2018). Polyamines and Cancer. In: Alcázar, R., Tiburcio, A. (eds) Polyamines. Methods in Molecular Biology, vol 1694. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7398-9_39
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7398-9_39
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7397-2
Online ISBN: 978-1-4939-7398-9
eBook Packages: Springer Protocols